Surgical Tourniquet Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
This report examines the surgical tourniquet market in detail, addressing market size, revenue estimates, competitive positioning, demand outlook, drivers of growth, market barriers, product development trends, supply chain dynamics, and investment opportunities.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Surgical Tourniquet Market size, market forecast and outlook by FMI

The surgical tourniquet market was valued at USD 573.40 million in 2025, projected to reach USD 603.22 million in 2026, and is forecast to expand to USD 1001.45 million by 2036 at a 5.2% CAGR. Increasing orthopedic procedure volumes, particularly knee arthroplasty and limb reconstruction surgeries, are sustaining baseline tourniquet consumption across hospital operating rooms. Trauma care protocol standardization in emergency departments is expanding pneumatic tourniquet deployment beyond elective surgical settings into acute hemorrhage control workflows.
Summary of Surgical Tourniquet Market
- Market Snapshot
- The surgical tourniquet market is valued at USD 573.40 million in 2025 and is projected to reach USD 1001.45 million by 2036.
- The industry is expected to grow at a 5.2% CAGR from 2026 to 2036, creating an incremental opportunity of USD 398.24 million.
- The market remains a surgical consumable and capital equipment hybrid where orthopedic procedure volumes, trauma care protocol compliance, and pneumatic system reliability define competitive dynamics.
- Demand and Growth Drivers
- Demand is sustained by rising knee arthroplasty and limb reconstruction surgical volumes that require pneumatic tourniquet application as a standard procedural step.
- Trauma care protocol standardization in emergency departments is expanding tourniquet deployment from elective surgical settings into acute hemorrhage management workflows.
- Growth is further supported by disposable cuff replacement cycles that generate recurring procurement tied directly to surgical case counts across orthopedic departments.
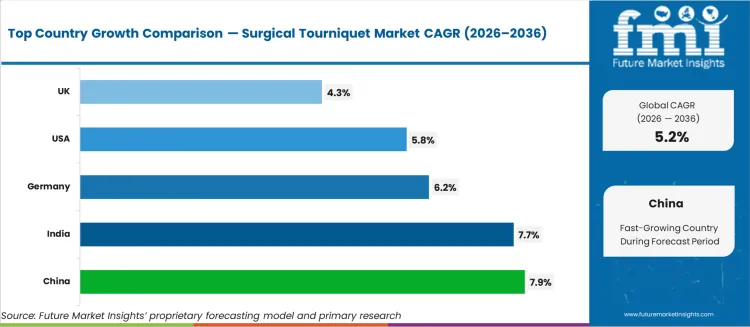
- Among key countries, China leads at 7.9% CAGR, India at 7.7%, Germany at 6.2%, USA at 5.8%, UK at 4.3%.
- Product and Segment View
- The market includes tourniquet cuffs (disposable and reusable) and tourniquet systems (pneumatic control units) used across knee arthroplasty, limb amputation, plastic surgery, and trauma care applications.
- Tourniquet cuffs leads by Product with 78.4% share in 2026.
- Knee arthroplasty leads by Application with 33.1% share in 2026.
- Scope includes Product, Application segmentation with regional coverage across seven global regions.
- Geography and Competitive Outlook
- China and India are the fastest-growing markets, while North America and Europe remain mature demand bases.
- Competition is shaped by product quality, distribution reach, and regulatory compliance, with key players including Stryker Corporation, Zimmer Biomet, Ulrich GmbH & Co.KG, AneticAid Ltd., Hammarplast Medical AB, Anetic Aid, VBM Medizintechnik GmbH, Delfi Medical Innovations, Daesung Maref, OHK Medical Devices, and Dessillons & Dutrillaux.
Based on FMI's report, the surgical tourniquet sector operates as a consumable and capital equipment hybrid market. Tourniquet cuffs represent the dominant revenue segment as disposable cuff replacement cycles generate recurring procurement volumes tied directly to surgical case counts. Tourniquet systems, comprising pneumatic control units and monitoring displays, contribute capital equipment revenues on longer replacement cycles. Knee arthroplasty procedures account for the largest application share, followed by amputation, plastic surgery, and trauma applications that collectively distribute demand across orthopedic and emergency surgical departments.
Country-level growth trajectories track orthopedic surgical volume expansion and trauma care infrastructure development. China at 7.9% CAGR leads as hospital construction programs embed pneumatic tourniquet systems into new operating room specifications. India follows at 7.7%, driven by expanding joint replacement procedure volumes in private hospital chains. Germany at 6.2% benefits from high orthopedic surgical density and capital equipment refresh cycles. USA at 5.8% and UK at 4.3% sustain steady replacement demand for tourniquet cuffs tied to elective surgery schedules.
Surgical Tourniquet Market key takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 603.22 million |
| Industry Value (2036) | USD 1001.45 million |
| CAGR (2026 to 2036) | 5.2% |
Source: Future Market Insights, 2026
Surgical Tourniquet Market definition
Surgical Tourniquet encompasses pneumatic limb occlusion devices used to create a bloodless surgical field during orthopedic, trauma, and plastic surgery procedures. The market covers tourniquet cuffs (disposable and reusable bladder assemblies) and tourniquet systems (electronic pneumatic control units with pressure monitoring). Primary applications include knee arthroplasty, limb amputation, reconstructive plastic surgery, and acute trauma hemorrhage control across hospital operating rooms and emergency departments.
Surgical Tourniquet Market inclusions
Market scope includes global and regional market sizes, a 2026 to 2036 forecast period, segment breakdowns by Product, Application, and regional analysis across North America, Latin America, Europe, East Asia, South Asia, Oceania, and Middle East and Africa. Pricing trend analysis and competitive positioning assessments are incorporated within the analytical framework.
Surgical Tourniquet Market exclusions
The scope excludes improvised field tourniquets, elastic bandage tourniquets without pneumatic regulation, and blood pressure cuffs not designed for surgical limb occlusion. Hemostatic agents and surgical drain systems are also excluded.
Surgical Tourniquet Market research methodology
- Primary Research: Analysts engaged with orthopedic department heads, operating room equipment managers, and trauma surgery coordinators to map tourniquet procurement cycles and product selection criteria.
- Desk Research: Data collection aggregated clinical practice guidelines, regulatory device approval databases, hospital procedure volume registries, and medical society publications relevant to surgical instrument and consumable markets.
- Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of product-level revenues, applying region-specific adoption curves to project future demand velocity.
- Data Validation and Update Cycle: Projections are tested against publicly reported expenditure guidance from leading industry participants and cross-referenced with regulatory filings.
Key Industry Highlights
Modern Technologies and Regulatory Approvals to Drive Growth
A number of public health initiatives have emphasized the importance of controlling bleeding during accidents and trauma in order to reduce mortality and morbidity. As obesity and its associated bariatric surgery prevalence increases, tourniquets are being used to manage blood flow during bariatric surgeries.
In addition to government funding and the development of healthcare infrastructures, the industry is also benefitting from the expansion of healthcare infrastructure.
In clinical settings, tourniquets are approved by regulatory agencies and certified for effectiveness and safety. As tourniquets become more accepted and approved, the industry is expected to grow. The introduction of modern technologies, such as Bluetooth-enabled tourniquets, can also be credited with making the industry more advanced.
Increasing demand for patient-specific tourniquet solutions, which are tailored to individual requirements and needs, is increasing due to their success in improving outcomes.
An increase in postoperative rehabilitation outcomes is associated with the use of tourniquets, particularly in orthopedic and limb surgery. As a result of these positive correlations, their use is increased.
Advanced Trauma Care to Provide Lucrative Opportunities
In recent years, advances in biomedical engineering have improved the ability to customize tourniquet systems according to surgeons and procedures, improving safety and efficiency.
A number of advancements in tourniquet technology have produced improved cuffs capable of creating a safe, low-pressure gradient on the underlying patient limbs, as well as systems capable of applying pressure automatically for a short period of time.
The popularity of aesthetic procedures and best-in-class trauma care drive demand in the market. Through the use of surgery tourniquets, surgeons can minimize perioperative blood loss and optimize antibiotic delivery.
A number of new tourniquet inflation devices, such as S-MART, aim to improve patient safety and reduce complications associated with tourniquet use. Trials and studies with a larger sample size will be conducted in order to prove the efficacy of these products.
Surgical Tourniquets Demand Side Trends
The healthcare industry is evolving, directed by catalysts like population growth, aging population, and innovation in medical technology. Within the industry, the demand for surgical tourniquets upswing as surgical processes become pervasive.
Adherence to regulatory standards is essential in the medical device sector. The vendors should be informed about shifts in rules and standards to warrant that products satisfy the essential needs of new entrants.
Training medical care professionals regarding the advantages and proper adoption of operative tourniquets is vital. The manufacturers can create learning materials or training projects to ensure that users comprehend the best activities and health precautions.
Tourniquet adoption during extremities surgery became widespread in orthopedics, as per a paper published in July 2025 in the Journal of Experimental Orthopedics. Around 90% of surgeons in the United States and the United Kingdom are thought to use tourniquets when doing total knee arthroplasty regularly.
Factors Adversely Affecting Surgical Tourniquet Industry Size Expansion
Economic fluctuations in medical care expenditure impede the growth for the market. The industry players should be prepared to traverse through periods of economic unreliability and modify their strategies correspondingly.
Adherence to strict regulatory standards and shifting guidelines are challenging for vendors functioning in the sector. Compliance with the regulations needs considerable investments in research, development, and quality control procedures.
As with any medical equipment, there are inherent threats related to the implementation of tourniquets in surgeries. The producers invest in extensive liability insurance and administer rigorous quality assurance standards to curb the hazard of product-related occurrences and legal actions.
2021 to 2025 Global Surgical Tourniquet Sales Outlook Compared to Demand Forecast from 2026 to 2036
The industry has witnessed notable growth from 2021 to 2025, ushered by factors like a spur in surgical processes, improvement in medical technology, and surging consciousness regarding patient safety. This period experienced an upsurge in demand, with prominent vendors launching novel products to satisfy the developing requirements of healthcare providers.
Between 2026 and 2036, the demand for surgical tourniquets is on an upward trajectory. Catalysts aiding this growth are the pervasiveness of chronic disorders compelling surgical interventions, proliferating healthcare institutions in developing countries, and an aging population needing surgical interventions.
Developments in surgical procedures and a pressing focus on patient safety augment the adoption of these devices. The soaring investments in healthcare research and development and the launch of advanced tourniquet systems stimulate the growth during the forecast period.
The industry is slated for considerable growth between 2026 and 2036, proffering lucrative opportunities for providers to cash on the evolving demand.
Market Concentration
A significant 28.7% share of the global market has been captured by companies in the Tier 1 category. Tourniquets in surgical procedures are part of the product offerings of these well-established medical device companies. With their strong financial positions, distribution channels, and research and development resources, they have an edge in the market.
Their innovative capabilities and ability to meet the diverse needs of customers around the world indicate that they will remain at the forefront. Prominent players include Stryker Corporation and Zimmer Biomet.
Tier 2 companies represent a 34.2% share of the industry is higher than that of tier 1 companies, demonstrating a strong presence overseas and an in-depth understanding of the industry. Surgical tourniquet companies in Tier 2 are specialized in this area.
Quality and innovation must remain at the forefront of this niche sectors. While niche players command a relatively small share of the market, they have a larger reach and financial clout than Tier 1 players. Their specialization and targeted approach enable them to deliver excellent results. Prominent tier 2 players include VBM Medizintechnik and Delfi Medical Innovations.
Most small firms in Tier 3 are regionally dominant and specialize in a narrow range of products. Offering a broad range of surgical products that are tailored to specific customer requirements in order to offer cost-effective solutions.
Players will perform exponentially in the industry if they comply with international standards and stringent regulations. Players include Ulrich Medical, Anetic Aid, Hammarplast Medical, Daesung Maref, OHK Medical Devices, and Dessillons & Dutrillaux.
Country-wise Analysis
The section below covers the forecast for emerging markets for surgical tourniquets in terms of countries. Information on key countries in several parts of the globe, including North America, Asia Pacific, Europe, and others, is provided.
The United States is anticipated to remain at the forefront in North America, with a CAGR of 5.8% through 2036. In Asia Pacific region, India experiences a CAGR of 7.7% by 2036.
.webp)
| Countries | CAGR 2026 to 2036 |
|---|---|
| United States | 5.8% |
| Germany | 6.2% |
| United Kingdom | 4.3% |
| India | 7.7% |
| China | 7.9% |
Growth Factors for The Surgical Tourniquet Market in the United States
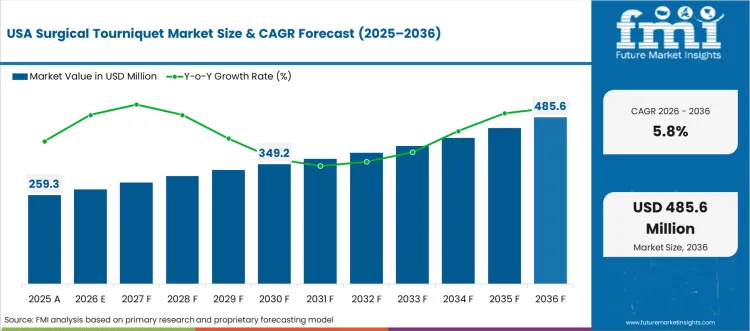
The United States is encouraged by modern medical care facilities and a strong demand for innovative healthcare devices. The flourishing consciousness concerning the advantages of minimally invasive surgical methods assists in the growth of the market.
The role of regulatory bodies in governing the surgical tourniquet sector market ushers the adoption of tourniquet technologies in the United States.
The pervasiveness of chronic illnesses and the inclining geriatric population ushered in the demand for the market in the United States. Technological innovations and partnerships among essential vendors advance the industry expansion in the United States.
Surgical Tourniquet Industry Growth and Trends in Germany
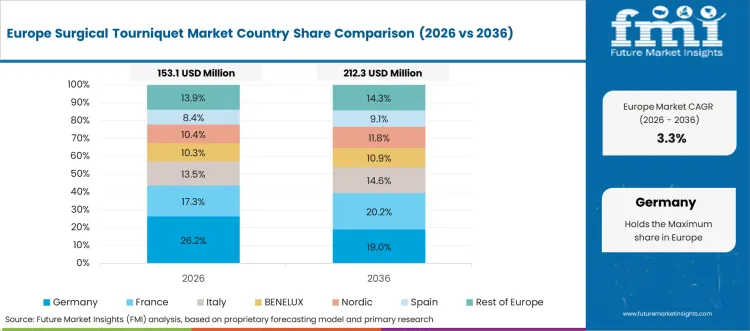
The market in Germany thrives on a focus on accuracy and quality, indicating the impact of new technologies like artificial intelligence on the market. The strict rules and emphasis on product efficiency direct innovation and expansion in Germany's medical tourniquet market.
Germany experiences growth amplified by a strengthening number of surgeries and proliferating investments in healthcare facilities. The commitment to patient safety and high standards of healthcare delivery in Germany stimulate the growth avenue of the market.
Future Outlook of The Surgical Tourniquet Market in India
The market in India witnessed steady growth characterized by a proliferating healthcare system and widening medical tourism. The adoption of digital health tech and telemedicine platforms augments the business in India, particularly in remote regions with constrained access to medical care amenities.
The disposable and environmental sustainability concerns catapult the use of the instruments in India. The towering investments in healthcare units and government schemes to encourage medical device production bolster the growth in the market.
Category-wise Outlook
The section contains information about the leading segments in the surgical tourniquet industry. In terms of product type category, the tourniquet cuffs segment garners a share of 78.4% by 2036. By application category, the knee arthroplasty segment takes precedence with a share of 33.1% in 2036.
Tourniquet Cuffs' Essential Role in Healthcare
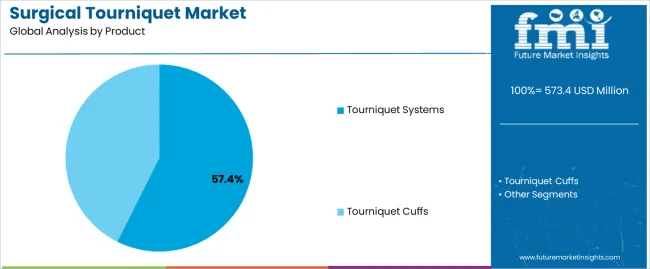
| Segment | Tourniquet Cuffs |
|---|---|
| Value Share (2036) | 78.4% |
Tourniquet cuffs are usually cheaper as compared to complete tourniquet systems, making them widely available and easier to purchase. They are replaced frequently because of wear and hygiene rules, spurring significant sales.
Hospitals and surgical centers prioritize storing tourniquet cuffs because of their vital role and standard demand in surgeries. Cuffs are used with diverse tourniquet systems, improving their flexibility and demand. As the number of surgeries escalates, the demand for tourniquet cuffs also rises, resulting in a steady boom in sales.
Enhanced Joint Replacement with Reduced Operating Time and Minimal Blood Loss to Drive Demand
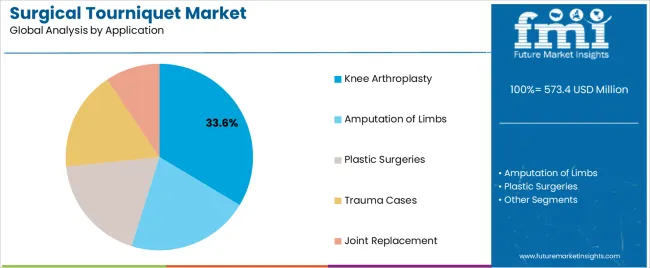
| Segment | Knee Replacement |
|---|---|
| Value Share (2036) | 33.1% |
A tourniquet is a widely accepted and effective practice during knee replacement surgery. In addition to preventing blood loss, it improves the visibility of the wound and stops the bleeding. The use of tourniquets during joint arthroplasty surgery further prevents bleeding from resurfaced bones, so bone-implant cementation can proceed more successfully.
Tourniquets may improve the long-term survival of knee implants by reducing bleeding from porous bone ends, thereby improving the bond between the soft cement and the bone. The tourniquet can reduce the frequency of suctioning and reduce complication risk during surgery.
Competition Outlook
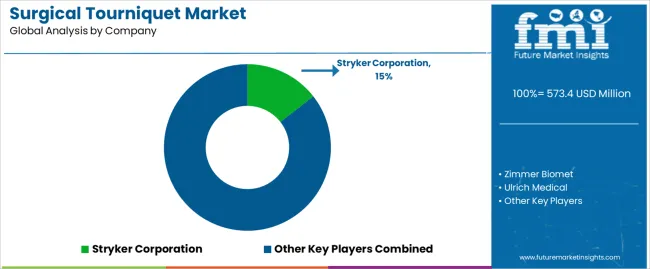
Key surgical tourniquet vendors use promotional and distribution procedures, like direct sales to medical care experts and sales to public health facilities. To keep up with demand globally, surgical tourniquet providers are strengthening their manufacturing capacity.
The majority of the leading producers are not only meeting the demand but also innovating. They are focusing on locating and making advantageous technologies and targeted products from internal as well as external sources.
This active approach is targeted at satisfying the unsatisfied demand in emerging economies, sustaining a positive sense of the industry's future. The manufacturers adopt partnerships and acquisitions as their primary strategies to intensify sales across regions.
Investment Analysis of Specific Surgical Tourniquet Companies
| Company | Stryker Corporation |
|---|---|
| Headquarter | United States |
| Recent Advancement | In February 2025, Stryker announced the acquisition of Vocera Communications, Inc. which is a pioneer in digital care collaboration and communication. |
| Company | Zimmer Biomet |
|---|---|
| Headquarter | United States |
| Recent Advancement | Zimmer Biomet Holdings, Inc. announced the acquisition of Embody, Inc. in January 2025, which is focused on soft tissue healing, for USD 155 million at closing and an additional USD 120 million determined to achieve regulatory and commercial milestones over three years. The acquisition is cumulative to the revenue growth and moderately dilutive to earnings per share. |
| Company | Ulrich GmbH & Co.KG |
|---|---|
| Headquarter | Germany |
| Recent Advancement | Bracco and Ulrich GmbH Co KG, a popular medical device producer specializing in media injectors and spinal implants, announced a long-term partnership in November 2025 that brings a Bracco-branded modern MR injector to the United States. |
Scope of the report
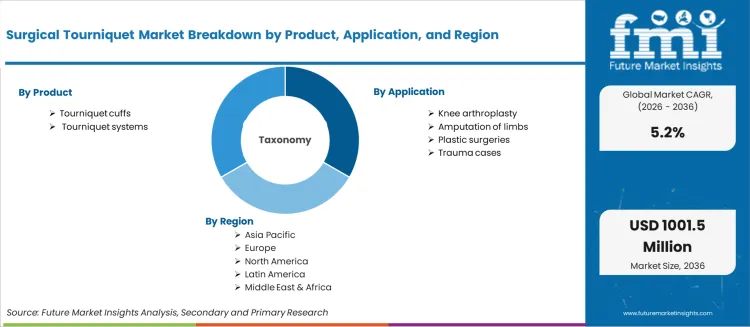
| Metric | Value |
|---|---|
| Quantitative Units | USD 603.22 million to USD 1001.45 million, at a CAGR of 5.2% |
| Market Definition | Surgical Tourniquet encompasses pneumatic limb occlusion devices used to create a bloodless surgical field during orthopedic, trauma, and plastic surgery procedures. The market covers tourniquet cuffs... |
| Product Segmentation | Tourniquet cuffs, Tourniquet systems |
| Application Segmentation | Knee arthroplasty, Amputation of limbs, Plastic surgeries, Trauma cases |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | China, India, Germany, USA, UK, and 40 plus countries |
| Key Companies Profiled | Stryker Corporation, Zimmer Biomet, Ulrich GmbH & Co.KG, AneticAid Ltd., Hammarplast Medical AB, Anetic Aid, VBM Medizintechnik GmbH, Delfi Medical Innovations, Daesung Maref, OHK Medical Devices, and Dessillons & Dutrillaux |
| Forecast Period | 2026 to 2036 |
| Approach | Forecasting models apply a bottom-up methodology starting with global installed base metrics and projecting the conversion rate to advanced product adoption. |
Key Segments of Surgical Tourniquet Market Report
By Product:
The industry is bifurcated into tourniquet systems and tourniquet cuffs. The tourniquet systems category is further bifurcated into single channel systems and dual channel systems.
The tourniquet cuffs category is further bifurcated into inflatable cuffs and non-inflatable cuffs. The inflatable cuffs sub-segment in further bifurcated into disposable cuffs and reusable cuffs. The non-inflatable cuffs sub-segment in further bifurcated into disposable cuffs and reusable cuffs
By Application:
The application category is classified into knee arthroplasty, amputation of limbs, plastic surgeries, and trauma cases.
By End-User:
The end user category is trifurcated into hospitals, ambulatory surgical centers, and specialized clinics.
Bibliography
- 1. World Health Organization. (2024). Global Health Expenditure Database. WHO.
- 2. USA Food and Drug Administration. (2024). Medical Device Databases. FDA.
- 3. European Commission. (2024). Regulation (EU) 2017/745 on Medical Devices, consolidated text. EUR-Lex.
- 4. Organisation for Economic Co-operation and Development. (2024). Health at a Glance 2024: OECD Indicators. OECD Publishing.
- 5. The World Bank. (2024). World Development Indicators: Health Systems. World Bank Group.
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
Frequently Asked Questions
How large is the demand for Surgical Tourniquet in the global market in 2026?
Demand for surgical tourniquet in the global market is estimated to be valued at USD 603.22 million in 2026.
What will be the market size of Surgical Tourniquet in the global market by 2036?
Market size for surgical tourniquet is projected to reach USD 1001.45 million by 2036.
What is the expected demand growth for Surgical Tourniquet between 2026 and 2036?
Demand for surgical tourniquet is expected to grow at a CAGR of 5.2% between 2026 and 2036.
Which Product is poised to lead global sales by 2026?
Tourniquet cuffs accounts for 78.4% share in 2026 as it addresses the primary demand requirements across global markets.
How significant is the role of Knee arthroplasty in driving surgical tourniquet adoption in 2026?
Knee arthroplasty represents 33.1% of segment share as end-user demand consolidates around established application pathways.
What is driving demand in China?
China is projected to grow at a CAGR of 7.9% during 2026 to 2036, driven by expanding infrastructure investment and regulatory modernization.
What does market forecast mean on this page?
The market forecast represents a model-based projection built on defined industry and supply chain assumptions for strategic planning purposes.
How does FMI build and validate the surgical tourniquet forecast?
Forecasting models apply a bottom-up methodology starting with global installed base metrics and cross-validate projections against quarterly industry revenue volumes.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2026 to 2036
- Tourniquet cuffs
- Tourniquet systems
- Tourniquet cuffs
- Y to o to Y Growth Trend Analysis By Product , 2021 to 2025
- Absolute $ Opportunity Analysis By Product , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Knee arthroplasty
- Amputation of limbs
- Plastic surgeries
- Trauma cases
- Knee arthroplasty
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Application
- Competition Analysis
- Competition Deep Dive
- Stryker Corporation
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Zimmer Biomet
- Ulrich GmbH & Co.KG
- AneticAid Ltd.
- Hammarplast Medical AB
- Anetic Aid
- VBM Medizintechnik GmbH
- Delfi Medical Innovations
- Daesung Maref
- OHK Medical Devices
- Dessillons & Dutrillaux
- Stryker Corporation
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Product
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Product
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Product
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

