Demand for Immunoassay CDMO in Japan
This report covers the Immunoassay CDMO demand landscape in Japan through analysis of market size, revenue forecast, competitive landscape, demand outlook, growth drivers, restraints, services trends, end user analysis, company size trends, supply chain developments, strategic growth opportunities.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Demand for Immunoassay CDMO in Japan Market Size, Market Forecast and Outlook By FMI
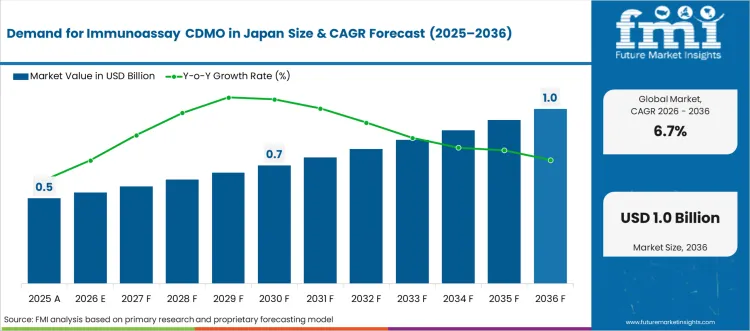
The Immunoassay CDMO in Japan market was valued at USD 0.50 billion in 2025, projected to reach USD 0.53 billion in 2026, and is forecast to expand to USD 1.02 billion by 2036 at a 6.7% CAGR. FMI analysts note that Japanese diagnostic and pharmaceutical companies are increasingly outsourcing immunoassay manufacturing to contract development and manufacturing organisations to manage rising quality compliance costs under PMDA regulatory scrutiny.
Summary of Demand for Immunoassay CDMO in Japan Market
- Market Overview
- The Immunoassay CDMO in Japan market is valued at USD 0.50 billion in 2025 and is projected to reach USD 1.02 billion by 2036.
- The industry is expected to grow at a 6.7% CAGR from 2026 to 2036, creating an incremental opportunity of USD 0.49 billion.
- The market is a quality-compliance-driven outsourcing category where PMDA regulatory scrutiny, GMP enforcement, and rising assay complexity define competitive positioning among Japanese CDMOs.
- Demand and Growth Drivers
- Demand is rising as Japanese diagnostic and pharmaceutical companies outsource immunoassay manufacturing to manage escalating quality compliance costs under PMDA oversight.
- Aging population diagnostics demand is generating sustained procurement cycles for specialised CDMO services across lateral flow, ELISA, and chemiluminescence platforms.
- Smaller biotech firms without internal manufacturing capacity rely on CDMO partnerships to meet regulatory filing timelines without capital expenditure.
- Kyushu and Okinawa lead at 8.3% CAGR, followed by Kanto at 7.7%, Kinki at 6.7%, Chubu at 5.9%, Tohoku at 5.2%, and Rest of Japan at 4.9%.
- Product and Segment View
- The market includes services, end user, company size as primary segmentation dimensions.
- Manufacturing and Production leads by Services with 53% share in 2026.
- Diagnostic Companies leads by End User with 48.2% share in 2026.
- Geography and Competitive Outlook
- Kyushu & Okinawa is the fastest-growing region at 8.3% CAGR, while Kanto anchors the largest demand base.
- Competition is shaped by technical specialisation, regulatory compliance capability, and platform integration depth, with key players including Firalis Molecular Precision, ProBio, Sanner GmbH, Biopharma Group, GTP Bioways.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant, opines: 'FMI's analysis reveals that the immunoassay cdmo in japan market is entering a phase where procurement decisions are increasingly dictated by regulatory compliance timelines and measurable performance thresholds. Organisations that delay platform investment risk permanent lockout from competitive procurement tiers their counterparts are actively securing.'
- Strategic Implications / Executive Takeaways
- Solution providers must secure certified integration pathways with tier-1 enterprise systems to qualify for forthcoming institutional procurement contracts.
- Capital project directors should phase out legacy approaches and mandate native platform compatibility in all new equipment and software procurement specifications.
- Technical teams must develop specialised competencies in services validation to demonstrate compliance parameters during the evaluation phase.
Demand for Immunoassay CDMO in Japan Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 0.53 billion |
| Industry Value (2036) | USD 1.02 billion |
| CAGR (2026 to 2036) | 6.7% |
Source: Future Market Insights, 2026
As per FMI, the convergence of aging population diagnostics demand and tightening GMP enforcement is creating sustained procurement cycles for specialised CDMO services across Japan. Smaller biotech firms without internal manufacturing capacity are structuring multi-year CDMO agreements to secure regulatory filing timelines without incurring fixed capital expenditure.
FMI is of the opinion that regional demand distribution across Japan reflects the concentration of pharmaceutical and diagnostics manufacturing infrastructure. Kyushu and Okinawa lead at 8.3% CAGR, supported by prefectural biotech cluster incentives and proximity to Asian export logistics hubs. Kanto follows at 7.7%, anchored by the concentration of diagnostic company headquarters and CDMO facilities in the greater Tokyo area. Kinki registers 6.7% growth as Osaka's pharmaceutical manufacturing base generates steady outsourcing demand. Chubu expands at 5.9%, driven by medical device manufacturers diversifying into companion diagnostic production. Tohoku and Rest of Japan maintain 5.2% and 4.9% growth respectively, with smaller biotech firms relying on CDMO partnerships to meet regulatory filing requirements. Based on FMI's report, prefectures with active biotech industrial zone designations attract disproportionate CDMO capacity expansions relative to their population share.
Demand for Immunoassay CDMO in Japan Market Definition
Based on FMI's report, the Immunoassay CDMO in Japan market covers contract development and manufacturing services for immunoassay-based diagnostic products, including ELISA kits, lateral flow assays, chemiluminescence immunoassays, and related reagent systems outsourced by diagnostic, biotech, and pharmaceutical companies operating within or supplying the Japanese market.
Demand for Immunoassay CDMO in Japan Market Inclusions
Future Market Insights analysis covers contract manufacturing and production services, assay development and optimization, formulation and stability testing, and analytical services for immunoassay products. The scope encompasses regional market sizes across Japanese prefectural groupings, forecast projections from 2026 to 2036, and segmentation by service type, end user, and company size.
Demand for Immunoassay CDMO in Japan Market Exclusions
The scope excludes in-house manufacturing by integrated diagnostic companies, contract services for non-immunoassay diagnostic platforms such as molecular diagnostics or clinical chemistry, and raw material supply without value-added development services.
Demand for Immunoassay CDMO in Japan Market Research Methodology
- Primary Research: Analysts conducted interviews with CDMO business development directors, diagnostic company outsourcing managers, and PMDA regulatory affairs consultants to map procurement decision criteria and quality compliance requirements.
- Desk Research: Data collection aggregated PMDA manufacturing inspection reports, MHLW pharmaceutical industry statistics, and published CDMO capacity expansion disclosures from listed contract manufacturers.
- Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of immunoassay CDMO contract revenues within Japan, applying end-user outsourcing penetration curves and regulatory compliance cost escalation assumptions.
- Data Validation and Update Cycle: Projections are cross-validated against publicly reported contract manufacturing revenue guidance from major listed CDMOs and quarterly outsourcing expenditure trends disclosed by Japanese diagnostic companies.
Why is the Demand for Immunoassay CDMO in Japan Growing?
The demand for immunoassay CDMO services in Japan is growing due to the increasing need for diagnostic solutions and the rising importance of personalized medicine. Immunoassays are essential for detecting and quantifying biomarkers, and with Japan’s aging population and the rise in chronic diseases, there is a greater demand for advanced diagnostic tests. CDMOs specializing in immunoassays offer critical services, including the development, manufacturing, and scaling of these tests, which are essential for detecting conditions such as cancer, cardiovascular diseases, and infectious diseases.
A significant driver behind this growth is the expansion of the biopharmaceutical industry in Japan, particularly in the field of immunotherapy and targeted treatments. As healthcare becomes more personalized, there is an increasing reliance on biomarkers to tailor treatments to individual patients. This has driven the need for high-quality immunoassays, which in turn is fueling the demand for CDMO services to support the development and manufacturing of diagnostic and therapeutic products.
Advancements in the technology used for immunoassay development, including the use of automation and data analytics, are enhancing the speed and efficiency of CDMO services. The need for compliance with rigorous regulatory standards in Japan also drives pharmaceutical companies to outsource immunoassay development and manufacturing to specialized CDMOs that have the expertise and infrastructure to meet these requirements. As Japan continues to prioritize health innovations and the growing demand for personalized medicine, the demand for immunoassay CDMO services is expected to grow steadily through 2035.
What is the Segment-Wise Analysis of Demand for Immunoassay CDMO in Japan?
Demand for immunoassay contract development and manufacturing organizations (CDMO) in Japan is segmented by services, end user, and region. By services, demand is divided into manufacturing and production, assay development and optimization, formulation and stability testing, analytical services, and others, with manufacturing and production leading at 53%. The demand is also segmented by end user, including diagnostic companies, biotech companies, and pharmaceutical companies, with diagnostic companies accounting for 48.2% of the demand. Regionally, demand is divided into Kyushu & Okinawa, Kanto, Kinki, Chubu, Tohoku, and the Rest of Japan.
Why Does Manufacturing and Production Lead the Service Demand for Immunoassay CDMO?
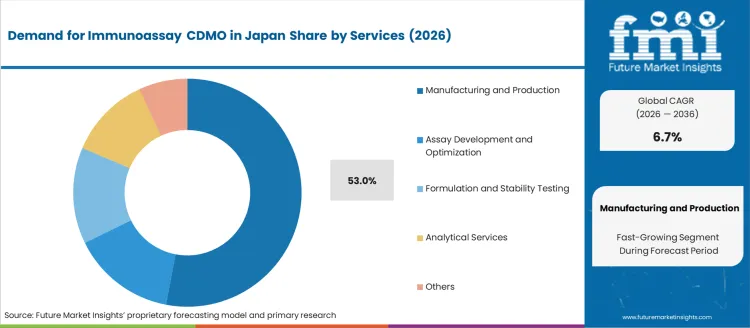
Manufacturing and production account for 53% of the demand for immunoassay CDMO services in Japan. This service is essential for the large-scale production of diagnostic assays and related products, which are in high demand due to the growing healthcare and diagnostics sector. Immunoassay products are widely used in clinical diagnostics, with applications ranging from disease detection to monitoring treatment efficacy. The demand for reliable and high-quality manufacturing services is driven by the need for consistency, regulatory compliance, and cost-effective production methods in the healthcare industry. As the need for diagnostics increases with Japan's aging population, the demand for CDMO services focused on manufacturing and production will continue to rise. Immunoassay CDMOs play a critical role in providing the necessary infrastructure and expertise for the large-scale production of diagnostic assays, ensuring their availability and quality.
How Do Diagnostic Companies Lead the End-User Demand for Immunoassay CDMO?
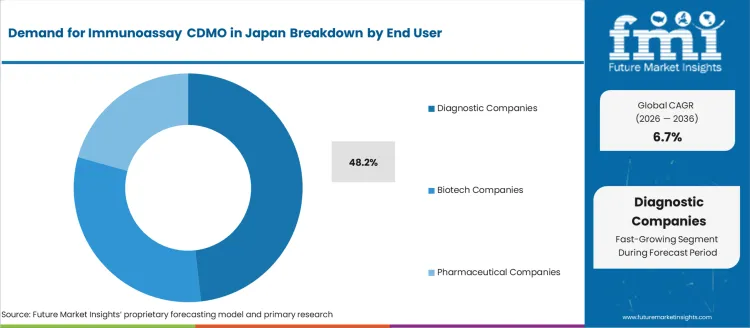
Diagnostic companies account for 48.2% of the demand for immunoassay CDMO services in Japan. These companies rely heavily on CDMOs for the development, optimization, and manufacturing of immunoassay products used in diagnostics. Immunoassays are crucial tools in clinical diagnostics, enabling the detection and quantification of biomarkers for a variety of medical conditions. The need for accurate and reliable diagnostic tests drives diagnostic companies to partner with CDMOs that can provide high-quality production and testing services. As the demand for advanced diagnostic solutions grows, especially in areas like infectious diseases, cancer, and chronic conditions, the role of CDMOs in supporting diagnostic companies with specialized services becomes increasingly critical. The continued growth of the healthcare industry, along with the rising demand for personalized medicine and point-of-care diagnostics, ensures that diagnostic companies will remain a leading end-user of immunoassay CDMO services in Japan.
What are the Key Trends, Drivers, and Restraints in Demand for Immunoassay CDMO in Japan?
In Japan, demand for immunoassay contract‑development & manufacturing services is rising alongside growth of the overall in‑vitro diagnostics (IVD) industry and increasing healthcare needs due to an aging population. As more diagnostic tests for chronic diseases, infections, cancer markers, and age‑related conditions are required, manufacturers and diagnostic firms seek CDMO partners to supply reagents, kits, and immunoassay components efficiently. Outsourcing to CDMOs lets companies avoid the high fixed costs of in‑house production while meeting regulatory and quality standards.
Why is Demand for Immunoassay CDMO Growing in Japan?
Demand for immunoassay CDMOs is growing in Japan because diagnostic firms and medical‑device companies increasingly rely on external partners to handle complex assay development, manufacturing, and regulatory compliance. As demand rises for accurate, high‑sensitivity immunoassays for early detection, health screening, chronic‑disease monitoring, and oncology diagnostics firms prefer outsourcing to specialized providers to achieve scalability, quality, and faster time‑to‑industry. The expanding elderly population, growing chronic and lifestyle‑related disease burden, and increased public‑health screening programs boost the need for large volumes of immunoassay kits, driving CDMO demand. Outsourcing also helps manage fluctuating production volumes and keeps capital burdens lower for diagnostic firms.
How are Technological and Industry Innovations Driving Growth of Immunoassay CDMO in Japan?
Technological advances in immunoassay development such as automation of assay platforms, high‑throughput manufacturing, improved reagent formulations, and multiplex testing are making CDMO services more attractive in Japan. CDMOs that invest in automated production lines and advanced process controls can deliver high‑quality, consistent immunoassays at scale, supporting large‑volume diagnostic demand. Growing complexity in diagnostics (e.g., biomarker panels, oncology markers, precision diagnostics) pushes firms to outsource to CDMOs with the necessary technical expertise and regulatory know‑how. As CDMOs expand their service portfolio from assay development and validation to mass production and quality control they become key enablers for diagnostic firms needing rapid, compliant, high‑volume output.
What are the Key Challenges Limiting Wider Adoption of Immunoassay CDMO in Japan?
Despite promising growth, adoption of immunoassay CDMO in Japan faces challenges. High capital expenditure is required to set up or upgrade facilities to meet stringent regulatory and quality standards, which may deter small‑ or medium‑sized players. Capacity limitations among existing CDMOs can lead to bottlenecks if demand surges. Regulatory compliance including validation protocols, quality‑assurance, and certification for diagnostic products adds complexity and time. Industry competition and an increasing number of CDMOs can pressure pricing and margins, possibly reducing profitability. Finally, for highly specialized or niche assays, firms may prefer in‑house development or partner with overseas CDMOs, limiting domestic growth potential.
What is the Regional Demand Outlook for Immunoassay CDMO in Japan?
.webp)
| Region | CAGR (%) |
|---|---|
| Kyushu & Okinawa | 8.3% |
| Kanto | 7.7% |
| Kinki | 6.7% |
| Chubu | 5.9% |
| Tohoku | 5.2% |
| Rest of Japan | 4.9% |
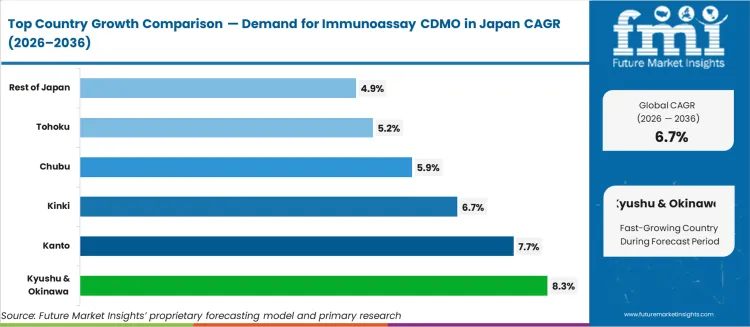
Demand for immunoassay CDMO in Japan is growing steadily, with Kyushu & Okinawa leading at an 8.3% CAGR, driven by increasing investments in pharmaceutical manufacturing and the development of contract manufacturing services. The Kanto region follows with a 7.7% CAGR, supported by its major pharmaceutical hubs and high demand for biopharmaceutical production. Kinki shows a 6.7% CAGR, fueled by strong industrial demand in the biotechnology and life sciences sectors. Chubu experiences a 5.9% CAGR, driven by regional investments in healthcare and pharmaceutical services. Tohoku and the Rest of Japan see moderate growth at 5.2% and 4.9%, respectively, as the demand for contract development and manufacturing services continues to expand in both urban and rural regions.
How is Demand for Immunoassay CDMO Growing in Kyushu & Okinawa?
Kyushu & Okinawa is experiencing the highest demand for immunoassay CDMO in Japan, with an 8.3% CAGR. This growth is primarily driven by the region’s increasing investments in pharmaceutical manufacturing and the expanding contract manufacturing sector. As part of a nationwide effort to strengthen biopharmaceutical production, Kyushu & Okinawa are receiving significant attention from pharmaceutical companies seeking to outsource their development and manufacturing services. The region’s focus on improving healthcare infrastructure and fostering pharmaceutical R&D is contributing to the rising demand for immunoassay services. The presence of pharmaceutical plants and biotechnology firms in Kyushu has further boosted the need for specialized manufacturing services. As the demand for outsourced immunoassay production increases, Kyushu & Okinawa is expected to maintain strong growth in the CDMO sector.
Why is Demand for Immunoassay CDMO Growing in Kanto?
Kanto, home to Tokyo and a major pharmaceutical hub, is witnessing significant demand for immunoassay CDMO services, with a 7.7% CAGR. The region’s pharmaceutical industry is one of the largest in Japan, with numerous global and local pharmaceutical companies located in the area. These companies rely heavily on contract development and manufacturing services to meet growing demand for immunoassay-based diagnostics and therapeutics. Kanto’s strong infrastructure and R&D focus are key drivers, as the region continuously innovates in biotechnology and biopharmaceuticals. Furthermore, the growing trend toward outsourcing production in the life sciences sector is increasing demand for immunoassay CDMO services. As the industry for biopharmaceuticals and diagnostic testing expands, Kanto’s demand for specialized manufacturing services will remain strong, contributing to its 7.7% CAGR in this sector.
What is Driving Immunoassay CDMO Demand in Kinki?
In Kinki, demand for immunoassay CDMO is growing steadily at a 6.7% CAGR, fueled by the region’s strong biotechnology and pharmaceutical sectors. Kinki, with major cities like Osaka and Kyoto, has long been a center for innovation in life sciences and pharmaceutical development. The region is home to a diverse range of pharmaceutical manufacturers, research institutions, and biotech companies, all of which drive the need for contract development and manufacturing services. As the demand for diagnostic tests and biologics rises, companies in Kinki are increasingly outsourcing their immunoassay manufacturing needs to specialized CDMOs. Furthermore, Kinki is focusing on advanced pharmaceutical R&D, which encourages the adoption of cutting-edge manufacturing processes. This combination of industrial growth and focus on R&D ensures steady demand for immunoassay CDMO services in Kinki.
How is Immunoassay CDMO Demand Growing in Chubu?
Chubu is seeing moderate growth in demand for immunoassay CDMO services, with a 5.9% CAGR. The region’s industrial base, which includes strong manufacturing and technology sectors, is driving the demand for specialized contract development and manufacturing services. Chubu’s pharmaceutical and biotechnology sectors are expanding as companies seek more efficient ways to produce immunoassays and other diagnostic products. The region’s focus on innovation in manufacturing processes and healthcare services further boosts the need for immunoassay CDMO services. Chubu’s central location in Japan makes it a key hub for logistical support, which is important for the distribution of pharmaceutical products. As pharmaceutical outsourcing continues to grow, the demand for immunoassay production services in Chubu is expected to continue at a steady pace, supported by local industry developments.
Why is Immunoassay CDMO Demand Growing in Tohoku?
Tohoku is experiencing moderate growth in immunoassay CDMO demand, with a 5.2% CAGR. The region’s pharmaceutical and biotechnology sectors are evolving, with a growing emphasis on improving healthcare infrastructure and outsourcing services to specialized manufacturers. Tohoku, traditionally known for its manufacturing and agricultural industries, is now expanding into biopharmaceuticals and diagnostic services, which is fueling demand for immunoassay CDMO. The government’s focus on promoting regional development and improving healthcare in rural areas is also contributing to the growth of the pharmaceutical outsourcing industry. As local pharmaceutical companies look to scale up production and innovate in diagnostics, demand for specialized immunoassay manufacturing services is expected to rise. Tohoku’s evolving pharmaceutical ecosystem presents opportunities for immunoassay CDMO services, driving steady demand in the region.
What is Driving Immunoassay CDMO Demand in Rest of Japan?
The Rest of Japan is seeing moderate growth in immunoassay CDMO services demand, with a 4.9% CAGR. This growth is driven by the continued expansion of pharmaceutical outsourcing in more rural and suburban areas outside the main industrial hubs. As the pharmaceutical and biotechnology industries grow in these regions, companies are increasingly turning to contract manufacturers to handle immunoassay production and other diagnostic services. The Rest of Japan’s efforts to improve healthcare infrastructure and increase access to specialized biopharmaceutical services are contributing to the rising demand. Regional manufacturers are becoming more reliant on outsourcing to meet growing needs for specialized manufacturing without making significant investments in in-house facilities. As the trend toward biopharmaceutical outsourcing continues, the demand for immunoassay CDMO services in the Rest of Japan is expected to grow steadily, though at a more moderate rate compared to more industrialized regions.
What is the Competitive Landscape of Immunoassay CDMO Demand in Japan?
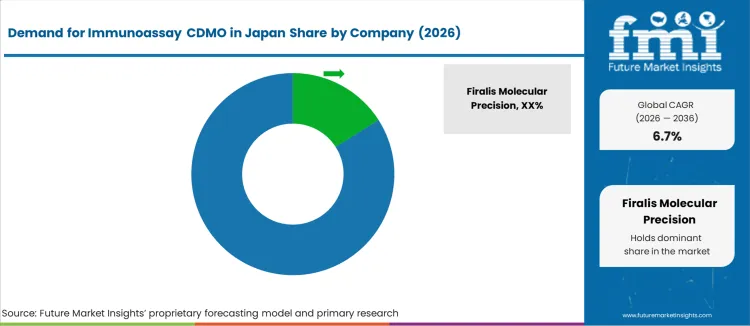
The demand for Immunoassay Contract Development and Manufacturing Organizations (CDMO) in Japan is increasing as the pharmaceutical and biotechnology sectors focus on the development of innovative diagnostics and therapeutics. Immunoassays are crucial for disease detection, monitoring, and personalized medicine, making them essential in medical diagnostics. With advancements in healthcare and the rising need for accurate, high-quality testing solutions, there is a growing need for reliable CDMOs to provide comprehensive services, from assay development to manufacturing. Japan's well-established healthcare infrastructure and growing biotech sector further contribute to the rising demand for these services.
Key players in the Immunoassay CDMO industry in Japan include Firalis Molecular Precision, ProBio, Sanner GmbH, Biopharma Group, and GTP Bioways©. Firalis Molecular Precision leads with a industry share of 15.7%, specializing in the development and manufacturing of precision immunoassays for both diagnostic and therapeutic applications. ProBio offers services in the production of immunoassay kits, focusing on high-quality, scalable manufacturing solutions. Sanner GmbH provides packaging solutions and components for immunoassays, ensuring reliable and efficient delivery of diagnostic kits. Biopharma Group offers integrated services for the production of immunoassay reagents and diagnostics, while GTP Bioways© provides custom manufacturing solutions for immunoassays, catering to both the diagnostic and research sectors.
Competition in the Immunoassay CDMO industry in Japan is driven by the growing demand for rapid, accurate, and cost-effective diagnostic solutions. The increasing focus on personalized medicine, as well as the rise in point-of-care testing, is pushing for more advanced and efficient immunoassay technologies. Companies compete by offering innovative, high-quality, and scalable manufacturing services, with a focus on regulatory compliance and customized solutions for various diagnostic needs. As Japan's healthcare industry continues to evolve, CDMOs that can provide flexibility, technical expertise, and faster turnaround times are gaining a competitive edge in the sector.
Key Players in Japan Immunoassay CDMO Demand
- Firalis Molecular Precision
- ProBio
- Sanner GmbH
- Biopharma Group
- GTP Bioways©
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 0.53 billion to USD 1.02 billion, at a CAGR of 6.7% |
| Market Definition | Based on FMI's report, the Immunoassay CDMO in Japan market covers contract development and manufacturing services for immunoassay-based diagnostic products, including ELISA kits, lateral flow assays, chemiluminescence immunoassays, and related reagent systems outsourced by diagnostic, biotech, and pharmaceutical companies operating within or supplying the Japanese market. |
| Segmentation | Services: Manufacturing and Production, Assay Development and Optimization, Formulation and Stability Testing, Analytical Services, Others; End User: Diagnostic Companies, Biotech Companies, Pharmaceutical Companies; Company Size: Small companies, Mid-size companies, Large companies |
| Regions Covered | Kyushu & Okinawa, Kanto, Kinki, Chubu, Tohoku, Rest of Japan |
| Key Companies Profiled | Firalis Molecular Precision, ProBio, Sanner GmbH, Biopharma Group, GTP Bioways |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up methodology combining installed base metrics with regulatory compliance adoption curves and primary research validation. |
Japan Immunoassay CDMO Demand by Key Segments
Services:
- Manufacturing and Production
- Assay Development and Optimization
- Formulation and Stability Testing
- Analytical Services
- Others
End User:
- Diagnostic Companies
- Biotech Companies
- Pharmaceutical Companies
Company Size:
- Small companies
- Mid-size companies
- Large companies
Region:
- Kyushu & Okinawa
- Kanto
- Kinki
- Chubu
- Tohoku
- Rest of Japan
Bibliography
- 1. Ministry of Health, Labour and Welfare. (2025). Pharmaceutical and Medical Device Act Enforcement Report. MHLW Japan.
- 2. Pharmaceuticals and Medical Devices Agency. (2025). Annual Report on CDMO and Outsourced Manufacturing Activities. PMDA.
- 3. World Health Organization. (2024). WHO Expert Committee on Biological Standardization: Technical Report Series. WHO.
- 4. International Council for Harmonisation. (2024). ICH Q5E: Comparability of Biotechnological/Biological Products. ICH.
- 5. Organisation for Economic Co-operation and Development. (2025). Health at a Glance: Pharmaceutical Sector Indicators. OECD Publishing.
- 6. Japan Pharmaceutical Manufacturers Association. (2025). Databook of Japanese Pharmaceutical Industry. JPMA.
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
Frequently Asked Questions
How large is the Immunoassay CDMO in Japan market in 2026?
The Immunoassay CDMO in Japan market is estimated to be valued at USD 0.53 billion in 2026.
What will be the market size of Immunoassay CDMO in Japan by 2036?
Market size for Immunoassay CDMO in Japan is projected to reach USD 1.02 billion by 2036.
What is the expected CAGR for Immunoassay CDMO in Japan between 2026 and 2036?
The Immunoassay CDMO in Japan market is expected to grow at a CAGR of 6.7% between 2026 and 2036.
Which Services leads the Immunoassay CDMO in Japan market in 2026?
Manufacturing and Production accounts for 53% share in 2026 by Services.
Which region is projected to grow fastest?
Kyushu & Okinawa is projected to grow at a CAGR of 8.3% during 2026 to 2036.
What does this report include in its scope?
The report covers market sizing, segmentation analysis, regional forecasts, competitive landscape evaluation, and forecast projections from 2026 to 2036.
How does FMI build and validate the Immunoassay CDMO in Japan forecast?
Forecasting models apply a bottom-up methodology starting with installed base metrics and cross-validate projections against quarterly revenue volumes and regulatory compliance adoption data.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Services
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Services , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Services , 2026 to 2036
- Manufacturing and Production
- Assay Development and Optimization
- Formulation and Stability Testing
- Analytical Services
- Others
- Manufacturing and Production
- Y to o to Y Growth Trend Analysis By Services , 2021 to 2025
- Absolute $ Opportunity Analysis By Services , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Diagnostic Companies
- Biotech Companies
- Pharmaceutical Companies
- Diagnostic Companies
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Company Size
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Company Size, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Company Size, 2026 to 2036
- Small companies
- Mid-size companies
- Large companies
- Small companies
- Y to o to Y Growth Trend Analysis By Company Size, 2021 to 2025
- Absolute $ Opportunity Analysis By Company Size, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Services
- By End User
- By Company Size
- By Country
- Market Attractiveness Analysis
- By Country
- By Services
- By End User
- By Company Size
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Services
- By End User
- By Company Size
- By Country
- Market Attractiveness Analysis
- By Country
- By Services
- By End User
- By Company Size
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Services
- By End User
- By Company Size
- By Country
- Market Attractiveness Analysis
- By Country
- By Services
- By End User
- By Company Size
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Services
- By End User
- By Company Size
- By Country
- Market Attractiveness Analysis
- By Country
- By Services
- By End User
- By Company Size
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Services
- By End User
- By Company Size
- By Country
- Market Attractiveness Analysis
- By Country
- By Services
- By End User
- By Company Size
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Services
- By End User
- By Company Size
- By Country
- Market Attractiveness Analysis
- By Country
- By Services
- By End User
- By Company Size
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Services
- By End User
- By Company Size
- By Country
- Market Attractiveness Analysis
- By Country
- By Services
- By End User
- By Company Size
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Services
- By End User
- By Company Size
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Services
- By End User
- By Company Size
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Services
- By End User
- By Company Size
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Services
- By End User
- By Company Size
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Services
- By End User
- By Company Size
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Services
- By End User
- By Company Size
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Services
- By End User
- By Company Size
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Services
- By End User
- By Company Size
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Services
- By End User
- By Company Size
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Services
- By End User
- By Company Size
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Services
- By End User
- By Company Size
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Services
- By End User
- By Company Size
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Services
- By End User
- By Company Size
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Services
- By End User
- By Company Size
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Services
- By End User
- By Company Size
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Services
- By End User
- By Company Size
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Services
- By End User
- By Company Size
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Services
- By End User
- By Company Size
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Services
- By End User
- By Company Size
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Services
- By End User
- By Company Size
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Services
- By End User
- By Company Size
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Services
- By End User
- By Company Size
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Services
- By End User
- By Company Size
- Competition Analysis
- Competition Deep Dive
- Firalis Molecular Precision
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- ProBio
- Sanner GmbH
- Biopharma Group
- GTP Bioways©
- Firalis Molecular Precision
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Services , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Company Size, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Services , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Company Size, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Services , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Company Size, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Services , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Company Size, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Services , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by Company Size, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Services , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by Company Size, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Services , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by Company Size, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Services , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by Company Size, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Services , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Services , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Services
- Figure 6: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by End User
- Figure 9: Global Market Value Share and BPS Analysis by Company Size, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Company Size, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Company Size
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Services , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Services , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Services
- Figure 26: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by End User
- Figure 29: North America Market Value Share and BPS Analysis by Company Size, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Company Size, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Company Size
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Services , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Services , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Services
- Figure 36: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by End User
- Figure 39: Latin America Market Value Share and BPS Analysis by Company Size, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Company Size, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Company Size
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Services , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Services , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Services
- Figure 46: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by End User
- Figure 49: Western Europe Market Value Share and BPS Analysis by Company Size, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by Company Size, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by Company Size
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Services , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Services , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Services
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by End User
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by Company Size, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by Company Size, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by Company Size
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Services , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Services , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Services
- Figure 66: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by End User
- Figure 69: East Asia Market Value Share and BPS Analysis by Company Size, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by Company Size, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by Company Size
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Services , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Services , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Services
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by Company Size, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by Company Size, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by Company Size
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Services , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Services , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Services
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by Company Size, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by Company Size, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by Company Size
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

