Surgical Heart Valves Market
This report covers the surgical heart valves market through analysis of market size, revenue forecast, competitive landscape, demand outlook, growth drivers, restraints, industry trends, supply chain developments, and strategic growth opportunities.
Surgical Heart Valves Market size, market forecast and outlook by FMI
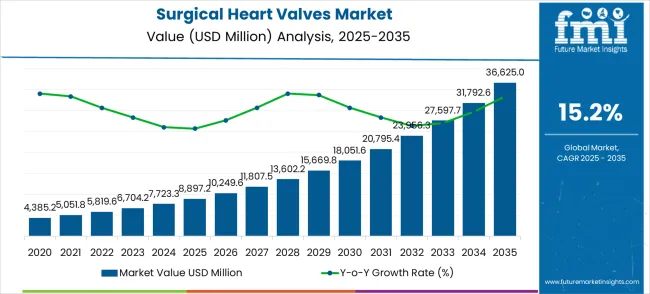
The surgical heart valves market was valued at USD 8.89 billion in 2025, projected to reach USD 10.24 billion in 2026, and is forecast to expand to USD 42.16 billion by 2036 at a 15.2% CAGR. Expanding prevalence of structural heart disease among aging populations, paired with the clinical migration from open surgical repair to catheter-based valve deployment, is compressing product replacement cycles and widening addressable patient pools. Hospital capital equipment budgets increasingly allocate dedicated line items for transcatheter procedure suites.
Summary of Surgical Heart Valves Market
- Market Snapshot
- The surgical heart valves market is valued at USD 8.89 billion in 2025 and is projected to reach USD 42.16 billion by 2036.
- The industry is expected to grow at a 15.2% CAGR from 2026 to 2036, creating an incremental opportunity of USD 31.92 billion.
- The sector is a high-growth, procedure-driven medical device category where clinical evidence generation, reimbursement access, and operator training networks define competitive positioning.
- Demand and Growth Drivers
- Demand is expanding as transcatheter valve replacement procedures gain clinical approval across broader patient risk categories, widening the addressable treatment population.
- Hospital capital investment in catheterization laboratory construction is accelerating valve device procurement as facilities add dedicated structural heart intervention capacity.
- Growth is supported by aging population demographics in mature markets and expanding cardiac surgery infrastructure in emerging economies.
- Among key countries, China leads at 20.5% CAGR, India at 19.0%, Germany at 17.5%, Brazil at 16.0%, USA at 14.4%, UK at 12.9%.
- Product and Segment View
- The market includes transcatheter and surgical heart valve prostheses, encompassing TAVR, TPVR, and TMVR devices deployed across hospital catheterization laboratories and surgical suites.
- TAVR leads by Procedure Type with 52.0% share in 2026.
- Hospitals leads by Application with 68.0% share in 2026.
- Scope includes Procedure Type, Application segmentation with regional coverage across seven global regions.
- Geography and Competitive Outlook
- China and India are the fastest-growing markets, while North America and Europe remain mature demand bases.
- Competition is shaped by product quality, distribution reach, and regulatory compliance, with key players including Edwards Lifesciences, Medtronic, Abbott, Boston Scientific, Venus Medtech, Meril Life Sciences, and Braile Biomedica.
As per FMI, the surgical heart valves sector is undergoing a procedural migration that is reshaping device mix and average selling prices. Transcatheter aortic valve replacement (TAVR) procedures now account for the majority of aortic valve interventions in many mature markets, displacing open surgical aortic valve replacement volumes. This shift elevates per-procedure device costs while reducing hospital length-of-stay metrics. Emerging markets are entering the TAVR adoption curve at different stages, with reimbursement framework development serving as the primary gating factor for procedure volume growth.
Country-level growth rates reflect the pace of transcatheter procedure adoption and reimbursement expansion. China leads at 20.5% CAGR as national cardiac surgery networks expand catheterization laboratory capacity. India follows at 19.0%, where public hospital systems are integrating TAVR into tier-2 city cardiac care programs. Germany at 17.5% benefits from established transcatheter reimbursement pathways and high procedural operator density. Brazil at 16.0% and USA at 14.4% maintain steady volumes anchored by aging population demographics and cardiology department capital refreshment cycles.
Surgical Heart Valves Market key takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 10.24 billion |
| Industry Value (2036) | USD 42.16 billion |
| CAGR (2026 to 2036) | 15.2% |
Source: Future Market Insights, 2026
Surgical Heart Valves Market definition
Surgical Heart Valves encompasses prosthetic valve devices designed to replace or repair damaged cardiac valves. The market covers transcatheter valve systems (TAVR, TPVR, TMVR) and surgical replacement valves deployed through catheterization laboratories, hospital surgical suites, and ambulatory surgical centers. These devices address aortic stenosis, mitral regurgitation, and pulmonary valve dysfunction across diagnostic and therapeutic applications.
Surgical Heart Valves Market inclusions
Market scope includes global and regional market sizes, a 2026 to 2036 forecast period, segment breakdowns by Procedure Type, Application, and regional analysis across North America, Latin America, Europe, East Asia, South Asia, Oceania, and Middle East and Africa. Pricing trend analysis and competitive positioning assessments are incorporated within the analytical framework.
Surgical Heart Valves Market exclusions
The scope excludes annuloplasty rings used in valve repair procedures without prosthetic replacement, cardiac pacemakers, and implantable defibrillators. Diagnostic echocardiography equipment and imaging systems used in pre-procedural planning are also excluded.
Surgical Heart Valves Market research methodology
- Primary Research: Analysts engaged with interventional cardiologists, hospital procurement directors, and clinical engineering leads to map device selection criteria and procedural adoption decision gates.
- Desk Research: Data collection aggregated clinical trial registries, regulatory approval databases, hospital capital expenditure disclosures, and procedure volume publications from medical societies and health ministries.
- Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of product-level revenues, applying region-specific adoption curves to project future demand velocity.
- Data Validation and Update Cycle: Projections are tested against publicly reported expenditure guidance from leading industry participants and cross-referenced with regulatory filings.
Why is the Surgical Heart Valves Market Growing?
Market expansion is being supported by the rapidly aging global population and the corresponding increase in age-related cardiovascular diseases requiring surgical intervention. Modern cardiac care requires advanced valve replacement technologies that provide superior clinical outcomes while minimizing procedural risks and recovery times. The excellent safety profiles and proven clinical efficacy of transcatheter heart valve procedures make them essential components in contemporary cardiovascular medicine where patient outcomes and quality of life improvement are paramount.
The growing emphasis focus on minimally invasive cardiac procedures and patient-centered care is driving demand for advanced valve technologies from certified manufacturers with proven track records of clinical safety and procedural success. Healthcare providers are increasingly investing in transcatheter valve systems that offer reduced procedural risks and improved patient experiences compared to traditional open-heart surgery approaches. Clinical evidence and regulatory guidelines are establishing treatment standards that favor minimally invasive valve replacement solutions with demonstrated long-term durability and hemodynamic performance.
Segmental Analysis
The market is segmented by procedure type, application, and region. By intervention approach, the market is divided into TAVR (Transcatheter Aortic Valve Replacement), TPVR (Transcatheter Pulmonary Valve Replacement), and TMVR (Transcatheter Mitral Valve Replacement). Based on application, the market is categorized into hospitals, ambulatory surgical centers, and cardiac catheterization laboratories. Regionally, the market is divided into North America, Europe, East Asia, South Asia & Pacific, Latin America, and Middle East & Africa.
By Procedure Type, TAVR Segment Accounts for 52% Market Share
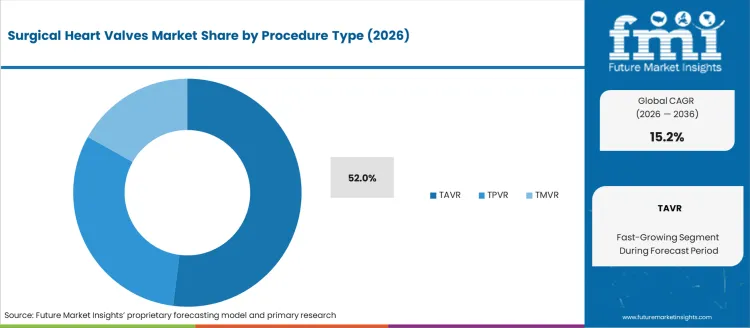
TAVR (Transcatheter Aortic Valve Replacement) procedures are projected to account for 52% of the surgical heart valves market in 2026. This leading share is supported by the well-established clinical evidence base for TAVR procedures and the large patient population with aortic stenosis requiring intervention. TAVR provides excellent clinical outcomes with reduced procedural risks compared to surgical aortic valve replacement, making it the preferred treatment approach for high-risk and intermediate-risk patients with severe aortic stenosis. The segment benefits from extensive clinical experience, proven long-term durability, and expanding indications that now include lower-risk patient populations.
Modern TAVR systems incorporate advanced valve designs, precise delivery mechanisms, and comprehensive imaging guidance that ensure optimal valve positioning and hemodynamic performance. These innovations have significantly improved procedural success rates and long-term clinical outcomes while reducing complications through minimally invasive access approaches and enhanced valve technologies. The cardiology and cardiac surgery communities particularly drive demand for TAVR solutions, as these procedures offer proven benefits for patients across multiple risk categories and age groups.
AdditionallyT, the aging population and increasing prevalence of degenerative aortic stenosis create sustained demand for TAVR procedures globally. The integration of advanced imaging technologies and artificial intelligence in valve sizing and positioning creates opportunities for further procedural optimization and outcome improvement, accelerating market adoption as cardiac centers seek the most effective treatment options for their patient populations.
By Application, Hospital Segment Accounts for 68% Market Share
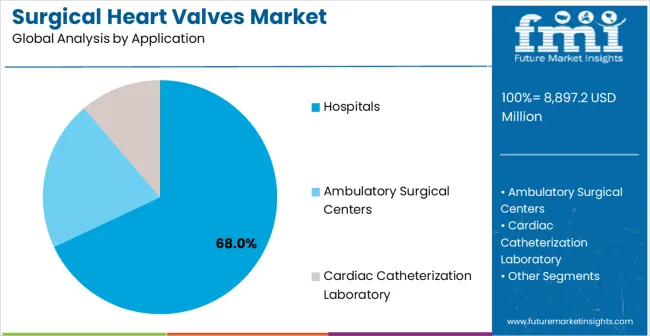
Hospital applications are expected to represent 68% of surgical heart valves demand in 2026. This dominant share reflects the complex nature of cardiac valve procedures and the institutional infrastructure required for comprehensive cardiovascular care. Hospital environments provide the necessary resources for pre-procedural planning, advanced imaging capabilities, surgical backup, and post-procedural monitoring required for successful valve interventions. The segment benefits from established cardiovascular service lines, specialized cardiac teams, and the capability to manage complex patient cases and potential complications.
Hospital applications demand exceptional clinical support capabilities to ensure optimal patient outcomes across diverse valve pathologies and patient risk profiles. These applications require comprehensive cardiovascular programs with interventional cardiology, cardiac surgery, anesthesiology, and critical care capabilities working collaboratively to deliver optimal patient care. The growing emphasis focus on cardiovascular excellence and patient outcome optimization drives consistent demand for advanced valve technologies in hospital settings that can demonstrate superior clinical results. Major medical centers and cardiovascular hospitals contribute significantly to market growth as institutions invest in leading-edge valve technologies to maintain clinical leadership and attract referring physicians.
AdditionallyT, the trend toward hybrid cardiovascular programs and heart team approaches in hospital systems creates opportunities for comprehensive valve treatment programs that combine surgical and transcatheter expertise. The segment also benefits from increasing hospital system consolidation and the development of specialized cardiovascular centers of excellence focused on valve disease management.
What are the Drivers, Restraints, and Key Trends of the Surgical Heart Valves Market?
The surgical heart valves market is advancing rapidly due to increasing cardiovascular disease prevalence and growing recognition of transcatheter valve intervention benefits in improving patient outcomes. However T, the market faces challenges including high device costs affecting healthcare budgets, reimbursement complexity across different healthcare systems, and the need for specialized training in transcatheter techniques. Clinical evidence development and physician education programs continue to influence adoption patterns and procedural volume growth.
Integration of Advanced Imaging and Artificial Intelligence Technologies
The growing implementation of 3D imaging systems, advanced echocardiography, and AI-powered valve sizing algorithms is enabling precise procedural planning and optimal valve selection in transcatheter interventions. Advanced imaging technologies provide comprehensive anatomical assessment and procedural guidance while optimizing valve positioning and reducing procedural complications. These technologies are particularly important for complex anatomical cases and patients requiring precise valve sizing for optimal hemodynamic outcomes.
Development of Next-Generation Valve Designs and Delivery Systems
Modern valve manufacturers are incorporating advanced materials science, refined valve leaflet designs, and innovative delivery mechanisms that improve valve hemodynamics while reducing procedural complexity and paravalvular regurgitation. Integration of biocompatible materials and enhanced valve durability enables superior long-term performance and significant clinical advantages compared to earlier-generation valve technologies. Advanced manufacturing techniques and quality control systems also support development of patient-specific valve solutions for diverse anatomical variations and clinical requirements.
Analysis of Surgical Heart Valves Market by Key Country
.webp)
| Country | CAGR |
|---|---|
| China | 20.5% |
| India | 19.0% |
| Germany | 17.5% |
| Brazil | 16.0% |
| United States | 14.4% |
| United Kingdom | 12.9% |
| Japan | 11.4% |
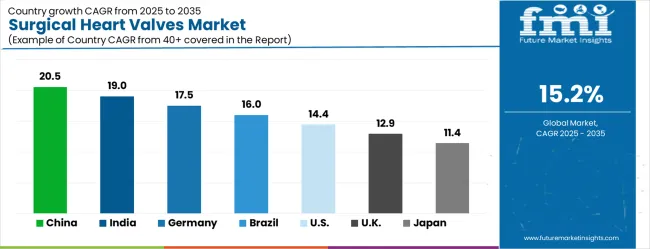
The global market is projected to grow at a CAGR of 15.2% between 2026 and 2036, driven by rising prevalence of cardiovascular diseases and increasing demand for advanced cardiac surgical procedures. China leads with 20.5% growth, supported by expanding healthcare infrastructure, rising surgical volumes, and adoption of innovative heart valve technologies. India follows at 19.0%, reflecting growing awareness of cardiovascular health, increasing hospital capacities, and advanced surgical adoption. Germany records 17.5%, driven by high standards in cardiac care and rapid integration of minimally invasive valve technologies. Brazil is projected at 16.0%, supported by improving healthcare access and rising cardiovascular procedure rates. The United States grows at 14.4% with steady demand for surgical valves and technological advancements, while the United Kingdom expands at 12.9% and Japan at 11.4%, reflecting gradual market growth and ongoing adoption of advanced cardiac interventions.
The surgical heart valves market is growing rapidly, with China leading at a 20.5% CAGR through 2036, driven by massive healthcare infrastructure development, increasing cardiovascular disease prevalence, and growing adoption of advanced cardiac interventions. India follows at 19.0%, supported by expanding healthcare access and rising awareness of transcatheter valve procedures in treating structural heart disease. Germany records strong growth at 17.5%, emphasizing clinical excellence, advanced cardiovascular care capabilities, and comprehensive healthcare coverage for cardiac interventions. Brazil grows steadily at 16.0%, integrating modern cardiac technologies into expanding healthcare systems and specialist cardiac centers. The United States shows strong growth at 14.4%, focusing on clinical innovation and expanding transcatheter procedure indications. The United Kingdom maintains robust expansion at 12.9%, supported by NHS cardiac services and clinical research programs. Japan demonstrates significant growth at 11.4%, emphasizing technological innovation and aging population cardiac care solutions.
The report covers an in-depth analysis of 40+ countries; seven top-performing countries are highlighted below.
Rapid Expansion of Surgical Heart Valves Market in China
China is projected to grow at a CAGR of 20.5% from 2026 to 2036, emerging as one of the fastest-growing surgical heart valve markets globally. Rising prevalence of cardiovascular diseases, aging population, and increasing number of cardiac surgeries are driving demand for advanced surgical valve solutions. Hospitals and specialty cardiac centers are adopting both mechanical and bioprosthetic valves to enhance patient outcomes. Local manufacturers are investing in research and development to produce high-performance, durable valves while complying with stringent medical regulations. Collaborations with international valve manufacturers are facilitating technology transfer and improving access to premium surgical solutions. Expanding health insurance coverage and rising patient awareness of minimally invasive procedures are further supporting growth in both urban and semi-urban regions.
- Over 60,000 cardiac valve procedures were performed in China in 2024
- Jiangsu Hengrui Medical expanded surgical valve production capacity by 30% in 2024
- Bioprosthetic valve adoption increased 22% year-on-year in 2024
Growth Forecast for Surgical Heart Valves Market in India
India is expected to grow at a CAGR of 19.0% from 2026 to 2036, fueled by increasing cardiovascular disease burden and expanding cardiac care infrastructure. Hospitals and specialty cardiac centers are adopting advanced mechanical and tissue valves for valve replacement surgeries. Government healthcare initiatives and rising health insurance penetration are improving accessibility for patients requiring surgical interventions. Domestic manufacturers are collaborating with international players to introduce innovative, cost-effective valve designs. Adoption is further enhanced by training programs for cardiologists and surgical teams, ensuring quality outcomes. Minimally invasive valve replacement procedures are becoming increasingly popular, especially in urban healthcare centers, boosting the market. Growth is supported by rising patient awareness, technological advancements, and expansion of private and public hospital networks across India.
- Over 35,000 valve replacement surgeries performed in 2024
- Indian manufacturers partnered with global firms to launch 10 new valve models in 2024
- Minimally invasive valve procedures grew by 18% year-on-year
Technological Advancements Driving Surgical Heart Valves Market in Germany
Germany is estimated to grow at a CAGR of 17.5% from 2026 to 2036, driven by adoption of high-performance surgical heart valves and technological innovation in cardiac care. Hospitals are increasingly preferring tissue and mechanical valves with advanced durability and biocompatibility. German manufacturers are focusing on research for improved hemodynamic performance, anti-calcification coatings, and minimally invasive deployment systems. Regulatory standards for device safety and efficacy encourage the production of high-quality valves, supporting sustained growth. The country’s well-established cardiac surgery infrastructure and growing elderly population contribute to increased procedure volumes. Collaborations between research institutes and medical device companies are fostering product innovation, ensuring Germany remains a leading market for advanced heart valve solutions in Europe.
- Germany performed over 28,000 valve replacement surgeries in 2024
- Adoption of anti-calcification tissue valves increased 15% in 2024
- Bioprosthetic valve manufacturers invested EUR 50 million in R&D in 2024
Expanding Surgical Heart Valves Market in Brazil
Brazil is projected to expand at a CAGR of 16.0% from 2026 to 2036, driven by rising cardiovascular disease incidence and increasing accessibility of cardiac surgical procedures. Adoption of both mechanical and tissue valves is growing in hospitals and specialty cardiac centers across urban and semi-urban regions. Local and international manufacturers are collaborating to provide cost-effective and durable valve solutions. Government initiatives to improve healthcare infrastructure and increase insurance coverage are supporting market growth. Minimally invasive and transcatheter procedures are increasingly implemented, reducing patient recovery time and boosting hospital adoption. Demand for valve replacement in high-risk patients, coupled with growing patient awareness, ensures steady growth across both public and private healthcare segments in Brazil.
- Over 22,000 valve replacement procedures performed in 2024
- Bioprosthetic valve adoption rose by 14% year-on-year in 2024
- Investments of USD 40 million made in advanced surgical valve production in 2024
High-Performance Surgical Heart Valves Driving Market Growth in the United States
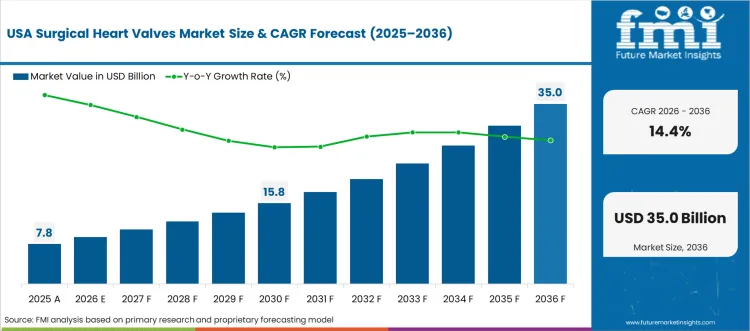
The United States is expected to grow at a CAGR of 14.4% during 2026-2036, driven by advanced healthcare infrastructure and rising prevalence of valvular heart diseases. Hospitals and specialty cardiac centers are adopting mechanical, tissue, and transcatheter valves for both adult and pediatric populations. Domestic manufacturers focus on innovative materials, minimally invasive deployment systems, and anti-calcification technology. Rising patient awareness and health insurance coverage contribute to higher procedure volumes. Adoption of transcatheter aortic valve replacement (TAVR) and mitral valve procedures is increasing steadily, offering alternatives to conventional open-heart surgeries. The USA market benefits from early adoption of medical innovations, clinical trial participation, and strong research funding in cardiovascular care.
- Over 120,000 valve replacement procedures performed in 2024
- Transcatheter valve adoption increased 20% year-on-year
- Medtronic and Edwards Lifesciences launched three new valve designs in 2024
Patient-Centric Trends Driving Surgical Heart Valves Market in the United Kingdom
The United Kingdom is projected to grow at a CAGR of 12.9% from 2026 to 2036, led by increasing demand for mechanical and tissue heart valves in hospitals and cardiac specialty centers. Adoption is driven by rising awareness of valvular heart disease, government healthcare programs, and improved access to surgical procedures. Manufacturers focus on high-performance valves with durability and biocompatibility, while import of premium transcatheter valves complements domestic offerings. Minimally invasive valve replacement procedures are gaining traction, reducing hospital stays and recovery time. Collaborations between NHS hospitals and medical device companies support clinical trials and technology adoption, reinforcing market growth. The UK market is shaped by patient-centric care, hospital modernization, and procedural innovation.
- Over 18,000 valve replacement surgeries conducted in 2024
- Minimally invasive valve replacement adoption grew by 13% in 2024
- Edwards Lifesciences and Abbott introduced new premium valve models in 2024
Surgical Heart Valve Adoption and Market Dynamics in Japan
Japan is expected to grow at a CAGR of 11.4% from 2026 to 2036, reflecting steady demand for mechanical, tissue, and transcatheter surgical heart valves. Hospitals and specialized cardiac centers are increasingly implementing minimally invasive procedures to improve patient outcomes. Domestic manufacturers focus on high-quality valve production and collaborate with international companies to ensure access to advanced valve technologies. Rising prevalence of valvular heart disease among the aging population supports consistent growth. Government healthcare programs and insurance coverage facilitate adoption, particularly in private and specialty hospitals. Patient awareness campaigns and training programs for surgical teams improve procedure efficiency and expand market acceptance. The market is characterized by technological adoption, regulatory compliance, and strong hospital infrastructure.
- Over 20,000 valve replacement procedures conducted in 2024
- Transcatheter valve adoption increased 12% year-on-year
- Sekisui Medical and Terumo Corporation expanded valve production capacity by 15% in 2024
China Leads Global Market Growth with Healthcare Infrastructure Expansion
Revenue from surgical heart valves systems in China is projected to exhibit the highest growth rate with a CAGR of 20.5% through 2036, driven by rapid healthcare infrastructure development and massive expansion of cardiovascular care capabilities across major metropolitan and regional medical centers. The country's growing elderly population and increasing cardiovascular disease prevalence are creating significant demand for advanced heart valve technologies. Major cardiac centers are establishing comprehensive structural heart disease programs to support growing patient populations and meet international care standards.
Healthcare modernization programs are supporting widespread adoption of transcatheter valve technologies across cardiovascular centers, driving demand for proven valve systems with demonstrated clinical efficacy and safety profiles.
Aging population demographics and cardiovascular disease prevalence are creating substantial opportunities for advanced valve applications in healthcare settings requiring sophisticated cardiac intervention capabilities.
India Demonstrates Strong Market Potential with Healthcare Access Expansion
Revenue from surgical heart valves systems in India is expanding at a CAGR of 19.0%, supported by increasing healthcare accessibility across diverse populations and growing investments in advanced cardiac care infrastructure. The country's expanding healthcare system and rising awareness of structural heart disease treatment options are driving demand for transcatheter valve technologies capable of treating complex patient populations. Healthcare facilities are investing in proven valve systems to improve cardiac care capabilities and expand treatment options for previously underserved patient populations.
Healthcare access expansion and cardiac care development are creating opportunities for advanced valve applications across diverse healthcare settings requiring reliable treatment solutions and clinical outcomes.
Infrastructure development programs and specialty care expansion are driving investments in modern valve technologies for cardiovascular applications throughout major medical centers and regional cardiac programs.
Germany Maintains Leadership with Clinical Excellence Standards
Demand for surgical heart valves systems in Germany is projected to grow at a CAGR of 17.5%, supported by the country's emphasis on clinical excellence and comprehensive cardiovascular care delivery standards. German cardiac centers are implementing advanced valve technologies that meet stringent clinical requirements and evidence-based treatment protocols. The market is characterized by focus on clinical outcomes, patient safety, and compliance with rigorous healthcare quality standards.
Cardiovascular excellence investments are prioritizing advanced valve technologies that demonstrate superior clinical performance and evidence-based outcomes while meeting German healthcare quality and safety standards.
Clinical research programs and evidence-based medicine initiatives are driving adoption of proven valve systems that support optimal patient outcomes and cardiovascular care excellence.
Brazil Expands Market with Cardiac Care Infrastructure Development
Revenue from surgical heart valves systems in Brazil is growing at a CAGR of 16.0%, driven by expanding cardiac care infrastructure and increasing adoption of transcatheter valve technologies across public and private healthcare sectors. The country's developing cardiovascular care capabilities are investing in advanced valve solutions to improve patient access to modern cardiac interventions. Healthcare facilities are adopting proven valve technologies to support growing cardiac patient populations and evolving treatment standards.
Cardiac care infrastructure expansion and healthcare system development are facilitating adoption of advanced valve technologies capable of supporting improved cardiovascular outcomes in expanding healthcare environments.
Healthcare modernization initiatives and specialty care development are driving demand for reliable valve solutions that meet evolving clinical standards and patient care requirements.
United States Focuses on Clinical Innovation and Procedural Excellence
Demand for surgical heart valves systems in the United States is expanding at a CAGR of 14.4%, driven by ongoing clinical innovation and comprehensive cardiovascular care programs across healthcare systems. Cardiac centers are investing in next-generation valve technologies that demonstrate superior clinical outcomes and expanded treatment indications. The market benefits from robust clinical research infrastructure and evidence-based adoption programs supporting advanced cardiovascular interventions.
Clinical innovation and cardiovascular excellence programs are driving adoption of advanced valve systems that demonstrate superior clinical benefits and evidence-based treatment advantages.
Healthcare quality initiatives and outcome-based care programs are supporting adoption of valve technologies that provide measurable improvements in patient outcomes and cardiovascular care delivery.
United Kingdom Emphasizes NHS Cardiac Services and Clinical Research
Demand for surgical heart valves systems in the United Kingdom is projected to grow at a CAGR of 12.9%, supported by comprehensive NHS cardiac services and national clinical research programs focused on cardiovascular innovation. Healthcare providers are implementing evidence-based valve technologies that provide consistent clinical outcomes and meet established treatment guidelines. The market is characterized by focus on clinical effectiveness, cost-effectiveness, and compliance with national healthcare standards.
NHS cardiac service initiatives and clinical excellence programs are supporting adoption of advanced valve systems that meet contemporary clinical standards and evidence-based treatment requirements.
Clinical research and healthcare innovation programs are creating demand for proven valve technologies that provide reliable clinical outcomes and healthcare system benefits.
Japan Demonstrates Technological Innovation and Aging Care Excellence
Demand for surgical heart valves systems in Japan is expanding at a CAGR of 11.4%, driven by the country's emphasis on technological innovation and comprehensive cardiac care solutions for aging populations. Japanese manufacturers and healthcare providers are developing advanced valve technologies that address the specific needs of elderly patients and complex cardiac conditions. The market benefits from focus on innovation, precision engineering, and continuous improvement in cardiovascular care delivery.
Aging population care programs and technological innovation initiatives are driving development of advanced valve systems that address specific cardiovascular needs of elderly patients and complex disease management.
Medical device innovation and precision healthcare programs are supporting adoption of high-quality valve solutions that optimize clinical outcomes and cardiovascular care effectiveness.
Europe Market Split by Country
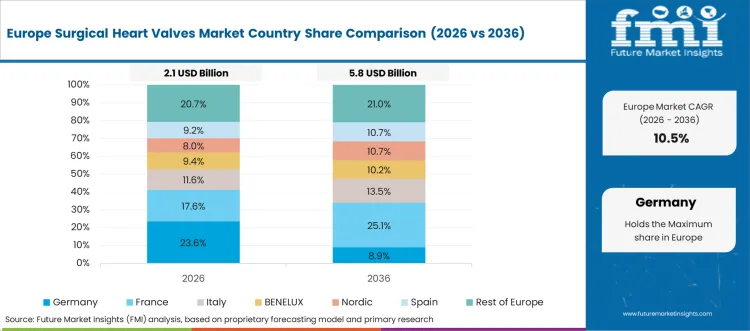
The surgical heart valves market in Europe is projected to grow from USD 2,399.5 million in 2026 to USD 9,873.8 million by 2036, registering a CAGR of 15.2% over the forecast period. Germany is expected to maintain its leadership with a 28.4% share in 2026, supported by its comprehensive cardiovascular healthcare system and advanced cardiac care infrastructure. The United Kingdom follows with 17.8% market share, driven by NHS cardiac services and clinical research programs. France holds 15.6% of the European market, benefiting from healthcare system modernization and cardiovascular care expansion. Italy and Spain collectively represent 23.2% of regional demand, with growing focus on transcatheter valve procedures and cardiac care development. The Rest of Europe region accounts for 15.0% of the market, supported by healthcare development in Eastern European countries and Nordic cardiovascular care systems.
Competitive Landscape of Surgical Heart Valves Market
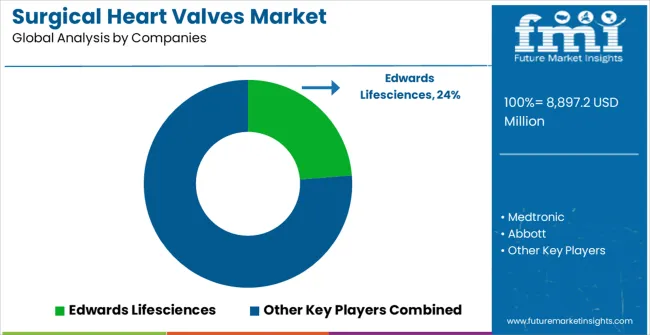
The market is highly competitive, driven by innovation, clinical reliability, and procedural efficiency. Edwards Lifesciences, Medtronic, and Abbott dominate with advanced transcatheter and mechanical valve technologies, focusing durability, biocompatibility, and minimally invasive delivery systems. Their competitive strength lies in extensive clinical data, global regulatory approvals, and comprehensive training programs for cardiologists and cardiac surgeons. Brochures highlight long-term hemodynamic performance, reduced complication rates, and compatibility with diverse patient anatomies, positioning these firms as leaders in both surgical and transcatheter valve replacement procedures.
Boston Scientific, Venus Medtech, and Meril Life Sciences compete by offering novel transcatheter heart valve systems with cost-effective solutions and tailored designs for emerging markets. Venus Medtech emphasizes innovation in self-expanding valves, while Meril Life Sciences focuses on accessibility and regional support, targeting hospitals seeking reliable yet affordable alternatives. Boston Scientific strengthens its position through integration with digital imaging and delivery systems, enhancing procedural precision and patient outcomes.
Braile Biomedica and other regional players differentiate through localized production, niche device designs, and flexible customer support. Product brochures emphasize valve durability, ease of implantation, and post-procedural safety. Competition is defined by clinical efficacy, procedural efficiency, and regulatory compliance. Companies invest in research, next-generation valve designs, and minimally invasive delivery technologies, while regional players leverage affordability and proximity to clients. Continuous innovation in material science, valve design, and implantation methods remains central to sustaining leadership in the market.The surgical heart valves market is defined by competition among established medical device manufacturers, specialized cardiac technology companies, and innovative cardiovascular solution providers. Companies are investing in advanced valve designs, clinical evidence development, physician training programs, and comprehensive patient support services to deliver safe, effective, and clinically superior valve solutions. Strategic partnerships, clinical research, and geographic expansion are central to strengthening product portfolios and market presence.
Edwards Lifesciences, operating globally, offers comprehensive transcatheter heart valve solutions with focus on clinical innovation, evidence generation, and physician education programs. Medtronic provides advanced structural heart technologies with emphasis on comprehensive valve portfolio and clinical support services. Abbott, multinational healthcare leader, delivers innovative cardiovascular solutions with focus on transcatheter valve advancement and patient outcomes. Boston Scientific offers specialized heart valve technologies with proven clinical performance and comprehensive training support.
Venus Medtech provides innovative transcatheter valve systems with emphasis on Asian market applications and clinical development. Meril Life Sciences delivers comprehensive cardiovascular technologies with focus on emerging market access and clinical evidence development. Braile Biomedica offers specialized heart valve solutions with regional expertise and clinical support capabilities.
Global Surgical Heart Valves Market -- Stakeholder Contribution Framework
The surgical heart valves market underpins cardiovascular care advancement, patient outcome optimization, minimally invasive treatment development, and healthcare quality improvement. With clinical evidence requirements, regulatory compliance mandates, and demand for improved patient outcomes, the sector must balance innovation leadership, clinical safety, and healthcare accessibility. Coordinated contributions from governments, healthcare systems, manufacturers, clinical organizations, and investors will accelerate the transition toward evidence-based, patient-centered, and clinically optimized heart valve technologies.
How Governments Could Spur Market Growth and Adoption
- Healthcare Access Incentives: Provide reimbursement coverage or healthcare funding for transcatheter valve procedures, particularly for high-risk patient populations and underserved communities.
- Clinical Standards: Establish cardiovascular care guidelines, quality metrics, and safety requirements, boosting adoption of evidence-based valve technologies and procedural excellence.
- Healthcare Programs: Embed advanced valve technologies into national cardiovascular health initiatives, aging population care programs, and specialty cardiac care development.
- Medical Device Regulation: Streamline approval processes for innovative valve technologies while maintaining rigorous safety and efficacy standards.
- Research Support: Recognize cardiovascular innovation within national healthcare research and medical technology advancement frameworks.
How Healthcare Systems Could Support Market Development
- Clinical Protocol Development: Create standardized care pathways, evidence-based treatment protocols, and comprehensive training programs for transcatheter valve procedures.
- Quality Metrics: Establish outcome measurements for procedural success rates, patient safety indicators, and long-term clinical effectiveness metrics.
- Professional Education: Develop specialized training programs for interventional cardiologists and cardiac surgeons on advanced valve technologies and procedural techniques.
- Evidence Generation: Support clinical research, registry development, and real-world evidence collection to demonstrate valve technology effectiveness.
How Manufacturers Could Strengthen the Ecosystem
- Clinical Innovation: Embed advanced materials, innovative valve designs, and precision delivery systems into heart valve products for enhanced clinical performance.
- Research Collaboration: Partner with leading cardiac centers to conduct clinical trials, develop evidence-based solutions, and advance valve technology innovation.
- Technology Advancement: Advance valve engineering, delivery system design, and procedural optimization technologies for improved clinical outcomes and procedural efficiency.
- Clinical Support: Bundle valves with comprehensive physician training, patient education, and clinical support services for successful program implementation.
How Clinical Organizations Could Navigate Opportunities
- Evidence Development: Conduct clinical studies, outcomes research, and comparative effectiveness studies to support evidence-based valve selection and procedural optimization.
- Practice Guidelines: Develop clinical practice guidelines, treatment protocols, and safety recommendations for transcatheter valve procedures and patient selection.
- Education Programs: Provide professional education, procedural training, and clinical mentorship to expand transcatheter valve expertise and procedural volumes.
- Quality Improvement: Establish benchmarking programs, best practice sharing, and continuous improvement initiatives for valve procedure outcomes.
How Investors and Financial Enablers Could Unlock Value
- Healthcare Innovation: Fund research and development of next-generation valve technologies, delivery system innovations, and clinical outcome optimization solutions.
- Market Access: Support market expansion initiatives, reimbursement advocacy, and healthcare system adoption programs in emerging cardiovascular markets.
- Clinical Evidence: Back clinical research programs, regulatory studies, and health economic research supporting valve technology adoption and procedural expansion.
- Technology Development: Invest in startups developing novel valve materials, smart monitoring systems, or AI-powered procedural guidance technologies.
- Global Expansion: Provide financing and infrastructure support for global manufacturing and distribution optimization to improve patient access to advanced valve technologies.
Key Players in the Surgical Heart Valves Market
- Edwards Lifesciences
- Medtronic
- Abbott
- Boston Scientific
- Venus Medtech
- Meril Life Sciences
- Braile Biomedica
Scope of the report
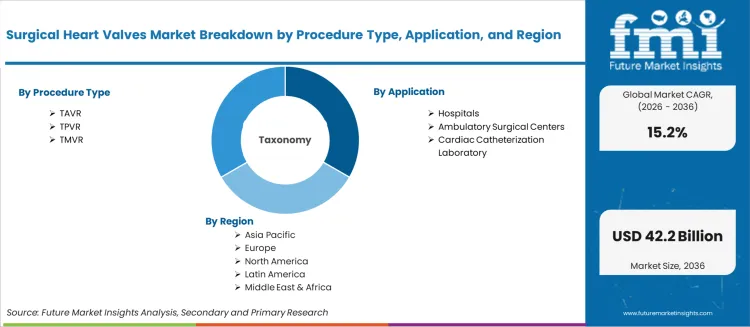
| Metric | Value |
|---|---|
| Quantitative Units | USD 10.24 billion to USD 42.16 billion, at a CAGR of 15.2% |
| Market Definition | Surgical Heart Valves encompasses prosthetic valve devices designed to replace or repair damaged cardiac valves. The market covers transcatheter valve systems (TAVR, TPVR, TMVR) and surgical replaceme... |
| Procedure Type Segmentation | TAVR, TPVR, TMVR |
| Application Segmentation | Hospitals, Ambulatory Surgical Centers, Cardiac Catheterization Laboratory |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | China, India, Germany, Brazil, USA, UK, Japan, and 40 plus countries |
| Key Companies Profiled | Edwards Lifesciences, Medtronic, Abbott, Boston Scientific, Venus Medtech, Meril Life Sciences, and Braile Biomedica |
| Forecast Period | 2026 to 2036 |
| Approach | Forecasting models apply a bottom-up methodology starting with global installed base metrics and projecting the conversion rate to advanced product adoption. |
Surgical Heart Valves Market by Segments
Procedure Type:
- TAVR
- TPVR
- TMVR
Application:
- Hospitals
- Ambulatory Surgical Centers
- Cardiac Catheterization Laboratory
Region:
- North America
- United States
- Canada
- Mexico
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- East Asia
- China
- Japan
- South Korea
- South Asia & Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia & Pacific
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- 1. World Health Organization. (2024). Global Health Expenditure Database. WHO.
- 2. USA Food and Drug Administration. (2024). Medical Device Premarket Approval Database. FDA.
- 3. European Commission. (2024). Regulation (EU) 2017/745 on Medical Devices, consolidated text. EUR-Lex.
- 4. Organisation for Economic Co-operation and Development. (2024). Health at a Glance 2024: OECD Indicators. OECD Publishing.
- 5. The World Bank. (2024). World Development Indicators: Health Systems. World Bank Group.
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
Frequently Asked Questions
How large is the demand for Surgical Heart Valves in the global market in 2026?
Demand for surgical heart valves in the global market is estimated to be valued at USD 10.24 billion in 2026.
What will be the market size of Surgical Heart Valves in the global market by 2036?
Market size for surgical heart valves is projected to reach USD 42.16 billion by 2036.
What is the expected demand growth for Surgical Heart Valves between 2026 and 2036?
Demand for surgical heart valves is expected to grow at a CAGR of 15.2% between 2026 and 2036.
Which Procedure Type is poised to lead global sales by 2026?
TAVR accounts for 52.0% share in 2026 as it addresses the primary demand requirements across global markets.
How significant is the role of Hospitals in driving surgical heart valves adoption in 2026?
Hospitals represents 68.0% of segment share as end-user demand consolidates around established application pathways.
What is driving demand in China?
China is projected to grow at a CAGR of 20.5% during 2026 to 2036, driven by expanding infrastructure investment and regulatory modernization.
What does market forecast mean on this page?
The market forecast represents a model-based projection built on defined industry and supply chain assumptions for strategic planning purposes.
How does FMI build and validate the surgical heart valves forecast?
Forecasting models apply a bottom-up methodology starting with global installed base metrics and cross-validate projections against quarterly industry revenue volumes.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Procedure Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Procedure Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Procedure Type , 2026 to 2036
- TAVR
- TPVR
- TMVR
- TAVR
- Y to o to Y Growth Trend Analysis By Procedure Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Procedure Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Hospitals
- Ambulatory Surgical Centers
- Cardiac Catheterization Laboratory
- Hospitals
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Procedure Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Procedure Type
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Procedure Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Procedure Type
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Procedure Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Procedure Type
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Procedure Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Procedure Type
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Procedure Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Procedure Type
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Procedure Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Procedure Type
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Procedure Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Procedure Type
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Procedure Type
- By Application
- Competition Analysis
- Competition Deep Dive
- Edwards Lifesciences
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Medtronic
- Abbott
- Boston Scientific
- Venus Medtech
- Meril Life Sciences
- Braile Biomedica
- Edwards Lifesciences
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Procedure Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Procedure Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Procedure Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Procedure Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Procedure Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Procedure Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Procedure Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Procedure Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Procedure Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Procedure Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Procedure Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Procedure Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Procedure Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Procedure Type
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Procedure Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Procedure Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Procedure Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Procedure Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Procedure Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Procedure Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Procedure Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Procedure Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Procedure Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Procedure Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Procedure Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Procedure Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Procedure Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Procedure Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Procedure Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Procedure Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Procedure Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Procedure Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

