Tumor mutational burden market is forecasted to reach USD 2,081.0 million in 2026 and expand to USD 6,757.7 million by 2036, at a CAGR of 12.5%. Value expansion reflects deeper incorporation of genomic metrics into immunotherapy decision pathways rather than broader diagnostic screening. Utilization intensity is tied to checkpoint inhibitor prescribing, where mutation load influences eligibility, sequencing, and combination strategies. Spending concentration remains within oncology centers and reference laboratories equipped with next generation sequencing and validated bioinformatics infrastructure. Cost structure is shaped by panel size, sequencing depth, data interpretation requirements, and quality assurance obligations, which sustain higher per test economics compared with single gene assays.
Laboratories prioritize harmonized scoring methodologies, reproducible cutoffs, and integration within comprehensive genomic profiling to reduce interpretation variability. Payer alignment and regulatory recognition influence test frequency and positioning as companion or complementary diagnostics. Tissue adequacy constraints, turnaround time sensitivity, and informatics expertise requirements limit indiscriminate use outside defined indications. Liquid biopsy approaches expand access but face sensitivity tradeoffs in low shedding tumors, reinforcing selective deployment. Growth persistence depends on guideline reinforcement, broader immunotherapy utilization, and continued reliance on mutation burden as a quantitative tool for managing treatment cost exposure and response uncertainty in precision oncology care.

| Metric | Value |
|---|---|
| Market Value (2026) | USD 2,081.0 million |
| Market Forecast Value (2036) | USD 6,757.7 million |
| Forecast CAGR 2026 to 2036 | 12.5% |
Demand for tumor mutational burden (TMB) testing is increasing as oncology practices integrate genomic profiling to better predict patient response to immunotherapy, particularly immune checkpoint inhibitors in cancers such as melanoma, lung, and urothelial carcinoma. TMB quantifies the number of somatic mutations per megabase in a tumor’s DNA, and higher mutation loads have been associated with greater neoantigen presentation that may improve responsiveness to immuno-oncology agents. Pathologists and molecular diagnostic laboratories are adopting TMB assays on next generation sequencing platforms to augment clinical decision-making when evaluating candidates for immune checkpoint blockade and combination immunotherapy strategies. Procurement teams in hospital systems and reference laboratories evaluate assay performance, platform interoperability, and bioinformatics pipelines because the ability to deliver reliable, reproducible TMB results influences treatment selection, clinical workflow efficiency, and integration with electronic health records.
Growth in evidence linking TMB with treatment outcomes and payer willingness to cover companion diagnostics further reinforces uptake of TMB testing in multidisciplinary oncology programs. Oncology research continues to refine TMB cutoffs, harmonize assay methodologies, and correlate burden metrics with progression-free and overall survival across diverse tumor types, which encourages oncologists to include TMB in comprehensive molecular profiles. Health systems emphasize quality assurance, standardized reporting, and participation in proficiency testing to maintain confidence in genomic risk stratification and immune response prediction. Expanded adoption of precision medicine initiatives and real-world data registries enhances the clinical utility of TMB measurements in routine care, contributing to continued demand growth in the tumor mutational burden market.
Demand for tumor mutational burden assessment is shaped by next-gen immunotherapy stratification needs, biomarker harmonization efforts, and integration into molecular oncology workflows. Clinical teams require reproducible metrics that align with treatment decision frameworks and trial eligibility. Adoption reflects expansion of next-generation sequencing, liquid biopsy utilization, and standardized reporting expectations. Selection priorities include analytical validity, turnaround time, and interpretive clarity. Segmentation outlines how test format, clinical purpose, and service delivery environment influence utilization patterns across precision oncology programs.
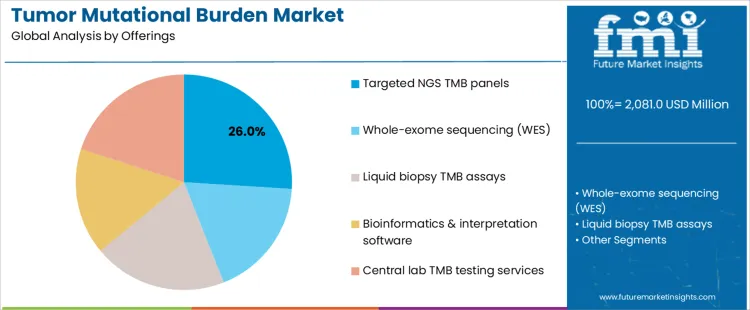
Targeted NGS TMB panels account for 26.0%, reflecting balance between sequencing depth, cost control, and clinical feasibility. These panels integrate efficiently into routine molecular profiling with manageable data complexity. Central lab TMB testing services and liquid biopsy TMB assays each represent 20.0%, supporting standardized execution and minimally invasive sampling, respectively. Whole-exome sequencing at 18.0% remains important for comprehensive analysis and research contexts. Bioinformatics and interpretation software at 16.0% enables harmonized scoring and reporting. Offering-based segmentation indicates preference for scalable solutions delivering actionable insights within clinical timelines.
Key Points

Immunotherapy patient selection represents 34.0%, reflecting reliance on TMB as a predictive biomarker for checkpoint inhibitor response. Comprehensive tumor profiling at 26.0% integrates TMB alongside genomic alterations to guide therapy planning. Clinical trials and translational research at 18.0% use TMB for cohort stratification and exploratory endpoints. Therapy response monitoring at 12.0% supports longitudinal assessment. Population genomics programs at 10.0% apply TMB to broader discovery efforts. Application-based segmentation highlights higher utilization where treatment selection, trial design, and biomarker validation intersect.
Key Points
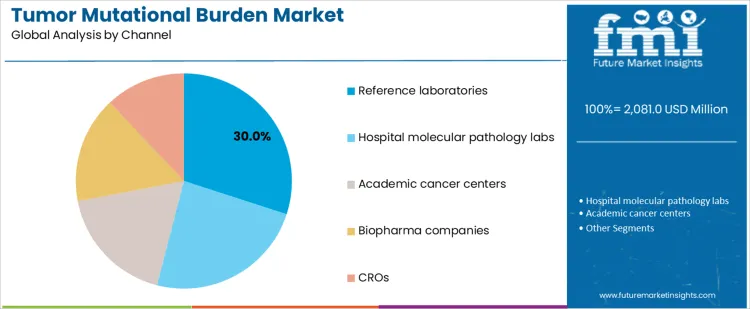
Reference laboratories account for 30.0%, reflecting centralized expertise, validated workflows, and scale efficiency. Hospital molecular pathology labs at 24.0% support integrated diagnostics within oncology care pathways. Academic cancer centers at 18.0% focus on research-driven testing and complex case evaluation. Biopharma companies at 16.0% apply TMB in drug development and companion diagnostics. CROs at 12.0% support trial execution and data management. Channel-based segmentation shows concentration where technical capability, regulatory alignment, and study infrastructure determine testing delivery.
Key Points
Demand for tumor mutational burden testing reflects clinical need to quantify genomic mutation load as a predictive biomarker for immunotherapy response. Adoption concentrates in oncology centers, molecular pathology laboratories, and reference diagnostics providers. Global scope aligns with expansion of immune checkpoint inhibitor use across solid tumors. Usage centers on next-generation sequencing assays performed on tissue and liquid biopsy samples to support treatment stratification.
Oncologists use tumor mutational burden to estimate likelihood of response to immune checkpoint inhibition when single-gene biomarkers prove insufficient. Demand increases where treatment decisions require broader genomic context beyond PD-L1 expression or specific mutations. Comprehensive sequencing panels enable simultaneous assessment of TMB alongside actionable alterations, conserving tissue and reducing testing delays. Clinical adoption depends on assay reproducibility, validated cutoffs, and correlation with outcome data across tumor types. Laboratories prioritize platforms compatible with existing workflows and bioinformatics pipelines. Turnaround time affects use in advanced disease with limited therapeutic windows. Adoption reflects clinical effort to refine patient selection for costly immunotherapies through quantitative genomic metrics.
Tumor mutational burden measurement varies by sequencing panel size, depth, and bioinformatic algorithms, complicating cross-platform comparability. Demand sensitivity rises where lack of harmonized thresholds creates uncertainty in clinical decision-making. Reimbursement policies remain inconsistent due to evolving evidence strength across indications. Data interpretation requires specialized expertise to contextualize results within tumor biology. Tissue adequacy constraints affect test success rates, particularly in small biopsies. Liquid biopsy approaches face sensitivity limitations in low-shedding tumors. Regulatory expectations differ by region for companion versus complementary diagnostics. Scalability remains constrained by standardization gaps, payer alignment challenges, and complexity of translating genomic data into consistent clinical action.
Demand for tumor mutational burden testing is expanding globally as immunotherapy selection increasingly relies on genomic predictors of response. Oncology guidelines incorporate TMB alongside PD-L1 and MSI to refine patient stratification. Broader access to next-generation sequencing platforms improves feasibility across care settings. Reimbursement clarity and assay standardization influence uptake consistency. Growth rates in India at 15.0%, Brazil at 14.5%, China at 14.0%, the USA at 11.0%, and Germany at 10.9% indicate sustained expansion driven by precision oncology adoption, biomarker-driven prescribing, and laboratory capacity scaling rather than incidence growth.
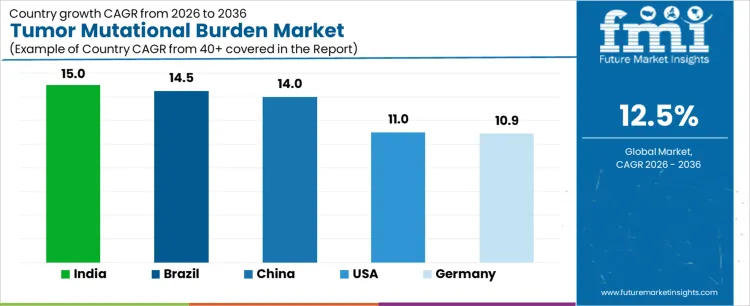
| Country | CAGR (%) |
|---|---|
| India | 15.0% |
| Brazil | 14.5% |
| China | 14.0% |
| USA | 11.0% |
| Germany | 10.9% |
Tumor mutational burden demand in India is growing at a CAGR of 15.0%, supported by expanding access to immuno-oncology therapies and molecular diagnostics. Private oncology networks integrate TMB within comprehensive NGS panels to guide checkpoint inhibitor use. Rising availability of sequencing services reduces turnaround times and cost barriers. Clinician awareness of biomarker-guided therapy selection increases testing frequency. Insurance pilots and patient assistance programs improve affordability. Growth reflects precision oncology integration and diagnostic infrastructure expansion rather than population-wide cancer screening changes.
Tumor mutational burden demand in Brazil is expanding at a CAGR of 14.5%, driven by increasing use of immunotherapies across solid tumors. Comprehensive cancer centers adopt TMB to refine treatment eligibility. Judicial access pathways facilitate availability within public systems. Regional laboratories expand sequencing capacity beyond major metros. Clinical trial participation increases familiarity with TMB interpretation. Growth reflects institutional adoption and therapy access mechanisms rather than decentralized testing proliferation.
Tumor mutational burden demand in China is growing at a CAGR of 14.0%, aligned with standardized precision oncology programs. Large tertiary hospitals integrate TMB within routine molecular profiling. Domestic sequencing platforms reduce cost and improve throughput. Reimbursement inclusion for selected biomarkers supports testing consistency. Alignment with domestic immunotherapy development reinforces clinical relevance. Growth reflects centralized protocol-driven adoption and scalable laboratory capacity rather than discretionary testing expansion.
Tumor mutational burden demand in the United States is expanding at a CAGR of 11.0%, supported by guideline recognition and payer coverage. Comprehensive genomic profiling commonly includes TMB scoring for advanced cancers. High penetration of NGS platforms ensures analytical consistency. FDA approvals linking TMB to therapy eligibility reinforce utilization. Integrated electronic records facilitate reporting and decision support. Growth reflects standardized precision oncology workflows and evidence maturation rather than increases in testing intensity per patient.
Tumor mutational burden demand in Germany is growing at a CAGR of 10.9%, shaped by evidence-based oncology and reimbursement governance. University hospitals and certified centers apply TMB within defined indications. Health technology assessments guide appropriate use. Central laboratory networks ensure assay standardization and quality control. Emphasis on clinical utility moderates indiscriminate testing. Growth remains steady, reflecting structured integration and policy oversight rather than rapid diffusion across all care settings.
Demand for tumor mutational burden testing is driven by immunotherapy eligibility assessment, precision oncology adoption, and regulatory recognition of TMB as a companion biomarker. Oncology laboratories and clinicians evaluate assay concordance, mutation coverage breadth, bioinformatics accuracy, sample input flexibility, and turnaround reliability. Buyer assessment includes regulatory clearance status, compatibility with tissue and liquid biopsy workflows, and alignment with approved checkpoint inhibitors. Procurement behavior reflects preference for validated platforms, centralized testing models, and long-term reagent agreements. Trend in the tumor mutational burden market shows increasing integration into comprehensive genomic profiling at diagnosis and disease progression stages.
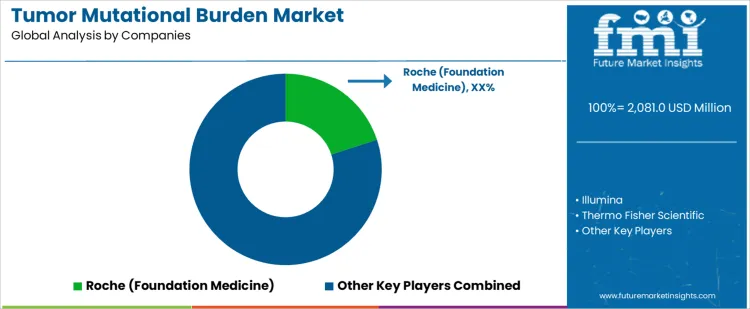
Roche Foundation Medicine leads competitive positioning through clinically validated TMB assays embedded within comprehensive genomic profiling services. Illumina competes by enabling high-throughput sequencing platforms supporting in-house TMB calculation and research-to-clinic translation. Thermo Fisher Scientific supports demand with flexible sequencing and informatics solutions suited for decentralized oncology laboratories. Guardant Health maintains relevance through blood-based TMB testing supporting noninvasive patient stratification. QIAGEN participates by offering sample-to-insight workflows facilitating standardized molecular testing. Competitive differentiation centers on clinical validation depth, assay reproducibility, regulatory alignment, and integration with immunotherapy decision pathways.
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Offerings | Targeted NGS TMB panels; Whole-exome sequencing (WES); Liquid biopsy TMB assays; Bioinformatics & interpretation software; Central lab TMB testing services |
| Applications | Immunotherapy patient selection; Comprehensive tumor profiling; Clinical trials & translational research; Therapy response monitoring; Population genomics programs |
| Channel | Reference laboratories; Hospital molecular pathology labs; Academic cancer centers; Biopharma companies; CROs |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, Brazil, China, USA, Germany, and 40+ countries |
| Key Companies Profiled | Roche (Foundation Medicine); Illumina; Thermo Fisher Scientific; Guardant Health; QIAGEN; Others |
| Additional Attributes | Dollar sales by offering, application, and channel; analytical concordance and reproducibility of TMB measurement across tissue and liquid biopsy platforms; role of bioinformatics pipelines in variant filtering, normalization, and reporting; clinical utility of TMB as a predictive biomarker for immunotherapy response; regulatory alignment and companion diagnostic considerations; procurement dynamics driven by centralized reference labs, hospital oncology adoption, and biopharma-sponsored clinical research programs. |
How big is the tumor mutational burden market in 2026?
The global tumor mutational burden market is estimated to be valued at USD 2,081.0 million in 2026.
What will be the size of tumor mutational burden market in 2036?
The market size for the tumor mutational burden market is projected to reach USD 6,757.7 million by 2036.
How much will be the tumor mutational burden market growth between 2026 and 2036?
The tumor mutational burden market is expected to grow at a 12.5% CAGR between 2026 and 2036.
What are the key product types in the tumor mutational burden market?
The key product types in tumor mutational burden market are targeted ngs tmb panels, whole-exome sequencing (wes), liquid biopsy tmb assays, bioinformatics & interpretation software and central lab tmb testing services.
Which applications segment to contribute significant share in the tumor mutational burden market in 2026?
In terms of applications, immunotherapy patient selection segment to command 34.0% share in the tumor mutational burden market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.