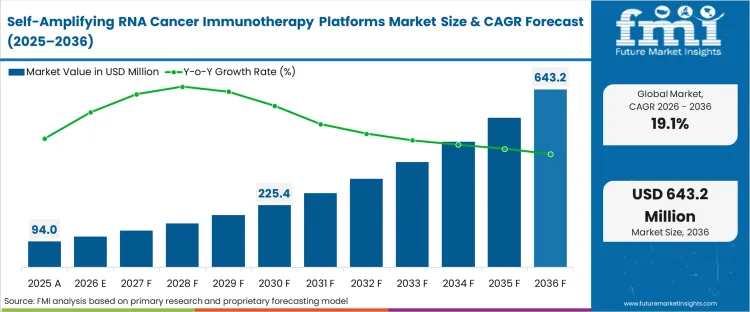
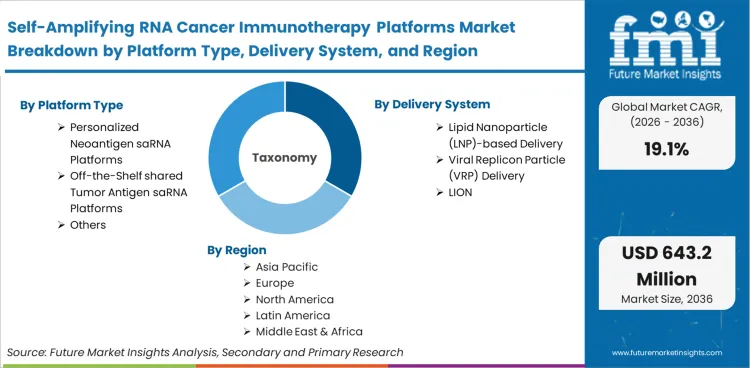
The self-amplifying RNA cancer immunotherapy platforms market was valued at USD 94.0 Million in 2025. The saRNA cancer immunotherapy platform market is poised to surpass USD 112.0 million in 2026 at a CAGR of 19.10% during the forecast period. Continued investment propels the valuation to USD 643.2 million through 2036 as the profound dose-sparing capability of self-amplifying RNA cancer therapeutics solves the systemic toxicity bottlenecks that have historically stalled conventional RNA-based solid tumour programs.
Oncology pipeline directors face an imperative to transition from standard linear constructs to a self-amplifying mRNA cancer platform model. This transition forces developers to abandon established delivery vectors in favour of novel platforms capable of protecting much larger transcripts. Organizations delaying this pivot risk stranding their mrna cancer vaccine biologic lines in phase I purgatory, where high-dose toxicity frequently outweighs therapeutic benefit. The actual bottleneck shaping the self-replicating RNA cancer immunotherapy market is not the immunogenicity of the replicon sequence itself, but the physical challenge of purifying 10kb-plus RNA transcripts at clinical grade without shearing the delicate molecular backbone.
Before the saRNA oncology platform market achieves self-reinforcing scale, developers must demonstrate that saRNA constructs can trigger durable T-cell memory responses without the continuous co-administration of checkpoint inhibitors. Clinical-stage biotechs pass this gate in the self-amplifying RNA cancer vaccine platform market when they successfully uncouple lipid delivery toxicity from the replicon payload itself. Once this separation is achieved, the rapid iteration of patient-specific neoantigen targets becomes commercially viable for a much wider tier of mid-sized biopharmaceutical developers mapping how big could the saRNA oncology platform market be by 2036.
The Japan saRNA cancer immunotherapy market advances at 21.4%, followed by the China at 20.8%, the United States market advances at 20.2%, Germany at 19.0%, the United Kingdom market advances at 18.7%, the Indian market grows at 18.3%, and South Korea at 17.9%. Japan's trajectory is set to record the fastest pace among saRNA cancer vaccine companies, driven by a first-of-its-kind commercial saRNA regulatory pathway and domestic manufacturing initiatives specifically targeting solid tumours. The divergence across these geographies stems from the varying capacity of localized biotech ecosystems to handle complex, large-format nucleic acid manufacturing compared to regions resting on older, conventional oncology installed bases.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 112.0 Million |
| Industry Value (2036) | USD 643.2 Million |
| CAGR (2026-2036) | 19.10% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
If asking what is a self-amplifying RNA cancer immunotherapy platform, the market encompasses the biological constructs, engineering technologies, and specialised delivery systems used to deploy viral-derived, self-replicating RNA in oncology. The functional boundary defining this market is the inclusion of genes encoding an RNA-dependent RNA polymerase, enabling the therapeutic payload to amplify intracellularly. This mechanism distinguishes the market from standard linear mRNA platforms, allowing for profound dose-sparing and extended antigen expression within the tumor microenvironment.
Inclusions cover personalized and off-the-shelf neoantigen cancer vaccines, cytokine-expressing replicon vectors forming a true saRNA dose-sparing oncology platform, and intratumoral saRNA cancer immunotherapy amplifiers utilized alongside cellular therapies. The scope explicitly captures the large-format lipid nanoparticles and engineered viral replicon particles designed to transport saRNA. Technologies supporting the formulation and stabilization of these highly complex, 9-to-12 kilobase structures are fully integrated within the market boundary.
Explicitly excluded are conventional, non-amplifying mRNA therapies evaluated in a saRNA vs mRNA cancer vaccine comparison, unmodified plasmid DNA vectors, and standard prophylactic viral vaccines for infectious diseases. These constructs lack the autonomous self-replication machinery required for inclusion. Furthermore, general-purpose liposomes and delivery vectors not optimized for massive transcript protection are excluded, as they physically cannot deliver the large saRNA payloads without triggering premature degradation.
When evaluating a personalized saRNA vaccine vs off-the-shelf tumor antigen vaccine, conventional shared antigen approaches consistently fail to generate sufficient T-cell infiltration against highly heterogeneous malignancies, forcing the transition toward personalized replicon architectures.
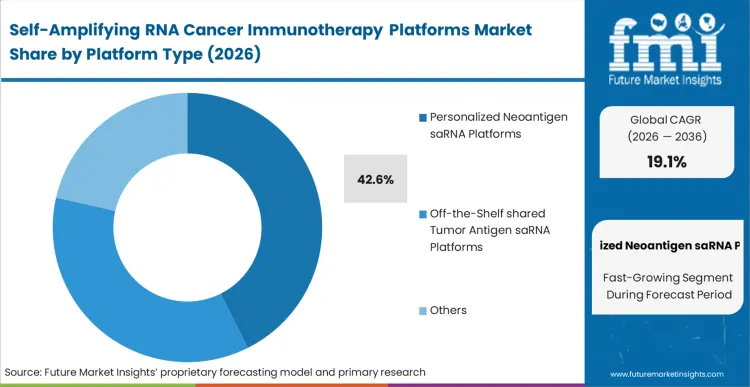
According to FMI's estimates, Personalized neoantigen saRNA platforms capture 42.6% share by aligning the extreme dose-sparing capability of alphavirus replicon cancer vaccine technology with patient-specific MHC-binding precision. Oncology pipeline managers utilize this platform to bypass the systemic toxicity that typically halts high-dose linear mRNA trials in phase II. By leveraging next generation immunotherapies, developers physically reduce the required payload size while sustaining antigen expression for weeks rather than days. Companies operating a saRNA cytokine cancer immunotherapy program that attempt to apply linear mRNA economics to personalized solid tumor vaccines face unsustainable per-patient manufacturing costs.
The 51.8% share held by Lipid nanoparticle (LNP)-based delivery stems entirely from the physical impossibility of transporting 10kb-plus transcripts through the bloodstream unprotected. Securing the best self-amplifying RNA delivery system for oncology is mandatory; without LNP encapsulation, ubiquitous ribonucleases destroy the delicate saRNA backbone before it reaches the targeted antigen-presenting cells. Translational directors seek a highly specialized saRNA LNP formulation partner for cancer vaccines utilizing advanced mrna synthesis raw materials to formulate lipid envelopes that accommodate massive payload sizes without triggering severe hepatotoxicity.
FMI analysts opine that the challenge for any immuno-oncology RNA delivery system lies in balancing the cationic lipid ratio to ensure endosomal escape without destroying the encapsulated replicon during formulation. Developers relying on older, first-generation liposomes inevitably experience catastrophic payload shearing during the extrusion process.
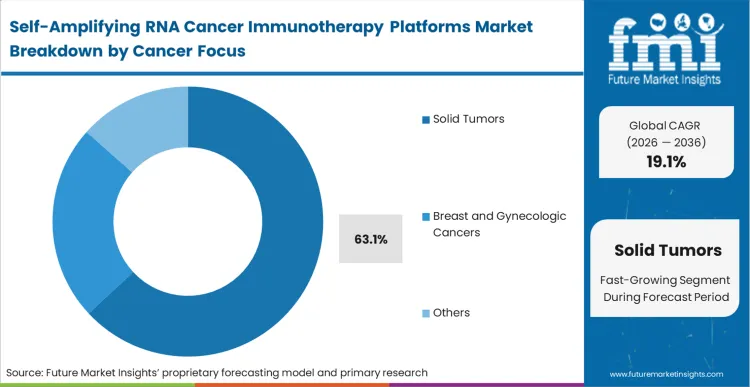
The profound failure of conventional biologics to penetrate dense fibrotic tissue in non-hematologic malignancies dictates the trajectory of this segment. Solid tumors represent 63.1% of the market because the localized immunosuppressive microenvironment actively neutralises standard systemic interventions. Whether operating a saRNA breast cancer vaccine platform, a saRNA gastrointestinal cancer vaccine, a saRNA melanoma immunotherapy platform, or a saRNA lung cancer immunotherapy program, Chief Medical Officers deploy saRNA constructs to force continuous, localized expression of neoantigens directly within the tumor mass.
Based on FMI's assessment, applying targeted oncology biologics in this format creates an extended therapeutic window that linear mRNA cannot match without repeated, toxic dosing. Programs that fail to achieve this localized amplification are rapidly outcompeted by therapies that break solid tumor tolerance without destroying the patient's broader immune function.
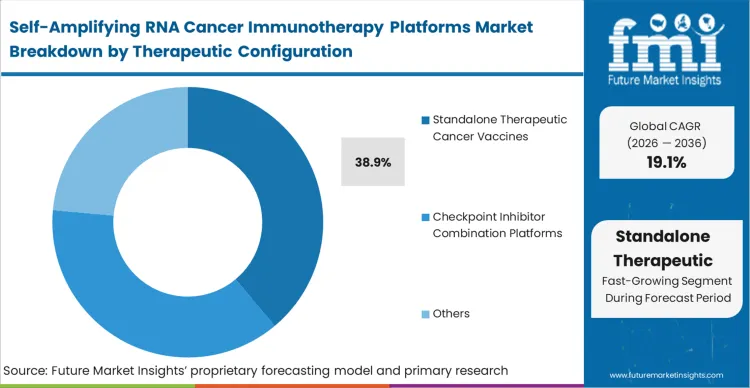
The clinical desire for standalone efficacy versus the biological reality that established tumours heavily express exhaustion markers. Standalone therapeutic cancer vaccines lead with 38.9% share, representing the initial wave of IND-cleared programs striving for monotherapy proof-of-concept. However, translational teams increasingly recognize that even a powerful saRNA CAR-T amplifier vaccine or a cell therapy adjunct saRNA vaccine cannot overcome deep PD-L1 expression alone without complementary modalities.
In FMI's view, this tension forces the integration of saRNA cancer biological therapy with established checkpoint inhibitors to unlock full T-cell functionality. Developers clinging rigidly to a monotherapy paradigm for advanced solid tumours risk stalling their pipelines against entrenched immunosuppressive barriers.
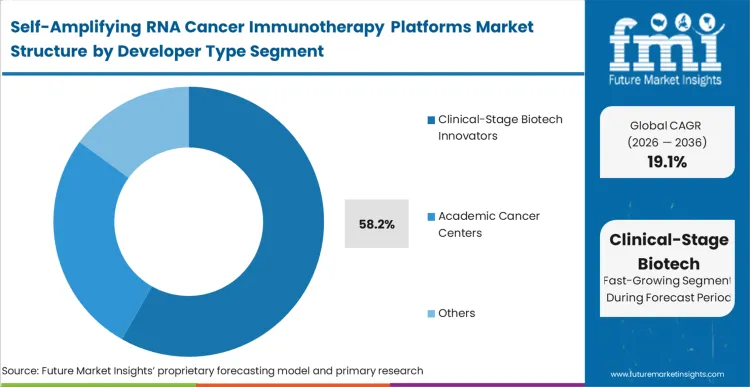
Large pharmaceutical organizations face an immediate decision: build internal large-format RNA infrastructure from scratch or secure self-amplifying RNA oncology platform licensing deals with specialized innovators. Clinical-stage biotech innovators dominate the landscape with 58.2% share because they possess the foundational intellectual property surrounding viral alphavirus backbones and large-transcript biologics contract manufacturing.
As per FMI's projection, massive incumbent organizations seeking oncology saRNA platform collaboration opportunities cannot easily pivot their established linear mRNA platforms to accommodate 12kb replicon structures without violating these existing patents. Consequently, biotech innovators serve as the primary supply node for translational know-how. Incumbents that delay licensing or acquiring these specialized developers find themselves completely locked out of the dose-sparing advantages critical for next-generation oncology pipelines.
When examining what drives adoption of saRNA cancer immunotherapy platforms, the severe systemic toxicity associated with high-dose linear mRNA compels translational oncology directors to transition to self-amplifying architectures. In solid tumor indications, achieving therapeutic T-cell activation requires consistent antigen presentation that conventional transcripts cannot provide without repeated, highly toxic dosing. This pressure forces pipeline managers to adopt saRNA constructs, which utilize cell and gene therapy manufacturing techniques to amplify a minute initial dose into a massive intracellular payload. Companies that aggressively implement these dose-sparing platforms capture a decisive commercial advantage by advancing through phase II safety gates that consistently halt linear mRNA solid tumor programs.
Conversely, assessing what are the main risks of self-amplifying RNA cancer platforms, the cancer vaccine manufacturing saRNA complexity of large replicon constructs creates a profound operational friction for personalized neoantigen developers. Unlike standard 2kb linear mRNA, saRNA transcripts span 9 to 12 kilobases, making them highly susceptible to physical shearing during standard cell therapy manufacturing and downstream purification. This friction is rooted in the sheer physical size and fragility of the molecule, severely limiting the speed of patient-specific iteration. While advanced low-shear microfluidic systems offer a partial solution, their throughput remains heavily constrained, bottlenecking clinical trial expansion.
Opportunities in the Self-Amplifying RNA Cancer Immunotherapy Platforms Market
According to the regional assessment, the Self-Amplifying RNA Cancer Immunotherapy Platforms Market is divided into Asia Pacific, North America, and Europe, covering more than 40 countries.
| Country | CAGR (2026 to 2036) |
|---|---|
| Japan | 21.4% |
| China | 20.8% |
| United States | 20.2% |
| Germany | 19.0% |
| United Kingdom | 18.7% |
| India | 18.3% |
| South Korea | 17.9% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
The regulatory environment shaping personalized immunotherapy across Asia Pacific actively prioritizes the commercialization of advanced nucleic acid technologies, bypassing the prolonged hesitancy seen in older regulatory frameworks. Health ministries in this region have established specific, accelerated pathways for self-amplifying architectures, treating them as functionally distinct from standard gene therapies.
FMI analysts suggests that this policy alignment allows regional biotechs to move from computational antigen design to IND clearance significantly faster than their Western counterparts. The market dynamic is driven by aggressive state-backed initiatives to build localized, large-format RNA manufacturing sovereignty, directly reducing the historical reliance on imported biologics.
FMI's report includes extensive analysis of emerging biomanufacturing hubs across Southeast Asia and Oceania. A consistent pattern across these additional markets is the aggressive implementation of technology-transfer agreements, allowing nascent biotech sectors to bypass early-generation linear mRNA completely and leapfrog directly to replicon capabilities.
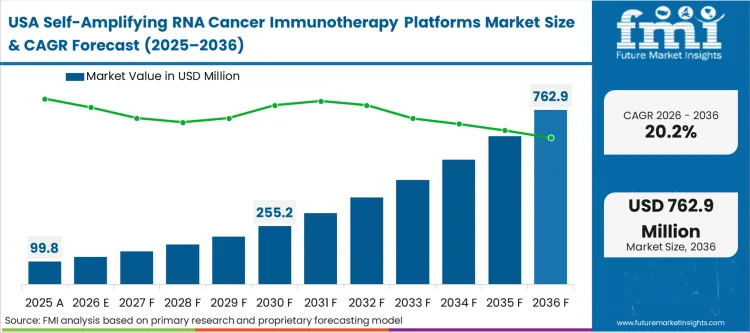
The sheer density of specialized venture capital and established translational oncology infrastructure defines North America's position. Unlike regions building capability from the ground up, the ecosystem here operates on a massive base of legacy mRNA experience, providing an unparalleled network of specialized LNP formulators, sequencing facilities, and clinical trial networks. As per FMI's assessment, this deep infrastructure forces a highly competitive environment where biotechs must prove not just safety, but superior dose-sparing economics to secure next-round funding. The fundamental condition is a mature, highly critical buyer base demanding verifiable evidence that saRNA can shatter the solid tumor barriers that linear transcripts failed to breach.
FMI's report includes analysis of the Canadian advanced biologics sector. The path of these adjacent markets reflects deep integration into the US supply chain, primarily acting as specialized nodes for lipid nanoparticle development and specialized raw material synthesis rather than full-scale independent platform development.
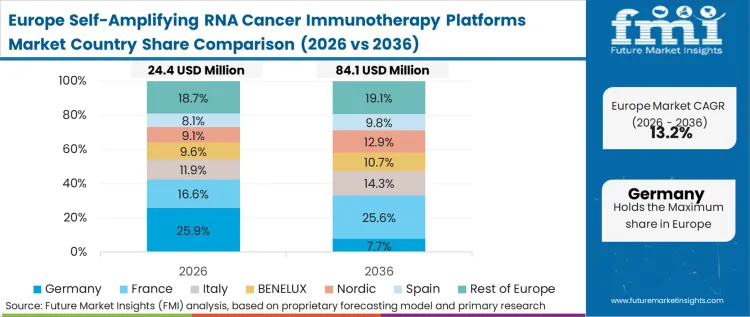
Cost structures and stringent cross-border clinical qualification standards drive adoption across Europe. Procurement cycles heavily scrutinise the total lifecycle cost of personalised oncology interventions, demanding therapies that minimise clinical administration burden. Based on FMI's projection, this economic pressure perfectly aligns with the dose-sparing nature of self-amplifying constructs, which require significantly less raw material and fewer clinical visits than conventional mRNA therapies. A landscape is revealed where massive incumbent oncology players are systematically absorbing agile saRNA innovators to defend their highly profitable, yet ageing, solid tumour portfolios from patent cliffs.
FMI's report includes an evaluation of specialised manufacturing clusters across Switzerland and the Nordics. The unifying pattern across these markets is their transition from broad-spectrum biological manufacturing toward highly specialized, low-volume, high-value pd-l1 biomarker testing linked saRNA synthesis hubs.
For executives analyzing which saRNA oncology companies look most advanced in 2026 or aiming to build a market map of self-amplifying RNA cancer platform developers, the competitive situation of the oncology biotech saRNA platform market remains highly concentrated because foundational intellectual property surrounding viral replicon backbones acts as an immense barrier. Companies such as BioNTech SE and Arcturus Therapeutics Holdings Inc. hold dominant positions not merely due to scale, but because they control the specific genetic architectures and delivery systems required to make saRNA functional. Buyers in this space, translational oncology directors and large pharmaceutical pipeline managers, use clinical-grade downstream purification capability as their primary variable to distinguish qualified innovators from computational generalists. An organization possessing an elegant algorithm but lacking the physical infrastructure to purify a 10kb transcript without shearing is systematically excluded from serious partnering discussions.
Incumbents like Immorna Biotherapeutics, Inc. possess a specific advantage: they have already navigated the complex IND clearance process for massive saRNA constructs and established immuno oncology assay validated delivery mechanisms. This advantage persists because the regulatory learning curve for large-format RNA is steep, and early movers have essentially written the quality control standards alongside regulators. To replicate this position, a challenger must build a highly specialized delivery capability, specifically, novel ionizable lipid libraries designed explicitly for large-transcript endosomal escape, rather than attempting to engineer around ironclad alphavirus patents using older, easily sheared nucleic acid isolation and purification technologies.
As the market progresses toward 2036, major pharmaceutical buyers actively resist lock-in by aggressively funding early-stage platform developers with alternative, non-alphavirus replicon designs. A tension exists between these massive buyers, who desire commoditized, modular rna targeted small molecules and RNA components, and the dominant saRNA vendors, whose incentives lie in maintaining closed, end-to-end proprietary platforms. Ultimately, the market trajectory points toward further concentration, as the extreme capital requirements of clinical-stage oncology trials force smaller innovators to be absorbed by major incumbents seeking to replace their failing linear mRNA solid tumor pipelines.
| Metric | Value |
|---|---|
| Quantitative Units | USD 112.0 Million to USD 643.2 Million, at a CAGR of 19.10% |
| Market Definition | The market covers the biological engineering, construct design, and lipid/viral delivery of self-replicating viral-derived RNA specific to oncology. It captures the technologies enabling intracellular payload amplification, functionally separating these dose-sparing platforms from conventional linear mRNA. |
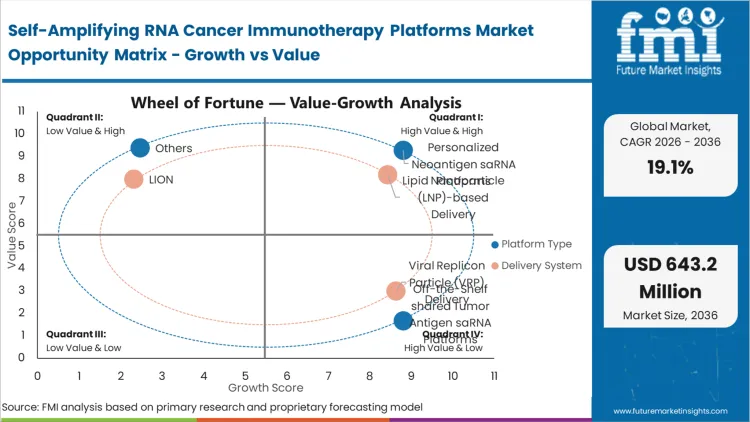
| Platform type Segmentation | Personalized neoantigen saRNA platforms, Off-the-shelf shared tumor antigen saRNA platforms, Cytokine-encoding saRNA immunotherapy platforms, saRNA amplifier or adjunct platforms for cell therapy |
| Delivery system Segmentation | Lipid nanoparticle (LNP)-based delivery, Viral replicon particle (VRP) delivery, LION / emulsion-based delivery, Polymeric or hybrid nanoparticle delivery |
| Cancer focus Segmentation | Solid tumors, Breast and gynecologic cancers, Gastrointestinal cancers, Thoracic and head & neck cancers, Virus-associated malignancies |
| Therapeutic configuration Segmentation | Standalone therapeutic cancer vaccines, Checkpoint inhibitor combination platforms, Cytokine-expressing immune activation platforms, CAR-T / cell-therapy amplification platforms |
| Developer type Segmentation | Clinical-stage biotech innovators, Academic cancer centers, Pharma-partnered translational programs, Government-backed research collaborations |
| Regions Covered | Asia Pacific, North America, Europe |
| Countries Covered | Japan, China, United States, Germany, United Kingdom, India, South Korea, and 40 plus countries |
| Key Companies Profiled | BioNTech SE, Arcturus Therapeutics Holdings Inc., Meiji Seika Pharma Co., Ltd., Immunomic Therapeutics, Inc., Immorna Biotherapeutics, Inc., HDT Bio Corp., AlphaVax, Inc. |
| Forecast Period | 2026 to 2036 |
| Approach | Chief Scientific Officers and Translational Oncology Directors were interviewed to assess pipeline maturity. The baseline anchors to verified IND clearances and active clinical evaluations. Forecasts were cross-validated against peer-reviewed technical assumptions regarding dose-sparing ratios and delivery platform viability. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the Self-Amplifying RNA Cancer Immunotherapy Platforms Market in 2026?Compare saRNA and mRNA for cancer immunotherapy use?
The industry value reaches USD 112.0 million in 2026, signalling the transition of self-amplifying platforms from academic computational models into heavily funded, IND-cleared clinical evaluations targeting intractable solid tumours.
What will it be valued at by 2036?
The market is projected to reach USD 643.2 million by 2036. This substantial accumulation reflects the widespread replacement of early-generation linear mRNA programs with replicon architectures capable of extreme dose-sparing economics.
What CAGR is projected?
The 19.10% CAGR reflects the pace at which developers are solving massive-transcript downstream purification bottlenecks, rather than the raw speed of antigen discovery. The rate is constrained by the physical difficulty of manufacturing 10kb-plus constructs without catastrophic batch shearing.
Which Platform type segment leads?
Personalized neoantigen saRNA platforms capture 42.6% share because off-the-shelf shared antigens consistently fail to overcome highly immunosuppressive, patient-specific tumor microenvironments. Pipeline managers utilize these specific platforms to align the replicon's amplification power with precise MHC-binding profiles.
Which Delivery system segment leads?
Lipid nanoparticle (LNP)-based delivery holds 51.8% of the market. This mechanism dominates because naked 12kb RNA transcripts are instantly destroyed by ubiquitous ribonucleases in the bloodstream, forcing reliance on highly specialized, low-shear lipid encapsulation protocols to achieve cellular entry.
Which Cancer focus segment leads?
Solid tumors dominate with 63.1% share. The unique capacity of saRNA to force continuous, localized antigen expression breaks the systemic tolerance barriers that deeply fibrotic malignancies utilize to neutralize conventional systemic biologics.
What drives rapid growth?
The severe systemic toxicity associated with high-dose conventional mRNA forces translational directors to pivot toward saRNA. Replicon vectors solve this bottleneck by amplifying a minute initial payload intracellularly, delivering sustained T-cell activation without the dose-limiting inflammation of linear transcripts.
What is the primary restraint?
The extreme physical fragility of massive 9-to-12 kilobase RNA molecules bottlenecks commercial scale-up. Contract manufacturers face profound friction when attempting to adapt legacy downstream purification systems, which routinely shear the delicate saRNA backbone under pressure.
Which country grows fastest?
Japan advances at 21.4%, outpacing China's 20.8% trajectory because Japan combines a first-in-class commercial saRNA regulatory framework with deeply integrated domestic manufacturing initiatives specifically optimized for solid tumor interventions.
How does delivery vector toxicity impact adoption?
Developers must definitively uncouple the inflammatory profile of the lipid envelope from the immunogenicity of the replicon itself. Platforms that fail to fine-tune their ionizable lipids trigger severe hepatotoxicity before the viral polymerase can initiate meaningful payload amplification.
What dictates the shift from standalone to combination therapies?
While standalone cancer vaccines prove baseline mechanism functionality, advanced solid tumors heavily express exhaustion markers that neutralize isolated T-cell responses. This biological reality forces clinical-stage biotechs to integrate their saRNA constructs alongside established checkpoint inhibitors to unlock full therapeutic efficacy.
Why is foundational IP a critical competitive aligner?
The specific genetic sequences governing alphavirus-derived replication machinery are locked behind dense patent thickets controlled by a few clinical-stage biotechs. Incumbent pharmaceutical giants must aggressively license these platforms, as engineering novel, non-infringing replicon backbones requires years of unproven development.
How does China's market position differ from the United States?
China leverages deep state-supported biotech zones and highly localized raw material synthesis to aggressively compress IND clearance timelines. In contrast, the US market is defined by a dense, highly competitive venture capital ecosystem where biotechs must prove superior dose-sparing economics to survive intense patent scrutiny.
What role do academic cancer centers play in developer type segmentation?
Academic centers provide the highly centralized genomic sequencing required for accurate neoantigen identification. Their deeply characterized patient cohorts act as the critical testing ground for algorithms before biotech innovators scale those designs into commercial manufacturing.
Why do polymeric nanoparticle deliveries trail LNP adoption?
Polymeric hybrid systems struggle to consistently encapsulate massive 12kb transcripts without triggering early degradation or immune rejection. Until these alternative vectors match the predictable endosomal escape rates of finely tuned ionizable lipids, LNP formats will retain their dominant commercial position.
What limits the expansion of saRNA in South Korea?
South Korean procurement teams face intense pressure to secure reliable supplies of proprietary enzymatic capping analogs vital for large-transcript stability. The market growth is dictated by the fierce competition between major domestic conglomerates attempting to internalize these supply chains before scaling globally.
How do extended producer responsibility frameworks influence this market?
Unlike rigid physical goods, the regulatory equivalent in this space involves total lifecycle tracking of custom biological materials. Developers must guarantee the exact traceability of patient biopsies through the entire localized manufacturing loop, forcing massive investments in secure, closed-system digital tracking.
Why are standard liposomes excluded from this market's definition?
General-purpose liposomes lack the precisely engineered cationic lipid ratios necessary to physically accommodate a massive saRNA molecule. Attempting to force a 10kb replicon into a standard liposome results in catastrophic shearing and zero therapeutic viability.
What changes when saRNA is applied as an in vivo CAR-T amplifier?
Utilizing a replicon to locally reprogram immune cells inside the patient eliminates the immense cost and complexity of ex vivo cell manipulation. This approach fundamentally transforms the market from a vaccine-centric model toward fully integrated, scalable cellular therapy.
How do low-shear microfluidic mixing protocols influence market dynamics?
Contract manufacturers who implement low-shear microfluidics capture a decisive advantage by drastically reducing batch failure rates. Without this specific hardware capability, developers cannot consistently formulate intact saRNA, creating a hard ceiling on their clinical trial expansion capabilities.
Explain the self-amplifying RNA cancer platform market in simple terms?
The mRNA cancer vaccine market is often the baseline for understanding this sector. While traditional mRNA delivers instructions that degrade quickly, self-amplifying RNA (saRNA) acts like a biological copy machine inside the cell. Once delivered, it copies itself repeatedly, producing cancer-fighting antigens for weeks from a very small initial dose, solving the toxic side effects of high-dose traditional therapies.
Compare saRNA and mRNA for cancer immunotherapy use?
In a direct saRNA vs mRNA cancer vaccine comparison, traditional mRNA requires massive, frequent doses to keep fighting a solid tumor, which frequently poisons the patient's liver with lipid accumulation before the tumor dies. In contrast, saRNA requires a fraction of the dose because it amplifies itself autonomously once inside the target cell.
What is a self-amplifying RNA cancer immunotherapy platform?
It is a highly engineered biological system, combining a viral-derived replicating RNA backbone, a cancer-specific payload, and a specialized lipid delivery envelope, designed to trigger a profound, long-lasting immune response against tumors without the dose-limiting toxicity of older technologies.
How is saRNA different from mRNA in cancer vaccines?
The difference is the inclusion of genes encoding an RNA-dependent RNA polymerase. This viral machinery allows the saRNA construct to autonomously copy its own instructions inside the cell, meaning developers can use drastically lower initial doses to achieve significantly longer therapeutic expression.
Are any saRNA cancer vaccines approved?
While self-amplifying RNA has reached commercial approval for infectious diseases (such as COVID-19 in Japan), the oncology applications remain strictly in clinical and translational evaluation phases. Biotechs are currently focused on proving safety and uncoupling delivery toxicity from payload efficacy in early-stage solid tumor trials.
Which companies are developing saRNA cancer therapies?
The landscape is heavily concentrated around foundational IP holders and specialized biotech innovators such as BioNTech SE, Arcturus Therapeutics Holdings Inc., Meiji Seika Pharma Co., Ltd., Immorna Biotherapeutics, Inc., and HDT Bio Corp.
Why is saRNA promising for oncology?
The core promise is dose-sparing. Because the RNA copies itself, translational teams can bypass the extreme systemic toxicity that halts conventional linear mRNA trials, allowing them to safely target dense, immunosuppressive solid tumors that require sustained T-cell activation over weeks rather than days.
How does a self-amplifying RNA vs DNA cancer vaccine comparison look functionally?
Unlike DNA vaccines, which must physically cross into the cell's nucleus to function (risking genomic integration), saRNA platforms operate entirely within the cytoplasm. This provides the consistent expression profile typically associated with DNA vectors without the severe regulatory and biological risks of nuclear entry.
How does saRNA compare in a saRNA vs viral vector cancer immunotherapy evaluation?
While viral vectors (like adenoviruses) excel at delivery, patients rapidly develop neutralizing antibodies against the viral shell, preventing repeated dosing. Self-amplifying RNA delivers the replication power of a virus but uses a synthetic lipid envelope, allowing for repeated patient dosing without triggering anti-vector immunity.
How does a saRNA platform vs dendritic cell vaccine platform evaluation shape procurement?
Dendritic cell platforms require extracting the patient's cells, engineering them in a specialized lab, and re-infusing them, an incredibly slow and expensive process. A personalized cancer vaccine market analysis shows saRNA platforms bypass this entirely by delivering the instructions directly into the patient's body in vivo, massively simplifying the manufacturing and supply chain.
What roles do the lipid nanoparticle oncology market play in saRNA advancement?
Without advanced LNP engineering, saRNA cannot exist as a commercial therapy. The sheer size of a 10kb-plus replicon requires highly specialized lipid ratios to physically protect the molecule in the bloodstream and ensure it escapes the endosome once inside the cell.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.