Orthopedic Contract Manufacturing Market
This report presents a comprehensive study of the orthopedic contract manufacturing market, examining market size, revenue forecast, competitive landscape, demand trends, growth drivers, restraints, technological developments, supply chain analysis, and future growth opportunities across key regions.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Orthopedic Contract Manufacturing Market Size, Market Forecast and Outlook By FMI
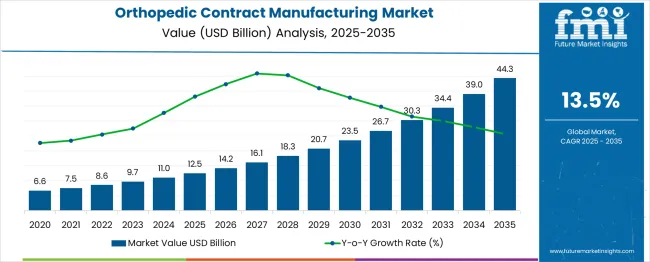
In 2025, the orthopedic contract manufacturing market was valued at USD 12.50 billion. Based on Future Market Insights' analysis, demand is estimated to grow to USD 14.13 billion in 2026 and USD 47.95 billion by 2036. FMI projects a CAGR of 13.00% during the forecast period.
Summary of Orthopedic Contract Manufacturing Market
- Orthopedic Contract Manufacturing Market Definition
- Orthopedic contract manufacturing encompasses outsourced production services for orthopedic implants (joint prostheses, spinal hardware, trauma fixation devices), surgical instruments, sterilization cases, and instrument trays, provided by specialized contract manufacturers offering forging, casting, CNC machining, finishing, coating, and assembly capabilities to orthopedic OEM device companies.
- Demand Drivers in the Market
- Orthopedic OEM outsourcing acceleration, with an estimated 40% to 50% of implant production now contracted externally, is driven by capital expenditure avoidance, capacity flexibility, and the regulatory compliance burden of maintaining FDA-registered and ISO 13485-certified manufacturing facilities.
- Additive manufacturing (3D printing) adoption for porous-coated implant surfaces and patient-specific instruments is creating demand for contract manufacturers with qualified metal AM (EBM, DMLS) production capability that most orthopedic OEMs have not yet built internally.
- Surgical instrument set complexity growth, with each new robotic-assisted surgery platform requiring dedicated instrument trays containing 40 to 80 specialized tools per set, is generating instrument contract manufacturing demand that scales with robotic surgery system installation base growth.
- Key Segments Analyzed in the FMI Report
- Implants type: 64.0% share in 2026, reflecting its structural position as the primary category across established procurement channels.
- Forging/Casting service: 33.0% share in 2026, reflecting its structural position as the primary category across established procurement channels.
- China: 18.20% compound growth through 2036, indicating concentrated demand acceleration.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant for Healthcare analysts observe that the orthopedic contract manufacturing market is an outsourced production services category experiencing accelerated growth as OEM outsourcing ratios increase and manufacturing technology complexity rises. FMI analysts observe that implants command 64.0% type share because implant production generates the highest per-unit contract manufacturing revenue due to the precision machining, surface finishing, and quality documentation requirements that differentiate medical device manufacturing from general industrial machining. Forging/casting at 33.0% service share leads as the primary near-net-shape forming operation for titanium and cobalt-chrome implant blanks. China leads at 18.2% CAGR because domestic orthopedic OEMs entering the market are outsourcing manufacturing rather than building vertically integrated production facilities, creating concentrated contract manufacturing demand.
- Strategic Implications / Executive Takeaways
- Contract manufacturers must invest in qualified metal additive manufacturing (EBM, DMLS) production lines with FDA-registered and ISO 13485-certified quality systems to capture the growing porous implant surface and patient-specific instrument segment.
- Orthopedic OEMs should consolidate contract manufacturing relationships to 2 to 3 qualified suppliers per product family to balance supply chain risk mitigation against the procurement efficiency of reduced vendor management complexity.
- Contract manufacturers serving the Chinese orthopedic OEM segment should pursue NMPA device master file registrations that enable their manufacturing sites to serve as qualified production facilities for multiple OEM customers simultaneously.
The market is set to add approximately USD 33.82 billion in absolute terms between 2026 and 2036. Orthopedic OEM outsourcing acceleration driven by manufacturing capacity constraints, capital expenditure avoidance, and regulatory compliance complexity sustains contract manufacturing growth. The shift toward personalized implants, additive manufacturing, and robotic-assisted surgery instrument sets is creating demand for contract manufacturers with advanced capabilities that mid-size OEMs cannot economically maintain in-house.
As per FMI, country-level growth rates through 2036 are projected as follows: China at 18.20%, India at 16.90%, Germany at 15.50%, France at 14.20%, UK at 12.80%, USA at 11.50%, Brazil at 10.10%. China records the fastest expansion driven by concentrated institutional investment.
Orthopedic Contract Manufacturing Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 14.13 Billion |
| Industry Value (2036) | USD 47.95 Billion |
| CAGR (2026 to 2036) | 13.00% |
Source: Future Market Insights, 2026
Orthopedic Contract Manufacturing Market Definition
The orthopedic contract manufacturing market encompasses outsourced production services for medical device companies requiring specialized manufacturing capabilities for orthopedic implants, surgical instruments, and related procedural hardware. Service categories span primary forming (forging, casting, metal injection molding), precision machining (multi-axis CNC, Swiss turning, EDM), surface finishing (polishing, blasting, plasma spraying), coating application (hydroxyapatite, titanium plasma spray, PVD), and final assembly with sterilization packaging. FMI is of the opinion that the market is in an outsourcing acceleration phase where OEM manufacturing insourcing ratios are declining as technology complexity (additive manufacturing, robotic instrument sets) and regulatory compliance costs (ISO 13485, FDA registration) favour specialized contract manufacturers.
Orthopedic Contract Manufacturing Market Inclusions
Market scope covers contract manufacturing services for orthopedic implants, surgical instruments, sterilization cases, and instrument trays, including forging/casting, precision CNC machining, surface finishing, coating application, and final assembly provided to orthopedic OEM companies.
Orthopedic Contract Manufacturing Market Exclusions
In-house manufacturing by vertically integrated orthopedic OEMs (Stryker, Zimmer Biomet, J&J DePuy Synthes), general industrial contract machining not qualified for medical device production, and orthopedic product design and R&D services sold independently are excluded.
Orthopedic Contract Manufacturing Market Research Methodology
- Primary Research: Analysts conducted structured interviews with procurement managers, product engineers, and distribution channel operators to map purchase decision triggers and specification requirements across key verticals.
- Desk Research: Data collection aggregated regulatory filings, industry standards documentation, patent registries, trade body publications, and company annual reports across all target geographies.
- Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of product shipment volumes and average selling prices, validated against publicly disclosed revenue figures from leading suppliers.
- Data Validation and Update Cycle: Projections are cross-referenced against quarterly earnings data, import-export trade statistics, and industry association production reports to maintain forecast integrity.
Why is the Orthopedic Contract Manufacturing Market Growing?
Market expansion is being supported by the increasing demand for specialized manufacturing expertise and the corresponding need for cost-effective production solutions among orthopedic device manufacturers. Modern medical device companies are increasingly focused on core competencies such as research and development, while outsourcing complex manufacturing processes to specialized contract manufacturers. The proven expertise of contract manufacturers in precision machining, advanced materials processing, and regulatory compliance makes them preferred partners for orthopedic device production.
The growing emphasis on quality assurance and regulatory compliance is driving demand for contract manufacturing services that maintain the highest standards of medical device production. Customer preference for comprehensive manufacturing solutions that combine multiple services including forging, machining, finishing, and assembly is creating opportunities for integrated service providers. The rising influence of cost pressures and the need for scalable manufacturing capacity is also contributing to increased adoption of contract manufacturing services across different orthopedic device categories and market segments.
Segmental Analysis
The market is segmented by type outlook, service outlook, company, and region. By type outlook, the market is divided into implants, instruments, cases, trays, and others. Based on service outlook, the market is categorized into forging/casting, machining & finishing, and other specialized services. The market includes leading companies such as Tecomet Inc., Orchid Orthopedic Solutions, Cretex Companies, Viant, ARCH Medical Solutions Corp., Avalign Technologies, LISI Medical, Paragon Medical, Norman Noble, Inc., and Autocam Medical. Regionally, the market is divided into North America, Europe, East Asia, South Asia & Pacific, Latin America, and Middle East & Africa.
By Type Outlook, Implants Segment Accounts for 64% Market Share
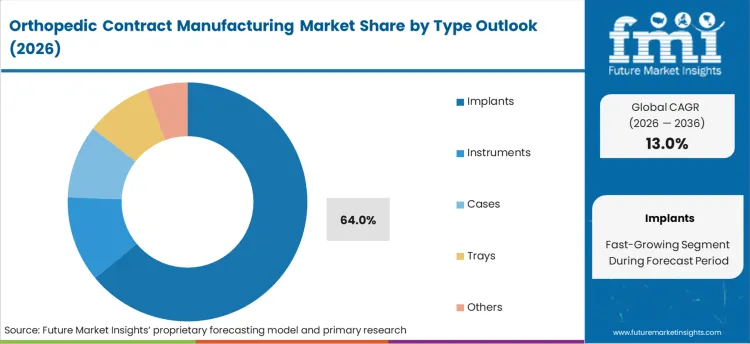
The implants segment is projected to account for 64% of the orthopedic contract manufacturing market in 2026, reaffirming its position as the category's core component. Healthcare providers increasingly understand the critical importance of high-quality implant manufacturing in patient outcomes and long-term device performance. The complexity of implant manufacturing, including precision requirements, biocompatibility standards, and regulatory compliance, makes contract manufacturing an attractive option for many orthopedic device companies.
This segment forms the foundation of most contract manufacturing relationships, as it represents the most technically demanding and quality-critical aspect of orthopedic device production. Surgeon preferences and ongoing clinical validation continue to strengthen demand for precision-manufactured implants. With healthcare systems focusing more on patient outcomes and device longevity, implant manufacturing quality aligns with both therapeutic and economic healthcare goals. Its broad appeal across different orthopedic specialties ensures sustained dominance, making it the central growth driver of orthopedic contract manufacturing demand.
By Service Outlook, Forging/Casting Segment Accounts for 33% Market Share
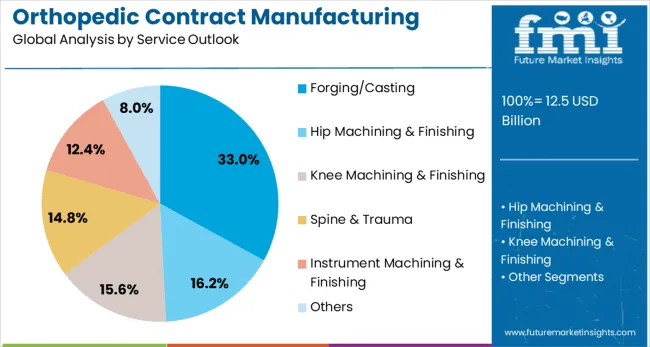
Forging and casting services are projected to represent 33% of orthopedic contract manufacturing demand in 2026, underscoring their role as fundamental manufacturing processes for orthopedic devices. Manufacturers gravitate toward specialized forging and casting services for their ability to create complex geometries, superior material properties, and cost-effective production of high-volume components. Positioned as critical foundation processes, these services offer both structural integrity benefits and economic advantages for large-scale orthopedic device production.
The segment is supported by the rising demand for lightweight yet strong orthopedic components, where advanced forging and casting techniques play a central role in material optimization strategies. Additionally, contract manufacturers are increasingly combining forging and casting capabilities with secondary operations like machining and finishing, enhancing the value proposition and justifying comprehensive service partnerships. As orthopedic device companies prioritize manufacturing efficiency and material performance, forging and casting services will continue to maintain strong demand, reinforcing their essential positioning within the contract manufacturing services spectrum.
Rising Outsourcing of Implant Production
The orthopedic contract manufacturing market is experiencing significant growth as medical device companies increasingly outsource implant production. Outsourcing allows firms to reduce operational costs, shorten product development timelines, and access specialized expertise in materials and precision engineering. Contract manufacturers provide scalability that helps original equipment manufacturers meet rising global demand without investing heavily in new facilities. They also ensure consistent quality through advanced testing and regulatory compliance support. The growing prevalence of orthopedic conditions such as arthritis and fractures has intensified the need for implants, further encouraging outsourcing. This shift has positioned contract manufacturing as a critical partner for companies seeking efficiency, cost control, and market expansion.
Strong Demand for Minimally Invasive Devices
Minimally invasive orthopedic procedures are gaining popularity because they reduce recovery time, lower infection risks, and improve patient outcomes. This trend has increased the demand for specialized surgical instruments, smaller implants, and precision components. Contract manufacturers are addressing these requirements by producing highly accurate and complex devices that support advanced surgical techniques. Their ability to work with innovative materials and provide cleanroom manufacturing environments ensures high-quality output. As hospitals and clinics adopt less invasive surgical methods, device makers rely on contract partners to deliver components that meet strict design and performance standards. This rising demand for minimally invasive solutions is shaping the market’s trajectory.
Expansion into Emerging Healthcare Markets
Another important dynamic in the orthopedic contract manufacturing market is expansion into emerging healthcare markets. Regions such as Asia, Latin America, and the Middle East are witnessing rising demand for orthopedic procedures due to aging populations, lifestyle changes, and growing access to healthcare services. Contract manufacturers are partnering with local distributors and healthcare providers to establish strong regional presence. By offering cost-effective production and adapting products to regional needs, they are capturing significant growth opportunities. This expansion also helps manufacturers diversify their customer base beyond mature markets in North America and Europe. Emerging markets are now central to the long-term strategies of leading orthopedic contract manufacturers.
Analysis of Orthopedic Contract Manufacturing Market by Key Country
.webp)
| Country | CAGR (2026-2036) |
|---|---|
| China | 18.2% |
| India | 16.9% |
| Germany | 15.5% |
| France | 14.2% |
| UK | 12.8% |
| USA | 11.5% |
| Brazil | 10.1% |
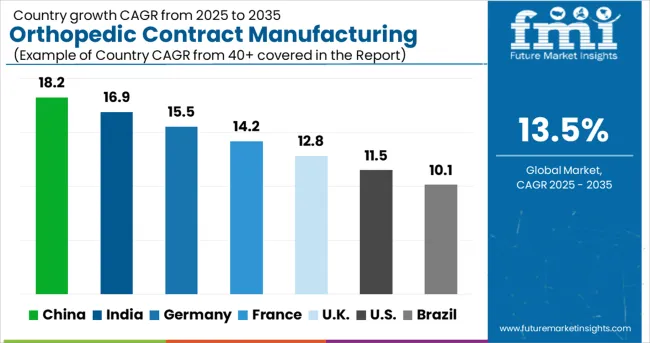
The orthopedic contract manufacturing market is experiencing robust growth globally, with China leading at an 18.2% CAGR through 2036, driven by expanding manufacturing capabilities, increasing medical device demand, and growing adoption of advanced production technologies. India follows closely at 16.9%, supported by strong cost advantages, skilled workforce availability, and increasing demand for quality manufacturing services. Germany shows steady growth at 15.5%, emphasizing precision manufacturing and advanced engineering capabilities. France records 14.2%, focusing on innovation-driven manufacturing and high-value service offerings. The UK shows 12.8% growth, prioritizing regulatory compliance and quality manufacturing standards.
The report covers an in-depth analysis of 40+ countries top-performing countries are highlighted below.
China Leads Asian Market Growth with Manufacturing Excellence
Orthopedic contract manufacturing in China is projected to grow at a CAGR of 18.2% through 2036, driven by expansion of manufacturing capacity, adoption of advanced production technologies, and the country’s rapidly growing medical device sector. Competition is strong among domestic and international manufacturers establishing comprehensive production facilities to serve orthopedic device companies across tier-1 and tier-2 cities. Compared with India and Brazil, adoption is accelerated due to government-backed incentives and large-scale industrial infrastructure. Investments in skilled workforce development, automation, and quality management systems are enhancing service capabilities and supporting China’s position as a leading hub for high-quality orthopedic device manufacturing.
- Government initiatives promoting medical device manufacturing and quality standards are supporting certified and compliant services across industrial regions.
- Adoption of advanced technologies and workforce skill enhancement is driving rapid service expansion and quality improvements nationwide.
India Demonstrates Exceptional Market Potential with Cost-Effective Manufacturing
Orthopedic contract manufacturing in India is expected to grow at a CAGR of 16.9%, supported by competitive manufacturing costs, skilled technical workforce, and rising capabilities in precision manufacturing services. Competition is shaped by domestic manufacturers and international medical device companies forming strategic partnerships to leverage India’s cost advantages. Compared with China, scale is smaller but growth potential remains high due to increasing adoption of quality standards and regulatory compliance. Investments in advanced production technologies, staff training, and process optimization are improving service efficiency. Indian manufacturers are increasingly delivering specialized solutions to support orthopedic device production, meeting domestic and international demand for precision, quality, and cost-effectiveness.
- Focus on certifications and regulatory compliance is creating opportunities for specialized contract manufacturing services across medical device markets.
- Investment in workforce development and advanced manufacturing technologies is driving service capability enhancement and quality improvement.
United States Focuses on Advanced Manufacturing and Regulatory Excellence
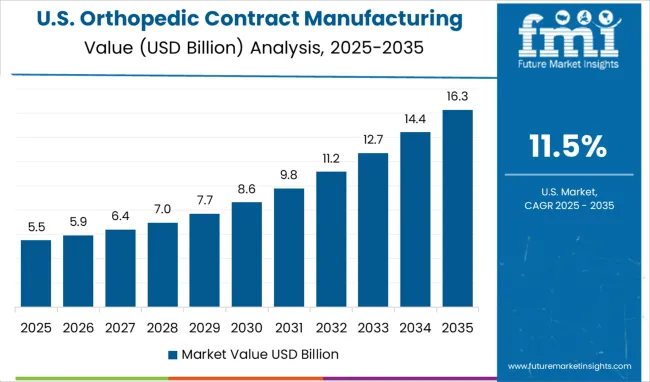
Orthopedic contract manufacturing in the USA is projected to grow at a CAGR of 11.5%, supported by the adoption of advanced manufacturing technologies and emphasis on regulatory compliance. Competition involves providers delivering comprehensive services combining precision machining, quality management systems, and supply chain reliability. Compared with China and India, adoption is more mature and technology-focused, prioritizing consistency and process optimization. Investments in automation, production precision, and regulatory expertise enhance service credibility and operational efficiency. USA manufacturers are increasingly relied upon for high-complexity orthopedic devices, integrating cutting-edge manufacturing processes with rigorous compliance protocols to ensure quality, safety, and reliability for both domestic and international markets.
- Adoption of advanced manufacturing technologies and automation integration is driving demand for high-precision contract manufacturing services.
- Regulatory expertise and quality system capabilities are reinforcing trust and reliability in orthopedic device production partnerships.
Germany Maintains Leadership with Precision Manufacturing Innovation
Orthopedic contract manufacturing in Germany is projected to grow at a CAGR of 15.5% through 2036, supported by precision engineering, advanced production technologies, and strong focus on quality excellence. Competition is shaped by providers delivering high-quality, reliable services meeting strict regulatory requirements. Compared with China and India, adoption is more quality-driven and technologically advanced, emphasizing operational reliability. Investments in next-generation manufacturing solutions, integrated quality management, and clinical partnerships are enhancing service capabilities. German contract manufacturers are increasingly relied upon for complex orthopedic device production, leveraging expertise in precision machining, advanced materials, and regulatory compliance to maintain leadership in high-performance, innovative contract manufacturing services.
- Investment in advanced manufacturing technologies is supporting next-generation contract manufacturing with superior precision and integrated quality management.
- Collaboration between manufacturing specialists and medical device companies is enhancing service innovation and reinforcing Germany’s market leadership.
United Kingdom Strengthens Market with Quality-Driven Manufacturing Services
Orthopedic contract manufacturing in the UK is projected to grow at a CAGR of 12.8%, supported by rising demand for quality-focused services and comprehensive regulatory compliance capabilities. Competition involves domestic providers and international partners offering specialized solutions for orthopedic devices. Compared with China and India, adoption is moderate but emphasizes reliability, compliance, and service integration. Investments in regulatory expertise, specialized manufacturing capabilities, and comprehensive service portfolios enhance operational efficiency. British contract manufacturers are increasingly supporting orthopedic device companies by delivering precise, compliant, and scalable production solutions while meeting both domestic and international quality expectations across a range of orthopedic applications.
- Expansion of specialized manufacturing capabilities and regulatory expertise is improving accessibility of advanced contract manufacturing services.
- Growing demand for integrated manufacturing solutions is encouraging providers to develop specialized capabilities for orthopedic device production.
France Anchors Growth with Innovation-Driven Manufacturing Excellence
Orthopedic contract manufacturing in France is projected to grow at a CAGR of 14.2% through 2036, supported by innovation-focused production, advanced materials processing, and integrated service offerings. Competition emphasizes technological advancement, quality management, and premium service delivery. Compared with Germany and China, adoption emphasizes high-value, niche orthopedic applications. Investments in next-generation manufacturing technologies, research collaboration, and service integration are enhancing operational efficiency. French contract manufacturers are increasingly delivering innovative solutions that combine precision production with regulatory compliance and advanced analytics, reinforcing their position as trusted providers of high-quality orthopedic contract manufacturing services for both domestic and international medical device companies.
- Integration of advanced manufacturing technologies and innovation-focused service development is driving comprehensive production capabilities.
- Specialized facilities, partnerships, and premium service platforms are enhancing market distribution and strengthening customer confidence in service effectiveness.
Europe Market Split by Country
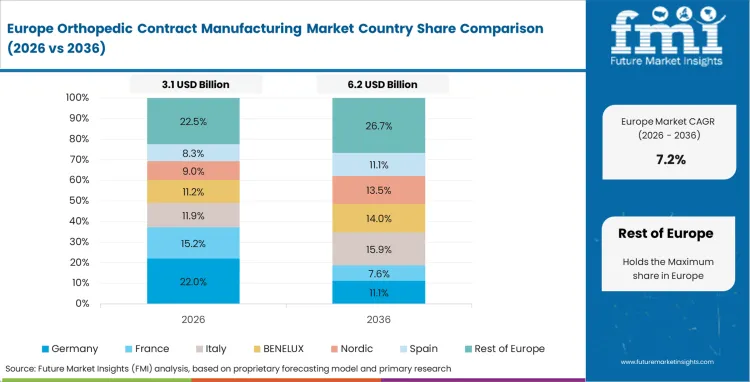
The orthopedic contract manufacturing market in Europe demonstrates mature development across major economies with Germany showing strong presence through its precision engineering capabilities and consumer appreciation for high-quality medical device manufacturing, supported by companies leveraging advanced manufacturing technologies to develop effective orthopedic contract services that address complex device requirements and stringent quality standards.
France represents a significant market driven by its medical device innovation heritage and sophisticated understanding of orthopedic manufacturing, with companies pioneering advanced contract manufacturing services that combine French engineering expertise with cutting-edge production technologies for enhanced device quality and manufacturing efficiency.
The UK exhibits considerable growth through its embrace of innovative manufacturing solutions and regulatory excellence, with companies leading the development of comprehensive contract manufacturing services and thorough customer education about manufacturing benefits and quality advantages. Italy and Spain show expanding interest in specialized manufacturing solutions, particularly in premium contract services targeting precision manufacturing and advanced materials processing. BENELUX countries contribute through their focus on technological innovation and eco-efficient manufacturing practices, while Eastern Europe and Nordic regions display growing potential driven by increasing manufacturing capabilities and expanding access to advanced orthopedic contract manufacturing services across diverse industrial sectors.
Competitive Landscape of Orthopedic Contract Manufacturing Market
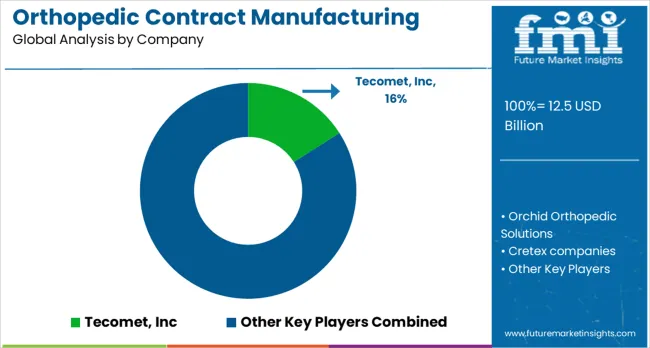
The orthopedic contract manufacturing market is characterized by precision engineering and specialized medical device expertise. Custom implants and surgical instruments are provided by Tecomet, Inc., with high-volume production capabilities. Integrated design and manufacturing workflows are applied by Orchid Orthopedic Solutions for spinal and joint implants. Cretex Companies leverages advanced machining, assembly, and finishing techniques across diverse orthopedic portfolios. Scalable manufacturing solutions for standard and patient-specific implants are offered by Viant. Quality assurance and regulatory-compliant production are emphasized by ARCH Medical Solutions Corp.
Competition is shaped by material selection, manufacturing precision, and process validation. Additive manufacturing and precision machining are utilized by Avalign Technologies to create complex implant geometries. LISI Medical delivers tight-tolerance implants and instrumentation with consistent performance. Turnkey solutions, including prototyping, tooling, and assembly, are provided by Paragon Medical. Engineering support and modular implant assembly are emphasized by Norman Noble, Inc. Automated machining and process monitoring are integrated by Autocam Medical to ensure reproducible results. Technical brochures highlight production capabilities, materials expertise, and assembly processes, guiding end users on operational reliability and product performance.
Differentiation is reinforced through process scalability, regulatory compliance, and design flexibility. Machining, forging, additive manufacturing, and assembly processes are consistently demonstrated in promotional material. Quality certifications, validation protocols, and customization options are highlighted for enhanced confidence. Modular designs, rapid prototyping, and monitored workflows are emphasized to showcase market positioning. Regional production capabilities, engineering expertise, and technical support are presented to reinforce reliability and mission-focused outcomes. Product brochures serve as concise evidence of competitive strategy, highlighting precision, process excellence, and consistent delivery across orthopedic applications.
Key Players in the Orthopedic Contract Manufacturing Market
- Tecomet, Inc.
- Orchid Orthopedic Solutions
- Cretex Companies
- Viant
- ARCH Medical Solutions Corp.
- Avalign Technologies
- LISI Medical
- Paragon Medical
- Norman Noble, Inc.
- Autocam Medical
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 14.13 Billion to USD 47.95 Billion, at a CAGR of 13.00% |
| Market Definition | Orthopedic contract manufacturing encompasses outsourced production services for orthopedic implants (joint prostheses, spinal hardware, trauma fixation devices), surgical instruments, sterilization cases, and instrument trays, provided by specialized contract manufacturers offering forging, casting, CNC machining, finishing, coating, and assembly capabilities to orthopedic OEM device companies. |
| Type Segmentation | Implants, Instruments, Cases, Trays, Others |
| Service Segmentation | Forging/Casting, Machining & Finishing, Other Services |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | China, India, Germany, France, UK, USA, Brazil, and 40 plus countries |
| Key Companies Profiled | Tecomet, Inc., Orchid Orthopedic Solutions, Cretex Companies, Viant, ARCH Medical Solutions Corp., Avalign Technologies, LISI Medical, Paragon Medical, Norman Noble, Inc., Autocam Medical |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down modeling validated against primary research inputs and publicly available industry data. |
Orthopedic Contract Manufacturing Market by Segments
Type Outlook:
- Implants
- Instruments
- Cases
- Trays
- Others
Service Outlook:
- Forging/Casting
- Machining & Finishing
- Other Services
Company:
- Tecomet, Inc.
- Orchid Orthopedic Solutions
- Cretex Companies
- Viant
- ARCH Medical Solutions Corp.
- Avalign Technologies
- LISI Medical
- Paragon Medical
- Norman Noble, Inc.
- Autocam Medical
Region:
- North America
- United States
- Canada
- Mexico
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- East Asia
- China
- Japan
- South Korea
- South Asia & Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia & Pacific
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- [1] USA Food and Drug Administration. (2024). FDA Device Establishment Registration Database: Contract Manufacturer Listings. FDA.
- [2] International Organization for Standardization. (2024). ISO 13485: Medical Devices, Quality Management Systems. ISO.
- [3] Tecomet Inc. (2024). Orthopedic Contract Manufacturing Capabilities Portfolio. Tecomet.
- [4] Orchid Orthopedic Solutions. (2024). Annual Report 2024: Orthopedic Manufacturing Services. Orchid.
- [5] Medical Device and Diagnostic Industry. (2024). Orthopedic OEM Outsourcing Survey: Manufacturing Strategy Trends. MDDI.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary research documentation.
Frequently Asked Questions
How large is the demand for Orthopedic Contract Manufacturing in the global market in 2026?
Demand for Orthopedic Contract Manufacturing in the global market is estimated to be valued at USD 14.13 billion in 2026.
What will be the market size of Orthopedic Contract Manufacturing by 2036?
Market size for Orthopedic Contract Manufacturing is projected to reach USD 47.95 billion by 2036.
What is the expected demand growth between 2026 and 2036?
Demand is expected to grow at a CAGR of 13.00% between 2026 and 2036.
Which Type is poised to lead by 2026?
Implants accounts for 64.0% in 2026.
How significant is Forging/Casting in driving adoption?
Forging/Casting represents 33.0% of segment share in 2026.
What country records the fastest growth?
China is projected to grow at a CAGR of 18.20% during 2026 to 2036.
What is the projected growth for India?
India is projected to expand at a CAGR of 16.90% during 2026 to 2036.
What is included in the scope of this report?
Market scope covers contract manufacturing services for orthopedic implants, surgical instruments, sterilization cases, and instrument trays, including forging/casting, precision CNC machining, surface finishing, coating application, and final assembly provided to orthopedic OEM companies.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Type Outlook
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Type Outlook , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Type Outlook , 2026 to 2036
- Implants
- Instruments
- Cases
- Trays
- Others
- Implants
- Y to o to Y Growth Trend Analysis By Type Outlook , 2021 to 2025
- Absolute $ Opportunity Analysis By Type Outlook , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Service Outlook
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Service Outlook, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Service Outlook, 2026 to 2036
- Forging/Casting
- Machining & Finishing
- Other Services
- Forging/Casting
- Y to o to Y Growth Trend Analysis By Service Outlook, 2021 to 2025
- Absolute $ Opportunity Analysis By Service Outlook, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Type Outlook
- By Service Outlook
- By Country
- Market Attractiveness Analysis
- By Country
- By Type Outlook
- By Service Outlook
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Type Outlook
- By Service Outlook
- By Country
- Market Attractiveness Analysis
- By Country
- By Type Outlook
- By Service Outlook
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Type Outlook
- By Service Outlook
- By Country
- Market Attractiveness Analysis
- By Country
- By Type Outlook
- By Service Outlook
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Type Outlook
- By Service Outlook
- By Country
- Market Attractiveness Analysis
- By Country
- By Type Outlook
- By Service Outlook
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Type Outlook
- By Service Outlook
- By Country
- Market Attractiveness Analysis
- By Country
- By Type Outlook
- By Service Outlook
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Type Outlook
- By Service Outlook
- By Country
- Market Attractiveness Analysis
- By Country
- By Type Outlook
- By Service Outlook
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Type Outlook
- By Service Outlook
- By Country
- Market Attractiveness Analysis
- By Country
- By Type Outlook
- By Service Outlook
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Type Outlook
- By Service Outlook
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Type Outlook
- By Service Outlook
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Type Outlook
- By Service Outlook
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Type Outlook
- By Service Outlook
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Type Outlook
- By Service Outlook
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Type Outlook
- By Service Outlook
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Type Outlook
- By Service Outlook
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Type Outlook
- By Service Outlook
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Type Outlook
- By Service Outlook
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Type Outlook
- By Service Outlook
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Type Outlook
- By Service Outlook
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Type Outlook
- By Service Outlook
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Type Outlook
- By Service Outlook
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Type Outlook
- By Service Outlook
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Type Outlook
- By Service Outlook
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Type Outlook
- By Service Outlook
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type Outlook
- By Service Outlook
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Type Outlook
- By Service Outlook
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Type Outlook
- By Service Outlook
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type Outlook
- By Service Outlook
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Type Outlook
- By Service Outlook
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Type Outlook
- By Service Outlook
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Type Outlook
- By Service Outlook
- Competition Analysis
- Competition Deep Dive
- Tecomet, Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Orchid Orthopedic Solutions
- Cretex Companies
- Viant
- ARCH Medical Solutions Corp.
- Avalign Technologies
- LISI Medical
- Paragon Medical
- Norman Noble, Inc.
- Autocam Medical
- Tecomet, Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Type Outlook , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Service Outlook, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Type Outlook , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Service Outlook, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Type Outlook , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Service Outlook, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Type Outlook , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Service Outlook, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Type Outlook , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Service Outlook, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Type Outlook , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Service Outlook, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Type Outlook , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Service Outlook, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Type Outlook , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Service Outlook, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Type Outlook , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Type Outlook , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Type Outlook
- Figure 6: Global Market Value Share and BPS Analysis by Service Outlook, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Service Outlook, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Service Outlook
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Type Outlook , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Type Outlook , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Type Outlook
- Figure 23: North America Market Value Share and BPS Analysis by Service Outlook, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Service Outlook, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Service Outlook
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Type Outlook , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Type Outlook , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Type Outlook
- Figure 30: Latin America Market Value Share and BPS Analysis by Service Outlook, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Service Outlook, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Service Outlook
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Type Outlook , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Type Outlook , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Type Outlook
- Figure 37: Western Europe Market Value Share and BPS Analysis by Service Outlook, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Service Outlook, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Service Outlook
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Type Outlook , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Type Outlook , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Type Outlook
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Service Outlook, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Service Outlook, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Service Outlook
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Type Outlook , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Type Outlook , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Type Outlook
- Figure 51: East Asia Market Value Share and BPS Analysis by Service Outlook, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Service Outlook, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Service Outlook
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Type Outlook , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Type Outlook , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Type Outlook
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Service Outlook, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Service Outlook, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Service Outlook
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Type Outlook , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Type Outlook , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Type Outlook
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Service Outlook, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Service Outlook, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Service Outlook
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

