The bladder cancer immunotherapy market is projected to reach USD 5,012.0 million in 2026 and expand to USD 14,489.7 million by 2036, at a CAGR of 11.2%. Value expansion reflects the structural repositioning of immunotherapy within bladder cancer treatment pathways rather than incremental therapy substitution. Utilization is closely linked to rising treatment duration, broader line-of-therapy use, and integration across non-muscle-invasive, muscle-invasive, and metastatic settings. Spending persistence is reinforced by high recurrence rates and limited durability of conventional treatments, which sustain repeated intervention cycles. Pricing behavior is shaped by biologic manufacturing complexity, infusion-based delivery, and outcome-linked reimbursement frameworks, positioning immunotherapy as a high-value, protocol-driven oncology segment.
Hospitals and cancer centers rely on immunotherapy to manage BCG-unresponsive disease and extend survival in advanced cases, increasing dependency on long-term administration and monitoring infrastructure. Response variability and immune-related toxicity management constrain indiscriminate use, reinforcing specialist-led prescribing and payer oversight. Cost exposure remains significant due to prolonged treatment courses and combination regimens under investigation. Value growth remains anchored to guideline expansion, earlier line incorporation, and sustained reliance on immune-based approaches to manage recurrence and progression risk in bladder cancer care.
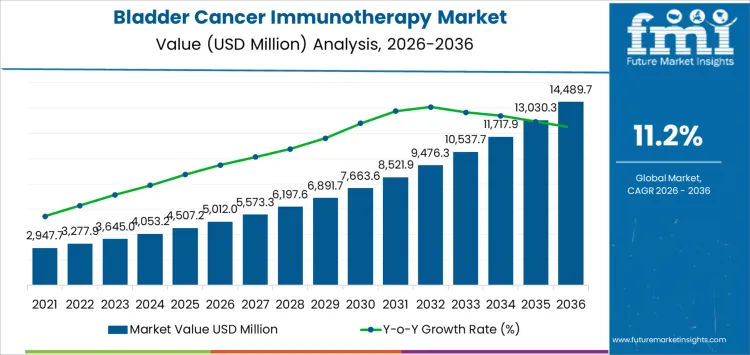
| Metric | Value |
|---|---|
| Market Value (2026) | USD 5,012.0 million |
| Market Forecast Value (2036) | USD 14,489.7 million |
| Forecast CAGR 2026 to 2036 | 11.2% |
Clinical practice in uro-oncology is shifting toward immunotherapy as an effective option for managing advanced and recurrent bladder cancer where conventional surgery and chemotherapy have limited durability. Biomarker limitations and evolving evidence bases slow rapid cancer biological therapy switching once protocols are established. Immune checkpoint inhibitors and targeted biologic agents engage the patient’s immune system to recognize and attack malignant urothelial cells, which expands therapeutic choice for patients who cannot tolerate or do not respond to cytotoxic regimens. Oncologists assess immune biomarker expression, prior treatment history, and performance status to select appropriate immunotherapy products that align with individual disease characteristics. Hospital pharmacies and specialty infusion centers invest in compatible administration infrastructure and monitoring protocols because safe delivery and management of immune-related adverse events are essential components of care. Procurement teams prioritize products that demonstrate consistent clinical benefit, manageable safety profiles, and predictable supply to support long-term treatment plans across outpatient and inpatient oncology services.
Population health strategies and cancer care guidelines are reinforcing adoption of bladder cancer immunotherapies that improve progression free survival and offer meaningful quality of life advantages for eligible patients. Health systems and payers incorporate real-world evidence of treatment outcomes into coverage frameworks that support access while balancing budget impact. Clinical research continues to expand indications and combination approaches, which encourages multidisciplinary tumor boards to include immunotherapy in treatment algorithms earlier in the disease course. Educational outreach and decision support tools help clinicians interpret evolving evidence and optimize sequencing of therapies. These clinical, operational, and evidence-driven trends are contributing to sustained demand growth in the bladder cancer immunotherapy market.
Demand for bladder cancer immunotherapy is shaped by survival improvement goals, durability of response, and treatment sequencing across disease stages. Clinical adoption reflects expansion beyond intravesical therapy toward systemic immune modulation. Personalized medicine strategies underpin optimized immunotherapeutic sequencing in advanced bladder cancer care. Decision pathways prioritize efficacy in refractory disease, tolerability in older populations, and integration with surgery or chemotherapy. Utilization patterns align with biomarker stratification, infusion infrastructure, and guideline updates.
Demand patterns in the immunomodulator market help contextualize immune response therapies in oncology. Segmentation clarifies how therapy modality, disease setting, and care delivery environment influence adoption intensity across bladder cancer management.
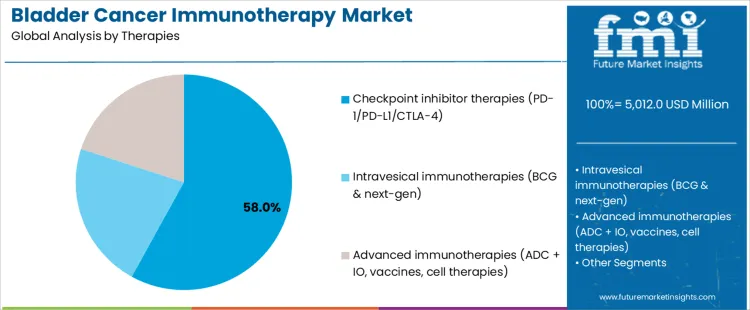
Checkpoint inhibitor therapies at 58.0% represent the largest share, reflecting established efficacy in advanced disease and broad regulatory approvals. These agents target PD-1, PD-L1, or CTLA-4 pathways to restore antitumor immune activity. Intravesical immunotherapies at 22.0% remain central for localized disease management, particularly in BCG-responsive pathways and next-generation formulations. Advanced immunotherapies at 20.0% include antibody drug conjugate combinations, vaccines, and cell-based approaches addressing resistance and relapse. Therapy-class segmentation indicates concentration where systemic efficacy, durability, and clinical familiarity drive utilization.
Key Points
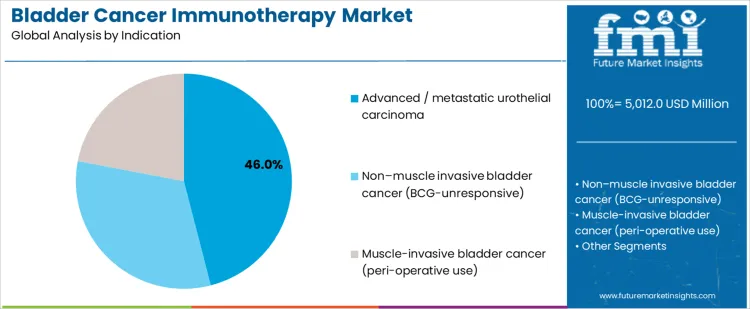
Advanced or metastatic urothelial carcinoma accounts for 46.0%, reflecting reliance on immunotherapy following platinum-based regimens. Non-muscle invasive bladder cancer unresponsive to BCG at 32.0% drives adoption of novel immunotherapeutic approaches to avoid cystectomy. Muscle-invasive disease at 22.0% applies immunotherapy in peri-operative settings to improve recurrence outcomes. Indication-based segmentation highlights stronger utilization where unmet need, recurrence risk, and survival benefit justify immune-based intervention.
Key Points
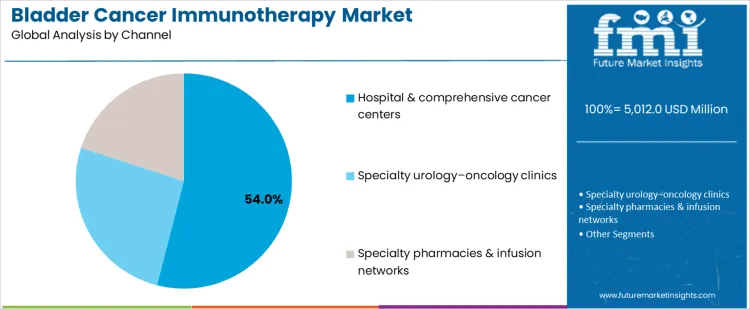
Hospital and comprehensive cancer centers represent 54.0%, reflecting infusion capacity, multidisciplinary care, and complex case management. The immuno oncology assay reinforces the importance of biomarker evaluation in immunotherapy efficacy. Specialty urology-oncology clinics at 26.0% provide focused care for localized and recurrent disease with coordinated treatment pathways. Specialty pharmacies and infusion networks at 20.0% support administration logistics, reimbursement handling, and patient access. The antineoplastic combinations provide context for multi-modal oncology regimens interacting with immunotherapeutics. Channel segmentation reflects concentration where infrastructure, clinical expertise, and coordination enable sustained immunotherapy delivery.
Key Points
Demand for bladder cancer immunotherapy reflects need to manage recurrent and advanced disease where conventional intravesical or cytotoxic treatments show limited durability. Adoption concentrates in urology clinics, oncology centers, and hospital infusion units. Global scope aligns with guideline incorporation of immune-based regimens across disease stages. Trends identified in lung cancer therapeutics provide broader therapeutic context for immunotherapy adoption. Usage centers on intravesical immunotherapies and systemic immune checkpoint inhibitors applied in non-muscle-invasive, muscle-invasive, and metastatic settings.
Bladder cancer presents high recurrence rates that drive repeated intervention across long disease courses. Demand increases where patients become unresponsive or intolerant to intravesical chemotherapy. Immune-based therapies stimulate antitumor response through localized or systemic immune activation, supporting durable disease control in selected patients. Non-muscle-invasive disease relies on intravesical immunotherapy to delay progression and avoid cystectomy. Advanced disease settings adopt systemic immunotherapy following platinum-based treatment or in cisplatin-ineligible patients. Treatment choice depends on disease stage, biomarker expression, prior therapy exposure, and patient fitness. Integration into stepwise treatment algorithms supports continuity of care. Adoption reflects clinical emphasis on reducing recurrence burden and extending survival without escalating surgical intervention.
Immunotherapy response rates vary significantly across patient subgroups, affecting predictability of outcomes. Demand sensitivity rises where lack of response delays transition to definitive surgical management. Immune-related adverse events require multidisciplinary monitoring and rapid intervention capability. Treatment duration and infusion schedules increase resource utilization in oncology settings. High therapy cost influences reimbursement approval and line-of-therapy positioning. Biomarker utility remains imperfect, limiting precise patient selection. Manufacturing complexity affects pricing stability. Long-term follow-up obligations add system burden. Scalability remains constrained by response heterogeneity, safety management demands, and payer controls tied to outcome justification.
Demand for bladder cancer immunotherapy is expanding globally as immune checkpoint inhibitors gain wider use across non-muscle-invasive and advanced disease settings. Treatment guidelines increasingly position immunotherapy following BCG failure and in metastatic lines. Improved diagnostic stratification and therapy sequencing support adoption consistency. Reimbursement decisions and infusion infrastructure influence utilization pace across regions. Growth rates in India at 12.6%, China at 12.4%, Brazil at 12.0%, the USA at 11.0%, and Germany at 10.8% indicate sustained expansion driven by therapy access broadening, protocol adoption, and oncology capacity scaling rather than incidence-led growth.
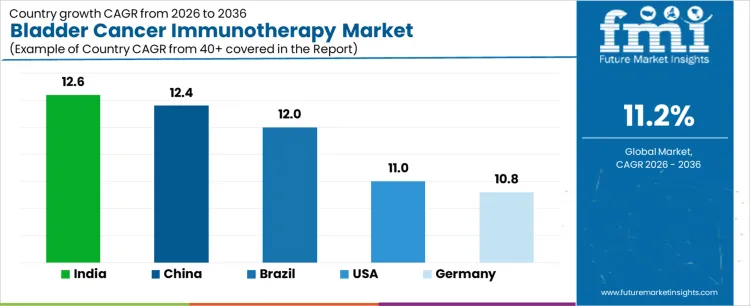
| Country | CAGR (%) |
|---|---|
| India | 12.6% |
| China | 12.4% |
| Brazil | 12.0% |
| USA | 11.0% |
| Germany | 10.8% |
Bladder cancer immunotherapy demand in India is growing at a CAGR of 12.6%, supported by increasing availability of checkpoint inhibitors across tertiary oncology centers. Rising diagnosis of advanced-stage disease increases reliance on systemic therapies. Private hospitals expand infusion capabilities to support immunotherapy delivery. Inclusion of immunotherapy within institutional treatment protocols improves adoption consistency. Gradual expansion of insurance coverage reduces out-of-pocket barriers. Growth reflects therapy accessibility improvements, protocol formalization, and oncology infrastructure expansion rather than shifts in screening intensity.
Bladder cancer immunotherapy demand in China is expanding at a CAGR of 12.4%, aligned with standardized oncology pathways and domestic biologics availability. National reimbursement inclusion for selected immunotherapies improves patient access. Large cancer hospitals integrate immunotherapy within bladder cancer treatment algorithms. Domestic manufacturers supply cost-competitive checkpoint inhibitors. Centralized procurement supports volume scaling. Growth reflects policy-backed access expansion and standardized clinical use rather than experimental treatment adoption.
Bladder cancer immunotherapy demand in Brazil is growing at a CAGR of 12.0%, driven by expansion of comprehensive cancer care networks. Public and private oncology providers adopt immunotherapy following guideline updates. Legal access mechanisms support availability within public systems. Increasing use after BCG failure elevates treatment volumes. Regional cancer centers improve infusion access outside major metros. Growth reflects institutional adoption and therapy access mechanisms rather than accelerated diagnostic rates.
Bladder cancer immunotherapy demand in the United States is expanding at a CAGR of 11.0%, supported by guideline-led sequencing and payer reimbursement. Immunotherapies are integrated across metastatic and high-risk non-muscle-invasive settings. Broad insurance coverage supports sustained utilization. Clinical trial activity expands therapeutic options and physician familiarity. Advanced infusion infrastructure ensures delivery capacity. Growth reflects protocol adherence and therapeutic innovation pace rather than changes in bladder cancer incidence.
Bladder cancer immunotherapy demand in Germany is growing at a CAGR of 10.8%, shaped by evidence-based oncology frameworks and reimbursement discipline. Certified cancer centers apply immunotherapy according to defined treatment pathways. Statutory insurance coverage ensures access following benefit assessment approvals. Emphasis on outcomes monitoring guides appropriate use. Centralized hospital systems support consistent infusion delivery. Growth remains steady, reflecting structured adoption and health system governance rather than rapid therapy diffusion.

Demand for bladder cancer immunotherapy is shaped by recurrence risk, progression management needs, and expanded use beyond refractory disease. Oncologists and urologists evaluate response durability, progression free survival data, safety in comorbid populations, and suitability for intravesical or systemic use. Buyer assessment includes regulatory approvals by stage, guideline inclusion, administration logistics, and sequencing with chemotherapy. Procurement behavior reflects hospital formulary decisions, reimbursement alignment, and infusion capacity planning. Trend in the personalized immunotherapy market shows broader adoption in non-muscle invasive and advanced disease settings, supported by real world outcome evidence and evolving treatment algorithms.
Merck and Company leads competitive positioning through checkpoint inhibitors with strong clinical adoption across advanced and earlier stage bladder cancer indications. Bristol Myers Squibb competes via immunotherapy regimens emphasizing durable responses and combination potential. Roche supports demand with immune based therapies integrated within oncology portfolios and supported by biomarker strategies. AstraZeneca maintains relevance through immuno oncology programs aligned with disease progression management and combination development. Pfizer participates through selective immunotherapy assets positioned within broader uro oncology strategies. Competitive differentiation centers on clinical trial depth, durability of response, access positioning, and alignment with evolving bladder cancer treatment pathways.
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Therapies | Checkpoint inhibitor therapies (PD-1/PD-L1/CTLA-4); Intravesical immunotherapies (BCG & next-gen); Advanced immunotherapies (ADC + IO, vaccines, cell therapies) |
| Indication | Advanced / metastatic urothelial carcinoma; Non-muscle invasive bladder cancer (BCG-unresponsive); Muscle-invasive bladder cancer (peri-operative use) |
| Channel | Hospital & comprehensive cancer centers; Specialty urology-oncology clinics; Specialty pharmacies & infusion networks |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, China, Brazil, USA, Germany, and 40+ countries |
| Key Companies Profiled | Merck & Co.; Bristol Myers Squibb; Roche; AstraZeneca; Pfizer; Others |
| Additional Attributes | Dollar sales by therapy class, indication, and channel; clinical efficacy and durability of response across checkpoint inhibitors, intravesical therapies, and advanced immunotherapy platforms; treatment sequencing influenced by BCG response status and disease stage; infusion logistics and intravesical administration requirements; reimbursement and regulatory pathways shaping access in hospital-based oncology care; procurement dynamics driven by specialty cancer centers and infusion network partnerships. |
How big is the bladder cancer immunotherapy market in 2026?
The global bladder cancer immunotherapy market is estimated to be valued at USD 5,012.0 million in 2026.
What will be the size of bladder cancer immunotherapy market in 2036?
The market size for the bladder cancer immunotherapy market is projected to reach USD 14,489.7 million by 2036.
How much will be the bladder cancer immunotherapy market growth between 2026 and 2036?
The bladder cancer immunotherapy market is expected to grow at a 11.2% CAGR between 2026 and 2036.
What are the key product types in the bladder cancer immunotherapy market?
The key product types in bladder cancer immunotherapy market are checkpoint inhibitor therapies (pd-1/pd-l1/ctla-4), intravesical immunotherapies (bcg & next-gen) and advanced immunotherapies (adc + io, vaccines, cell therapies).
Which indication segment to contribute significant share in the bladder cancer immunotherapy market in 2026?
In terms of indication, advanced / metastatic urothelial carcinoma segment to command 46.0% share in the bladder cancer immunotherapy market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.