The demand for electrophoresis equipment and supplies in the USA is valued at USD 0.7 billion in 2025 and is forecast to reach USD 1.2 billion by 2035, recording a CAGR of 4.6%. Demand is supported by continued utilization of electrophoresis workflows in protein and nucleic acid separation across clinical diagnostics, academic laboratories, and R&D environments. Investments in life sciences, expansion of molecular testing, and increased biopharmaceutical development contribute to equipment procurement and consumable replenishment. Standardization of laboratory processes and quality-assurance compliance reinforces sustained supply requirements.
Gel electrophoresis systems lead adoption because they remain widely used for genetic analysis, routine biomarker screening, and protein-characterization protocols. These systems support reproducible sample preparation, cost-efficient operation, and compatibility with staining reagents and imaging platforms used in analytical laboratories. Enhancements in gel-casting formats, buffer chemistry, and detection sensitivity continue to influence system upgrades.
Demand is strongest in the West, South, and Northeast, supported by established research universities, biotechnology firms, and diagnostic-testing facilities. Key suppliers include Thermo Fisher Scientific, Bio-Rad Laboratories, Agilent Technologies, Cytiva, and Analytik Jena. Their offerings include electrophoresis chambers, power supplies, precast gels, and buffers used in genomic and proteomic research workflows.
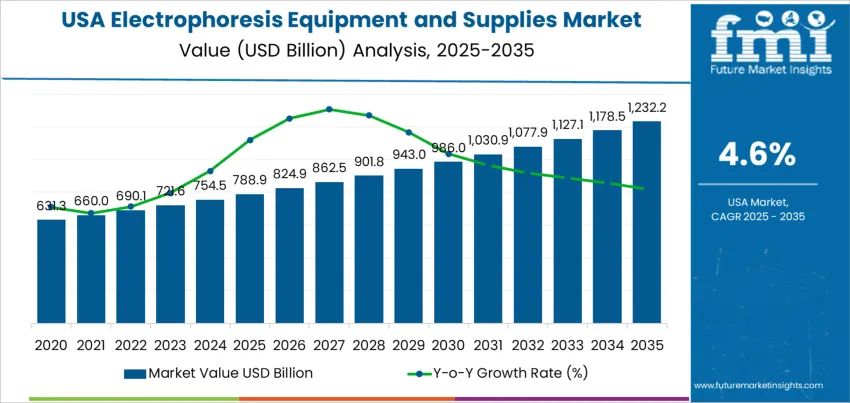
Saturation for demand for electrophoresis equipment and supplies in the United States reflects a mature footprint across academic laboratories, clinical diagnostics, and biopharmaceutical research. Most institutions with molecular biology operations already maintain installed systems, which reduces the pace of first-time purchases during the later forecast period. Replacement and maintenance cycles become the main source of equipment demand, while consumables continue to generate recurring volume tied to ongoing experiment activity.
Growth potential remains higher in segments adopting advanced separation methods such as capillary electrophoresis and automated platforms. These formats support improved throughput and accuracy in applications including protein characterization and genetic testing. Smaller research organizations and emerging biotechnology firms provide additional expansion by increasing capacity in early-stage discovery.
Saturation pressures hold back steep acceleration because fundamental electrophoresis techniques are well established and change gradually. Activity does not plateau completely. Expanding clinical applications, education-based procurement, and continuous supply turnover sustain a stable progression. The overall pattern indicates high institutional penetration with moderated equipment growth, balanced by consistent demand for gels, buffers, and reagents used in routine laboratory workflows.
| Metric | Value |
|---|---|
| USA Electrophoresis Equipment and Supplies Sales Value (2025) | USD 0.8 billion |
| USA Electrophoresis Equipment and Supplies Forecast Value (2035) | USD 1.2 billion |
| USA Electrophoresis Equipment and Supplies Forecast CAGR (2025 to 2035) | 4.6% |
Demand for electrophoresis equipment and supplies in the USA is increasing because research laboratories, biotechnology companies and clinical testing facilities continue expanding work in genomics, proteomics and molecular diagnostics. Electrophoresis supports DNA and protein separation for applications including disease biomarker discovery, forensic analysis and quality control in biologic drug development. Academic institutions frequently procure gel systems, buffers and imaging tools to support teaching and federally funded research programs.
Growth in biopharmaceutical manufacturing encourages regular use of electrophoresis during process validation and product release testing. Hospitals and reference laboratories perform protein electrophoresis to help evaluate blood disorders and monitor therapeutic responses. Single use consumables such as precast gels and sample preparation reagents generate ongoing demand because they streamline workflows and reduce setup time.
Automation and digital imaging improvements support faster data interpretation, which benefits high throughput clinical and industrial environments. Supply chain availability through established laboratory distributors maintains consistent access for large networks and smaller regional labs. Constraints include cost considerations for advanced capillary systems and training requirements for technicians unfamiliar with more automated platforms. Some laboratories transition part of their workflows to alternative separation or sequencing technologies, which influences purchasing priorities over time.
Demand for electrophoresis equipment and supplies in the United States is supported by clinical diagnostics adoption, biomarker evaluation, infectious disease testing, and proteomic research. Healthcare providers and laboratory networks prioritize high-throughput, precision-driven separation methods to support accurate molecular detection. Growth aligns with increased chronic disease testing, drug development activity, and automation in clinical workflows. Procurement trends reflect reliability, reagent compatibility, and regulatory-compliant performance across diagnostic and scientific settings.
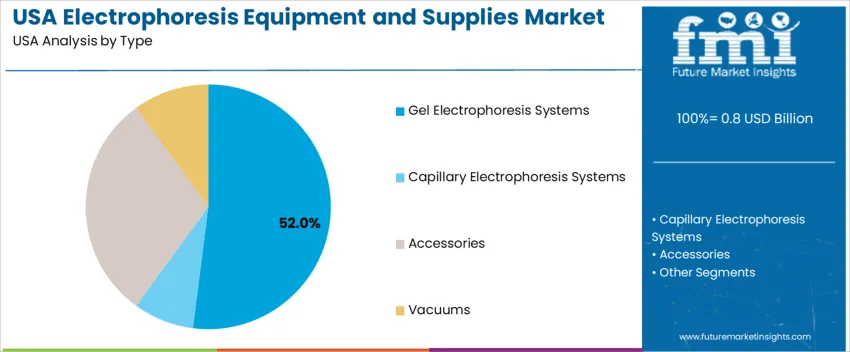
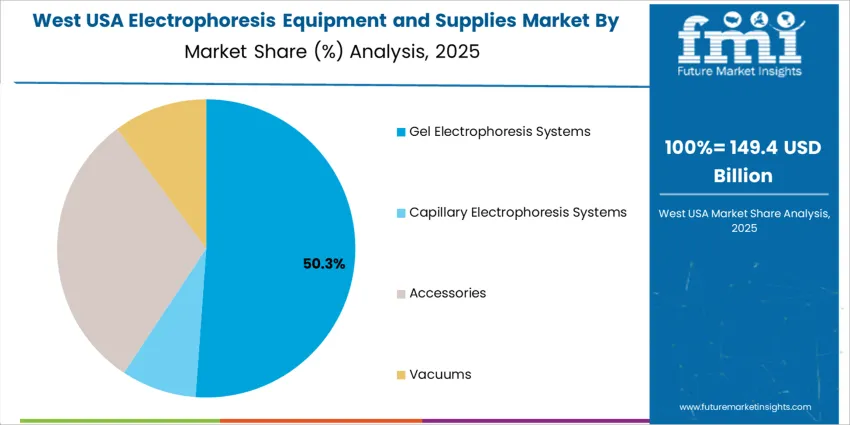
Gel electrophoresis systems account for 52.0%, driven by their continued utility in protein and nucleic acid separation within clinical laboratories and academic research. Accessories represent 30.0%, including gels, buffers, power supplies, and imaging systems required for recurring testing workflows. Vacuums hold 10.0%, primarily as supplementary laboratory components supporting sample handling. Capillary electrophoresis systems represent 8.0%, used in specialty molecular diagnostics requiring high resolution and reduced reagent volumes. Equipment selection in the United States reflects operational familiarity, cost-effective maintenance, and the need for flexible configurations adaptable to varied research and diagnostic protocols.
Key Points:
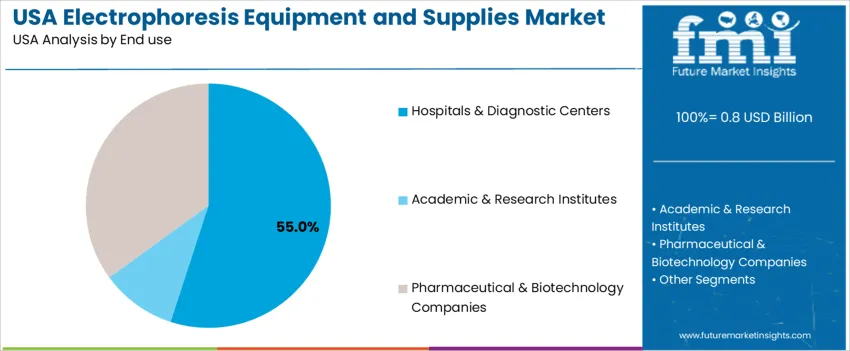
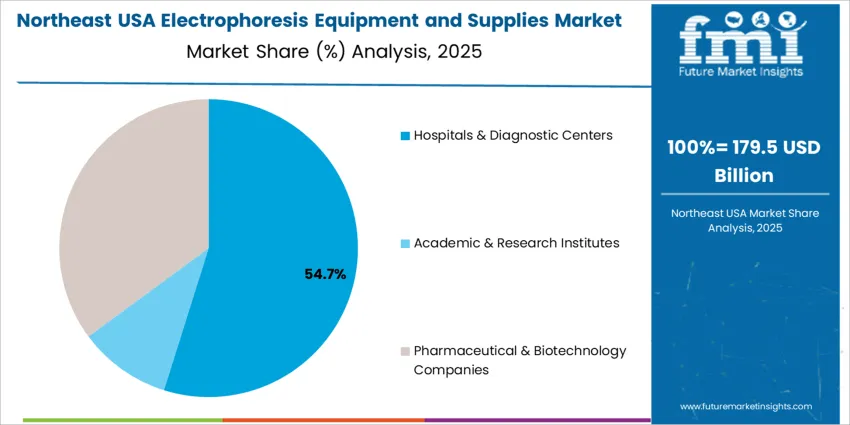
Hospitals and diagnostic centers represent 55.0%, driven by increased utilization of electrophoretic assays for blood disorders, cancer monitoring, and infectious disease diagnostics. Pharmaceutical and biotechnology companies hold 35.0%, reflecting research investments in biopharmaceutical development and protein characterization. Academic and research institutes account for 10.0%, supporting molecular biology education and innovation pipelines. End-user priorities focus on accuracy, procedural standardization, consumable availability, and scalability across automated laboratory settings.
Key Points:
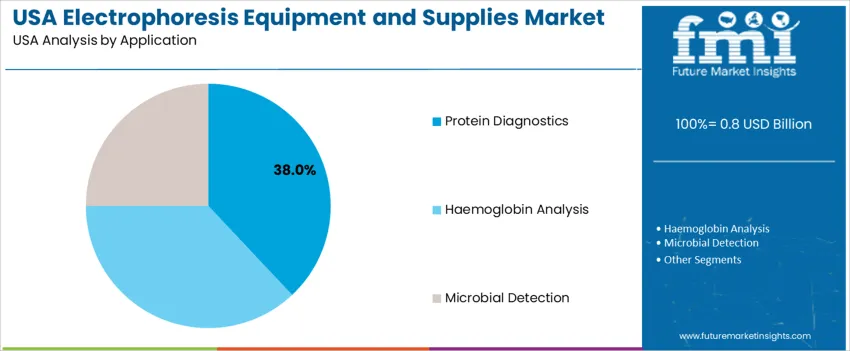
Protein diagnostics account for 38.0%, driven by widespread testing for monoclonal gammopathies, enzyme disorders, and therapeutic protein monitoring. Hemoglobin analysis holds 37.0%, supported by sickle cell screening, thalassemia monitoring, and anemia-related investigations. Microbial detection represents 25.0%, used in food safety testing, antibiotic resistance research, and pathogen surveillance. U.S. application trends emphasize early disease detection, precision therapeutic management, and expanded use of automated electrophoretic workflows.
Key Points:
Growth of molecular diagnostics, increased academic and biotech research funding and rising demand for DNA and protein analysis in clinical laboratories are driving demand.
In the United States, electrophoresis systems remain essential for genetic testing, forensic identification and protein characterization performed in hospital labs and research institutions. Federal and private investment in biotechnology and personalized medicine increases laboratory capacity for genomic studies in cancer and rare disease detection. Universities and biotech startups in states such as California, Massachusetts and North Carolina purchase electrophoresis equipment to support early-stage drug discovery and academic research programs. Clinical labs use gel and capillary electrophoresis for confirmation testing to support quality assurance under regulatory requirements. Consumables including precast gels, stain kits and imaging accessories generate recurring purchases linked to routine workflow volume.
High equipment cost for advanced systems, workflow competition from next-generation sequencing and staffing shortages in laboratory settings restrain demand.
Capillary and automated electrophoresis instruments require capital investment and regular maintenance; which smaller labs evaluate carefully before upgrading from manual systems. Expanding adoption of sequencing platforms can reduce gel-based DNA separation in some genetic workflows, lowering incremental equipment growth in facilities focused on high-throughput molecular diagnostics. Staffing shortages in clinical labs limit operational bandwidth for new system onboarding and validation. Budget oversight in publicly funded institutions leads to careful procurement cycles. These economic and operational pressures slow broad modernization across all research segments.
Shift toward automated capillary electrophoresis, increased demand in biopharmaceutical quality control and rising integration with digital documentation define key trends.
Biopharma manufacturers adopt advanced electrophoresis systems to monitor product purity and charge variants during monoclonal antibody development and batch release. Automated capillary platforms gain visibility due to improved reproducibility and reduced hands-on technician time aligned with lean-lab initiatives. Digital gel documentation tools are integrated with laboratory information systems to support compliance, traceability and secure data storage. Education-focused suppliers expand compact, easy-to-use systems for teaching labs to ensure skill development in future researchers. These developments indicate sustained, method-driven demand for electrophoresis equipment and supplies across the United States clinical and life-science research landscape.
Demand for electrophoresis equipment and consumables in the United States is expanding as molecular diagnostics, genomic research, and protein analysis applications increase across clinical, academic, and biopharmaceutical laboratories. Adoption aligns with investments in precision-medicine workflows, higher testing throughput, and consumable USAge tied to recurring sample processing. Universities and biotech clusters remain major buyers of gel systems, capillary platforms, imaging units, and single-use reagents such as gels, buffers, and stains. West USA leads growth at 5.2% CAGR, followed by South USA at 4.7%, Northeast USA at 4.2%, and Midwest USA at 3.6%. Regional expansion reflects infrastructure maturity, funding availability, and procurement cycles within scientific organizations.
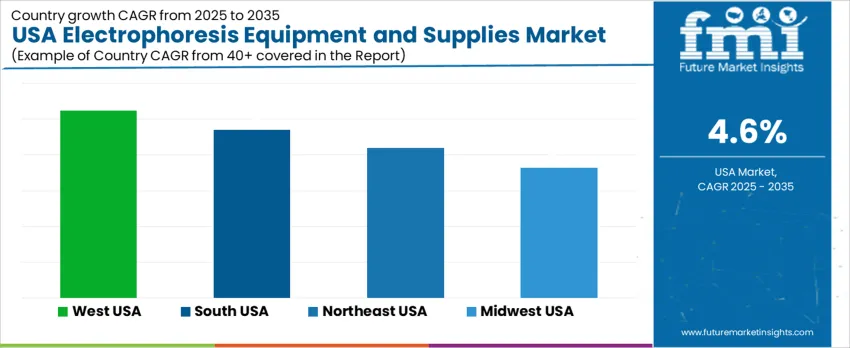
| Region | CAGR (2025 to 2035) |
|---|---|
| West USA | 5.2% |
| South USA | 4.7% |
| Northeast USA | 4.2% |
| Midwest USA | 3.6% |
West USA expands at 5.2% CAGR supported by concentrated genomics and life-science innovation hubs in California and Washington. Biomedical research facilities prioritize enhanced resolution systems for proteomic characterization and nucleic-acid separation. Funding from venture-backed biotech startups strengthens procurement of mid- to high-end instruments with automated workflows that reduce manual variability. Clinical laboratories adopt standardized consumable kits aligned with diagnostic testing requirements in oncology and infectious diseases. Local manufacturing and distribution nodes help reduce reagent stockouts, enabling dependable replenishment cycles. Procurement emphasizes compatibility with digital documentation software for traceability and reproducibility across regulated environments.
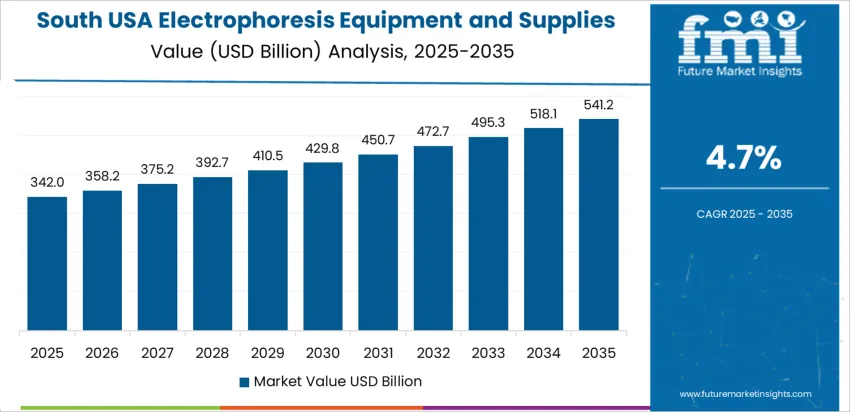
South USA shows 4.7% CAGR, linked to expanding medical-research universities, biomanufacturing corridors, and hospital laboratory networks across Texas, North Carolina, and Florida. Adoption focuses on clinically oriented electrophoresis such as serum-protein separation for metabolic and cancer-related diagnostics. CDMO facilities incorporate electrophoresis QC methods for biologics development. Regional purchasing prioritizes operational reliability and service availability due to dispersed lab locations. Workforce-training collaborations strengthen USAge consistency by standardizing gel and capillary protocols.
Northeast USA grows at about 4.2% CAGR, shaped by high R&D expenditures from leading pharmaceutical companies and academic institutions in New York, New Jersey, and Massachusetts. Procurement decisions focus on validated technologies necessary for regulatory-aligned research. Advanced laboratories expand capillary electrophoresis for higher throughput analysis in monoclonal-antibody development. Consumables USAge remains high due to continuous method optimization in preclinical drug discovery. Buyers evaluate systems for long-term data integration into electronic laboratory records.
Midwest USA maintains 3.6% CAGR, led by clinical-diagnostic centers and university research programs across Illinois, Ohio, and Michigan. Demand supports protein gel electrophoresis used in autoimmune disorder detection and academic training labs across undergraduate and graduate programs. Buyers seek cost-efficient instruments with proven durability for repeated instructional use. Industrial manufacturing of diagnostic products reinforces consumable sourcing near production hubs, ensuring fulfillment stability. Decision-making prioritizes affordability and compatibility with legacy platforms retained across long equipment cycles.
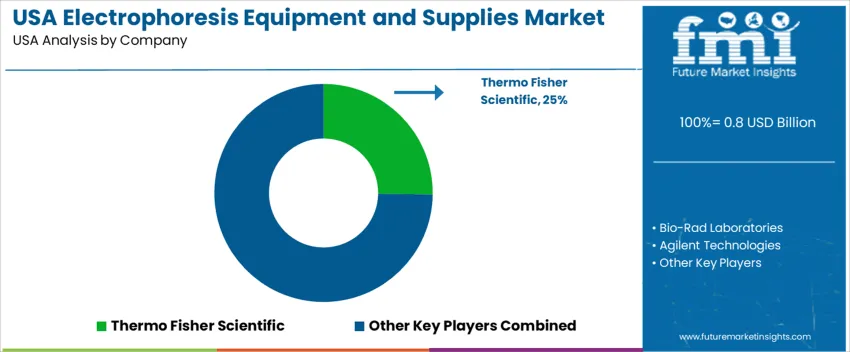
Electrophoresis equipment and supplies in the United States are used in molecular biology, clinical diagnostics, academic research, and biopharmaceutical development. Demand centers on protein and nucleic-acid separation systems, along with validated consumables that support repeatable results in high-throughput laboratory environments. Buyers emphasize workflow reliability, gel and buffer consistency, and compatibility with automated imaging and data-analysis tools. Institutions prioritize suppliers with technical field support and stable reagent availability to reduce experimental variability.
Thermo Fisher Scientific holds an estimated 25.2% share. Its platforms and ready-to-use consumables support nationwide research universities, clinical labs, and biopharma facilities. Broad distribution and integrated software contribute to continued adoption. Bio-Rad Laboratories maintains strong participation through electrophoresis instruments and consumables widely used for protein separation across biomedical research labs. Agilent Technologies provides capillary electrophoresis systems aligned with analytical workflows in pharmaceutical development and regulated testing environments.
Cytiva (formerly part of GE Healthcare) engages in specialized separation systems used in bioprocess analysis and advanced research programs. Analytik Jena contributes capital equipment to laboratories requiring repeatable gel-based workflows in genomics and proteomics. Competitive positioning in the United States depends on accuracy of separation, gel-format flexibility, regulatory compliance for clinical applications, and dependable service coverage supporting continuous research output and diagnostic reliability.
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Type | Gel Electrophoresis Systems, Capillary Electrophoresis Systems, Accessories, Vacuums |
| End Use | Hospitals & Diagnostic Centers, Academic & Research Institutes, Pharmaceutical & Biotechnology Companies |
| Application | Protein Diagnostics, Haemoglobin Analysis, Microbial Detection |
| Regions Covered | West USA, South USA, Northeast USA, Midwest USA |
| Key Companies Profiled | Thermo Fisher Scientific, Bio-Rad Laboratories, Agilent Technologies, Cytiva, Analytik Jena |
| Additional Attributes | Dollar sales by system type, end-use sector, and diagnostic application; spending focus on advanced protein and DNA/RNA separation platforms; procurement growth in translational research and clinical labs; increased automation in gel documentation and capillary systems; demand driven by precision medicine initiatives and infectious disease diagnostics; cluster concentration in Midwest and Northeast research corridors; competitive differentiation based on throughput, resolution, and reagent ecosystem. |
How big is the demand for electrophoresis equipment and supplies in USA in 2025?
The demand for electrophoresis equipment and supplies in USA is estimated to be valued at USD 0.8 billion in 2025.
What will be the size of electrophoresis equipment and supplies in USA in 2035?
The market size for the electrophoresis equipment and supplies in USA is projected to reach USD 1.2 billion by 2035.
How much will be the demand for electrophoresis equipment and supplies in USA growth between 2025 and 2035?
The demand for electrophoresis equipment and supplies in USA is expected to grow at a 4.6% CAGR between 2025 and 2035.
What are the key product types in the electrophoresis equipment and supplies in USA?
The key product types in electrophoresis equipment and supplies in USA are gel electrophoresis systems, capillary electrophoresis systems, accessories and vacuums.
Which end use segment is expected to contribute significant share in the electrophoresis equipment and supplies in USA in 2025?
In terms of end use, hospitals & diagnostic centers segment is expected to command 55.0% share in the electrophoresis equipment and supplies in USA in 2025.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.