Paediatric Spasticity Treatment Market : Global Industry Analysis 2015 - 2024 and Opportunity Assessment 2025 - 2035
Paediatric Spasticity Treatment Market Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
Paediatric Spasticity Treatment Market Size and Share Forecast Outlook 2025 to 2035
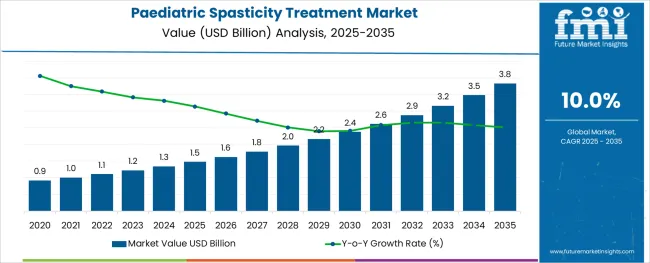
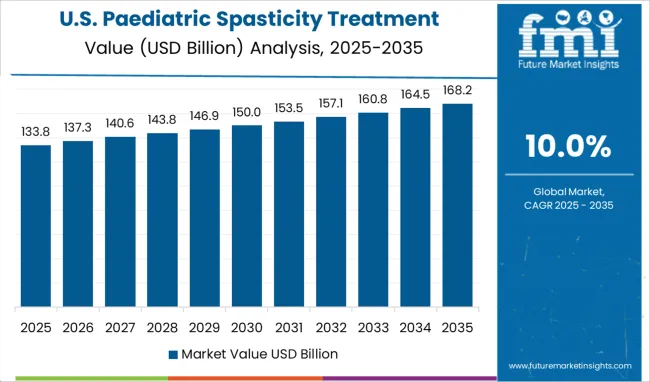
The Paediatric Spasticity Treatment Market is estimated to be valued at USD 1.5 billion in 2025 and is projected to reach USD 3.8 billion by 2035, registering a compound annual growth rate (CAGR) of 10.0% over the forecast period.
Quick Stats for Paediatric Spasticity Treatment Market
- Paediatric Spasticity Treatment Market Industry Value (2025): USD 1.5 billion
- Paediatric Spasticity Treatment Market Forecast Value (2035): USD 3.8 billion
- Paediatric Spasticity Treatment Market Forecast CAGR: 10.0%
- Leading Segment in Paediatric Spasticity Treatment Market in 2025: Botox (38.6%)
- Key Growth Region in Paediatric Spasticity Treatment Market: North America, Asia-Pacific, Europe
- Top Key Players in Paediatric Spasticity Treatment Market: Allergan plc, Ipsen Biopharmaceuticals, Almirall, Piramal Critical Care, Genentech, Sanofi, Saol Therapeutics, USA WorldMeds
Rationale for Segmental Growth in the Paediatric Spasticity Treatment Market
Market Overview
The paediatric spasticity treatment market is experiencing steady expansion as clinical understanding of childhood neuromuscular disorders evolves and treatment protocols advance. There has been growing attention from healthcare professionals on managing spasticity to improve mobility and prevent long-term complications in children with cerebral palsy and other neurological disorders. Scientific publications have emphasized the importance of early therapeutic intervention to minimize musculoskeletal deformities and enhance quality of life.
Drug development in this space has focused on formulations tailored for paediatric patients with a strong emphasis on safety and tolerability. Additionally, healthcare systems have expanded specialized rehabilitation and neurology care services which has increased treatment accessibility.
Looking ahead, the market is expected to grow with advancements in therapeutic techniques and a rising focus on multidisciplinary care models. Segmental growth is anticipated to be led by Botox in drug type, Injectable as the preferred route of administration, and Hospitals Pharmacies in the distribution channel reflecting clinical adoption patterns and patient care requirements.
Segmental Analysis
The market is segmented by Drug Type, Route of Administration, and Distribution Channel and region. By Drug Type, the market is divided into Botox, Baclofen or Muscle Relaxants, Intrathecal Baclofen, Clonazepam, Diazepam, Tizanidine, and Others. In terms of Route of Administration, the market is classified into Injectable and Oral. Based on Distribution Channel, the market is segmented into Hospitals Pharmacies, Drug Stores, Retail Pharmacies, and Mail Order Pharmacies. Regionally, the market is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
The market is segmented by Drug Type, Route of Administration, and Distribution Channel and region. By Drug Type, the market is divided into Botox, Baclofen or Muscle Relaxants, Intrathecal Baclofen, Clonazepam, Diazepam, Tizanidine, and Others. In terms of Route of Administration, the market is classified into Injectable and Oral. Based on Distribution Channel, the market is segmented into Hospitals Pharmacies, Drug Stores, Retail Pharmacies, and Mail Order Pharmacies. Regionally, the market is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the Drug Type Segment: Botox
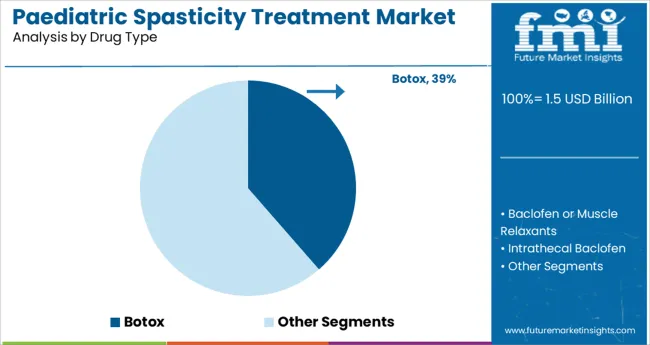
The Botox segment is projected to account for 38.6% of the paediatric spasticity treatment market revenue in 2025 making it the leading drug type. This segment has been driven by the proven clinical effectiveness of botulinum toxin in reducing muscle tone and improving functional mobility in children with spasticity. Healthcare providers have selected Botox for its targeted action which allows localized muscle relaxation without systemic side effects.
Treatment guidelines have recommended botulinum toxin as a first-line intervention in managing focal spasticity supporting its adoption in clinical practice. Additionally, Botox injections have been associated with reduced surgical interventions and improved therapy outcomes when used alongside physiotherapy.
As clinical experience with paediatric dosing protocols grows the Botox segment is expected to remain the drug of choice for spasticity management in children.
Insights into the Route of Administration Segment: Injectable
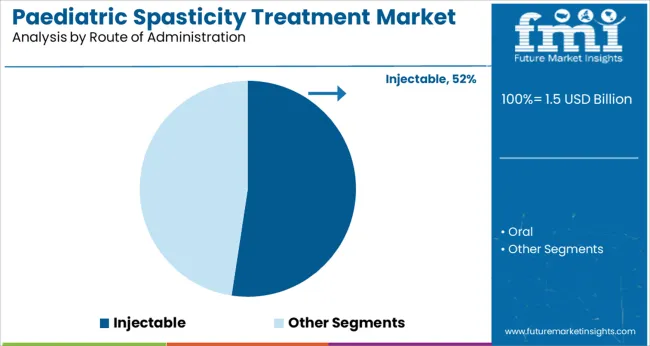
The Injectable segment is projected to contribute 52.4% of the paediatric spasticity treatment market revenue in 2025 holding its leadership in route of administration. This segment has expanded due to the preference for targeted drug delivery methods that provide localized symptom relief with predictable effects. Injectable formulations such as botulinum toxin have allowed clinicians to deliver therapy directly to overactive muscles ensuring better control over treatment outcomes.
The rapid onset of action and extended duration of symptom relief have further supported the widespread use of injectables in paediatric spasticity care. Additionally, advancements in injection techniques and improved patient comfort measures have enhanced the acceptability of this route among children and caregivers.
As personalized treatment plans become more common the Injectable segment is expected to continue its growth as the preferred method of administration.
Insights into the Distribution Channel Segment: Hospitals Pharmacies
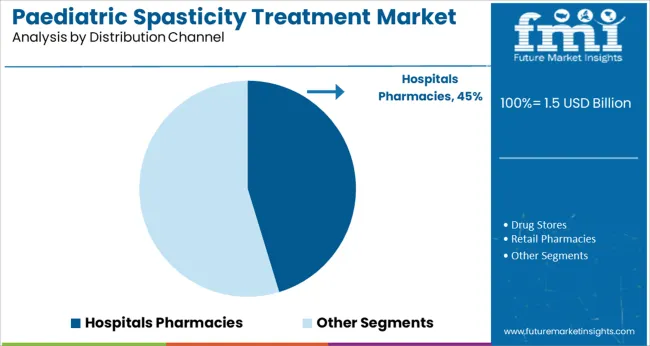
The Hospitals Pharmacies segment is projected to capture 45.3% of the paediatric spasticity treatment market revenue in 2025 maintaining its dominance among distribution channels. This segment has grown as specialized treatments like botulinum toxin injections are typically administered in hospital settings where multidisciplinary care teams oversee paediatric neurology and rehabilitation. Hospitals Pharmacies have served as the primary point for procuring and dispensing these therapies ensuring compliance with safety protocols and dosage requirements.
Additionally, hospitals have expanded paediatric specialty clinics and outpatient departments improving access to spasticity management services. The role of Hospitals Pharmacies has also included supporting patient education and follow-up care coordinating between physicians therapists and families.
With the ongoing focus on comprehensive paediatric care the Hospitals Pharmacies segment is expected to remain the key distribution channel for spasticity treatment medications.
What are the Key Dynamics to the Paediatric Spasticity Treatment Market Share?
Increasing awareness, growing research and development for drug development drive the sales of paediatric spasticity treatment in the near future. Changing lifestyle, developing new and innovative technology, and improving healthcare infrastructure are expected to fuel the demand for paediatric spasticity treatment in the forecast period.
The launch of novel therapeutics and growing per capita income in emerging economies such as China and Brazil is further driving the sales of paediatric spasticity treatment.
Some key factors that might hinder the growth of paediatric spasticity treatment market include the high cost of treatment and low awareness in underdeveloped and developing countries, and rigorous regulations for the approval of drug.
The progress and development of new therapeutic treatments, as well as the growing number of clinical trials for paediatric spasticity treatment, provide a significant opportunity for the demand for paediatric spasticity treatment to grow over the forecast period.
Collaboration and partnership among leading companies for the development of novel medications, as well as increased research funding, will boost the sales of paediatric spasticity treatment in the near future.
Which is the Key Paediatric Spasticity Treatment Market?
North America is expected to contribute to a major share in the global paediatric spasticity treatment market, owing to the increasing population, increasing number of screening procedures, presence of major manufacturers, and growing clinical trials.
Well-developed healthcare facilities and government funding for drug development further drive the sales of paediatric spasticity treatment.
Europe contributes the second-largest share with significant growth in the demand for paediatric spasticity treatment owing to technological advancement in the treatment coupled with growing adoption of a new treatments.
Asia Pacific paediatric spasticity treatment market is primarily driven by the factors such as growing prevalence, increasing population, and growing awareness of new treatment options.
China is expected to register rapid growth in the sales of paediatric spasticity treatment due to the large patient pool and availability of new drugs for the paediatric spasticity treatment.
Latin America and Middle East & Africa are projected to show a slow rise in demand for paediatric spasticity treatment due to less awareness, lack of adequate healthcare facilities, and less developed infrastructure.
How is the Competitive Landscape in the Paediatric Spasticity Treatment Market?
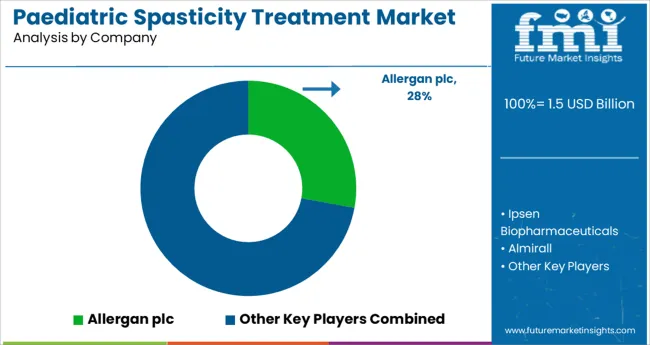
Some of the key players present in the global paediatric spasticity treatment are Allergan plc, Ipsen Biopharmaceuticals, Inc., Almirall, S.A, Piramal Critical Care, Inc., Genentech, Inc, Sanofi, Saol Therapeutics, and US WorldMeds, LLC among others.
The demand for paediatric spasticity treatment is moderately competitive, with several significant players. In terms of sales of paediatric spasticity treatment, just a few of the market's top firms are currently producing new products to solve unmet market challenges.
Recent Developments in the paediatric spasticity treatment market include:
The US Food and Drug Administration (FDA) accepted a supplementary Biologics License Application for XEOMIN in August 2024. (incobotulinumtoxinA).
The United States Food and Drug Administration (FDA) approved the expanded use of Dysport (abobotulinumtoxinA) for the treatment of Spasticity in paediatric patients in July 2024, according to Ipsen Biopharmaceuticals.
The report consists of key players, contributing to the paediatric spasticity treatment market share. It also consists of organic and inorganic growth strategies adopted by market players to improve their market positions. This exclusive report analysis the competitive landscape and paediatric spasticity treatment market share acquired by players to strengthen their market position.
Scope Of Report
| Report Attribute | Details |
|---|---|
| Growth rate | CAGR of 10% from 2025 to 2035 |
| Base year for estimation | 2024 |
| Historical data | 2020 to 2024 |
| Forecast period | 2025 to 2035 |
| Quantitative units | Revenue in million, volume in kilotons, and CAGR from 2025 to 2035 |
| Report coverage | Revenue forecast, volume forecast, company ranking, competitive landscape, growth factors, and trends, Pricing Analysis |
| Segments covered | Drug type, route of administration, distribution channel, region |
| Regional scope | North America; Western Europe; Eastern Europe; Middle East; Africa; ASEAN; South Asia; Rest of Asia; Australia and New Zealand |
| Country scope | USA, Canada, Mexico, Germany, UK, France, Italy, Spain, Russia, Belgium, Poland, Czech Republic, China, India, Japan, Australia, Brazil, Argentina, Colombia, Saudi Arabia, UAE, Iran, South Africa |
| Key companies profiled | Allergan plc; Ipsen Biopharmaceuticals Inc.; Almirall; S.A; Piramal Critical Care, Inc.; Genentech Inc; Sanofi; Saol Therapeutics; USA WorldMeds, LLC among others. |
| Customization scope | Free report customization (equivalent to up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope. |
| Pricing and purchase options | Avail customized purchase options to meet your exact research needs. |
Key Segments
By Drug Type:
- Baclofen or Muscle Relaxants
- Botox
- Intrathecal Baclofen
- Clonazepam
- Diazepam
- Tizanidine
- Others
By Route of Administration:
- Oral
- Injectable
By Distribution Channel:
- Hospitals Pharmacies
- Drug Stores
- Retail Pharmacies
- Mail Order Pharmacies
By Region:
- North America
- Latin America
- Asia Pacific
- Europe
- MEA
Frequently Asked Questions
How big is the paediatric spasticity treatment market in 2025?
The global paediatric spasticity treatment market is estimated to be valued at USD 1.5 billion in 2025.
What will be the market size for the paediatric spasticity treatment market in 2035?
It is projected to reach USD 3.8 billion by 2035.
How much will be the CAGR of the paediatric spasticity treatment market between 2025 and 2035?
The market is expected to grow at a 10.0% CAGR between 2025 and 2035.
What are the key product types in the paediatric spasticity treatment market?
The key product types are botox, baclofen or muscle relaxants, intrathecal baclofen, clonazepam, diazepam, tizanidine and others.
Which injectable segment is expected to dominate the paediatric spasticity treatment market in 2025?
injectable segment is expected to dominate with a 52.4% industry share in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020-2024 and Forecast, 2025-2035
- Historical Market Size Value (USD Million) Analysis, 2020-2024
- Current and Future Market Size Value (USD Million) Projections, 2025-2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020-2024 and Forecast 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By Drug Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Drug Type , 2020-2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Drug Type , 2025-2035
- Botox
- Baclofen or Muscle Relaxants
- Intrathecal Baclofen
- Clonazepam
- Diazepam
- Tizanidine
- Others
- Botox
- Y-o-Y Growth Trend Analysis By Drug Type , 2020-2024
- Absolute $ Opportunity Analysis By Drug Type , 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By Route of Administration
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Route of Administration, 2020-2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Route of Administration, 2025-2035
- Injectable
- Oral
- Injectable
- Y-o-Y Growth Trend Analysis By Route of Administration, 2020-2024
- Absolute $ Opportunity Analysis By Route of Administration, 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2020-2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2025-2035
- Hospitals Pharmacies
- Drug Stores
- Retail Pharmacies
- Mail Order Pharmacies
- Hospitals Pharmacies
- Y-o-Y Growth Trend Analysis By Distribution Channel, 2020-2024
- Absolute $ Opportunity Analysis By Distribution Channel, 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2020-2024
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2025-2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- USA
- Canada
- Mexico
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Type
- By Route of Administration
- By Distribution Channel
- Key Takeaways
- Latin America Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Type
- By Route of Administration
- By Distribution Channel
- Key Takeaways
- Western Europe Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Type
- By Route of Administration
- By Distribution Channel
- Key Takeaways
- Eastern Europe Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Type
- By Route of Administration
- By Distribution Channel
- Key Takeaways
- East Asia Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- China
- Japan
- South Korea
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Type
- By Route of Administration
- By Distribution Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Type
- By Route of Administration
- By Distribution Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Type
- By Route of Administration
- By Distribution Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Type
- By Route of Administration
- By Distribution Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Type
- By Route of Administration
- By Distribution Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Type
- By Route of Administration
- By Distribution Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Type
- By Route of Administration
- By Distribution Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Type
- By Route of Administration
- By Distribution Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Type
- By Route of Administration
- By Distribution Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Type
- By Route of Administration
- By Distribution Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Type
- By Route of Administration
- By Distribution Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Type
- By Route of Administration
- By Distribution Channel
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Type
- By Route of Administration
- By Distribution Channel
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Type
- By Route of Administration
- By Distribution Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Type
- By Route of Administration
- By Distribution Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Type
- By Route of Administration
- By Distribution Channel
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Type
- By Route of Administration
- By Distribution Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Type
- By Route of Administration
- By Distribution Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Type
- By Route of Administration
- By Distribution Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Type
- By Route of Administration
- By Distribution Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Type
- By Route of Administration
- By Distribution Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Type
- By Route of Administration
- By Distribution Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Type
- By Route of Administration
- By Distribution Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Type
- By Route of Administration
- By Distribution Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Drug Type
- By Route of Administration
- By Distribution Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Drug Type
- By Route of Administration
- By Distribution Channel
- Competition Analysis
- Competition Deep Dive
- Allergan plc
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Ipsen Biopharmaceuticals
- Almirall
- Piramal Critical Care
- Genentech
- Sanofi
- Saol Therapeutics
- US WorldMeds
- Allergan plc
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2020-2035
- Table 2: North America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 3: Latin America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 4: Western Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 5: Eastern Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 6: East Asia Market Value (USD Million) Forecast by Country, 2020-2035
- Table 7: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020-2035
- Table 8: Middle East & Africa Market Value (USD Million) Forecast by Country, 2020-2035
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020-2035
- Figure 3: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 4: Global Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 5: Global Market Attractiveness Analysis by Region
- Figure 6: North America Market Incremental $ Opportunity, 2025-2035
- Figure 7: Latin America Market Incremental $ Opportunity, 2025-2035
- Figure 8: Western Europe Market Incremental $ Opportunity, 2025-2035
- Figure 9: Eastern Europe Market Incremental $ Opportunity, 2025-2035
- Figure 10: East Asia Market Incremental $ Opportunity, 2025-2035
- Figure 11: South Asia and Pacific Market Incremental $ Opportunity, 2025-2035
- Figure 12: Middle East & Africa Market Incremental $ Opportunity, 2025-2035
- Figure 13: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 14: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 15: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 16: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 17: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 18: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 19: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 20: Global Market - Tier Structure Analysis
- Figure 21: Global Market - Company Share Analysis

