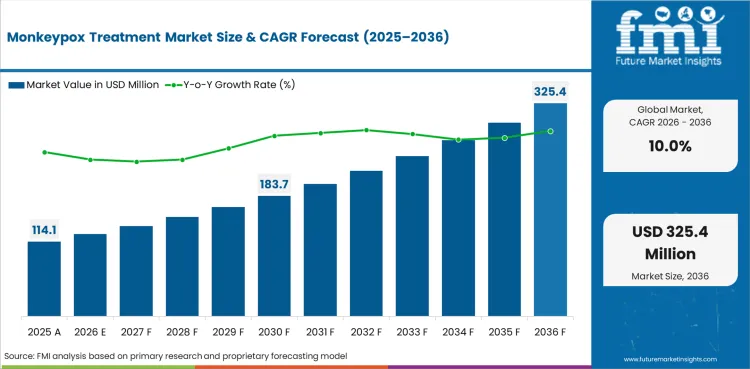
The Monkeypox Treatment Market covers therapeutic products, prophylactic vaccines, immunoglobulin therapies, and supportive care products used for the treatment and prevention of monkeypox (mpox) infections.
Market scope covers all commercially available products and services categorized by treatment type (Treatment Type, Therapeutic, Prophylactic Vaccines), administration (Administration, Oral, Injectable), technology (Technology, Pharmaceutical Therapy, Biotechnology-Based Therapy, Genetic Engineering-Based Therapy), treatment category (Treatment Category, Antiviral Medications, Vaccination, Immunoglobulin Therapy, Supportive Care), end user (End User, Hospitals, Specialty Clinics, Research Institutes), distribution channel (Distribution Channel, Hospital Pharmacies, Retail Pharmacies, Online Pharmacies). Revenue coverage spans the 2026 to 2036 forecast period.
The scope does not include general antiviral products not indicated for orthopoxvirus infections, diagnostic test kits, or infection control equipment not classified as therapeutic products.
The Monkeypox Treatment Market is growing as public health systems maintain preparedness programs following the 2022 to 2024 outbreak cycle. Therapeutic products account for 68.4% of the treatment type segment, with prophylactic vaccines contributing the preventive component. Oral administration leads at 52.0%, reflecting the preference for tecovirimat tablet formulations in outpatient management. Pharmaceutical therapy holds 46.5% of the technology segment, with biotechnology-based and genetic engineering-based therapies representing emerging modalities.
Antiviral medications account for 48.5% of the treatment category, with vaccination, immunoglobulin therapy, and supportive care serving distinct clinical roles. Hospitals lead end-user demand at 49.0%, with infectious disease clinics and research institutes contributing additional procurement volume. Hospital pharmacies hold 44.0% of distribution, consistent with the specialist prescribing and institutional dispensing patterns for orthopoxvirus treatments.
Public health stockpiling programs in the US, EU, and Japan are a structural demand driver, creating government procurement volumes that are linked to pandemic preparedness budgets rather than active case counts. Clinical trial activity for next-generation antivirals is expanding the pipeline, with several compounds in Phase I and Phase II development targeting improved efficacy and reduced side effect profiles.
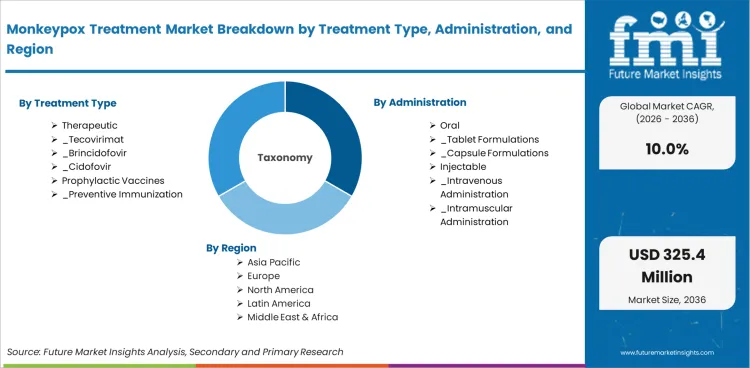
The Monkeypox Treatment Market is organized across 6 segmentation dimensions: Treatment Type, Administration, Technology, Treatment Category, End User, Distribution Channel.
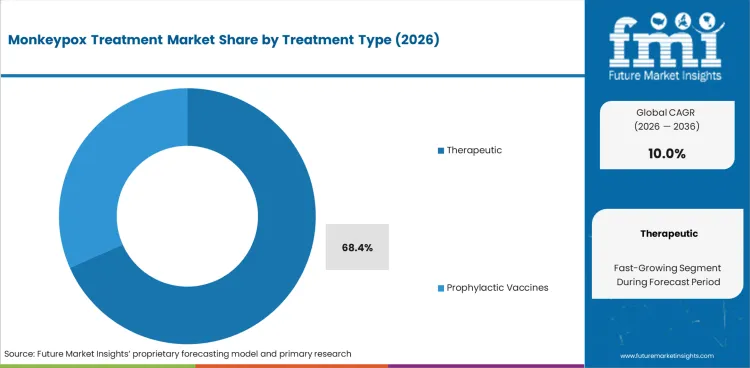
In 2026, Therapeutic is expected to account for 68.4% of the Treatment Type segment. Key sub-categories include Tecovirimat, Brincidofovir, Cidofovir.
Therapeutic holds 68.4% of the segment, contributing to segment diversity.
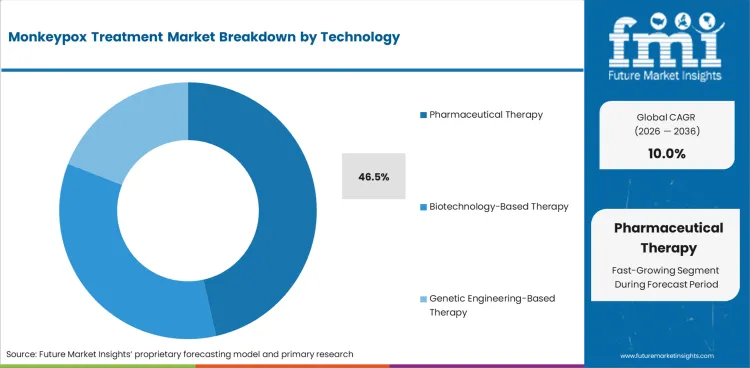
In 2026, Pharmaceutical Therapy is expected to account for 46.5% of the Technology segment. Key sub-categories include Small Molecule Drugs.
Pharmaceutical Therapy holds 46.5% of the segment, contributing to segment diversity.
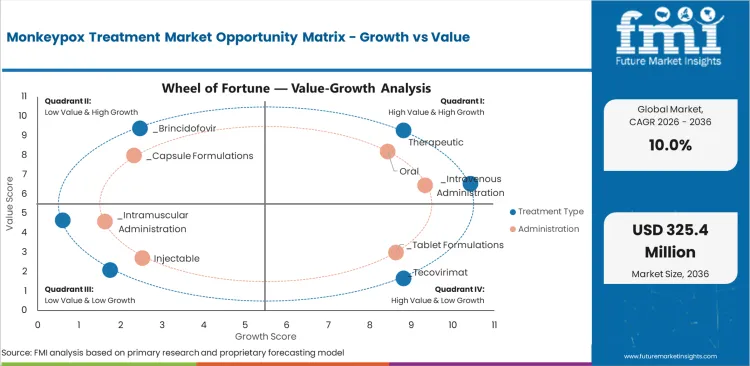
The Monkeypox Treatment Market is shaped by clinical demand, technology development, regulatory frameworks, and access dynamics. Growth reflects the balance between demand drivers and adoption constraints.
Government health agencies maintain strategic stockpiles of tecovirimat and MVA-BN vaccine as part of biodefense and pandemic preparedness programs. This procurement is budget-driven and occurs independently of active outbreak levels, providing baseline demand stability.
The monkeypox treatment pharmacopeia is narrow, with tecovirimat as the primary approved antiviral and brincidofovir and cidofovir available under emergency or compassionate use protocols. Clinical trials evaluating new antiviral compounds and combination regimens are expanding the development pipeline.
Pre-exposure vaccination for high-risk populations and post-exposure ring vaccination during outbreaks create recurring procurement demand for MVA-BN (Jynneos/Imvanex). Vaccine supply constraints and cold chain requirements influence distribution patterns across regions.
Demand for therapeutics and diagnostics is influenced by monkeypox outbreak cycles, creating volume variability. Sustained endemic transmission in certain regions and periodic outbreaks in non-endemic countries maintain ongoing clinical demand above pre-2022 levels.
.webp)
| Country | CAGR |
|---|---|
| USA | 10.5% |
| UK | 9.8% |
| Japan | 10.3% |
| EU | 10.2% |
| South Korea | 10.4% |
Source: FMI analysis based on primary research and proprietary forecasting model
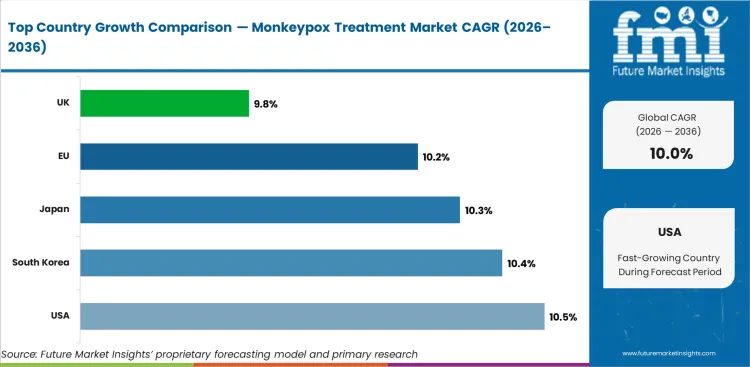
The global Monkeypox Treatment Market is projected to expand at a CAGR of 10.0% from 2026 to 2036. The analysis covers more than 30 countries.
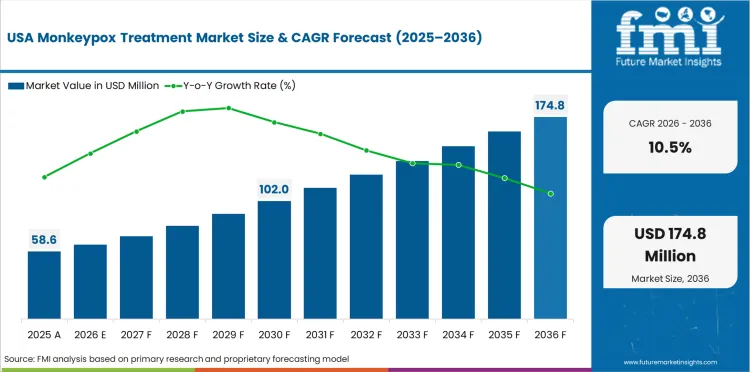
The United States is projected to grow at 10.5% through 2036. The largest biodefense stockpiling program and clinical trial infrastructure are the primary factors shaping demand.
The United Kingdom is projected to grow at 9.8% through 2036. NHS infectious disease treatment programs and public health preparedness are the primary factors shaping demand.
Japan is projected to grow at 10.3% through 2036. Biodefense procurement and advanced infectious disease treatment infrastructure are the primary factors shaping demand.
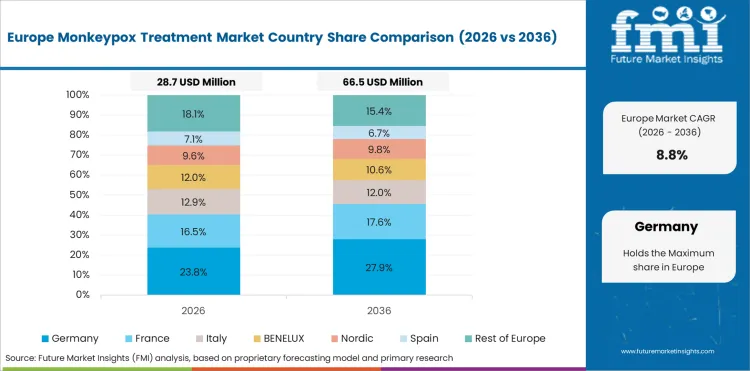
The European Union is projected to grow at 10.2% through 2036. Harmonized procurement frameworks and ECDC coordination for outbreak response are the primary factors shaping demand.
South Korea is projected to grow at 10.4% through 2036. Infectious disease preparedness investment and clinical research capacity are the primary factors shaping demand.
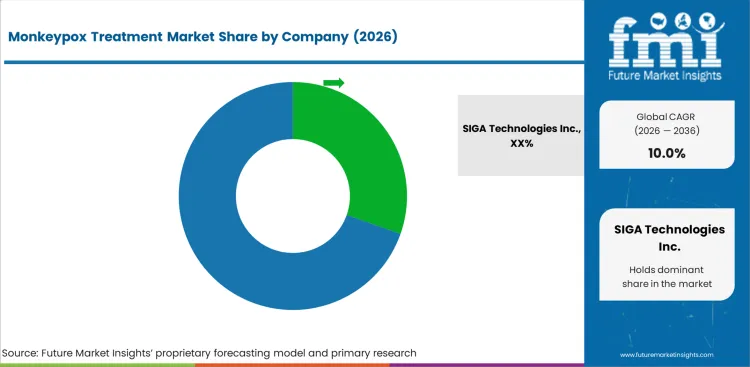
SIGA Technologies Inc. holds a leading position through its antiviral portfolio and vaccine platform. Chimerix, Inc. maintains a strong position through complementary capabilities.
Bavarian Nordic, Sanofi SA, Emcure Pharmaceuticals compete on specialized capabilities and Geographic Footprint. These companies serve distinct market segments through targeted strategies.
Market entry requires investment in antiviral portfolio, regulatory compliance, and distribution infrastructure. Established companies benefit from customer relationships and track records.
Key global companies leading the monkeypox treatment market include:
| Company | Antiviral Portfolio | Vaccine Platform | Stockpiling Contracts | Geographic Footprint |
|---|---|---|---|---|
| SIGA Technologies Inc. | High | High | Strong | Global |
| Chimerix, Inc. | High | High | Strong | Global |
| Bavarian Nordic | High | High | Strong | Global |
| Sanofi SA | Medium | High | Moderate | Regional |
| Emcure Pharmaceuticals | Medium | High | Moderate | Regional |
| Jinan Jinda Pharmaceutical Chemistry Co., Ltd. | Medium | High | Moderate | Regional |
| Emergent Pharmaceuticals | Medium | Medium | Low | Regional |
| Gilead Sciences, Inc. | Medium | Medium | Low | Regional |
| Hetero | Medium | Medium | Low | Regional |
| Piramal Pharma Solutions | Medium | Medium | Low | Regional |
Source: Future Market Insights competitive analysis, 2026.
Key Developments in Monkeypox Treatment Market
Major Global Players:
Emerging Players/Startups
| Parameter | Details |
|---|---|
| Quantitative Units | USD 125.47 million to USD 325.43 million, at a CAGR of 10.0% |
| Market Definition | The Monkeypox Treatment Market covers therapeutic products, prophylactic vaccines, immunoglobulin therapies, and supportive care products used for the treatment and prevention of monkeypox (mpox) infections. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | USA, UK, Japan, EU, South Korea, Italy, South Korea, 30 plus countries |
| Key Companies Profiled | SIGA Technologies Inc., Chimerix, Inc., Bavarian Nordic, Sanofi SA, Emcure Pharmaceuticals, Jinan Jinda Pharmaceutical Chemistry Co., Ltd., Emergent Pharmaceuticals, Gilead Sciences, Inc., Hetero, Piramal Pharma Solutions |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down methodology starting with verified data, projecting adoption velocity across segments and regions. |
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with publication dates, URLs, and supporting data for all cited works.
What is the global market demand for Monkeypox Treatment in 2026?
In 2026, the global monkeypox treatment market is expected to be worth USD 125.47 million.
How big will the Monkeypox Treatment Market be in 2036?
By 2036, the monkeypox treatment market is expected to be worth USD 325.43 million.
How much is demand expected to grow between 2026 and 2036?
Between 2026 and 2036, demand is expected to grow at a CAGR of 10.0%.
Which Treatment Type segment is expected to lead in 2026?
Therapeutic is expected to account for 68.4% of the Treatment Type segment in 2026.
What is driving demand in USA?
USA is projected to grow at 10.5% through 2036, supported by the largest biodefense stockpiling program and clinical trial infrastructure.
What is driving demand in South Korea?
South Korea is projected to grow at 10.4% through 2036, supported by infectious disease preparedness investment and clinical research capacity.
What does this report mean by 'Monkeypox Treatment Market definition'?
The Monkeypox Treatment Market covers therapeutic products, prophylactic vaccines, immunoglobulin therapies, and supportive care products used for the treatment and prevention of monkeypox (mpox) infections.
How does FMI forecast and validate the data?
Forecasting models use a hybrid bottom-up and top-down approach, starting with verified data and cross-checking against industry production data, regulatory databases, and manufacturer disclosures.
What is the global market demand for Monkeypox Treatment in 2026?
In 2026, the global monkeypox treatment market is expected to be worth USD 125.47 million.
How big will the Monkeypox Treatment Market be in 2036?
By 2036, the monkeypox treatment market is expected to be worth USD 325.43 million.
How much is demand expected to grow between 2026 and 2036?
Between 2026 and 2036, demand is expected to grow at a CAGR of 10.0%.
Which Treatment Type segment is expected to lead in 2026?
Therapeutic is expected to account for 68.4% of the Treatment Type segment in 2026.
What is driving demand in USA?
USA is projected to grow at 10.5% through 2036, supported by the largest biodefense stockpiling program and clinical trial infrastructure.
What is driving demand in South Korea?
South Korea is projected to grow at 10.4% through 2036, supported by infectious disease preparedness investment and clinical research capacity.
What does this report mean by 'Monkeypox Treatment Market definition'?
The Monkeypox Treatment Market covers therapeutic products, prophylactic vaccines, immunoglobulin therapies, and supportive care products used for the treatment and prevention of monkeypox (mpox) infections.
How does FMI forecast and validate the data?
Forecasting models use a hybrid bottom-up and top-down approach, starting with verified data and cross-checking against industry production data, regulatory databases, and manufacturer disclosures.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.