High Barrier Packaging Films for Pharmaceuticals Market
The high barrier packaging films for pharmaceuticals market is segmented by Material Type (Metallized Films, Aluminum Oxide Coated Films, Silicon Oxide Coated Films, PVDC Coated Films), Barrier Level (Ultra-High Barrier, High Barrier, Medium Barrier, Standard Barrier), Application (Blister Packaging, Strip Packaging, Sachet Packaging, Pouch Packaging), Thickness Range (Below 50 Microns, 50 to 100 Microns, 100 to 200 Microns, Above 200 Microns), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
High Barrier Packaging Films for Pharmaceuticals Market Size, Market Forecast and Outlook By FMI
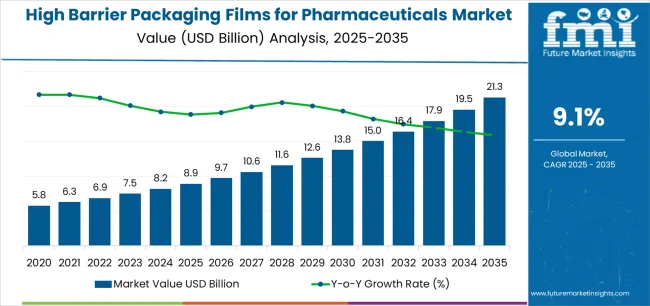
The high barrier packaging films for pharmaceuticals market is expected to expand from USD 9.7 billion in 2026 to USD 22.8 billion by 2036. The market is anticipated to register a 8.9% CAGR during the forecast period. Metallized films are likely to lead with a 43.0% share, while blister packaging is projected to account for 48.0% of application-level demand in 2026.
Summary of the High Barrier Packaging Films for Pharmaceuticals Market
- Demand and Growth Drivers
- Drug stability needs are increasing demand for films with strong moisture and oxygen protection.
- Specialty medicines require packaging films that protect product quality across long supply chains.
- Pharmaceutical quality rules are pushing converters toward validated barrier film structures.
- Product and Segment View
- Metallized films are expected to lead material demand due to reliable barrier strength and scalable production.
- Blister packaging is likely to remain the main application as unit-dose drug formats expand.
- Oxide-coated films are anticipated to gain interest where clarity and high barrier performance are needed.
- Geography and Competitive Outlook
- China is expected to lead growth as domestic drug production and export packaging scale increase.
- The USA is likely to remain a major value market due to strict drug packaging standards.
- Suppliers with coating precision and validation support are anticipated to gain stronger pharmaceutical accounts.
- Analyst Opinion
- Ismail Sutaria, Senior Consultant for Packaging at FMI, suggests, “The high barrier packaging films for pharmaceuticals market is moving toward stronger protection and cleaner validation. Pharmaceutical customers want films that protect sensitive products without slowing filling operations. Suppliers that combine barrier performance with regulatory support will be better placed to win long-term contracts.”
- High Barrier Packaging Films for Pharmaceuticals Market Value Analysis
- The high barrier packaging films for pharmaceuticals market is becoming central to drug shelf-life protection.
- Demand is likely to rise as biologics and specialty medicines need stronger pack integrity.
- Blister packaging is expected to keep leading demand through dose protection and patient-use convenience.
- Spending is anticipated to improve where suppliers offer reliable sealing and documented barrier performance.
Blister packaging remains the dominant application area for high barrier films in pharmaceuticals, followed by strip, sachet, and pouch packaging formats. The United States leads overall market demand, driven by large-scale pharmaceutical production and advanced packaging standards. China demonstrates strong growth momentum due to expanding domestic pharmaceutical manufacturing. Germany, the United Kingdom, and Japan show steady demand supported by their established pharmaceutical and healthcare sectors.
Key manufacturers operating in the high barrier pharmaceutical packaging films market include Amcor plc, Klöckner Pentaplast Group, Tekni-Plex Inc., Constantia Flexibles, Wipak Group, Uflex Ltd., Mondi Group, Cosmo Films Limited, Toray Industries Inc., and Mitsubishi Chemical Corporation.
High Barrier Packaging Films for Pharmaceuticals Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 9.7 billion |
| Market Forecast Value (2036) | USD 22.8 billion |
| Forecast CAGR (2026-2036) | 8.9% |
Why is the High Barrier Packaging Films for Pharmaceuticals Market Expanding?
The high barrier packaging films for pharmaceuticals market is developing as drug manufacturers strengthen their packaging strategies to ensure long term product stability and compliance with strict global quality standards. Pharmaceutical formulations are increasingly sensitive to moisture, oxygen, and temperature variation, leading companies to adopt advanced film structures that can protect active ingredients throughout storage, transport, and dispensing.
Demand is rising from producers of oral solid dosages, injectable therapies, and biologic drugs that require controlled barrier environments. These films play a central role in preserving drug potency and preventing contamination, especially for formulations distributed across complex and climate diverse supply chains. Pharmaceutical companies are prioritizing packaging materials that can maintain integrity under extended shelf life requirements while supporting efficient production and conversion processes.
Growth in the biologics and specialty drug segment is a major contributor. High value and temperature sensitive therapies require multilayer barrier films that deliver consistent sealing, puncture resistance, and protection from external environmental factors. This has increased reliance on high barrier films in blister packs, pouches, and sachets used for both prescription and over the counter medicines.
Manufacturers are also focused on operational flexibility. High barrier films that offer compatibility with high speed filling lines, improved machinability, and reliable print performance are gaining preference among pharmaceutical packaging converters. These features allow brands to maintain quality while optimizing packaging efficiency and minimizing waste.
Regulatory frameworks are reinforcing adoption. Compliance standards related to drug safety, packaging validation, and global transport conditions are encouraging pharmaceutical companies to implement barrier film systems that deliver dependable protective performance. These factors continue to support the steady advancement of the high barrier packaging films for pharmaceuticals market across international pharmaceutical manufacturing and distribution networks.
How Is the High Barrier Packaging Films for Pharmaceuticals Market Segmented by Material Type, Barrier Level, Application, Thickness Range, and Region?
The high barrier packaging films for pharmaceuticals market is segmented by material type, barrier level, application, thickness range, and region to reflect performance requirements and end-use adoption. Material types include metallized films, aluminum oxide coated films, silicon oxide coated films, and PVDC coated films, each designed for specific barrier functions. Barrier level classification consists of ultra-high barrier, high barrier, medium barrier, and standard barrier, addressing varying protection needs for pharmaceutical products. Application segmentation includes blister packaging, strip packaging, sachet packaging, and pouch packaging. Thickness range is categorized into below 50 microns, 50 to 100 microns, 100 to 200 microns, and above 200 microns. Regional demand spans North America, Europe, Asia Pacific, Latin America, Middle East and Africa, and other regions, reflecting differences in pharmaceutical production and packaging standards.
Why Do Metallized Films Hold the Largest Share in the High Barrier Packaging Films for Pharmaceuticals Market by Material Type?
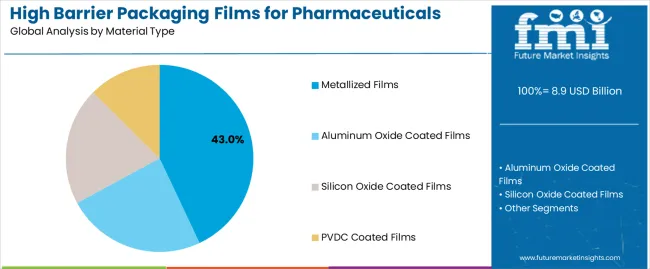
Metallized films account for 43% of the high barrier packaging films for pharmaceuticals market in 2026, reflecting their strong adoption across moisture and oxygen-sensitive drug packaging. Their dominance is supported by consistent barrier performance, scalable manufacturing capability, and compatibility with standard pharmaceutical packaging lines. These films are widely used for protecting tablets, capsules, and unit-dose medications during storage and transport. Manufacturers prefer metallized films due to their cost efficiency and reliable sealing performance. Continued investment in vacuum metallization and material quality control supports this segment’s leadership. This positions metallized films as the primary material type within the pharmaceutical high barrier films market.
What Drives the Dominance of Blister Packaging in the High Barrier Packaging Films for Pharmaceuticals Market by Application?
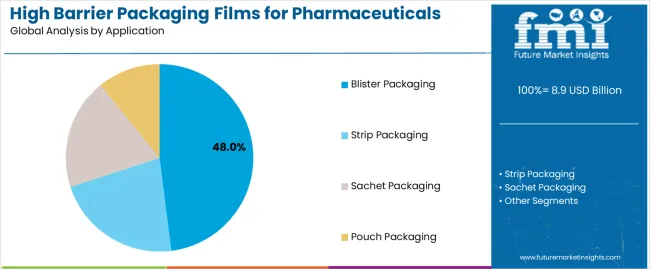
Blister packaging represents 48% of the high barrier packaging films for pharmaceuticals market in 2026, maintaining its role as the leading application format. Its dominance is due to its ability to protect unit-dose medications while making sure they are dispensed correctly and can't be tampered with. Pharmaceutical companies adopt blister packaging to support patient adherence and product stability across treatment cycles. Continuous improvement in cold-form blister technology enhances barrier effectiveness and material strength. The segment benefits from established regulatory acceptance and compatibility with automated filling systems. This secures blister packaging as the largest application segment within the pharmaceutical high barrier films market.
What are the Key Drivers in the High Barrier Packaging Films for Pharmaceuticals Market?
Demand for high barrier packaging films is increasing as pharmaceutical manufacturers prioritize drug stability, extended shelf life, and reliable protection for biologics and specialty formulations. Rising regulatory focus on sterility assurance and packaging validation is strengthening the need for materials that provide strong resistance to moisture, oxygen, and light exposure. Growth in biologics and biosimilar drug production is accelerating the adoption of advanced lamination and coating film structures that maintain product integrity throughout distribution. Manufacturers are also investing in multilayer film technology to support complex dosage formats and strict compliance requirements. These drivers are expanding the role of high barrier films in ensuring safe and efficient pharmaceutical packaging operations.
What are the Key Restraints in the High Barrier Packaging Films for Pharmaceuticals Market?
The market faces cost limitations related to high-performance resins, coatings, and multilayer film processing required for pharmaceutical-grade barrier materials. Maintaining consistent barrier strength during lamination, forming, and sealing operations adds technical complexity for converters. Regulatory approval and documentation standards increase product development time and restrict flexibility for manufacturers operating in multiple global markets. Smaller pharmaceutical companies face budget constraints that limit adoption of advanced barrier packaging. Recycling challenges associated with multilayer structures also create environmental compliance pressure. These factors slow market penetration and reduce the pace of technology upgrades across pharmaceutical packaging operations that demand both performance and affordability.
What are the Key Trends in the High Barrier Packaging Films for Pharmaceuticals Market?
Manufacturers are expanding the use of high barrier films for biologics and biosimilar drug packaging that requires superior moisture and oxygen protection. Integration of active packaging systems such as moisture-scavenging layers and stability-enhancing coatings is increasing across pharmaceutical applications. Intelligent packaging features, including temperature monitoring and serialization compatibility, are being incorporated to improve traceability and patient safety. Development of lightweight and flexible multilayer film structures is gaining attention to support portability and ease of handling. Advances in coating and lamination technologies are also improving performance while supporting compliance standards. These trends reflect the market’s shift toward higher precision, enhanced protection, and smarter pharmaceutical packaging solutions.
How Is the Global High Barrier Packaging Films for Pharmaceuticals Market Moving Forward?
The high barrier packaging films for pharmaceuticals market is growing as drug manufacturers rely on stronger and more protective film materials for safe medicine storage and transport. These films are used in blister packs, sachets, and strip packaging where consistent sealing and resistance to moisture and oxygen are critical. Manufacturers are improving multilayer film performance so pharmaceutical products remain stable throughout their shelf life. Rising production of tablets, capsules, and vaccines is supporting market expansion across regulated healthcare supply chains. The market continues to develop as pharmaceutical companies seek dependable packaging materials that balance durability, accuracy, and large-scale operational efficiency.
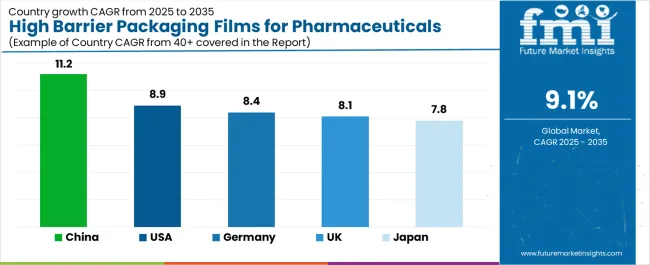
| Country | CAGR |
|---|---|
| USA | 8.9% |
| Germany | 8.4% |
| UK | 8.1% |
| China | 11.2% |
| Japan | 9.9% |
What Is Driving Demand for High Barrier Packaging Films in the USA?
Demand for the high barrier packaging films for pharmaceuticals market in the USA is progressing at an 8.9% CAGR as pharmaceutical companies expand the use of advanced multilayer films for product protection. Drug manufacturers require high-quality barrier films to ensure medicine stability during shipping and long-term storage. Suppliers are enhancing film strength and sealing accuracy to meet strict pharmaceutical packaging standards. The market benefits from the country’s strong pharmaceutical manufacturing and healthcare distribution networks. Demand for high barrier pharmaceutical films in the USA remains led by large pharmaceutical producers and contract packaging firms focused on reliability and regulatory compliance.
How Is Germany Supporting Growth in the High Barrier Pharmaceutical Films Market?
Demand for the high barrier packaging films for pharmaceuticals market in Germany is progressing at an 8.4% CAGR. The country’s advanced pharmaceutical and medical device industries rely on high-barrier films that deliver stable protection for sensitive drug formulations. Manufacturers are focusing on laminated structures that provide strong moisture and oxygen resistance while maintaining product safety. Growth reflects the expansion of pharmaceutical exports and the modernization of healthcare packaging. Demand for high barrier pharmaceutical films in Germany is primarily driven by pharmaceutical producers and specialized packaging converters supplying both domestic and European healthcare markets.
Why Is the High Barrier Pharmaceutical Films Market Expanding in the UK?
Demand for the high barrier packaging films for pharmaceuticals market in the UK is progressing at an 8.1% CAGR as pharmaceutical manufacturers increase the use of reliable barrier films for blister packs and strip packaging. Companies are strengthening packaging operations to meet rising healthcare demand and maintain consistent drug safety standards. Suppliers are improving film durability and printing quality for regulated pharmaceutical applications. Growth also reflects the expansion of pharmaceutical processing and packaging facilities. Demand for high barrier pharmaceutical films in the UK remains concentrated among pharmaceutical companies and contract packaging organizations serving the broader European market.
What Is Driving Rapid Adoption of High Barrier Packaging Films in China?
Demand for the high barrier packaging films for pharmaceuticals market in China is progressing at an 11.2% CAGR, supported by the country’s large pharmaceutical manufacturing and export operations. Drug producers are increasing the use of multilayer barrier films to protect tablets, capsules, and vaccines during storage and global distribution. Suppliers are expanding local production of aluminum-based and polymer laminates to meet industrial demand. Growth is also linked to the modernization of pharmaceutical packaging standards across the country. Demand for high barrier pharmaceutical films in China is led by pharmaceutical manufacturers and healthcare packaging suppliers focused on high-volume production and supply chain reliability.
How Is the High Barrier Pharmaceutical Films Market Performing in Japan?
Demand for the high barrier packaging films for pharmaceuticals market in Japan is progressing at a 9.9% CAGR as pharmaceutical companies adopt advanced packaging materials for precision drug storage and protection. Manufacturers require compact and highly durable barrier films that perform consistently within automated packaging environments. Suppliers are refining multilayer film structures to meet strict quality and performance expectations. Growth reflects rising production of specialty medicines and increasing healthcare packaging modernization. Demand for high barrier pharmaceutical films in Japan remains concentrated among pharmaceutical manufacturers and high-technology packaging companies that prioritize product integrity and long-term reliability.
What Is the Competitive Landscape of Demand for the High Barrier Packaging Films for Pharmaceuticals Market Globally?
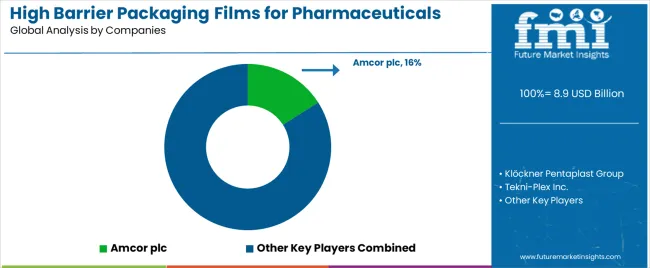
The high barrier pharmaceutical packaging films market reflects a technology intensive and moderately consolidated structure driven by the need for product stability, sterility assurance, and long term protection of sensitive drug formulations. Demand is expanding as pharmaceutical and biologic manufacturers seek multilayer films that provide superior resistance against moisture, oxygen, light, and chemical contamination. Competitive positioning is influenced by barrier performance capability, compliance with global pharmaceutical packaging standards, production scale, and the ability to co-develop specialized films for evolving drug delivery formats.
Global leaders such as Amcor plc, Klöckner Pentaplast Group, and Tekni-Plex Inc. maintain strong market positions through advanced blister films, aluminum laminate structures, and high performance thermoformable packaging materials designed for oral solids and sterile drug packaging. Their strength lies in global manufacturing networks, proven reliability, and the ability to support pharmaceutical customers with regulatory documentation and validation services.
Constantia Flexibles and Wipak Group compete by advancing sustainable high barrier solutions that reduce material weight while maintaining protection levels required for pharmaceutical compliance. These companies focus on recyclable and PVdC free film structures that align with the pharmaceutical sector’s transition toward circular economy packaging materials.
Asian and global suppliers including Uflex Ltd., Mondi Group, Cosmo Films Limited, Toray Industries Inc., and Mitsubishi Chemical Corporation are expanding their pharmaceutical film portfolios to meet rising regional demand. These players leverage raw material expertise, multilayer film processing strength, and growing production footprints to offer high clarity, multilayer barrier films that support both generic and specialty drug packaging.
Competitive dynamics in this market balance scale driven supply reliability with innovation led differentiation. Companies compete on barrier film durability, coating precision, pharmaceutical safety certifications, and their ability to integrate films into broader drug packaging workflows. Suppliers that align advanced barrier performance with sustainable material development and strong validation support are positioned to capture long term growth as pharmaceutical companies prioritize both compliance and supply chain efficiency.
Key Players in the High Barrier Packaging Films for Pharmaceuticals Market
- Amcor plc
- Klöckner Pentaplast Group
- Tekni-Plex Inc.
- Constantia Flexibles
- Wipak Group
- Uflex Ltd.
- Mondi Group
- Cosmo Films Limited
- Toray Industries Inc.
- Mitsubishi Chemical Corporation
Scope of the Report
| Item | Value |
|---|---|
| Quantitative Units | USD billion |
| Material Type | Metallized Films; Aluminum Oxide Coated Films; Silicon Oxide Coated Films; PVDC Coated Films |
| Barrier Level | Ultra-High Barrier; High Barrier; Medium Barrier; Standard Barrier |
| Application | Blister Packaging; Strip Packaging; Sachet Packaging; Pouch Packaging |
| Thickness Range | Below 50 Microns; 50-100 Microns; 100-200 Microns; Above 200 Microns |
| Regions Covered | North America; Europe; Asia Pacific; Latin America; Middle East and Africa; Other Regions |
| Countries Covered | United States; Germany; United Kingdom; China; Japan; and 40+ additional countries |
| Key Companies Profiled | Amcor plc; Klöckner Pentaplast Group; Tekni-Plex Inc.; Constantia Flexibles; Wipak Group; Uflex Ltd.; Mondi Group; Cosmo Films Limited; Toray Industries Inc.; Mitsubishi Chemical Corporation |
| Technology Type | Vacuum Metallization; Oxide Coating Technology; PVDC Barrier Coating; Multi-Layer Film Extrusion |
| End Use | Solid Dosage Packaging; Liquid Dosage Packaging; Medical Devices; Diagnostic Kits |
| Performance Features | Moisture Protection; Oxygen Barrier; Chemical Resistance; Heat Sealability; Durability |
| Additional Attributes | Dollar sales by material type, barrier level, application, thickness range, and region; regional demand trends; competitive landscape; technological advancements in coating engineering; barrier enhancement initiatives; moisture protection programs; premium product development strategies; adoption of recyclable and mono-material barrier films; integration with smart packaging and traceability systems; and supply chain optimization strategies |
High Barrier Packaging Films for Pharmaceuticals Market by Segments
Material Type:
- Metallized Films
- Aluminum Oxide Coated Films
- Silicon Oxide Coated Films
- PVDC Coated Films
Barrier Level:
- Ultra-High Barrier
- High Barrier
- Medium Barrier
- Standard Barrier
Application:
- Blister Packaging
- Strip Packaging
- Sachet Packaging
- Pouch Packaging
Thickness Range:
- Below 50 Microns
- 50-100 Microns
- 100-200 Microns
- Above 200 Microns
Region:
- North America
- United States
- Canada
- Mexico
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic Countries
- BENELUX
- Rest of Europe
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Rest of Middle East & Africa
- Other Regions
- Oceania
- Central Asia
Other Markets
Bibliography
- World Health Organization. 2023. Guidance on pharmaceutical packaging materials and barrier film performance requirements for drug stability and sterility assurance.
- U.S. Food and Drug Administration. 2024. Standards for blister packaging and high-barrier film validation in pharmaceutical manufacturing and distribution.
- International Organization for Standardization. 2023. ISO specifications for multilayer pharmaceutical packaging films and sterilization barrier properties.
- United States Pharmacopeia. 2023. Requirements for moisture and oxygen barrier films used in pharmaceutical blister and strip packaging.
- European Medicines Agency. 2024. Technical recommendations for pharmaceutical packaging film materials and regulatory compliance for drug shelf-life protection.
Frequently Asked Questions
How big is the high barrier packaging films for pharmaceuticals market in 2026?
The global high barrier packaging films for pharmaceuticals market is valued at USD 9.7 billion in 2026.
What will be the size of the high barrier packaging films for pharmaceuticals market in 2036?
The high barrier packaging films for pharmaceuticals market is projected to reach USD 22.7 billion by 2036.
How much will the high barrier packaging films for pharmaceuticals market grow between 2026 and 2036?
The high barrier packaging films for pharmaceuticals market is expected to grow at a CAGR of 8.9% between 2026 and 2036.
Which material type holds the largest share in the high barrier packaging films for pharmaceuticals market?
Metallized films lead the market with a 43% share in 2026.
Which application segment contributes the largest share in the high barrier packaging films for pharmaceuticals market?
Blister packaging dominates the market with a 48% share in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Material Type:
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Material Type: , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Material Type: , 2026 to 2036
- Metallized Films
- Aluminum Oxide Coated Films
- Silicon Oxide Coated Films
- PVDC Coated Films
- Metallized Films
- Y to o to Y Growth Trend Analysis By Material Type: , 2021 to 2025
- Absolute $ Opportunity Analysis By Material Type: , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application:
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application:, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application:, 2026 to 2036
- Blister Packaging
- Strip Packaging
- Sachet Packaging
- Pouch Packaging
- Blister Packaging
- Y to o to Y Growth Trend Analysis By Application:, 2021 to 2025
- Absolute $ Opportunity Analysis By Application:, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Material Type:
- By Application:
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type:
- By Application:
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Material Type:
- By Application:
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type:
- By Application:
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Material Type:
- By Application:
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type:
- By Application:
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Material Type:
- By Application:
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type:
- By Application:
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Material Type:
- By Application:
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type:
- By Application:
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Material Type:
- By Application:
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type:
- By Application:
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Material Type:
- By Application:
- By Country
- Market Attractiveness Analysis
- By Country
- By Material Type:
- By Application:
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type:
- By Application:
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type:
- By Application:
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type:
- By Application:
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type:
- By Application:
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type:
- By Application:
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type:
- By Application:
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type:
- By Application:
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type:
- By Application:
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type:
- By Application:
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type:
- By Application:
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type:
- By Application:
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type:
- By Application:
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type:
- By Application:
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type:
- By Application:
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type:
- By Application:
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type:
- By Application:
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type:
- By Application:
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type:
- By Application:
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type:
- By Application:
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type:
- By Application:
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type:
- By Application:
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Material Type:
- By Application:
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Material Type:
- By Application:
- Competition Analysis
- Competition Deep Dive
- Amcor plc
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Klöckner Pentaplast Group
- Tekni-Plex Inc.
- Constantia Flexibles
- Wipak Group
- Uflex Ltd.
- Mondi Group
- Cosmo Films Limited
- Toray Industries Inc.
- Mitsubishi Chemical Corporation
- Amcor plc
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Material Type: , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application:, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Material Type: , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application:, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Material Type: , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application:, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Material Type: , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application:, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Material Type: , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application:, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Material Type: , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application:, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Material Type: , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application:, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Material Type: , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application:, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Material Type: , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Material Type: , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Material Type:
- Figure 6: Global Market Value Share and BPS Analysis by Application:, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application:, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application:
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Material Type: , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Material Type: , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Material Type:
- Figure 23: North America Market Value Share and BPS Analysis by Application:, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Application:, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Application:
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Material Type: , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Material Type: , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Material Type:
- Figure 30: Latin America Market Value Share and BPS Analysis by Application:, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application:, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Application:
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Material Type: , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Material Type: , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Material Type:
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application:, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application:, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application:
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Material Type: , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Material Type: , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Material Type:
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application:, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application:, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application:
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Material Type: , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Material Type: , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Material Type:
- Figure 51: East Asia Market Value Share and BPS Analysis by Application:, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application:, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Application:
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Material Type: , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Material Type: , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Material Type:
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application:, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application:, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application:
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Material Type: , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Material Type: , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Material Type:
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application:, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application:, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application:
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

