Antimicrobial Susceptibility Testing Market
Antimicrobial Susceptibility Testing Market Growth - Industry Forecast 2026-2036
Antimicrobial Susceptibility Testing Market Forecast and Outlook 2026 to 2036
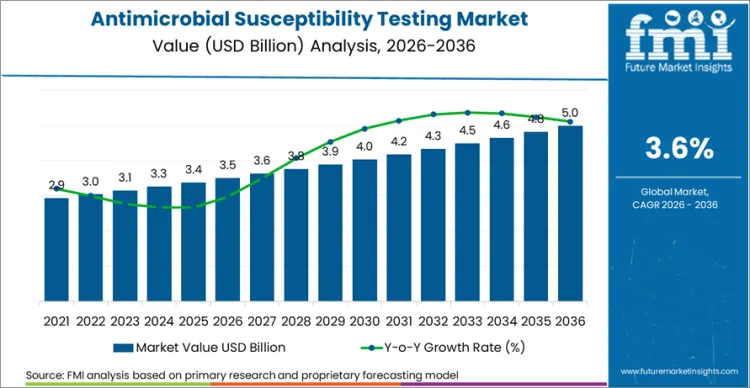
The global antimicrobial susceptibility testing sector is on track to achieve a valuation of USD 5.0 billion by 2036, accelerating from USD 3.5 billion in 2026 at a CAGR of 3.6%. As per Future Market Insights, expansion is structurally underpinned by the World Health Organization's designation of antimicrobial resistance as one of the top 10 global public health threats facing humanity, with the WHO estimating in its 2024 Global Report on Antimicrobial Resistance that AMR directly caused 1.27 million deaths in 2019 and was associated with 4.95 million deaths globally. This mortality burden compels healthcare systems to invest in diagnostic infrastructure that enables targeted antibiotic prescribing. Simultaneously the regulatory landscape is advancing as the FDA formally recognised the CLSI M100 36th edition Performance Standards for Antimicrobial Susceptibility Testing in February 2026, updating the consensus benchmark that clinical laboratories across the United States use to interpret susceptibility results and calibrate testing systems.
Dr. Sophia E. Shanko, Founder and CEO of ShanX Medtech, stated: 'Our vision is to equip every clinician with the ability to act decisively, guided by diagnostic evidence in real-time.' This statement, made in January 2026 following the company's EUR 24 million (USD 28 million) seed funding round and a European Commission contract (HERA), confirms that the next generation of AST innovation is targeting point-of-care deployment with sub-1-hour turnaround. FMI opines that ultra-rapid AST platforms will capture 15 to 20% of the hospital-based AST market by value within five years, driven by antimicrobial stewardship programme mandates and the quantifiable reduction in length of hospital stay associated with same-day targeted prescribing.
The competitive landscape in 2025 and 2026 is defined by FDA clearance activity and automation platform launches. Becton Dickinson received FDA 510(k) clearance for the BD Phoenix M50 system combined with the BDXpert System within the BD Synapsys Informatics suite in April 2025, integrating automated identification and AST with informatics-driven clinical decision support. BioMerieux obtained FDA 510(k) clearance for the VITEK COMPACT PRO system in March 2025, automating microbial identification and AST to improve turnaround times for high-volume clinical laboratories. ShanX Medtech advanced clinical validation of its ultra-rapid 1-hour AST platform through partnerships with Erasmus Medical Centre and AMR GLOBAL, initially targeting urinary tract infections. The FDA recognised the M100 standard for Amikacin injection susceptibility against Enterobacterales and Pseudomonas aeruginosa in October 2025, updating clinical breakpoints. As per FMI, this convergence of automation platform clearances, ultra-rapid technology investment, and regulatory standard updates confirms that the AST market is transitioning from a laboratory efficiency tool into a clinical intervention platform that directly impacts antibiotic prescribing decisions and patient outcomes.
Summary of Antimicrobial Susceptibility Testing Market
What Is Growth Outlook for Antimicrobial Susceptibility Testing Market as per Future Market Insights Projection?
Future Market Insights projects the antimicrobial susceptibility testing industry to expand at a CAGR of 3.6% from 2026 to 2036, increasing from USD 3.5 Billion in 2026 to USD 5.0 Billion by 2036.
FMI Research Approach: FMI proprietary forecasting model based on clinical laboratory test volumes, hospital antimicrobial stewardship programme adoption rates, and AST platform installed base tracking.
How Do FMI Analysts Perceive Antimicrobial Susceptibility Testing Market to Evolve?
FMI analysts perceive the market evolving toward ultra-rapid, point-of-care AST platforms that deliver actionable results within 1 to 7 hours, enabling same-day targeted antibiotic prescribing and replacing the traditional 48 to 72 hour culture-based workflow.
FMI Research Approach: WHO Global Report on Antimicrobial Resistance and FDA CLSI M100 recognition timeline analysis.
Which Country Holds Largest Share in Global Antimicrobial Susceptibility Testing Market?
The United States holds a significant share of the global antimicrobial susceptibility testing market by value which is supported by the FDA's active 510(k) clearance pipeline, the scale of its clinical laboratory infrastructure, and the enforcement of antimicrobial stewardship programme mandates.
FMI Research Approach: FMI country-level revenue modeling by clinical laboratory counts, hospital antibiotic stewardship programme penetration, and FDA 510(k) clearance tracking.
How Large Will Antimicrobial Susceptibility Testing Market Be by 2036?
The global antimicrobial susceptibility testing market is projected to reach USD 5.0 Billion by 2036.
FMI Research Approach: FMI long-term revenue forecast derived from global AMR surveillance investment projections and hospital diagnostic budget allocation trends.
What Is Definition of Antimicrobial Susceptibility Testing Market?
The antimicrobial susceptibility testing market includes automated and manual laboratory systems, reagents, consumables, and software used to determine the susceptibility of bacterial and fungal isolates to antimicrobial agents, enabling targeted antibiotic prescribing in clinical settings.
FMI Research Approach: FMI market taxonomy aligned with CLSI and EUCAST AST methodology classifications.
What Are Globally Unique Trends Shaping Antimicrobial Susceptibility Testing Market?
Globally unique trends include the development of sub-1-hour AST platforms (ShanX Medtech), the FDA's recognition of the CLSI M100 36th edition as the consensus clinical standard, and the European Commission's HERA contracts funding rapid AST technology development for AMR preparedness.
FMI Research Approach: FDA regulatory notices and European Commission HERA contract tracking.
Antimicrobial Susceptibility Testing Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 3.5 Billion |
| Industry Value (2036) | USD 5.0 Billion |
| CAGR (2026 to 2036) | 3.6% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Drivers
Rise in Bacterial Infections
Bacterial infections have been on the rise for a while, and a large population in the world is suffering from some infection or another. People in families with low or moderate incomes are prone to suffering from various diseases. Antimicrobial susceptibility testing is used in hospitals to test out infections such as tuberculosis or those due to hygiene problems, and the results are generally accurate.
Some individuals are given a treatment that may not suit them; therefore, they require a personalized treatment plan. Antimicrobial susceptibility testing helps recommend precision medicines. The government also plays a vital role in increasing research in this field by funding research programs and organizing awareness campaigns. Additionally, the market is getting increasingly competitive.
| Countries | Healthcare Expenditure (% of GDP) |
|---|---|
| United States | 16.80% |
| Germany | 12.50% |
| France | 11.20% |
| Sweden | 11.00% |
| Japan | 10.90% |
| Canada | 10.80% |
| Switzerland | 10.80% |
| United Kingdom | 10.00% |
| Netherlands | 10.00% |
| Austria | 10.00% |
Trends
Innovations in Antimicrobial Susceptibility Testing to Propel Adoption
New strains of bacteria and the mechanism of their resistance are being studied, and this will be possible as improvements keep happening in antimicrobial susceptibility testing methods. Incorporating tailored approaches leads to enhanced precision diagnostics, which will be a major driver behind the market growth.
Additionally, steps are expected to be taken in regions with resource constraints. The areas that are not equipped with the best healthcare facilities will also get access to this testing due to faster and cheaper testing methods.
Advancements in Genomic and Molecular Testing
With growing research in the field of molecular and genomic testing, antimicrobial susceptibility testing methodologies have become more advanced. Moreover, technologies, including next-generation sequencing (NGS), PCR, and CRISPR-based diagnostics, allow for quicker detection of strains, which streamlines healthcare processes.
Additionally, these techniques are growing in popularity in antimicrobial susceptibility testing workflows for the formulation of precision medicines. Moreover, CRISPR-based tools can offer results within minimal time, which considerably decreases the diagnostic timeline.
Challenges
High Initial Investment and Problems with Standardization May Limit Uptake
The laboratories that test for antibiotic susceptibility need a lot of finance due to the requirement for sophisticated testing machines. Thus, it is quite expensive to buy and maintain them. Additionally, the testing time frame might be long because of processing, which might lead to postponement of results, and in certain conditions, the test must be repeated several times for better accuracy. Moreover, upgrades to the processors can also be costly and complicated.
Furthermore, different labs can have different testing protocols, and there is a possibility of errors because of such a lack of standardization. The handling of extensive data can be tedious, too. Additionally, the setup of the labs is quite a challenge in places where the infrastructure and funding are already very poor.
Segment-wise Analysis
By Product
Ongoing Efforts for the Development of Innovative Instruments to Foster Segment Revenue
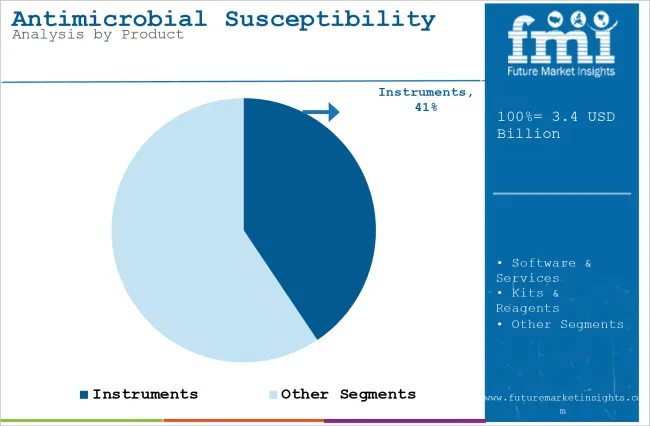
| Attributes | Details |
|---|---|
| Top Product Type | Instruments |
| Market Share in 2025 | 40.6% |
By product, the market is segmented into instruments, software & services, and kits & reagents. The instruments segment is expected to account for nearly 40.6% market share in 2025. New-generation antimicrobial susceptibility testing techniques are progressing toward more automatic and less manual labor instruments.
The instruments ensure that the testing happens at high speed and give the doctors a chance to monitor the testing process in real-time. These instruments are vital in big hospitals and labs, where there is a good investment for them. Moreover, there is an ongoing effort to develop more powerful devices that have more advanced features. The instruments would see constant development to gain higher accuracy.
By Application
Surging Sepsis Occurrence to Drive Uptake
By application, the market is segmented into sepsis, respiratory infections, gastrointestinal infections, meningitis & encephalitis, endocarditis, and others. The testing method is being widely used for the treatment of sepsis owing to its accuracy in identifying pathogens. Moreover, the higher prevalence of the disease in developed and developing regions globally is contributing to segment expansion.
By End User
Rising Hospital Admissions to Bolster Demand
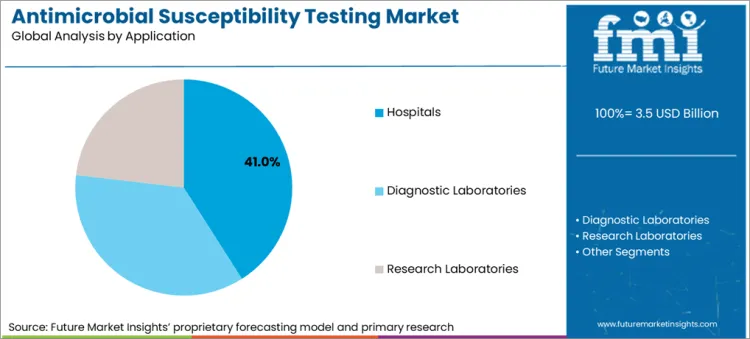
| Attributes | Details |
|---|---|
| Top End User Type | Hospitals |
| Market Share in 2025 | 41% |
By end user, the antimicrobial susceptibility testing market is segmented into diagnostic laboratories, hospitals, and research laboratories. The hospitals segment is expected to account for nearly 41% of the market share in 2025, driven by an increasing number of hospitalizations. Additionally, diagnostic tests are needed by many of the patients, mainly those who suffer from bacterial infections. Sometimes, when infections are severe, the patients also need to be admitted to the intensive care units (ICUs).
In such settings, the patients might need to be tested several times at different times of the day to check if the infection has become less severe. Owing to growing hospital-acquired infections, these settings also have facilities to conduct antibiotic sustainability testing. Even governments keep antimicrobial susceptibility testing as a priority and fund these projects regularly. Due to this, the hospitals contribute to a significant share of revenue in the market.
Regional Analysis
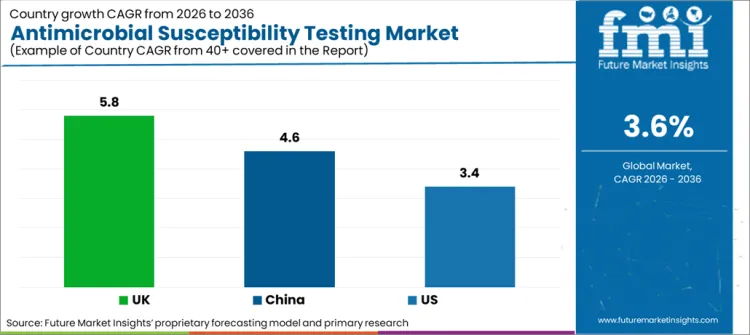
| Country/Region | CAGR (2025 to 2035) |
|---|---|
| USA | 3.4% |
| UK | 5.80% |
| China | 4.60% |
North America Antimicrobial Susceptibility Testing Industry Analysis USA
The antimicrobial susceptibility testing market in the USA is expected to grow at a high growth rate and its CAGR is expected to be 3.4% during the forecast period 2025 to 2035. Antibiotic resistance cases can be seen to be increasing in the country, due to which there is a constant need to develop new treatment strategies to deal with infections.
Additionally, healthcare facilities are very well developed in the USA, and a large amount of money is invested in developing good technologies for diagnosis. Moreover, the government ensures that the research programs are well-funded.
Europe Antimicrobial Susceptibility Testing Industry Analysis
The UK is expected to grow at a CAGR of 5.8% during the forecast period. It can be seen that Europe is well developed in terms of academic and research institutions, and there is a strong motivation for research, as the government funds the research programs pretty well. Hospital-acquired infections are a major issue, and to ensure proper treatment, infections within the hospital need to be tackled. In this regard, constant efforts are being put into reducing them with the help of antimicrobial susceptibility testing.
Asia Pacific Antimicrobial Susceptibility Testing Industry Outlook
China is poised to grow at a CAGR of 4.6% during the forecast period, which is the highest among all the Asia Pacific countries under study (the countries from the Asia Pacific include China, India, Japan, Australia, and South Korea). This CAGR is pretty high, given the size and population of the country. China is one of the countries where antibiotic consumption is the highest. Due to this, it is normal to develop resistance to antibiotics.
To ensure that the entire population is healthy, high investments are being made in developing the healthcare infrastructure. China is also experiencing rapid urbanization because of its large population. Due to the integration of artificial intelligence, test results can be analyzed and produced faster, with some parts of the process being automated.
Competitive Landscape
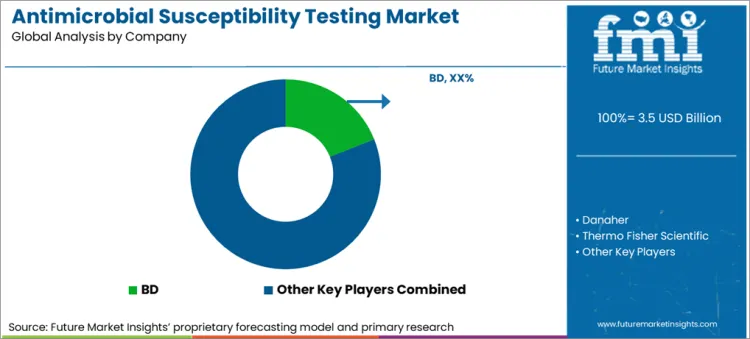
The focus of key market players is on making the testing procedures smoother and more efficient. The profits depend upon the instruments and products associated with testing used in hospitals and laboratories. Because of that, manufacturers supply the best products for testing. Moreover, the antimicrobial susceptibility testing market is expected to expand along with the healthcare sector.
| Key Companies | Area of Focus |
|---|---|
| iFAST Diagnostics | Providing rapid antimicrobial susceptibility testing solutions in under three hours. They do this by analyzing bacteria on a microchip. This technology aims to enhance patient treatment and fight antibiotic resistance. |
| Selux Diagnostics | Started a rapid antimicrobial susceptibility testing platform named Next Generation Phenotyping System. This testing platform provides results that are faster than the current testing procedures. |
| ClinLab International | With the help of digital culture technology, ClinLab delivers the testing results in under four hours, focusing on improving the test accuracy and speed |
Apart from the above-mentioned companies, there are several medical startups that are working tirelessly to provide tools for diagnosis and testing. The focus of these startups is to provide solutions to improve the speed and accuracy of testing. However, more research needs to be conducted as antibiotic resistance is a major problem in the medical sciences.
Key Market Players
- BD
- Danaher
- Thermo Fisher Scientific
- Bio-Rad
- Creative Diagnostics
- Accelerate Diagnostics
- Alifax
- Roche Diagnostics Limited
- Merck Group
- Synbiosis
Market Definition
The antimicrobial susceptibility testing market represents revenue generated from the manufacture and sale of automated systems, manual testing kits, reagents, consumables, and informatics software used to determine the susceptibility of microbial isolates to antimicrobial agents. The market measures the value of AST instruments, reagent consumables, software subscriptions, and service contracts sold to clinical microbiology laboratories, hospital pharmacies, and reference laboratories.
Inclusions cover automated AST platforms (BD Phoenix, bioMerieux VITEK, Q-linea ASTar), manual disk diffusion and broth microdilution methods, rapid and ultra-rapid AST systems, AST-specific reagent panels, CLSI and EUCAST calibrated consumables, and laboratory informatics systems that integrate AST results with antimicrobial stewardship decision support.
Exclusions include standalone microbiology culture media without AST functionality, molecular-only resistance gene detection assays (PCR-based) without phenotypic susceptibility confirmation, environmental AMR surveillance testing not conducted in clinical settings, and veterinary AST for animal health applications.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 3.5 Billion |
| Product Type | Automated AST Platforms, Manual Disk Diffusion, Broth Microdilution, Rapid/Ultra-Rapid AST Systems, Reagent Panels, Informatics Software |
| Testing Method | Phenotypic (MIC Determination, Disk Diffusion), Genotypic (Resistance Gene Panels), Combination |
| End User | Hospital Clinical Labs, Reference Laboratories, Public Health Labs, Antimicrobial Stewardship Programmes |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Countries Covered | USA, Germany, Netherlands, UK, France, Japan, China, India, and 40+ countries |
| Key Companies Profiled | Becton Dickinson, bioMerieux, Q-linea, ShanX Medtech, Bruker (Hain Lifescience), Thermo Fisher Scientific |
Recent Developments
- In January 2026, ShanX Medtech secured EUR 24 million (USD 28 million) in seed funding and a European Commission HERA contract to accelerate its ultra-rapid 1-hour AST platform, initially targeting urinary tract infections through clinical validation with Erasmus Medical Centre and AMR GLOBAL.
- In March 2025, bioMerieux obtained FDA 510(k) clearance for the VITEK COMPACT PRO system, automating microbial identification and AST to improve turnaround times for high-volume clinical laboratories.
- In February 2026, the FDA formally recognised the CLSI M100 36th edition Performance Standards, updating the consensus benchmark for clinical laboratory susceptibility interpretation across the United States.
Market Segmentation
Globally, the antimicrobial susceptibility testing market is divided into three main segments: product, application, and end user.
By Product:
By product, the market is segmented into instruments, software & services, and kits & reagents.
By Application:
By application, the market is segmented into sepsis, respiratory infections, gastrointestinal infections, meningitis & encephalitis, endocarditis, and others.
By End User:
By end user, the market is segmented into diagnostic laboratories, hospitals, and research laboratories.
Bibliography
- World Health Organization. (2024). Global Report on Antimicrobial Resistance: Surveillance and Epidemiological Data. WHO.
- Clinical and Laboratory Standards Institute. (2026, January). M100: Performance Standards for Antimicrobial Susceptibility Testing, 36th Edition. CLSI.
- United States Food and Drug Administration. (2026, February). Notices and updates: Recognition of CLSI M100 36th edition. FDA.gov.
- ShanX Medtech. (2026, January). ShanX Medtech secures EUR 24 million to transform 1-hour antimicrobial susceptibility testing. Innovation News Network.
Frequently Asked Questions
What is the current global market size for Antimicrobial Susceptibility Testing?
The global market is valued at USD 3.5 Billion in 2026, driven by WHO-designated AMR urgency and the FDA's active 510(k) clearance pipeline for automated and rapid AST platforms.
What is the projected Compound Annual Growth Rate (CAGR) for the market over the next 10 years?
The market is projected to grow at a CAGR of 3.6% from 2026 to 2036.
Which regions are experiencing the fastest expansion?
North America leads by value driven by FDA clearance activity and antimicrobial stewardship mandates, while Europe is the fastest-growing region for ultra-rapid AST technology adoption supported by European Commission HERA funding.
What are the primary market drivers?
WHO-designated AMR mortality burden (1.27 million direct deaths), FDA and CLSI regulatory standard updates, and European Commission HERA investment in rapid diagnostic preparedness are the primary growth catalysts.
Who are the leading suppliers in the industry?
Becton Dickinson, bioMerieux, Q-linea, and ShanX Medtech are key players, differentiating through automation scale, FDA clearance breadth, and ultra-rapid turnaround time capabilities.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2026 to 2036
- Instruments
- Software & Services
- Kits & Reagents
- Instruments
- Y to o to Y Growth Trend Analysis By Product , 2021 to 2025
- Absolute $ Opportunity Analysis By Product , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Hospitals
- Diagnostic Laboratories
- Research Laboratories
- Hospitals
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Application
- Competition Analysis
- Competition Deep Dive
- BD
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Danaher
- Thermo Fisher Scientific
- Bio-Rad
- Creative Diagnostics
- Accelerate Diagnostics
- BD
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Product
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Product
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Product
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

