Gaucher and Pompe Diseases Enzyme Replacement Therapy (ERT) Market
Gaucher and Pompe Diseases Enzyme Replacement Therapy (ERT) Market - Growth & Forecast 2025 to 2035
Gaucher and Pompe Diseases Enzyme Replacement Therapy (ERT) Market Outlook 2025 to 2035
Market growth of Gaucher and Pompe disease enzyme replacement therapy (ERT) will be substantial from 2025 to 2035 as healthcare organizations focus on both heightened disease awareness and biotechnology innovations and treatment accessibility for rare conditions.
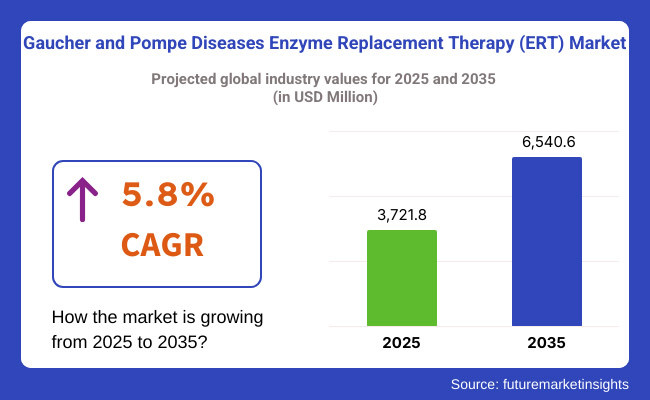
Enzyme replacement therapy, which enhances acute care and general lifestyle, is the primary treatment for Gaucher and Pompe diseases. It is analysed that the market valuation is USD 3,721.8 million in 2025, and will reach USD 6,540.6 million by 2035 with a compound annual growth rate (CAGR) of 5.8% between the given periods of forecast.
Market expansion results from increasing lysosomal storage disorder cases combined with advanced research funding and regulatory support for orphan drug development. Pharmaceutical firms concentrate on sharpening ERT performance measures while working to shorten infusion procedures and establish new disease management solutions for better therapy results.
The market expansion is supported by improved government-backed rare disease management infrastructure and enhanced diagnostic testing capabilities, which lead to faster diagnosis and treatment availability.
Several barriers stand in the way of market expansion, including treatment expenses that are expensive, payment limitations, and restricted availability of patients. Industry stakeholders work with healthcare providers, together with patient advocacy groups, alongside policymakers to enhance accessibility and affordability across the market.
Demand is due to the growing need for hospitals, specialty clinics, and research institutions for the Gaucher and Pompe diseases, the market treatment type, and the end-user application segment. Recombinant vaccine enzymes and next-generation biologics. For systemic processing disorders, such as Gaucher disease and Pompe disease, recombinant enzyme therapies like imiglucerase for the former and alglucosidase alfa for the latter are still the primary treatment options and remain the most commonly used agents because of their well-established efficacy and safety profiles.
New generation biologics, such as more advanced enzyme formulations, as well as the gene therapies currently in the pipeline, are built to yield more durable treatment and be more convenient for patients.
Hospitals and specialty clinics are major contributors to the demand among end users as they provide dedicated infusion therapy services and focused disease management. Research institutes have a critical role in driving ERT innovations and facilitating clinical trials to optimise treatment approaches.
With the landscape for rare diseases ever-changing, pharmaceutical companies have sought to create innovative delivery methods- intrathecal and oral ERT options to increase compliance and subsequently enhance patient outcomes.
Regional Market Trends
North America
The North American gastrointestinal and pulmonary diseases market stands strong on the back of high healthcare expenditures, advanced biotech research, and favourable regulatory policies. This is supported by transplant activities, high clinical trials, rapid drug approvals, and robust reimbursement framework in the USA and Canada. Furthermore, growing partnership initiatives between biopharmaceutical companies and academic organizations foster innovation in enzyme replacement therapy.
Europe
Financial support from government-funded rare disease initiatives, a well-established healthcare structure, and numerous patient advocacy programs act as part of the Europe Gaucher and Pompe diseases ERT market drivers.
Germany, France, and the UK drive market growth, showing high rates of orphan drug adoption and advanced treatment centres. The European Medicines Agency (EMA) is in favour of the development and approval of novel ERTs to better access patients and incentivise investment in next-generation enzyme therapies.
Asia-Pacific
The highest growth rate is seen in the Asia-Pacific region, with the steady rise in health care investments, a growing number of patients being aware of the disease, and more and more advanced diagnostic technologies.
In Asia-Pacific countries, including China, India, Japan, and South Korea, ERT demand is driven by increased access to healthcare as well as the governments' encouragement of the treatment of rare diseases. Despite affordability challenges, collaborations between international pharma companies and local care providers expand treatment reach and cost-effectiveness, facilitating the long-term growth of the market.
Challenges and Opportunities
Challenge
High treatment costs and limited accessibility
Low global accessibility and high treatment cost across the globe are some challenges in Gaucher and Pompe diseases enzyme replacement therapy (ERT) market. ERT is still the mainstay of treatment of these rare lysosomal storage diseases; however, its cost is prohibitive for patients and healthcare systems.
The problem is compounded by a lack of insurance coverage and government reimbursement programs in many areas that further limit access to lifesaving treatments. Further, the need for frequent intravenous infusions adds additional peril to healthcare infrastructure, especially in low- and middle-income nations wherein specialized medical facilities are limited.
Opportunity
Advancements in gene therapy and alternative treatment approaches
An array of possible gene therapy solutions and new formulations of drugs that have the potential to improve treatment efficacy and availability in the Gaucher and Pompe disease ERT landscape. Next-generation enzyme replacement formulations, including longer-acting enzyme replacement therapies (ERTs) and fusion proteins, may improve patient compliance and quality of life by reducing the frequency of infusions.
Gene therapy studies are also investigating whether a single lifetime therapy can avert the need for lifelong infusions of enzyme replacement. Market expansion is propelled by surges in government and private sector funding for rare disease treatments and accelerated orders for orphan drug regulatory approvals.
Shifts in the Gaucher and pompe diseases market from 2020 to 2024 and future trends 2025 to 2035
From 2020 to 2024, currently available enzyme replacement therapies are expected to become more widely available to patients due to expanded access programs from both government agencies and pharmaceutical companies alike.
Major approaches for enzyme engineering included improved infusion efficiency, decreased immunogenic response, and better enzyme stability. Yet high costs of treatment and a lack of global access to certain drugs continued to hamper the leadership of ERT in developing healthcare markets.
The market will shift, between 2025 and 2035, towards gene therapy, oral therapies, and the use of novel enzymes with greater bioavailability and sustained therapeutic benefit. Big Pharma will work more closely with biotech companies to target drugs more effectively and improve enzyme uptake with fewer side effects.
Moreover, as public authorities began to adopt orphan drug policies before 2010, they improved the surrounding environment for the faster entry of orphan drugs and stimulated ERT innovation and accessibility on a global scale.
Market Shifts: A Comparative Analysis 2020 to 2024 vs. 2025 to 2035
| Market Shift | 2020 to 2024 |
|---|---|
| Regulatory Landscape | Orphan drug approvals accelerated, but high costs limited widespread availability. |
| Technological Advancements | Improvements in enzyme stability and reduced infusion reactions. |
| Sustainability Trends | Efforts to improve affordability through patient assistance programs. |
| Treatment Accessibility | High costs and limited reimbursement programs restricted access. |
| Industry Adoption | ERT remained the standard care for Gaucher and Pompe diseases. |
| Consumer Preferences | Patients sought less invasive and more convenient treatment options. |
| Market Growth Drivers | Rising awareness of rare diseases and advances in biologic manufacturing. |
| Market Shift | 2025 to 2035 |
|---|---|
| Regulatory Landscape | Increased regulatory support for gene therapy and alternative formulations. |
| Technological Advancements | Development of gene therapy, oral ERT options, and targeted enzyme delivery. |
| Sustainability Trends | Expansion of government-funded rare disease treatment initiatives. |
| Treatment Accessibility | Global expansion of treatment centres and insurance coverage improvements. |
| Industry Adoption | Shift toward gene therapy and advanced biologic drug alternatives. |
| Consumer Preferences | Demand for long-acting and curative therapies with fewer side effects. |
| Market Growth Drivers | Market expansion driven by precision medicine, biopharmaceutical innovation, and policy reforms. |
Country-Wise Outlook
United States
The report on the United States Gaucher and Pompe diseases enzyme replacement therapy (ERT) market provides an analysis and a competitive landscape on the rare lysosomal storage disorders (LSDs), highlighting the market drivers, challenges, and key players involved. The growth of the biopharmaceutical industry and a well-established healthcare sector have facilitated the R&D of ERTs for these genetic conditions.
The FDA's Orphan Drug Designation and Rare Disease Priority Review programs should incentivize drug manufacturers to develop next-generation ERT formulations that optimize clinical outcomes. In addition, the expanding availability of insurance coverage and reimbursement programs is enhancing patient access to these expensive therapies. Additionally, the growing newborn screening programs and the development of genetic testing are leading to early diagnosis and timely intervention for Gaucher and Pompe diseases.
| Country | CAGR (2025 to 2035) |
|---|---|
| USA | 6.0% |
United Kingdom
The UK market for Gaucher and Pompe disease enzyme replacement medications is expanding, notably propelled by government-supported rare disease efforts, more clinical studies, and expanded access to innovative therapies. They are being supported by the National Health Service (NHS) and the National Institute for Health and Care Excellence (NICE) for early diagnosis and subsidized access to orphan drugs, which will allow quite a few patients to receive treatment.
The availability of specialized metabolic disorder treatment centres is also high, and the collaborations between biotech firms and academic institutions are propelling innovation mechanisms for the ERT sector. Research continues in gene therapy alternatives for lysosomal storage disorders, and may soon change the landscape of this market; gene therapy may eventually be able to replace or enhance existing enzyme replacement therapies.
| Country | CAGR (2025 to 2035) |
|---|---|
| UK | 5.7% |
European Union
The growing development of ERT for Gaucher and Pompe diseases in 13 European countries is fuelled by orphan drug regulatory incentives, rare disease funding, and sustained investments from pharmaceutical stakeholders. Germany, France and Italy lead the way, being at the vanguard of biopharmaceutical innovations for ERTs.
The recent European Medicines Agency (EMA) Orphan Drug Program has accelerated the approval and/or market access for innovative ERTs, resulting in improved patient access within the EMA's member states.
Moreover, increasing newborn screening programs and patient engagement campaigns have raised awareness and made diagnosis more likely in the early stages of the disease. With the incorporation of personalized medicine strategies and the establishment of combination therapies, the effectiveness of ERT treatments would be supplemented further.
| Region | CAGR (2025 to 2035) |
|---|---|
| European Union | 5.9% |
Japan
Japan’s Gaucher and Pompe diseases are expected to continue growing at a steady rate, driven by government-backed rare disease policies, burgeoning clinical research landscape in lysosomal storage disorder, and completion of translational research to precision medicine. MHLW actively promotes early diagnosis and subsidized access to orphan drugs, so that patients can receive treatment as early as possible.
The organic biotech sector and flagship pharmaceutical companies are investing in next-generation enzyme replacement therapies that will be longer-lasting and more effective formulations. The increasing use of AI and big data analytics in personalized medicine in Japan also serves to enhance ERT patient monitoring and treatment customization.
| Country | CAGR (2025 to 2035) |
|---|---|
| Japan | 5.8% |
South Korea
The market for Gaucher and Pompe disease ERT in South Korea is set to grow a lot because of the government initiatives to fund rare disease research, healthcare spending is increasing, and investments in the biotech industry are rising. With gene therapy and biologics manufacturing advancing rapidly in the country, they will have new enzyme replacement therapies with better efficacy.
Insurance reimbursement and financial assistance programs are helping patients in South Korea access ERT treatments through national rare disease programs. Moreover, the increasing acceptance of telemedicine and digital health platforms act as a facilitator for disease management as the effectiveness of enzyme replacement therapy among patient can be monitored effectively.
The growing biotechnology market, coupled with the rising number of strategic partnership agreements among global biotechnology companies offering contract research services, is predicted to spur the market growth.
| Country | CAGR (2025 to 2035) |
|---|---|
| South Korea | 5.8% |
Segmentation Outlook - Gaucher and pompe diseases enzyme replacement therapy (ert) market
Type 1 Gaucher disease and late-onset Pompe disease segments drive market growth as demand for enzyme replacement therapy (ert) expands
The segment of Type 1 Gaucher disease and late-onset Pompe disease dominates the Gaucher and Pompe disease ERT market, as an increasing population of lysosomal storage disorders and developing ERT. The majority of patients, however, have type 1 Gaucher disease which is also, at the same time, the most common and treatable form of Gaucher disease, with well-established ERT (Enzyme Replacement Therapy) approaches that can alleviate symptoms and improve the quality of life for patients with Gaucher.
In contrast, late-onset Pompe disease (LOPD) is associated with a substantial treatment gap while raising the demand for long-term ERT solutions, which is due to awareness from patients in addition to a growing breadth of diagnostic capabilities. These segments support market expansion as research advances and new treatment methods arise.
Type 1 Gaucher disease is a broader patient population with non-neuropathic symptoms responsive to ERT. Haematological and visceral symptoms have improved remarkably in individuals receiving regular enzyme replacement therapy. The late-onset form of Pompe disease, causing progressive weakness of skeletal and respiratory muscles, requires long-term ERT to manage progression and maintain ambulation.
The market growth is further driven by healthcare systems bolstering frameworks of management of rare diseases and pharmaceutical companies investing in targeted therapies, which is expected to ensure the continuous evolution of the market for effective ERT solutions for rare diseases.
Most Treatable and Highly Managed Condition in ERT Market Is Type 1 Gaucher Disease
The most common form of Gaucher disease is Type 1 Gaucher disease, constituting the most diagnosed worldwide cases. In contrast to the neuropathic forms, Type 1 Gaucher disease does not involve the central nervous system, providing a larger clinical window, and a greater response to enzyme replacement therapy. ERT effectively ameliorates hepatosplenomegaly, anaemia, thrombocytopenia, and any associated bone complications, greatly improving the patients' quality of life.
Several ERT options have emerged from the pharmaceuticals field, including imiglucerase, velaglucerase alfa, and taliglucerase alfa. These recombinant enzymes work by restoring the low levels of glucocerebrosidase, preventing glucocerebroside from building up to toxic levels in cells.
Regular ERT treatment has been shown in clinical studies to stabilize disease progression and reduce subsequent long-term complications, thus reaffirming its place as first-line therapy in Type 1 Gaucher disease.
Furthermore, convenient patient administration schedules and home infusion options have increased therapy adherence and accessibility. Many providers implement personalized ERT regimens based on disease severity, optimizing treatment efficacy.
As the focus of research continues on next-generation ERT formulations that provide better bioavailability and lower immunogenicity, Type 1 Gaucher disease management shows continued promise. It will ensure ongoing investment in this area.
Newer Developments in Type 1 Gaucher Disease ERT and Their Impact on Patient Long-Term Management
Recombinant technologies for enzyme replacement therapy greatly improved the efficacy and provided patients greater convenience for the treatment of Type 1 Gaucher disease. Recombinant enzyme engineering techniques allow for the preparation of more stable formulations with an extended half-life, leading to decreased infusion frequency and thus to increased patient compliance. New drug delivery systems are also being developed to promote the uptake of the enzymes in target cells, thereby increasing therapeutic efficacy.
With the integration of digital health monitoring and patient management platforms in healthcare systems, real-time tracking of the ERT response and symptom progression is now more possible. By utilizing these technologies, physicians can personalize the treatment regimen for each patient to achieve effective control of the disease.
Additionally, new avenues of treatment are being explored through clinical trials, including substrate reduction therapy (SRT) and gene therapy, which build upon the efficacy of the current options available.
This has created an impetus for rare disease therapy among key regulators, while several government-backed reimbursement programs have positively impacted patient access to ERT for rare diseases. The increasing incidence of Type 1 Gaucher disease ERT is also one of the major driving factors behind the key player's dominance in the market.
Late-Onset Pompe Disease Drives ERT Adoption as Diagnostic and Treatment Options Expand
Among these, late-onset Pompe disease (LOPD) has become the main driver in the ERT market, accompanied by increased awareness of its progressive neuromuscular symptoms that require timely intervention.
In contrast, classic infantile-onset Pompe disease appears during the first few months of life. It has an aggressive course, while LOPD [late-onset Pompe disease] manifests later with progressive muscle weakness, respiratory insufficiency, and limited ambulation. Enzyme replacement therapy is key in stabilizing disease progression and preserving muscle function in patients.
For late-onset Pompe disease, ERT, mainly alglucosidase alfa and alglucosidase alfa, gives the required enzyme activity to prevent the accumulation of glycogen in lysosomes. Frequent application of these therapies increases pulmonary function, muscle strength, and improves mobility in patients.
The most commonly used ERTs remain limited by their dependence on uptake by the mannose-6-phosphate (M6P) receptor in the lack of adequate levels of enzyme delivery to affected tissues, and as a result, there are ongoing clinical trials evaluating new formulations that can improve enzyme uptake and tissue penetration.
Although efficacious for LOPD, ERT requires lifelong therapy and is associated with significant treatment adherence challenges and accessibility issues. In conjunction with ERT, multidisciplinary care approaches involving physical therapy, respiratory support, and dietary management are highlighted by healthcare providers to maximize patient outcomes.
With advancements in technology driving the development of gene therapy and new treatment modalities, the management of late-onset Pompe disease will likely expand beyond traditional ERT.
University Contact Network Expands Along with the Rise of Research in the Treatment and Support of Late-Onset Pompe Disease
A wealth of research on late-onset Pompe disease has facilitated great strides in terms of diagnosis and therapeutic intervention. Improvements in biomarkers and genetic screening have allowed for earlier recognition of disease, which has led to better timing of ERT initiation and improved long-term outcomes. “Muscle biopsy, creatine kinase testing, and pulmonary function testing are now used by physicians to more specifically refine the diagnosis and tailor treatment accordingly.”
Patient-centric healthcare models to empower those on chronic ERT for LOPD have also been developed. Such therapies have become increasingly difficult to navigate, but telemedicine services, remote infusion monitoring, and patient education programs have all improved therapy adherence and minimized treatment discontinuation rates. These pharmaceutical companies work with patient advocacy organizations to spread the word and help patients access specialized Pompe disease care centres.
Further investigation of adjunctive therapies, such as enzyme stabilizers and chaperone molecules, has focused on improving the bioavailability of exogenously administered enzymes, thereby possibly decreasing the frequency and dosage needs of ERT components. As investigational therapies advance toward clinical implementation, they have the potential to facilitate this as well as offer replacement therapies for traditional ERT in the future.
Gene therapy has attracted considerable attention as an alternative one-time treatment to lifelong enzyme replacement therapy (ERT). The proof-of-concept is supported by ongoing clinical trials that investigate the efficacy of AAV-based gene delivery systems, which seek to avert the disease burden by establishing more persistent levels of enzymatic activity. If successfully developed, these therapies could transform the treatment landscape by providing a curative option for patients harbouring Gaucher and Pompe diseases.
Competitive Outlook
The gaucher and pompe diseases enzyme replacement therapy (ert) market is a highly competitive and dynamic market in the biopharmaceutical field, cantered on lifesaving therapies to treat pennies of lysosomal storage disorders. Enzyme-replacement therapy (ert) is the first-line treatment for these disorders, assisting them in making up for the lack of essential enzymes, and improving patient outcomes.
We are capitalizing on this by bringing in leading innovators focused on developing new-generation biologics, discovery of gene therapy, and next-generation delivery mechanisms that can maximize treatment benefit while minimizing side effects. This market encompasses global biopharmaceutical companies, rare disease specialists, and emerging biotech companies that are working on innovative therapeutics.
Market Share Analysis by Company
| Company Name | Estimated Market Share (%) |
|---|---|
| Sanofi (Genzyme) | 35-40% |
| Takeda Pharmaceutical Company Limited | 20-25% |
| Pfizer Inc. | 12-16% |
| Amicus Therapeutics, Inc. | 8-12% |
| AVROBIO, Inc. | 5-9% |
| Other Companies (combined) | 10-20% |
Key Company Offerings and Activities
| Company Name | Key Offerings/Activities |
|---|---|
| Sanofi (Genzyme) | Manufactures Cerezyme® (imiglucerase) for Gaucher disease and Nexviazyme® (avalglucosidase alfa) for Pompe disease, focusing on next-generation ERTs. |
| Takeda Pharmaceutical Company Limited | Develops VPRIV® (velaglucerase alfa) for Gaucher disease, prioritizing enzyme engineering and long-term efficacy studies. |
| Pfizer Inc. | Invests in novel ERT formulations and next-generation delivery mechanisms for lysosomal storage disorders, including Pompe disease. |
| Amicus Therapeutics, Inc. | It specializes in chaperone therapy and next-gen ERT solutions and is working on combination therapies for improved enzyme stability. |
| AVROBIO, Inc. | Focuses on gene therapy research as a potential alternative to traditional ERT for Gaucher and Pompe diseases. |
Key Company Insights
Sanofi (Genzyme) (35-40%)
Sanofi dominates the market with Cerezyme® and Nexviazyme®, offering proven ERTs for Gaucher and Pompe diseases, with ongoing research in next-generation biologics.
Takeda Pharmaceutical Company Limited (20-25%)
Takeda develops VPRIV® for gaucher disease, emphasizing long-term safety, efficacy, and enzyme engineering for better patient outcomes.
Pfizer Inc. (12-16%)
Pfizer is investing in next-generation ERTs and gene therapy approaches, targeting improved enzyme half-life and delivery methods.
Amicus Therapeutics, Inc. (8-12%)
Amicus is a pioneer in chaperone therapy and enzyme stability technologies, aiming to enhance ERT effectiveness for lysosomal storage disorders.
AVROBIO, Inc. (5-9%)
AVROBIO is developing gene therapy alternatives to traditional ERT, with a focus on long-term solutions for Gaucher and Pompe diseases.
Other Key Players (10-20% Combined)
Several other companies contribute to the ERT market for Gaucher and Pompe diseases, focusing on next-gen therapies, biosimilars, and novel enzyme formulations:
- JCR Pharmaceuticals Co., Ltd.
- Freeline Therapeutics Holdings plc
- Sangamo Therapeutics, Inc.
- Orchard Therapeutics plc
- Ultragenyx Pharmaceutical Inc.
- REGENXBIO Inc.
- Sigilon Therapeutics, Inc.
Gaucher and Pompe Diseases Enzyme Replacement Therapy (ERT) Market Segmentation
By Therapeutic Condition:
- Gaucher Disease
- Type 1 (Non-neuropathic)
- Type 2 (Acute Infantile Neuronopathic)
- Type 3 Gaucher Disease (Chronic Neuronopathic)
- Perinatal Lethal Gaucher Disease
- Cardiovascular Gaucher Disease
- Pompe Disease
- Classic Infantile-onset
- Late-onset
By Route of Administration:
- Oral
- Parental
By Distribution Channel:
- Hospital Pharmacies
- Specialty Treatment Pharmacies
- Retail Pharmacies
- Online Pharmacies
By Region:
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia Pacific
- Middle East and Africa
Frequently Asked Questions
What was the overall size of the gaucher and pompe diseases enzyme replacement therapy (ERT) market in 2025?
The overall market size for the gaucher and pompe diseases enzyme replacement therapy (ERT) market was USD 3,721.8 million in 2025.
How big is the gaucher and pompe diseases enzyme replacement therapy (ERT) market expected to be in 2035?
The gaucher and pompe diseases enzyme replacement therapy (ERT) market is expected to reach USD 6,540.6 million in 2035.
What will drive the demand for the gaucher and pompe diseases enzyme replacement therapy (ERT) market during the forecast period?
The increasing prevalence of lysosomal storage disorders, rising advancements in enzyme replacement therapies, and growing focus on early diagnosis and treatment fuel the gaucher and pompe diseases enzyme replacement therapy (ERT) market during the forecast period.
List the top 5 countries contributing to the gaucher and pompe diseases enzyme replacement therapy (ERT) market.
The top 5 countries driving the development of the gaucher and pompe diseases enzyme replacement therapy (ERT) market are the USA, UK, European Union, Japan, and South Korea.
Which segment in type is expected to lead in the gaucher and pompe diseases enzyme replacement therapy (ERT) market?
Type 1 gaucher disease and late-onset pompe disease segments lead market growth to command a significant share over the assessment period.
Table of Content
- Executive Summary
- Industry Introduction, including Taxonomy and Market Definition
- Market Trends and Success Factors, including Macro-economic Factors, Market Dynamics, and Recent Industry Developments
- Global Market Demand Analysis 2020 to 2024 and Forecast 2025 to 2035, including Historical Analysis and Future Projections
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035
- Therapeutic Condition
- Route of Administration
- Distribution Channel
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Therapeutic Condition
- Gaucher Disease
- Type 1 (Non-neuropathic)
- Type 2 (Acute Infantile Neuronopathic)
- Type 3 Gaucher Disease (Chronic Neuronopathic)
- Perinatal Lethal Gaucher Disease
- Cardiovascular Gaucher Disease
- Pompe Disease
- Classic Infantile-onset
- Late-onset
- Gaucher Disease
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Route of Administration
- Oral
- Parental
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Distribution Channel
- Hospital Pharmacies
- Specialty Treatment Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia Pacific
- Middle East and Africa
- North America Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Latin America Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Western Europe Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Eastern Europe Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- East Asia Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- South Asia Pacific Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Middle East and Africa Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Sales Forecast 2025 to 2035 by Therapeutic Condition and Route of Administration for 30 Countries
- Competition Outlook, including Market Structure Analysis, Company Share Analysis by Key Players, and Competition Dashboard
- Company Profile
- Alexion Pharmaceuticals, Inc.
- Pfizer
- BioMarin Pharmaceuticals Inc
- Ultragenyx Pharmaceutical Inc.
- Janssen Pharmaceuticals, Inc (J&J)
- Sigma-Tau Pharmaceuticals Inc.
- AbbVie Inc.
- Sanofi S.A.
- Shire Plc
- Merck KGaA
List of Tables
Table 1: Global Market Value (US$ Million) Forecast by Region, 2018 to 2033
Table 2: Global Market Value (US$ Million) Forecast by Disease, 2018 to 2033
Table 3: Global Market Value (US$ Million) Forecast by Route of Administration, 2018 to 2033
Table 4: Global Market Value (US$ Million) Forecast by Distribution Channel , 2018 to 2033
Table 5: North America Market Value (US$ Million) Forecast by Country, 2018 to 2033
Table 6: North America Market Value (US$ Million) Forecast by Disease, 2018 to 2033
Table 7: North America Market Value (US$ Million) Forecast by Route of Administration, 2018 to 2033
Table 8: North America Market Value (US$ Million) Forecast by Distribution Channel , 2018 to 2033
Table 9: Latin America Market Value (US$ Million) Forecast by Country, 2018 to 2033
Table 10: Latin America Market Value (US$ Million) Forecast by Disease, 2018 to 2033
Table 11: Latin America Market Value (US$ Million) Forecast by Route of Administration, 2018 to 2033
Table 12: Latin America Market Value (US$ Million) Forecast by Distribution Channel , 2018 to 2033
Table 13: Europe Market Value (US$ Million) Forecast by Country, 2018 to 2033
Table 14: Europe Market Value (US$ Million) Forecast by Disease, 2018 to 2033
Table 15: Europe Market Value (US$ Million) Forecast by Route of Administration, 2018 to 2033
Table 16: Europe Market Value (US$ Million) Forecast by Distribution Channel , 2018 to 2033
Table 17: South Asia Market Value (US$ Million) Forecast by Country, 2018 to 2033
Table 18: South Asia Market Value (US$ Million) Forecast by Disease, 2018 to 2033
Table 19: South Asia Market Value (US$ Million) Forecast by Route of Administration, 2018 to 2033
Table 20: South Asia Market Value (US$ Million) Forecast by Distribution Channel , 2018 to 2033
Table 21: East Asia Market Value (US$ Million) Forecast by Country, 2018 to 2033
Table 22: East Asia Market Value (US$ Million) Forecast by Disease, 2018 to 2033
Table 23: East Asia Market Value (US$ Million) Forecast by Route of Administration, 2018 to 2033
Table 24: East Asia Market Value (US$ Million) Forecast by Distribution Channel , 2018 to 2033
Table 25: Oceania Market Value (US$ Million) Forecast by Country, 2018 to 2033
Table 26: Oceania Market Value (US$ Million) Forecast by Disease, 2018 to 2033
Table 27: Oceania Market Value (US$ Million) Forecast by Route of Administration, 2018 to 2033
Table 28: Oceania Market Value (US$ Million) Forecast by Distribution Channel , 2018 to 2033
Table 29: MEA Market Value (US$ Million) Forecast by Country, 2018 to 2033
Table 30: MEA Market Value (US$ Million) Forecast by Disease, 2018 to 2033
Table 31: MEA Market Value (US$ Million) Forecast by Route of Administration, 2018 to 2033
Table 32: MEA Market Value (US$ Million) Forecast by Distribution Channel , 2018 to 2033
List of Figures
Figure 1: Global Market Value (US$ Million) by Disease, 2023 to 2033
Figure 2: Global Market Value (US$ Million) by Route of Administration, 2023 to 2033
Figure 3: Global Market Value (US$ Million) by Distribution Channel , 2023 to 2033
Figure 4: Global Market Value (US$ Million) by Region, 2023 to 2033
Figure 5: Global Market Value (US$ Million) Analysis by Region, 2018 to 2033
Figure 6: Global Market Value Share (%) and BPS Analysis by Region, 2023 to 2033
Figure 7: Global Market Y-o-Y Growth (%) Projections by Region, 2023 to 2033
Figure 8: Global Market Value (US$ Million) Analysis by Disease, 2018 to 2033
Figure 9: Global Market Value Share (%) and BPS Analysis by Disease, 2023 to 2033
Figure 10: Global Market Y-o-Y Growth (%) Projections by Disease, 2023 to 2033
Figure 11: Global Market Value (US$ Million) Analysis by Route of Administration, 2018 to 2033
Figure 12: Global Market Value Share (%) and BPS Analysis by Route of Administration, 2023 to 2033
Figure 13: Global Market Y-o-Y Growth (%) Projections by Route of Administration, 2023 to 2033
Figure 14: Global Market Value (US$ Million) Analysis by Distribution Channel , 2018 to 2033
Figure 15: Global Market Value Share (%) and BPS Analysis by Distribution Channel , 2023 to 2033
Figure 16: Global Market Y-o-Y Growth (%) Projections by Distribution Channel , 2023 to 2033
Figure 17: Global Market Attractiveness by Disease, 2023 to 2033
Figure 18: Global Market Attractiveness by Route of Administration, 2023 to 2033
Figure 19: Global Market Attractiveness by Distribution Channel , 2023 to 2033
Figure 20: Global Market Attractiveness by Region, 2023 to 2033
Figure 21: North America Market Value (US$ Million) by Disease, 2023 to 2033
Figure 22: North America Market Value (US$ Million) by Route of Administration, 2023 to 2033
Figure 23: North America Market Value (US$ Million) by Distribution Channel , 2023 to 2033
Figure 24: North America Market Value (US$ Million) by Country, 2023 to 2033
Figure 25: North America Market Value (US$ Million) Analysis by Country, 2018 to 2033
Figure 26: North America Market Value Share (%) and BPS Analysis by Country, 2023 to 2033
Figure 27: North America Market Y-o-Y Growth (%) Projections by Country, 2023 to 2033
Figure 28: North America Market Value (US$ Million) Analysis by Disease, 2018 to 2033
Figure 29: North America Market Value Share (%) and BPS Analysis by Disease, 2023 to 2033
Figure 30: North America Market Y-o-Y Growth (%) Projections by Disease, 2023 to 2033
Figure 31: North America Market Value (US$ Million) Analysis by Route of Administration, 2018 to 2033
Figure 32: North America Market Value Share (%) and BPS Analysis by Route of Administration, 2023 to 2033
Figure 33: North America Market Y-o-Y Growth (%) Projections by Route of Administration, 2023 to 2033
Figure 34: North America Market Value (US$ Million) Analysis by Distribution Channel , 2018 to 2033
Figure 35: North America Market Value Share (%) and BPS Analysis by Distribution Channel , 2023 to 2033
Figure 36: North America Market Y-o-Y Growth (%) Projections by Distribution Channel , 2023 to 2033
Figure 37: North America Market Attractiveness by Disease, 2023 to 2033
Figure 38: North America Market Attractiveness by Route of Administration, 2023 to 2033
Figure 39: North America Market Attractiveness by Distribution Channel , 2023 to 2033
Figure 40: North America Market Attractiveness by Country, 2023 to 2033
Figure 41: Latin America Market Value (US$ Million) by Disease, 2023 to 2033
Figure 42: Latin America Market Value (US$ Million) by Route of Administration, 2023 to 2033
Figure 43: Latin America Market Value (US$ Million) by Distribution Channel , 2023 to 2033
Figure 44: Latin America Market Value (US$ Million) by Country, 2023 to 2033
Figure 45: Latin America Market Value (US$ Million) Analysis by Country, 2018 to 2033
Figure 46: Latin America Market Value Share (%) and BPS Analysis by Country, 2023 to 2033
Figure 47: Latin America Market Y-o-Y Growth (%) Projections by Country, 2023 to 2033
Figure 48: Latin America Market Value (US$ Million) Analysis by Disease, 2018 to 2033
Figure 49: Latin America Market Value Share (%) and BPS Analysis by Disease, 2023 to 2033
Figure 50: Latin America Market Y-o-Y Growth (%) Projections by Disease, 2023 to 2033
Figure 51: Latin America Market Value (US$ Million) Analysis by Route of Administration, 2018 to 2033
Figure 52: Latin America Market Value Share (%) and BPS Analysis by Route of Administration, 2023 to 2033
Figure 53: Latin America Market Y-o-Y Growth (%) Projections by Route of Administration, 2023 to 2033
Figure 54: Latin America Market Value (US$ Million) Analysis by Distribution Channel , 2018 to 2033
Figure 55: Latin America Market Value Share (%) and BPS Analysis by Distribution Channel , 2023 to 2033
Figure 56: Latin America Market Y-o-Y Growth (%) Projections by Distribution Channel , 2023 to 2033
Figure 57: Latin America Market Attractiveness by Disease, 2023 to 2033
Figure 58: Latin America Market Attractiveness by Route of Administration, 2023 to 2033
Figure 59: Latin America Market Attractiveness by Distribution Channel , 2023 to 2033
Figure 60: Latin America Market Attractiveness by Country, 2023 to 2033
Figure 61: Europe Market Value (US$ Million) by Disease, 2023 to 2033
Figure 62: Europe Market Value (US$ Million) by Route of Administration, 2023 to 2033
Figure 63: Europe Market Value (US$ Million) by Distribution Channel , 2023 to 2033
Figure 64: Europe Market Value (US$ Million) by Country, 2023 to 2033
Figure 65: Europe Market Value (US$ Million) Analysis by Country, 2018 to 2033
Figure 66: Europe Market Value Share (%) and BPS Analysis by Country, 2023 to 2033
Figure 67: Europe Market Y-o-Y Growth (%) Projections by Country, 2023 to 2033
Figure 68: Europe Market Value (US$ Million) Analysis by Disease, 2018 to 2033
Figure 69: Europe Market Value Share (%) and BPS Analysis by Disease, 2023 to 2033
Figure 70: Europe Market Y-o-Y Growth (%) Projections by Disease, 2023 to 2033
Figure 71: Europe Market Value (US$ Million) Analysis by Route of Administration, 2018 to 2033
Figure 72: Europe Market Value Share (%) and BPS Analysis by Route of Administration, 2023 to 2033
Figure 73: Europe Market Y-o-Y Growth (%) Projections by Route of Administration, 2023 to 2033
Figure 74: Europe Market Value (US$ Million) Analysis by Distribution Channel , 2018 to 2033
Figure 75: Europe Market Value Share (%) and BPS Analysis by Distribution Channel , 2023 to 2033
Figure 76: Europe Market Y-o-Y Growth (%) Projections by Distribution Channel , 2023 to 2033
Figure 77: Europe Market Attractiveness by Disease, 2023 to 2033
Figure 78: Europe Market Attractiveness by Route of Administration, 2023 to 2033
Figure 79: Europe Market Attractiveness by Distribution Channel , 2023 to 2033
Figure 80: Europe Market Attractiveness by Country, 2023 to 2033
Figure 81: South Asia Market Value (US$ Million) by Disease, 2023 to 2033
Figure 82: South Asia Market Value (US$ Million) by Route of Administration, 2023 to 2033
Figure 83: South Asia Market Value (US$ Million) by Distribution Channel , 2023 to 2033
Figure 84: South Asia Market Value (US$ Million) by Country, 2023 to 2033
Figure 85: South Asia Market Value (US$ Million) Analysis by Country, 2018 to 2033
Figure 86: South Asia Market Value Share (%) and BPS Analysis by Country, 2023 to 2033
Figure 87: South Asia Market Y-o-Y Growth (%) Projections by Country, 2023 to 2033
Figure 88: South Asia Market Value (US$ Million) Analysis by Disease, 2018 to 2033
Figure 89: South Asia Market Value Share (%) and BPS Analysis by Disease, 2023 to 2033
Figure 90: South Asia Market Y-o-Y Growth (%) Projections by Disease, 2023 to 2033
Figure 91: South Asia Market Value (US$ Million) Analysis by Route of Administration, 2018 to 2033
Figure 92: South Asia Market Value Share (%) and BPS Analysis by Route of Administration, 2023 to 2033
Figure 93: South Asia Market Y-o-Y Growth (%) Projections by Route of Administration, 2023 to 2033
Figure 94: South Asia Market Value (US$ Million) Analysis by Distribution Channel , 2018 to 2033
Figure 95: South Asia Market Value Share (%) and BPS Analysis by Distribution Channel , 2023 to 2033
Figure 96: South Asia Market Y-o-Y Growth (%) Projections by Distribution Channel , 2023 to 2033
Figure 97: South Asia Market Attractiveness by Disease, 2023 to 2033
Figure 98: South Asia Market Attractiveness by Route of Administration, 2023 to 2033
Figure 99: South Asia Market Attractiveness by Distribution Channel , 2023 to 2033
Figure 100: South Asia Market Attractiveness by Country, 2023 to 2033
Figure 101: East Asia Market Value (US$ Million) by Disease, 2023 to 2033
Figure 102: East Asia Market Value (US$ Million) by Route of Administration, 2023 to 2033
Figure 103: East Asia Market Value (US$ Million) by Distribution Channel , 2023 to 2033
Figure 104: East Asia Market Value (US$ Million) by Country, 2023 to 2033
Figure 105: East Asia Market Value (US$ Million) Analysis by Country, 2018 to 2033
Figure 106: East Asia Market Value Share (%) and BPS Analysis by Country, 2023 to 2033
Figure 107: East Asia Market Y-o-Y Growth (%) Projections by Country, 2023 to 2033
Figure 108: East Asia Market Value (US$ Million) Analysis by Disease, 2018 to 2033
Figure 109: East Asia Market Value Share (%) and BPS Analysis by Disease, 2023 to 2033
Figure 110: East Asia Market Y-o-Y Growth (%) Projections by Disease, 2023 to 2033
Figure 111: East Asia Market Value (US$ Million) Analysis by Route of Administration, 2018 to 2033
Figure 112: East Asia Market Value Share (%) and BPS Analysis by Route of Administration, 2023 to 2033
Figure 113: East Asia Market Y-o-Y Growth (%) Projections by Route of Administration, 2023 to 2033
Figure 114: East Asia Market Value (US$ Million) Analysis by Distribution Channel , 2018 to 2033
Figure 115: East Asia Market Value Share (%) and BPS Analysis by Distribution Channel , 2023 to 2033
Figure 116: East Asia Market Y-o-Y Growth (%) Projections by Distribution Channel , 2023 to 2033
Figure 117: East Asia Market Attractiveness by Disease, 2023 to 2033
Figure 118: East Asia Market Attractiveness by Route of Administration, 2023 to 2033
Figure 119: East Asia Market Attractiveness by Distribution Channel , 2023 to 2033
Figure 120: East Asia Market Attractiveness by Country, 2023 to 2033
Figure 121: Oceania Market Value (US$ Million) by Disease, 2023 to 2033
Figure 122: Oceania Market Value (US$ Million) by Route of Administration, 2023 to 2033
Figure 123: Oceania Market Value (US$ Million) by Distribution Channel , 2023 to 2033
Figure 124: Oceania Market Value (US$ Million) by Country, 2023 to 2033
Figure 125: Oceania Market Value (US$ Million) Analysis by Country, 2018 to 2033
Figure 126: Oceania Market Value Share (%) and BPS Analysis by Country, 2023 to 2033
Figure 127: Oceania Market Y-o-Y Growth (%) Projections by Country, 2023 to 2033
Figure 128: Oceania Market Value (US$ Million) Analysis by Disease, 2018 to 2033
Figure 129: Oceania Market Value Share (%) and BPS Analysis by Disease, 2023 to 2033
Figure 130: Oceania Market Y-o-Y Growth (%) Projections by Disease, 2023 to 2033
Figure 131: Oceania Market Value (US$ Million) Analysis by Route of Administration, 2018 to 2033
Figure 132: Oceania Market Value Share (%) and BPS Analysis by Route of Administration, 2023 to 2033
Figure 133: Oceania Market Y-o-Y Growth (%) Projections by Route of Administration, 2023 to 2033
Figure 134: Oceania Market Value (US$ Million) Analysis by Distribution Channel , 2018 to 2033
Figure 135: Oceania Market Value Share (%) and BPS Analysis by Distribution Channel , 2023 to 2033
Figure 136: Oceania Market Y-o-Y Growth (%) Projections by Distribution Channel , 2023 to 2033
Figure 137: Oceania Market Attractiveness by Disease, 2023 to 2033
Figure 138: Oceania Market Attractiveness by Route of Administration, 2023 to 2033
Figure 139: Oceania Market Attractiveness by Distribution Channel , 2023 to 2033
Figure 140: Oceania Market Attractiveness by Country, 2023 to 2033
Figure 141: MEA Market Value (US$ Million) by Disease, 2023 to 2033
Figure 142: MEA Market Value (US$ Million) by Route of Administration, 2023 to 2033
Figure 143: MEA Market Value (US$ Million) by Distribution Channel , 2023 to 2033
Figure 144: MEA Market Value (US$ Million) by Country, 2023 to 2033
Figure 145: MEA Market Value (US$ Million) Analysis by Country, 2018 to 2033
Figure 146: MEA Market Value Share (%) and BPS Analysis by Country, 2023 to 2033
Figure 147: MEA Market Y-o-Y Growth (%) Projections by Country, 2023 to 2033
Figure 148: MEA Market Value (US$ Million) Analysis by Disease, 2018 to 2033
Figure 149: MEA Market Value Share (%) and BPS Analysis by Disease, 2023 to 2033
Figure 150: MEA Market Y-o-Y Growth (%) Projections by Disease, 2023 to 2033
Figure 151: MEA Market Value (US$ Million) Analysis by Route of Administration, 2018 to 2033
Figure 152: MEA Market Value Share (%) and BPS Analysis by Route of Administration, 2023 to 2033
Figure 153: MEA Market Y-o-Y Growth (%) Projections by Route of Administration, 2023 to 2033
Figure 154: MEA Market Value (US$ Million) Analysis by Distribution Channel , 2018 to 2033
Figure 155: MEA Market Value Share (%) and BPS Analysis by Distribution Channel , 2023 to 2033
Figure 156: MEA Market Y-o-Y Growth (%) Projections by Distribution Channel , 2023 to 2033
Figure 157: MEA Market Attractiveness by Disease, 2023 to 2033
Figure 158: MEA Market Attractiveness by Route of Administration, 2023 to 2033
Figure 159: MEA Market Attractiveness by Distribution Channel , 2023 to 2033
Figure 160: MEA Market Attractiveness by Country, 2023 to 2033
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

