The enzyme replacement therapy market is projected to reach USD 12.2 billion in 2026, supported by stronger genetic testing capabilities and wider newborn screening coverage across developed healthcare systems. FMI estimates the market will expand at a 6.7% CAGR from 2026 to 2036, taking total revenue to USD 23.1 billion by 2036. Rising diagnosis rates for lysosomal storage disorders and improved identification of eligible patients remain the primary factors underpinning market growth.
Biopharmaceutical companies are investing in next generation ERTs designed to improve tissue uptake and reduce immunogenic responses, which can strengthen treatment durability and patient adherence. Market momentum is also supported by a shift toward more individualized care, with specialist centers adopting dosing strategies guided by clinical response and pharmacokinetic monitoring. This is increasing demand for integrated patient support services, infusion management, and long-term monitoring tools that improve therapy continuity.
Early intervention is becoming more central to market expansion as pediatric protocols gain emphasis and healthcare providers aim to initiate treatment earlier in the disease course. Clinical practice is also evolving toward combination approaches, including adjunct therapies that enhance enzyme activity or reduce substrate accumulation, which can broaden addressable use across selected indications. Interest in integrating emerging modalities, such as gene therapy, is shaping competitive positioning and pipeline development, although ERT remains a core standard of care in multiple rare disease categories.
Rare disease advocacy and orphan drug development continue to sustain investment and innovation. Regulatory pathways that accelerate review and approval are supporting faster commercialization of novel therapies. Emerging markets offer additional upside as healthcare infrastructure improves and awareness of inherited metabolic disorders expands among clinicians and diagnostic networks.
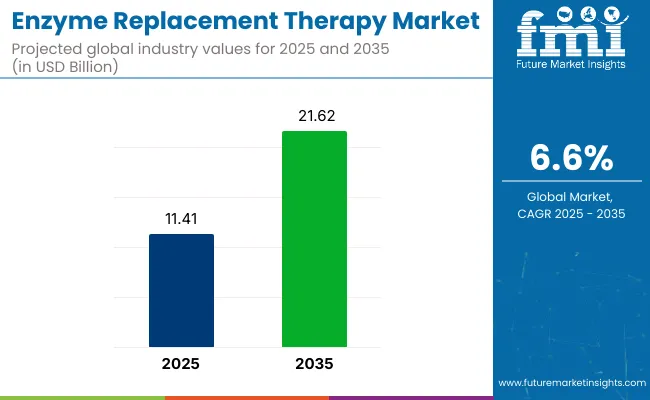
| Metric | Value |
|---|---|
| Industry Size (2026) | USD 12.2 Billion |
| Industry Value (2036) | USD 23.1 Billion |
| CAGR (2026-2036) | 6.7% |
Source: Future Market Insights analysis, supported by a proprietary forecasting model and primary research
Precision medicine adoption in rare diseases is rising as treatment centers shift toward personalized approaches that better match therapy to individual patient biology. Progress in pharmacogenomics testing is helping clinicians assess enzyme activity and genetic profiles more accurately, which supports dosing decisions and improves confidence in treatment planning for complex lysosomal disorders. At the same time, stronger biomarker discovery is expanding how providers monitor disease progression and therapy response across different patient groups, enabling more consistent follow-up and earlier adjustments when outcomes vary.
Awareness and training initiatives are also influencing adoption. Education around genetic counseling is strengthening the role of specialist clinics, improving diagnosis pathways and guiding families through treatment choices and long-term care planning. Pharmaceutical companies are responding by developing companion diagnostics that support patient selection, therapy monitoring, and outcome tracking alongside enzyme therapies.
Another important shift is the wider use of patient-reported outcomes in rare disease management. These measures add visibility into daily functioning and symptom burden, complementing laboratory endpoints and helping clinicians evaluate real-world impact. Together, these drivers are making precision medicine a more routine part of rare disease care without weakening clinical evidence requirements.
Global enzyme replacement therapy landscape demonstrates sophisticated segmentation addressing specialized requirements of the rare disease treatment ecosystem, categorizing the sector by therapeutic condition, administration route, distribution channel, and patient demographics. Detailed classification enables stakeholders to target specific disease populations, such as increasing demand for intrathecal delivery systems in neurologic conditions or growing adoption of home infusion services in stable patients. Through analyzing these segments, manufacturers can optimize therapeutic development to serve distinct requirements of pediatric specialists versus adult metabolic disease centers. Segmentation reveals progression from single-enzyme treatments toward multi-modal therapeutic approaches. As the industry matures, precise segmentation becomes essential for understanding regulatory pathways and reimbursement criteria, ensuring product development aligns with evolving rare disease treatment standards.
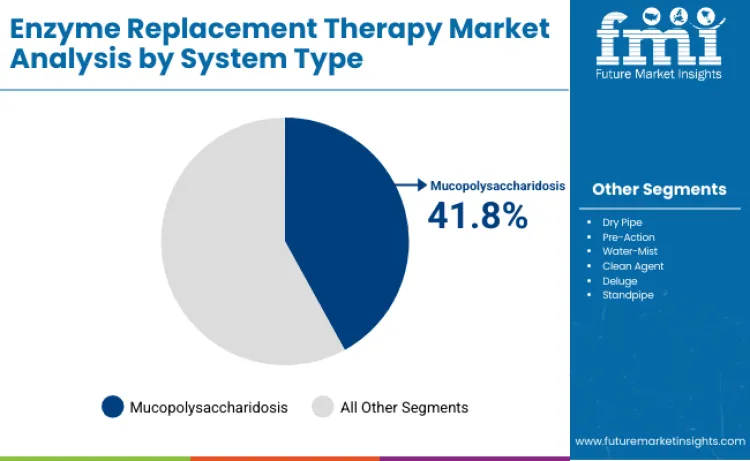
Mucopolysaccharidosis enzyme therapies secure 41.8% of therapeutic condition market share in 2026, driven by multiple distinct enzyme deficiencies requiring targeted replacement strategies across diverse MPS subtypes. Market position benefits from established treatment protocols for MPS I, MPS II, MPS IVA, and MPS VI conditions with proven clinical efficacy data. Segment advantages include comprehensive disease management approaches addressing skeletal, cardiac, and respiratory manifestations through specific enzyme formulations. Research concentrated on central nervous system penetration enhancement renews interest among neurologic MPS variants. Critical role in pediatric rare disease treatment strengthens market position, as these therapies provide life-extending benefits for children with progressive multi-system disorders. Growing emphasis on newborn screening programs creates sustained demand for early intervention enzyme replacement protocols.
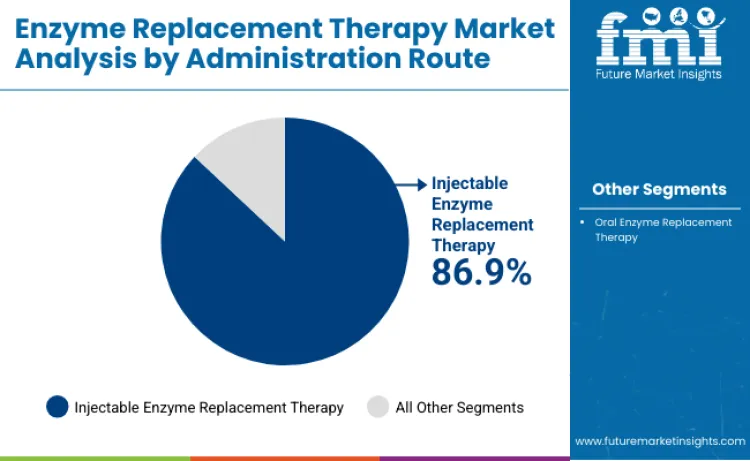
Injectable enzyme replacement therapies maintain 86.9% share of administration route segment in 2026, reflecting proven bioavailability advantages and established clinical infrastructure for intravenous infusion protocols. Administration dominance connects to superior enzyme stability profiles and predictable pharmacokinetic characteristics achieved through parenteral delivery systems. Healthcare providers depend on specialized infusion centers to monitor patient responses and manage potential hypersensitivity reactions during treatment administration. These formulations remain fundamental for achieving therapeutic enzyme concentrations in target tissues and organs. Continuous improvement in infusion pump technologies ensures injectable therapies designed for extended dosing intervals maintain competitive patient convenience factors. Strong regulatory approval pathways require extensive safety documentation, supporting consistent market access for established injectable enzyme products.
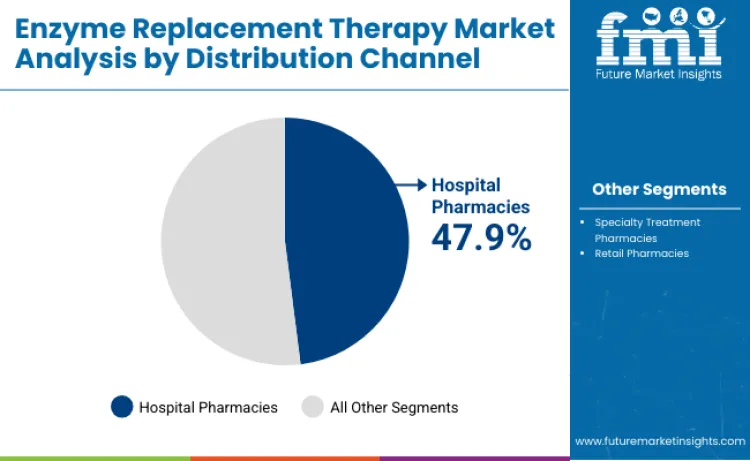
Hospital pharmacy distribution captures 47.9% of distribution channel market share in 2026, attributed to complex handling requirements and specialized storage conditions necessary for enzyme replacement therapies. Channel dominance stems from comprehensive patient monitoring capabilities and immediate access to emergency medical interventions during infusion procedures. Healthcare systems prioritize these distribution points for insurance authorization processing and prior approval coordination with specialty care teams. Application in high-acuity patient populations drives steady volume growth among tertiary care medical centers. Compatibility with clinical research protocols allows for seamless integration of investigational enzyme therapies into established treatment programs. Distributors focusing on cold-chain logistics and regulatory compliance for rare disease medications achieve substantial market penetration among academic medical centers.
Gene therapy is emerging as a major disruptor to enzyme replacement therapy by offering disease-modifying potential that targets the root genetic defect instead of long-term symptom control. In the near term, the market is likely to evolve toward hybrid care models, where patients may receive ERT as a bridge before or after gene therapy, particularly during immune management phases or when durable expression is still being established. Viral vector platforms also expand the addressable opportunity for tissue-specific enzyme expression, which can complement or reduce dependence on infusion-based regimens. As more genetic medicine programs and clinical trials expand, treatment centers with trial access may influence adoption patterns and accelerate demand for integrated care pathways. Market differentiation will increasingly depend on patient stratification tools that help identify who should transition to gene therapy, who should remain on ERT, and when switching is clinically appropriate.
Greater regulatory harmonization across regions can shorten approval timelines by enabling shared reliance on clinical evidence and coordinated review practices. Fast-track and breakthrough-type pathways are expected to remain important for novel ERTs in high unmet need indications, improving speed to market. At the same time, stronger post-market safety obligations will expand real-world datasets, which can support label expansion and broader pediatric use. Orphan drug policies continue to shape competitive dynamics through exclusivity incentives that sustain investment in ultra-rare conditions. Pediatric development requirements will also drive demand for age-appropriate formulations and dosing validation, expanding product lifecycle opportunities.
Reimbursement is increasingly being shaped by value-based frameworks that assess long-term outcomes, not just short-term clinical response. Real-world evidence is becoming a key market tool to demonstrate that early ERT can reduce irreversible organ damage and lower downstream healthcare costs. Payers are also building rare disease management programs that combine therapy access with patient support, adherence services, and care coordination. Health technology assessment decisions in many markets will continue to rely on comparative effectiveness and quality-of-life measures, influencing pricing and coverage. Biosimilar development may introduce pricing pressure over time, potentially expanding access while intensifying competition in established ERT indications.
Global enzyme replacement therapy marketplace exhibits diverse healthcare delivery systems influenced by rare disease policies, reimbursement frameworks, and specialized care infrastructure across different healthcare economies. Developed markets emphasize comprehensive genetic testing and early intervention strategies, while emerging healthcare systems focus on basic diagnostic capacity building and treatment access expansion. East Asia emerges as a rapid growth region due to advancing healthcare infrastructure and increasing awareness of inherited metabolic disorders. North American markets concentrate on innovative therapeutic development supporting precision medicine and combination treatment approaches. Government initiatives in developing countries promoting rare disease recognition ensure expanding enzyme therapy demand across multiple therapeutic areas.
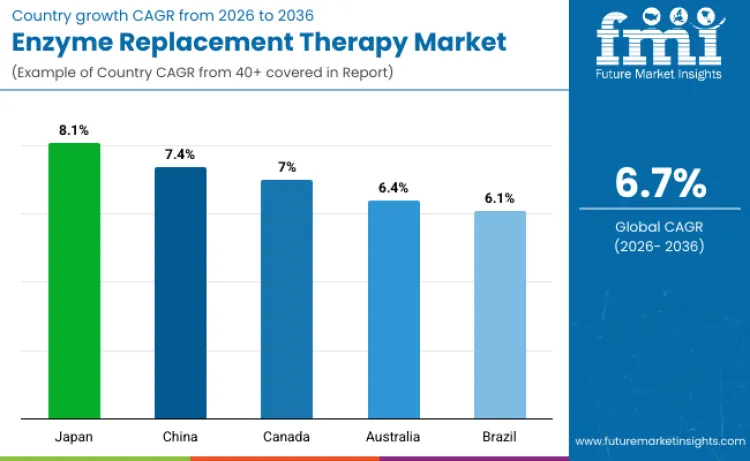
| Country | CAGR (2026 to 2036) |
|---|---|
| Japan | 8.1% |
| China | 7.4% |
| Canada | 7.0% |
| Australia | 6.4% |
| Brazil | 6.1% |
Source: Future Market Insights’ proprietary forecasting model and primary research
Enzyme replacement therapy utilization in Japan projects growth at 8.1% CAGR through 2036. Market expansion results from comprehensive aging population demographics creating increased prevalence of late-onset lysosomal storage disorders and metabolic enzyme deficiencies. Growth benefits from national health insurance coverage for rare disease treatments and established specialized medical centers providing multidisciplinary enzyme therapy management. Rising healthcare investment in precision medicine drives demand for genetic testing and early enzyme deficiency diagnosis among pediatric and adult populations. Presence of advanced biotechnology research institutions and clinical trial infrastructure creates substantial capacity for novel enzyme therapy development supporting scalable market growth. Focus on pharmaceutical innovation and regulatory excellence continues supporting Japan's position as a leading market for advanced enzyme replacement technologies.
Requirements for specialized enzyme therapies in China advance at 7.4% CAGR during the forecast period. Healthcare system modernization emphasizing rare disease recognition and treatment accessibility generates increasing demand for enzyme replacement protocols across urban medical centers. Growing investment in genetic testing capabilities and newborn screening programs strengthens early diagnosis rates for treatable enzyme deficiency conditions. Government policies supporting orphan drug importation and domestic biotechnology development accelerate access to international enzyme therapy standards. Strategic partnerships between Chinese healthcare providers and multinational pharmaceutical companies enable technology transfer programs, reinforcing China's enzyme replacement therapy market expansion through improved clinical expertise.
Enzyme replacement therapy sales in Canada expect growth at 7.0% CAGR. Market advancement benefits from universal healthcare coverage including rare disease treatment provisions and specialized enzyme therapy reimbursement programs. Development supports established genetic medicine infrastructure and comprehensive metabolic disease centers providing coordinated care for complex enzyme deficiencies. Advanced clinical research capabilities enable participation in international enzyme therapy trials and early access programs for investigational treatments. Technology leadership in personalized medicine and pharmacogenomic testing creates unique treatment optimization opportunities requiring customized enzyme therapy approaches. Emphasis on patient-centered care and health technology assessment continues establishing Canada as a premium market for evidence-based enzyme replacement innovations.
Enzyme replacement therapy market in Australia anticipates expansion at 6.4% CAGR. Growth attributes to comprehensive rare disease registry systems and coordinated national approaches to enzyme deficiency diagnosis and treatment management. Healthcare infrastructure development including specialized pediatric metabolic centers creates substantial treatment capacity for complex enzyme replacement protocols. Increasing collaboration between Australian medical researchers and international biotechnology companies ensures access to cutting-edge enzyme therapies through clinical trial participation and compassionate use programs. Economic evaluation frameworks supporting cost-effective rare disease treatments enhance long-term market access for innovative enzyme replacement technologies. Robust regulatory oversight ensuring therapeutic quality and safety standards enhances healthcare provider confidence in emerging enzyme therapy options.
Revenue from enzyme replacement therapies in Brazil projects growth at 6.1% CAGR. Market expansion connects to increasing healthcare investment in rare disease diagnosis and treatment infrastructure across major metropolitan medical centers. Growth stems from growing awareness among healthcare providers regarding treatable lysosomal storage disorders and available enzyme therapy options. Moderate adoption of genetic testing and metabolic screening programs increases early diagnosis rates for enzyme deficiency conditions requiring immediate therapeutic intervention. Presence of regional pharmaceutical manufacturing capabilities ensures consistent enzyme therapy supply chains and potential cost reduction through local production partnerships. Emphasis on healthcare accessibility and patient advocacy continues positioning Brazil as an emerging market for comprehensive enzyme replacement therapy programs.
Competitive positioning in the enzyme replacement therapy market is increasingly influenced by the strength of patient services that sit alongside clinical performance. Companies are differentiating by offering end to end support that helps patients start and stay on therapy, given the complexity of long-term infusion schedules and monitoring requirements. Major players such as Sanofi Genzyme and Takeda Pharmaceuticals are investing in disease management programs, access assistance, and patient navigation models that improve adherence and continuity of care. A clear market trend is the buildout of integrated care ecosystems that link therapy supply with genetic counseling, coordination with specialist centers, and real-world outcomes tracking to demonstrate value over time. Partnerships with rare disease advocacy groups and patient registries are also expanding, improving diagnosis awareness, supporting earlier referrals, and strengthening evidence generation.
Innovation remains a central competitive lever. Manufacturers are advancing enzyme engineering to improve stability and activity, while exploring delivery approaches that enhance tissue uptake and reduce immunogenic risk. Competitive intensity is further shaped by portfolio expansion activity, as rare disease focused biotech firms acquire complementary assets and large pharmaceutical companies pursue targeted deals to broaden orphan drug coverage. Sanofi Genzyme continues to hold a leading position, supported by a broad set of approved enzyme therapies and established global distribution capabilities across multiple lysosomal storage disorder indications.
| Items | Values |
|---|---|
| Quantitative Units | USD Billion |
| Therapeutic Condition Segments | Mucopolysaccharidosis; Gaucher Disease; Fabry Disease; Pompe Disease; Lysosomal Acid Lipase Deficiency; Others |
| Administration Route Categories | Injectable Enzyme Replacement Therapy; Oral Enzyme Replacement Therapy |
| Distribution Channel Categories | Hospital Pharmacies; Specialty Treatment Pharmacies; Retail Pharmacies |
| Patient Age Categories | Pediatric; Adult; Geriatric |
| Regions Covered | North America, Europe, East Asia, South Asia, Latin America, Middle East & Africa |
| Key Countries | Japan, China, Canada, Australia, Brazil |
| Key Companies Profiled | Sanofi Genzyme; Takeda Pharmaceuticals; BioMarin Pharmaceutical Inc.; Amicus Therapeutics; Alexion Pharmaceuticals; Ultragenyx Pharmaceutical Inc.; Sarepta Therapeutics; Pfizer Inc.; Novartis AG; CSL Behring |
| Additional Attributes | Dollar sales measured for enzyme replacement therapies used in lysosomal storage disorder treatment, specified by therapeutic condition (mucopolysaccharidosis, Gaucher, Fabry, Pompe diseases), administration route (injectable, oral), distribution channel (hospital, specialty, retail pharmacies), and regulatory compliance with evolving orphan drug standards and rare disease treatment protocols. |
How big is the enzyme replacement therapy market in 2026?
The enzyme replacement therapy industry is valued at USD 12.2 billion in 2026.
What will be the size of the enzyme replacement therapy market in 2036?
The enzyme replacement therapy market is projected to reach USD 23.1 billion by 2036.
How much will the enzyme replacement therapy market grow between 2026 and 2036?
The market is expected to expand at a CAGR of 6.7% during the 2026 to 2036 forecast period.
Which therapeutic condition segment leads the enzyme replacement therapy market?
Mucopolysaccharidosis treatments lead the therapeutic condition category, accounting for 41.8% share in 2026.
Which administration route segment holds the dominant share in the enzyme replacement therapy market?
Injectable formulations dominate the administration route landscape with an 86.9% share in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.