Orthopedic Consumables Market
This report provides an in-depth analysis of the orthopedic consumables market, covering market size, revenue projections, competitive landscape, demand outlook, key growth drivers, market restraints, emerging trends, supply chain dynamics, and strategic opportunities for stakeholders.
Orthopedic Consumables Market Size, Market Forecast and Outlook By FMI
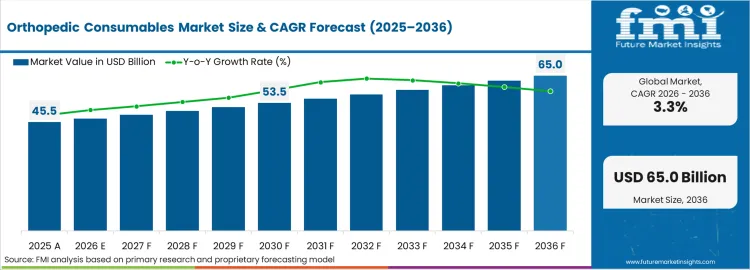
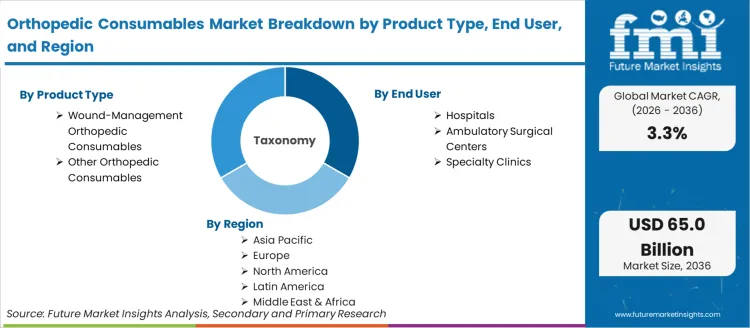
In 2025, the orthopedic consumables market was valued at USD 45.45 billion. Based on Future Market Insights' analysis, demand is estimated to grow to USD 46.95 billion in 2026 and USD 64.96 billion by 2036. FMI projects a CAGR of 3.30% during the forecast period.
Summary of Orthopedic Consumables Market
- Orthopedic Consumables Market Definition
- Orthopedic consumables are single-use and limited-reuse surgical supplies consumed during orthopedic procedures, including wound management products (surgical dressings, negative pressure wound therapy kits, hemostatic agents), casting and splinting materials, surgical drapes, arthroscopic shaver blades, suture anchors, and procedural disposables used in hospitals, ambulatory surgical centres, and specialty orthopedic clinics.
- Demand Drivers in the Market
- Orthopedic surgical procedure volumes exceeding 30 million annually across trauma, joint replacement, spine, and sports medicine generate proportional consumable consumption, with each procedure consuming USD 200 to USD 2,000 in disposable surgical supplies depending on procedure complexity.
- Single-use arthroscopic instrument adoption for infection prevention and guaranteed sharpness is converting reusable shaver blades, burrs, and graspers to disposable formats, increasing per-procedure consumable spend by 15% to 25%.
- Negative pressure wound therapy (NPWT) adoption for post-operative surgical site management in joint replacement and open fracture cases is creating a growing wound-management consumable segment with per-patient treatment costs of USD 500 to USD 3,000 over the wound healing period.
- Key Segments Analyzed in the FMI Report
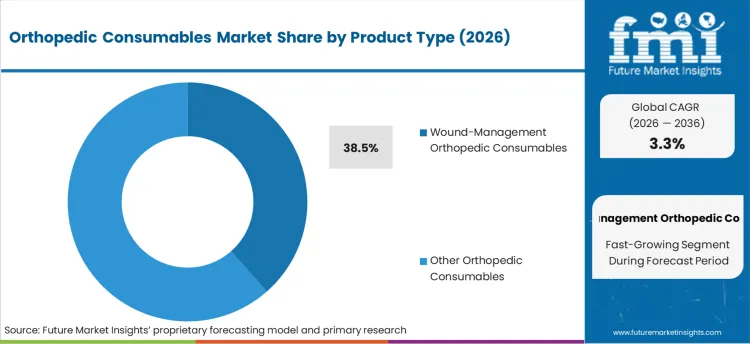
- Wound-Management Orthopedic Consumables product type: 38.5% share in 2026, reflecting its structural position as the primary category across established procurement channels.
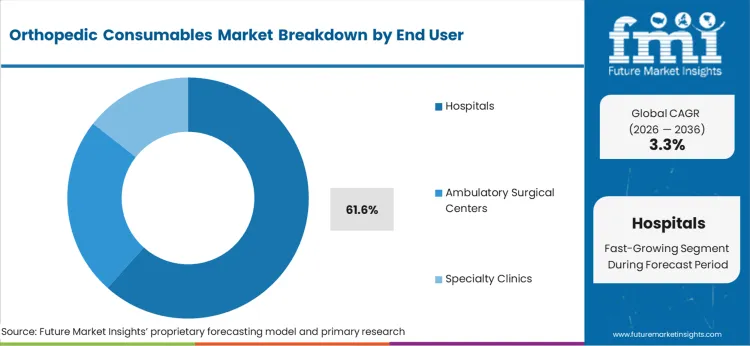
- Hospitals end user: 61.6% share in 2026, reflecting its structural position as the primary category across established procurement channels.
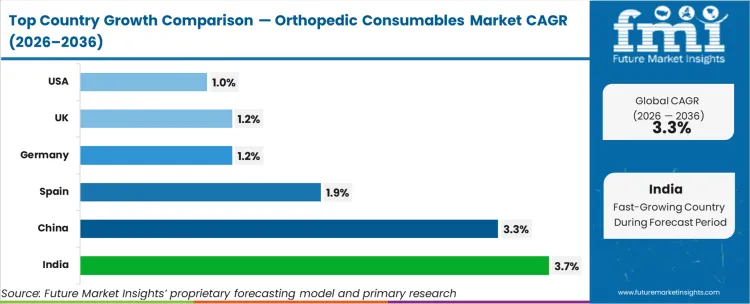
- India: 3.70% compound growth through 2036, indicating concentrated demand acceleration.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant for Healthcare analysts observe that the orthopedic consumables market is a high-volume surgical supply category where procedure volumes determine demand with minimal cyclical variation. FMI analysts observe that wound-management consumables command 38.5% product type share because surgical dressing, hemostasis, and NPWT products are consumed on every orthopedic procedure regardless of implant type or surgical approach. Hospitals at 61.6% end-user share confirms that inpatient orthopedic procedures generate the highest per-case consumable spend, though ambulatory surgical centres are growing faster in joint replacement and sports medicine volumes. The moderate 3.3% CAGR reflects the mature developed-market base where procedure volumes are growing slowly, while India (3.7%) and China (3.3%) contribute higher growth rates from expanding surgical access.
- Strategic Implications / Executive Takeaways
- Consumable suppliers should develop procedure-specific orthopedic kits bundling wound management, draping, and hemostasis products into single-SKU packages that simplify hospital procurement and reduce operating room setup time.
- Ambulatory surgical centres should negotiate volume-based consumable pricing contracts linked to annual procedure volume commitments to capture the per-unit cost reductions available from consolidated purchasing.
- Hospital value analysis committees should evaluate total cost of ownership for single-use versus reusable arthroscopic instruments, as the per-procedure sterile processing cost of reusable instruments increasingly narrows the cost gap with disposable alternatives.
The market is set to add approximately USD 18.01 billion in absolute terms between 2026 and 2036. Orthopedic surgical procedure volume growth and single-use device adoption for infection control sustain consumable demand. Each orthopedic procedure generates a defined consumable spend covering wound closure, dressing, hemostasis, and procedural disposables, creating per-procedure revenue that multiplies with surgical volume growth.
As per FMI, country-level growth rates through 2036 are projected as follows: India at 3.70%, China at 3.30%, Spain at 1.90%, Germany at 1.20%, UK at 1.20%, USA at 1.00%. India records the fastest expansion driven by concentrated institutional investment.
Orthopedic Consumables Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 46.95 Billion |
| Industry Value (2036) | USD 64.96 Billion |
| CAGR (2026 to 2036) | 3.30% |
Source: Future Market Insights, 2026
Orthopedic Consumables Market Definition
The orthopedic consumables market encompasses single-use and limited-reuse surgical supplies consumed during orthopedic procedures across all surgical subspecialties. Product categories span wound-management consumables (surgical dressings, hemostatic agents, negative pressure wound therapy kits), procedural disposables (surgical drapes, arthroscopic shaver blades, suture anchors, casting materials), and related single-use surgical supplies. FMI is of the opinion that the market is driven by orthopedic surgical procedure volume growth and the ongoing conversion from reusable to single-use surgical instruments for infection prevention, creating per-procedure consumable spend increases that compound with surgical volume expansion.
Orthopedic Consumables Market Inclusions
Market scope covers wound-management consumables (surgical dressings, NPWT kits, hemostatic agents) and other orthopedic procedural consumables (casting materials, surgical drapes, arthroscopic disposables, suture anchors) used across hospital, ambulatory surgical centre, and specialty clinic settings.
Orthopedic Consumables Market Exclusions
Durable orthopedic implants (joint prostheses, spinal hardware, fracture fixation plates), capital surgical equipment (power tools, navigation systems), and rehabilitation devices (braces, orthotics) are excluded.
Orthopedic Consumables Market Research Methodology
- Primary Research: Analysts conducted structured interviews with procurement managers, product engineers, and distribution channel operators to map purchase decision triggers and specification requirements across key verticals.
- Desk Research: Data collection aggregated regulatory filings, industry standards documentation, patent registries, trade body publications, and company annual reports across all target geographies.
- Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of product shipment volumes and average selling prices, validated against publicly disclosed revenue figures from leading suppliers.
- Data Validation and Update Cycle: Projections are cross-referenced against quarterly earnings data, import-export trade statistics, and industry association production reports to maintain forecast integrity.
Key Industry Highlights
The Rising Prevalence of Osteoarthritis is Significantly Contributing to the Growing Demand for Orthopedic Consumables across Healthcare Markets.
One of the major factors driving the market growth is the rising prevalence of musculoskeletal conditions like osteoarthritis across the globe. The incidence of osteoarthritis is higher among older age and female patient populations.
In 2019, according to the WHO 2023, 528 million people were living with osteoarthritis across the world, with an increase of 113% from 1990. About 73% of the symptomatic people were aged over 55 years and 60% of them were females. Out of all the joints affected, the knee was affected the most joint out of all three osteoarthritis knees, hip, and hand, with a total of 365 million patients.
Also, symptomatic knee [183 million] osteoarthritis affected more people than hip [115 million] and hand [38 million] symptomatic sites. Continuing the same trend, the patient pool in the future elderly is set to increase and is expected to increase the need for successful therapeutic measures, thus benefiting the orthopedic consumables market.
Given the above-mentioned burden of the active adult osteoarthritis population and the pain control issues, many people would ultimately get surgery, hence elevating demand in the overall demand for orthopedic consumables. Therefore, the Global Osteoarthritis Burden drives the growth of the orthopedic consumables market.
Growing Demand for Single-Use Solutions Presents Market Opportunity in Orthopedic Consumables
The orthopedic consumables market is set to have substantial growth potential as a result of the greater emphasis on single-use devices. Products such as these provide increased safety and convenience to healthcare providers and patients alike.
A greater emphasis on infection control and the elimination of cross-contamination risk has cemented single-use consumables as a favorable solution during surgical procedures. This paradigm shift is vital in orthopedic surgeries due to the importance of sterile conditions to ensure a favorable patient prognosis.
The potential for demand for single-use consumables and new entrants into the orthopedic consumables space represent important market opportunities. Several manufacturers see the market opportunity for developing disposable consumables that are constructed to the required standards of safety within modern health care.
The trend toward single-use sterile surgical supplies is also fostering new economic and product innovations as manufacturers are scorning the balance between reliability and low-cost consumables in terms of construction to the needs of orthopedic surgeons and surgical centers. Healthcare systems are expected to favor single-use orthopedic consumables, driving firms to develop and customize these devices for orthopedic surgeries.
Resorbable and Biocompatible Materials Transforming and Creating New Opportunities for the Orthopedic Consumables Market
A notable new market opportunity for orthopedic consumables exists in resorbable and biocompatible materials. Due to advancements in biomaterials, there is a growing interest in consumables including screws, pins, and plates that would degrade and be replaced by tissue once they have fulfilled their mechanical function.
This is especially critical to orthopedic surgeries in pediatric and young patients, as these devices would otherwise need to be removed via a second surgery if they were made from metal.
Resorbable materials lead to better patient outcomes while decreasing healthcare costs due to the non-necessity for secondary procedures.
The trend is supported by materials science breakthroughs in the field of poly(α-hydroxy acid) polymers, as many of the polymeric resorbable materials used nowadays, such as polylactic acid (PLA) and polyglycolic acid (PGA) have been shown to possess excellent biocompatibility, mechanical strength, and controlled degradation rates.
Moreover, resorbable soft tissue orthopedic devices such as screws, pins, and plates meet society's demand for more sustainable healthcare options for improved environmental health and diminished long-term use of non-degradable metals. The developers of the medical orthopedic consumption market with traditional resorbable materials are faced with a combination of clinical, economic, and sustainable benefits.
Regulatory and Compliance as Major Barrier to Growth in the Orthopedic Consumables Market
A significant constraint within the orthopedic consumables market is the increasing regulatory uncertainty often arising from the complex and rapidly evolving regulatory framework related to product approval and/or appeals, and compliance with international standards.
Orthopedic implants and consumables are multisystem, high risk devices, often utilizing novel materials, or complex designs. In short, they are often defined by complex manufacturing processes that require companies to conform to rigid regulatory requirements dictated by extension groups such as the USA FDA, the European Medicines Agency (EMA) or other global co-contributors.
For example, the FDA classifies many orthopedic consumables within the Class III device scope, thus they effectively require premarket approval that's typically supported by significant clinical data and strong evidence of benefit/risk ratios, which can cause significant delays in product approval.
This issue is compounded by variable regulatory standards across regions and markets. Organizations also have to comply with different rules of compliance depending on the market numerous players find this to cause further delays for new products and market uptake.
Moreover, once commercially available if an organization fails to comply, or there is a notifiable incident following a commercial sale, organizational recalls or other forms of litigation response can be overwhelming.
Therefore, these bespoken regulatory burdens are often difficult and burdensome for players involved in orthopedic consumables market and do create significant barriers to entry and/or long-term market countries in new orthopedic consumables.
2021 to 2025 Global Orthopedic Consumables Sales Outlook Compared to Demand Forecast from 2026 to 2036
Over time, the orthopedic consumables marketplace has undergone structural change, like many of the other areas of health care due to rising healthcare costs, increasing aging and demographic change, and advances in technology. Advances in materials and technology in consumable products (suture anchors, plates, screws, etc.) are designed and manufactured have driven the biggest change in orthopedic treatment.
The line between traditional surgical techniques and modern surgical techniques used for care is rapidly diminishing, as developers integrate biocompatible and resorbable substrates into previously surgical video-guided methods, to provide a safer means of care for patients.
Many advances in the surgical field utilize roboskills, like three-dimensional printing, to produce tailored implants that resolve surgical conditions that were challenging to address. Additionally, we have seen an increase in the use of minimally invasive surgical techniques that, in tandem with the advanced substrates, have increased the demand for consumable products that are more sophisticated and encompass simpler designs in all variables of surgical delivery.
Increased awareness of injuries related to sports, especially in youth, has added to a rise in the utilization of orthopedic consumables related to reconstructive surgeries.
The orthotic peripheral marketplace has also observed an emphasis on sustainable, cost-effective products with minimal direct post-surgical use. Development of cost-effective orthopedic products and consumables has accelerated to replace the need for alternate or post-operative surgical procedures, enabling care systems to find reduced costs.
This is related to regulatory pathways that have seen heightened levels of administrative files with exit documents for safety, biocompatibility, and effectiveness six months to two years later also reducing other alternate care commitments.
The globalization of healthcare practices has also provided opportunities for access and use in multiple arenas of healthcare an organized orthopedic marketplace; however, new opportunities have also confirmed challenges with regional differences with policy changes and developing and providing regulation of burgeoning global practice potentials and healthcare infrastructure issues.
Market Concentration
Tier 1 companies are the major companies as they hold a 25.7% share worldwide. These companies have large product portfolios and a high production capacity. Companies with the diverse portfolio also has global presence along with strong research and development capabilities.
Their vast range of applications and innovative solutions help them maintain industry leadership. Prominent companies within tier 1 include ConMed Corporation, Stryker Corporation and Johnson & Johnson
Tier 2 companies are relatively smaller as compared with tier 1 players. The tier 2 companies hold a share of 29.7% worldwide. Firms in this tier offer a wide range of orthopedic consumables, with a particular emphasis on minimally invasiveness. Their devotion to innovation and diverse product lines contributes to their prominent position. Key Companies under this category include Zimmer Biomet, Medtronic plc., 3M Company, and DSM Biomedical.
Compared to Tiers 1 and 2, Tier 3 companies offer orthopedic consumables, but with smaller revenue spouts and less influence. These companies mostly operate in one or two countries and have a limited customer base. Tier 3 companies, although, smaller are essential for the market. The companies such as Wright Medical Group, Inc., Bio Tek Instruments, Inc. and other fall under this category.
Country-wise Insights
The section below covers the industry analysis for the orthopedic consumables market for different countries. Market demand analysis on key countries in several regions of the globe, including North America, Asia Pacific, Europe, and others, is provided. The United States is anticipated to remain at the forefront in North America, with a CAGR of 1.0% through 2036. In Asia Pacific, India is projected to witness the highest CAGR in the market of 3.7% by 2036.
.webp)
| Countries | Value CAGR (2026 to 2036) |
|---|---|
| USA | 1.0% |
| Germany | 1.2% |
| India | 3.7% |
| UK | 1.2% |
| China | 3.3% |
| Spain | 1.9% |
Advancements in Orthopedic Surgery Driving Market Growth in USA
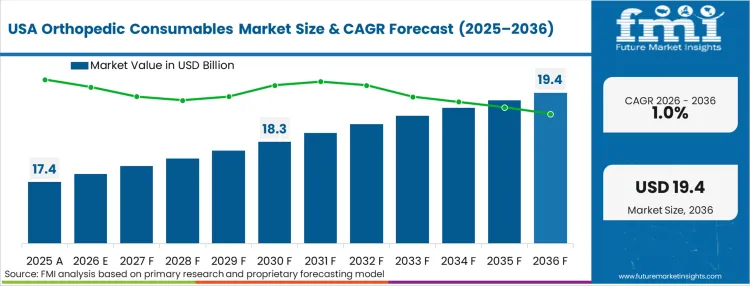
The USA dominates the global market with high share in 2026. The USA is expected to exhibit a CAGR of 1.0% throughout the forecast period (2026 to 2036).
The United States encompasses a considerable portion of the global orthopedic consumables, propelled by the increasing adoption of sophisticated orthopedic surgeries, particularly interventional procedures. A foremost catalyst of this trend is the prominently developed healthcare infrastructure that engenders the development of advanced surgical procedures using robotic-assisted and minimally invasive procedures.
The USA healthcare system is distinguished by relatively broad-based access to advanced specialty orthopedic care, which is highly localized in leading hospital and surgical center markets, allowing the surgical community to use advanced solutions to musculoskeletal disorders of bone, joint, and spine.
Orthopedic surgeons are becoming more and more receptive to interventional orthopedic surgeries and procedures, like arthroscopy and spine interventions, because of the positive results, which include shorter recovery times following surgery, lower rates of morbidity from complications, and improved outcomes over time. This leads to an increased demand for consumables, like sutures, bone graft alternatives, and fixation devices.
Rising Adoption of Disposable Devices Fueling Growth in Germany
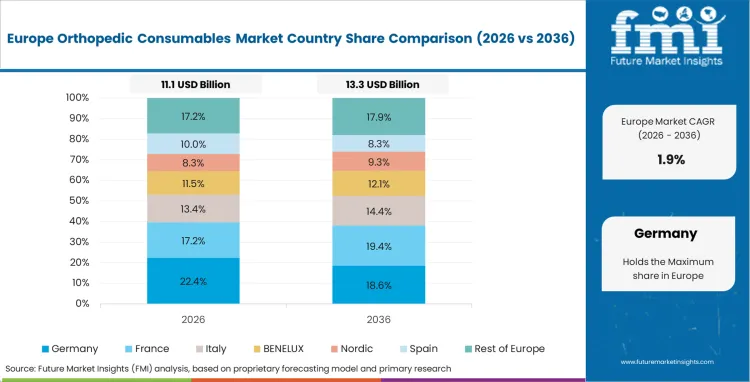
In 2026, Germany held a dominant revenue in the Western Europe market and is expected to grow with a CAGR of 1.2%. Germany is driven mainly by an emphasis on the use of disposable devices and systems in surgical procedures.
The increased acceptance of disposable devices in surgical procedures such as single-use sutures, sterile drapes, and wound care materials has developed based on considerations for infection control, operational efficiency, and cost-effective measures for orthopedic surgeries.
Disposable devices offer hospitals less risk of cross-contamination during the surgery, which provides for reduced length of stay and eliminates the need for sterilization, which is in alignment with Germany's diligent protocols on safety in health care. Due to the shorter recovery times and decreased complication rates associated with these less invasive techniques, orthopedic surgeons in Germany are increasingly employing consumables made for them.
Germany's orthopedic consumables market is anticipated to grow quickly as demand increases for safer and more effective surgical treatments, especially in the areas of trauma and reconstructive procedures.
Growing Sales Fueled by Local Manufacturing and Increased Usage in China
China occupies a leading value share in East Asia market in 2026 and is expected to grow with a CAGR of 3.3% during the forecasted period.
China's orthopedic consumables market is growing rapidly due to access to healthcare and increasing orthopedic product applications within surgery. One of the most influential factors is the strong domestic manufacturing base. Chinese manufacturers produce continuously developed, high-quality consumables at ideological, competitive prices.
Certain local companies are satisfying domestic demand and taking the next steps to develop cheaper alternatives to imported consumables in the global market. As a consequence, product acceptance has been widespread throughout Chinese hospitals and clinics, especially in regions that are experiencing a rise in orthopedic case volumes.
Healthcare infrastructure is expanding, supported by the government, leading to an increase in orthopedic surgery -specifically joint replacements and trauma cases. Increased cases of orthopedic surgeries are evident with an aging population, subsequent to increasing sports injuries and degenerative conditions. In addition, China's continued demand and prevalent manufacturing capacity facilitate the orthopedic consumables to rise.
Category-wise Insights
The section contains information about the leading segments in the industry. Based on product type, the wound-management orthopedic consumables is expected to account 38.5% of the global share in 2024.
Surge in Demand for Wound-Management Orthopedic Consumables due to its Enhancing Post-Operative Care and Patient Outcomes.
| Product Type | Wound-Management Orthopedic Consumables |
|---|---|
| Value Share (2026) | 38.5% |
By product type, wound-management orthopedic consumables is expected to hold a share of 38.5% in 2026. Wound-management orthopedic consumables are leading in the market, as these products are critical to post-operative care. These products act by preventing infections, controlling bleeding, and offering rapid healing along with considerably reducing the scarring post-orthopedic surgeries.
Antimicrobial dressings along with advanced wound care technology including bioactive products such as hydrogels have considerably enhanced the patient outcome, thus influencing the demand for them. With the growing complexity of orthopedic procedures, there is increasing demand for individualized trauma and elective surgery consumables, attributed to their higher demand as compared to other product types.
Laboratory Devices is expected to be a dominant segment in terms of Value
| By End User | Hospitals |
|---|---|
| Value Share (2026) | 61.6% |
Hospitals will account for 61.6% of the indication segment in 2026, and exhibit the highest CAGR in the forecast period. Hospitals would continue to lead the end user segment as they perform a considerable number of orthopedic surgeries in their facilities. Hospitals offer all-encompassing care with a well-developed infrastructure, modern technological tools, and effective treatment due to the presence of skilled surgeons and high patient inflow.
Hospitals also procure new consumables through their procurement facilities post-approval from respective authority for use in surgeries, thus contributing to a larger share for consumables enterprises. Significant numbers of trauma centers and elective surgery centers are based in the hospital premises which also drive the demand for orthopedic consumables.
Competitive Landscape
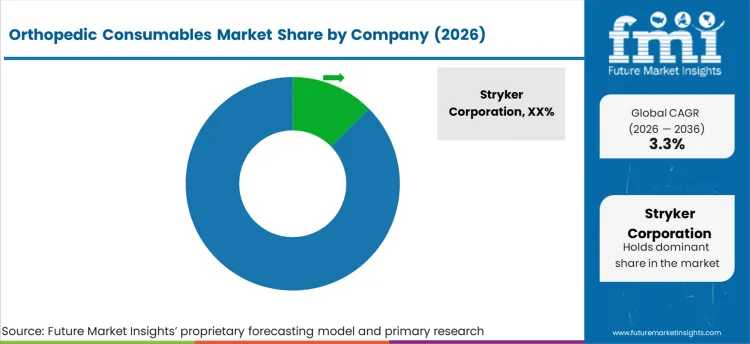
The orthopedic consumables market is highly competitive, with key players like Stryker, Zimmer Biomet, and Johnson and Johnson dominating through product innovation, mergers, and acquisitions. Companies focus on advanced technologies, such as robotic-assisted surgeries, and expanding portfolios to meet the growing demand for minimally invasive procedures and joint replacements.
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 46.95 Billion to USD 64.96 Billion, at a CAGR of 3.30% |
| Market Definition | Orthopedic consumables are single-use and limited-reuse surgical supplies consumed during orthopedic procedures, including wound management products (surgical dressings, negative pressure wound therapy kits, hemostatic agents), casting and splinting materials, surgical drapes, arthroscopic shaver blades, suture anchors, and procedural disposables used in hospitals, ambulatory surgical centres, and specialty orthopedic clinics. |
| Product Type Segmentation | Wound-Management Orthopedic Consumables, Other Orthopedic Consumables |
| End User Segmentation | Hospitals, Ambulatory Surgical Centers, Specialty Clinics |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | India, China, Spain, USA, Germany, UK, and 40 plus countries |
| Key Companies Profiled | Stryker Corporation, ConMed Corporation, Johnson & Johnson, Zimmer Biomet, Medtronic plc., 3M Company, DSM Biomedical, Wright Medical Group, Inc., Bio Tek Instruments, Inc. |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down modeling validated against primary research inputs and publicly available industry data. |
Key Segments of Orthopedic Consumables Industry
By Product Type:
In terms of service, the industry is divided into wound-management orthopedic consumables, surgical orthopedic consumables and others.
By End User:
In terms of end user, the industry is segregated into hospitals, ambulatory surgical centers, clinics and others
By Region:
Key countries of North America, Latin America, East Asia, South Asia & Pacific, Western Europe, Eastern Europe Middle East and Africa (MEA) have been covered in the report.
Bibliography
- [1] American Academy of Orthopaedic Surgeons. (2024). AAOS Orthopaedic Procedure Volume and Consumable Utilization Report. AAOS.
- [2] Stryker Corporation. (2024). Annual Report 2024: MedSurg and Neurotechnology Division. Stryker.
- [3] 3M Company. (2024). Medical Solutions Division: Surgical Consumables Product Portfolio. 3M.
- [4] Association for the Advancement of Medical Instrumentation. (2024). AAMI TIR12: Designing, Testing, and Labeling Reusable Medical Devices. AAMI.
- [5] World Health Organization. (2024). Global Guidelines on Prevention of Surgical Site Infection. WHO.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary research documentation.
Frequently Asked Questions
How large is the demand for Orthopedic Consumables in the global market in 2026?
Demand for Orthopedic Consumables in the global market is estimated to be valued at USD 46.95 billion in 2026.
What will be the market size of Orthopedic Consumables by 2036?
Market size for Orthopedic Consumables is projected to reach USD 64.96 billion by 2036.
What is the expected demand growth between 2026 and 2036?
Demand is expected to grow at a CAGR of 3.30% between 2026 and 2036.
Which Product Type is poised to lead by 2026?
Wound-Management Orthopedic Consumables accounts for 38.5% in 2026.
How significant is Hospitals in driving adoption?
Hospitals represents 61.6% of segment share in 2026.
What country records the fastest growth?
India is projected to grow at a CAGR of 3.70% during 2026 to 2036.
What is the projected growth for China?
China is projected to expand at a CAGR of 3.30% during 2026 to 2036.
What is included in the scope of this report?
Market scope covers wound-management consumables (surgical dressings, NPWT kits, hemostatic agents) and other orthopedic procedural consumables (casting materials, surgical drapes, arthroscopic disposables, suture anchors) used across hospital, ambulatory surgical centre, and specialty clinic settings.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Wound-Management Orthopedic Consumables
- Other Orthopedic Consumables
- Wound-Management Orthopedic Consumables
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- Ambulatory Surgical Centers
- Specialty Clinics
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By End User
- Competition Analysis
- Competition Deep Dive
- Stryker Corporation
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- ConMed Corporation
- Johnson & Johnson
- Zimmer Biomet
- Medtronic plc.
- 3M Company
- DSM Biomedical
- Wright Medical Group, Inc.
- Bio Tek Instruments, Inc.
- Stryker Corporation
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by End User
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Product Type
- Figure 23: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by End User
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Product Type
- Figure 30: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by End User
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by End User
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by End User
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Product Type
- Figure 51: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by End User
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

