The Cardiovascular Disease Treatment category captures 25.0% of the application segment, generating the largest procurement volumes from established buyers. This application dominance reflects structural demand tied to industry operations rather than discretionary purchasing decisions, creating revenue stability that persists through economic cycles.
The ablation technology market encompasses the full spectrum of medical ablation energy platforms including radiofrequency, laser, ultrasound, electrical, cryogenic, microwave, and hydrothermal modalities for cardiovascular, oncological, and interventional applications.
Market scope includes all commercially traded products segmented by product, application, end use. Revenue sizing spans the 2026 to 2036 forecast period.
The scope excludes related but distinct product categories and components manufactured exclusively for captive internal consumption.
The ablation technology market is expanding as core application sectors increase procurement volumes and new use cases emerge from technology development. Radiofrequency Ablators holds the largest share at 46.0% of the product segment, reflecting established market preference for proven product configurations that deliver reliable performance. Demand growth tracks with end-use industry investment cycles and capacity expansion programmes.
The Cardiovascular Disease Treatment category captures 25.0% of the application segment, generating the largest procurement volumes from established buyers. This application dominance reflects structural demand tied to industry operations rather than discretionary purchasing decisions, creating revenue stability that persists through economic cycles.
Geographic demand distribution reflects industrial development maturity and sector-specific investment patterns. Japan leads growth at 10.6% CAGR, driven by industrial capacity expansion and infrastructure development. Developed markets contribute steady replacement and upgrade demand that anchors base revenue, while emerging markets provide growth acceleration from new installation and capacity expansion.
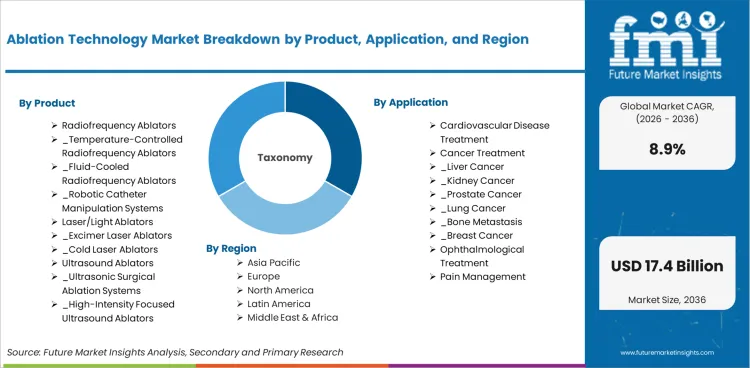
The ablation technology market is segmented by product, application, end use, and geographic regions. By product, the market is segmented as Radiofrequency Ablators, Laser/Light Ablators, Ultrasound Ablators, Electrical Ablators, Cryoablation Devices, Microwave Ablators, Hydrothermal/Hydromechanical Ablators. By application, the market is classified by Cardiovascular Disease Treatment, Cancer Treatment, Ophthalmological Treatment, Pain Management, Urological Treatment, Orthopedic Treatment, Cosmetic/Aesthetic Surgery, Gynecological Treatment, Others.
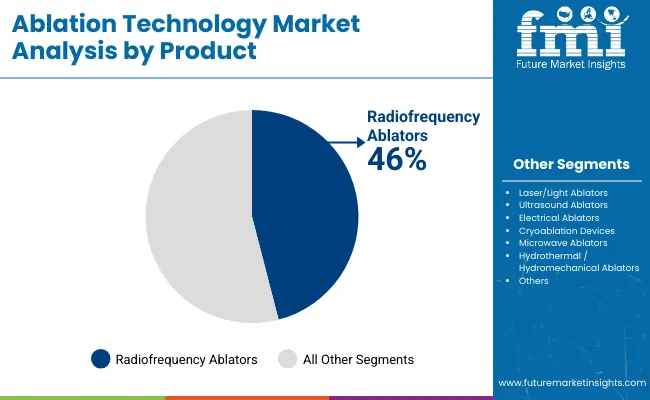
Radiofrequency Ablators is projected to lead the market with a 46.0% share of the product segment in 2026. This leadership position reflects established performance credentials, broad application suitability, and competitive economics that sustain procurement preference. Growth trajectory correlates with overall market expansion as demand from primary application sectors continues to increase with industry investment and capacity expansion.
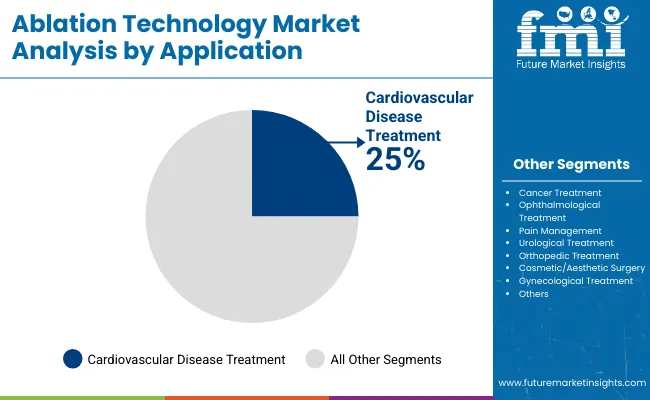
Cardiovascular Disease Treatment is expected to capture 25.0% of the application segment in 2026. This segment generates the largest procurement volumes from established industrial and institutional buyers who maintain standardized procurement specifications. Demand growth tracks with end-use industry investment and capacity expansion programmes.
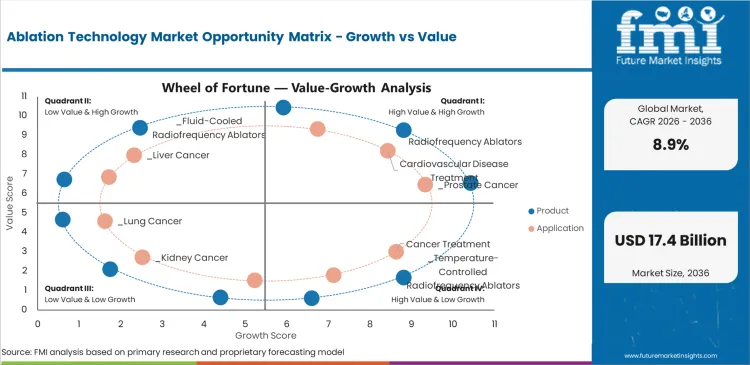
The ablation technology market is advancing as primary application sectors expand and technology development creates new opportunities. Manufacturers are investing in production capacity, product innovation, and market expansion. Despite challenges including cost volatility, competitive pressure, and regulatory complexity, growth opportunities are emerging from technology advancement, sustainability requirements, and geographic expansion.
Demand is shaped by investment expansion across core application sectors. Each major end-use industry is expanding capacity, modernizing facilities, and upgrading specifications in ways that generate procurement volumes. The Cardiovascular Disease Treatment application alone accounts for 25.0% of demand, creating a concentrated revenue base tied to industry operational requirements.
Raw material pricing fluctuations and supply chain constraints create margin variability. Companies with integrated supply chains or long-term procurement contracts maintain more stable cost structures. Product mix optimization toward premium specifications improves average selling prices.
Growth reflects adoption of advanced materials, improved designs, and enhanced performance specifications. Manufacturers investing in R&D create differentiated products commanding pricing premiums. Customer demand for improved performance and sustainability compliance is driving innovation across the competitive landscape.
.webp)
| Country | CAGR |
|---|---|
| Japan | 10.6% |
| UK | 9.8% |
| China | 9.7% |
| USA | 9.3% |
| South Korea | 6.7% |
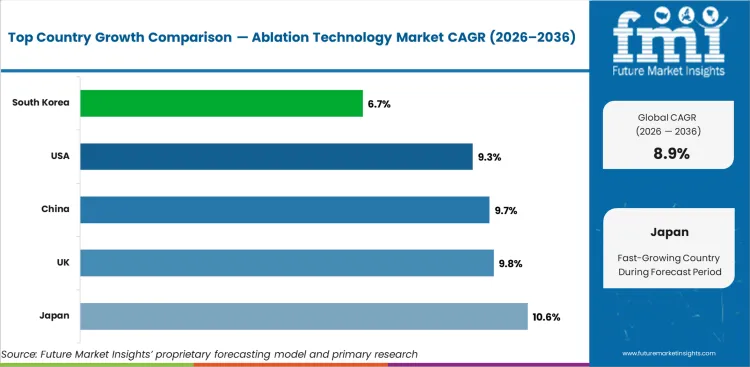
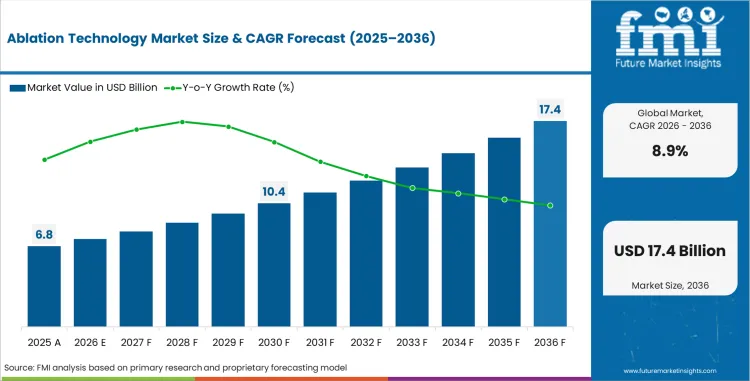
The ablation technology market is projected to grow globally at a CAGR of 8.9% from 2026 to 2036. The analysis spans over 30 countries, with the leading markets detailed below.
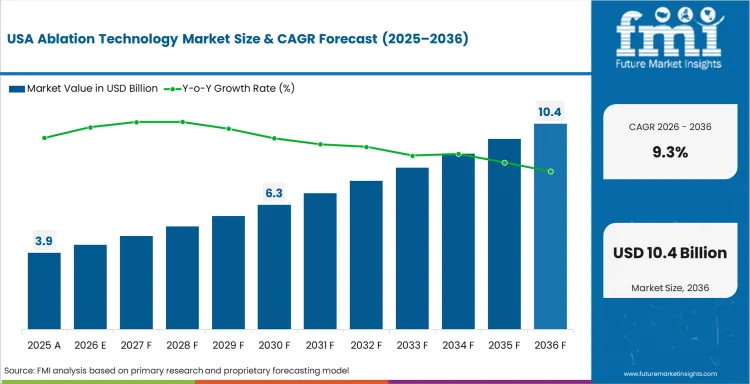
The United States is projected to grow at a CAGR of 9.3% through 2036, shaped by established industrial infrastructure, ongoing capacity investment, and growing procurement from primary application sectors.
The United Kingdom is projected to grow at a CAGR of 9.8% through 2036, shaped by established industrial infrastructure, ongoing capacity investment, and growing procurement from primary application sectors.
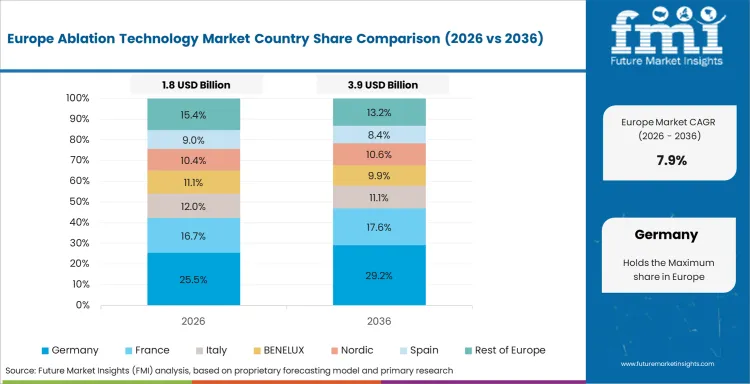
Germany is projected to grow at a CAGR of 5.0% through 2036, shaped by established industrial infrastructure, ongoing capacity investment, and growing procurement from primary application sectors.
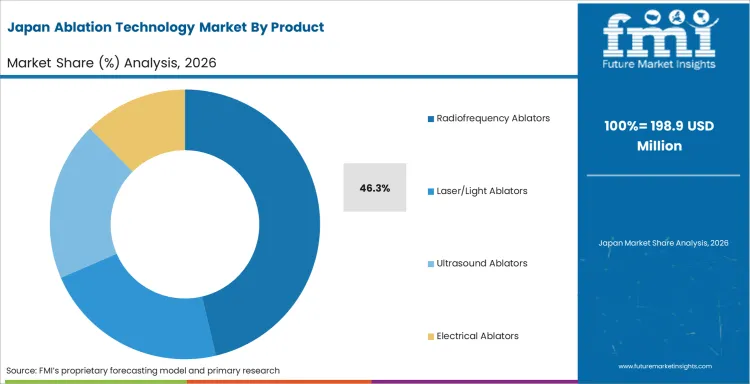
Japan is projected to grow at a CAGR of 10.6% through 2036, shaped by established industrial infrastructure, ongoing capacity investment, and growing procurement from primary application sectors.
China is projected to grow at a CAGR of 9.7% through 2036, shaped by established industrial infrastructure, ongoing capacity investment, and growing procurement from primary application sectors.
India is projected to grow at a CAGR of 5.0% through 2036, shaped by established industrial infrastructure, ongoing capacity investment, and growing procurement from primary application sectors.
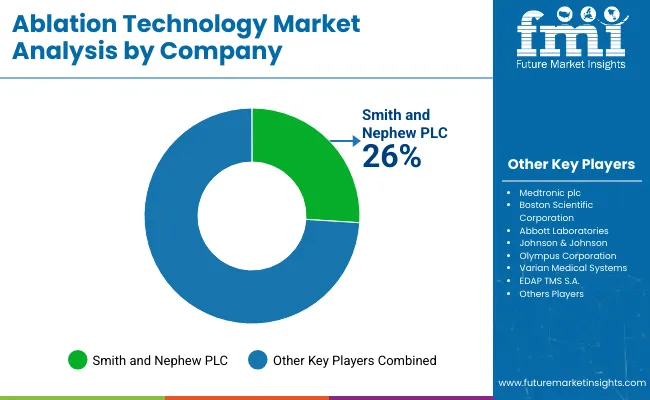
The ablation technology market is defined by competition among established manufacturers and specialty suppliers. Smith and Nephew plc maintains market leadership through comprehensive product portfolios and established distribution infrastructure serving primary application markets.
Medtronic plc competes on product specialization and regional expertise. Boston Scientific Corporation serves application-specific requirements through targeted product development.
Emerging companies and regional manufacturers expand market presence through competitive pricing, localized service, and niche application focus, creating a multi-tier competitive structure.
Key global companies leading the ablation technology market include:
Competitive Benchmarking: Ablation Technology Market
| Company | Product Range | Technical Capability | Market Access | Innovation |
|---|---|---|---|---|
| Smith and Nephew plc | High | Strong | Strong | High |
| Medtronic plc | High | Strong | Strong | High |
| Boston Scientific Corporation | High | Strong | Medium | Medium |
| Abbott Laboratories | Medium | Medium | Medium | Medium |
| Johnson and Johnson | Medium | Medium | Medium | Medium |
| Olympus Corporation | Medium | Medium | Medium | Medium |
Source: Future Market Insights competitive analysis, 2026.
Key Developments in Ablation Technology Market
Major Global Players:
Emerging Players/Startups
| Metric | Value |
|---|---|
| Quantitative Units | USD 7.4 billion to USD 17.4 billion, at a CAGR of 8.9% |
| Market Definition | The ablation technology market encompasses the full spectrum of medical ablation energy platforms including radiofrequency, laser, ultrasound, electrical, cryogenic, microwave, and hydrothermal modalities for cardiovascular, oncological, and interventional applications. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | India, China, USA, UK, Germany, Japan, and 30+ countries |
| Key Companies Profiled | Smith and Nephew, Medtronic, Boston Scientific, Abbott Laboratories, Johnson and Johnson |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down methodology starting with verified transaction data, projecting adoption velocity across segments and regions. |
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary research documentation.
How large is the demand for Ablation Technology in the global market in 2026?
Demand for ablation technology in the global market is estimated to be valued at USD 7.41 billion in 2026, supported by rising procedure volumes across cardiovascular disease treatment, oncology tumour ablation, and pain management, along with expanding minimally invasive therapy adoption in mature and emerging healthcare markets.
What will be the market size of Ablation Technology by 2036?
Market size for ablation technology is projected to reach USD 17.37 billion by 2036, driven by continued shift toward minimally invasive therapies, rising incidence of atrial fibrillation and solid tumours, and ongoing innovation in energy-based ablation modalities.
What is the expected demand growth for Ablation Technology between 2026 and 2036?
The market is expected to grow at a CAGR of 8.9% between 2026 and 2036, reflecting growing global procedure volumes, adoption of advanced energy modalities, and expanded clinical indications across cardiac, oncology, and chronic pain applications.
Which Product is poised to lead global sales by 2026?
Radiofrequency Ablators account for 46.0% in 2026, reflecting their long-established clinical track record, broad applicability across cardiac and tumour ablation procedures, and the wide installed base of RF generator systems across electrophysiology labs and interventional suites.
Which Application holds the largest share of the market?
Cardiovascular Disease Treatment represents 25.0% of the market in 2026, supported by rising atrial fibrillation prevalence, growing cardiac electrophysiology procedure volumes, and expanded clinical adoption of catheter-based ablation as a first-line treatment option.
What is driving demand in Japan?
Japan registers a CAGR of 10.6% through 2036, propelled by an ageing population, high cardiovascular disease burden, well-established reimbursement pathways for ablation procedures, and continued hospital investment in advanced electrophysiology and oncology capabilities.
What does the ablation technology market definition mean in this report?
This report covers ablation technology products used to destroy targeted tissue through thermal, electrical, or chemical energy, including radiofrequency ablators, cryoablation systems, microwave ablators, laser, and ultrasound ablation platforms across cardiac, oncology, gynaecology, and pain management applications.
How does FMI build and validate the Ablation Technology forecast?
Forecasting models apply a hybrid bottom-up methodology with verified data cross-validated against industry statistics, including procedure volume data, hospital capital equipment purchasing records, and device manufacturer shipment disclosures.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.