AI-enabled Wound Analysis Market
AI-enabled Wound Analysis Market Size and Share Forecast Outlook 2026 to 2036
AI-enabled Wound Analysis Market Forecast and Outlook 2026 to 2036
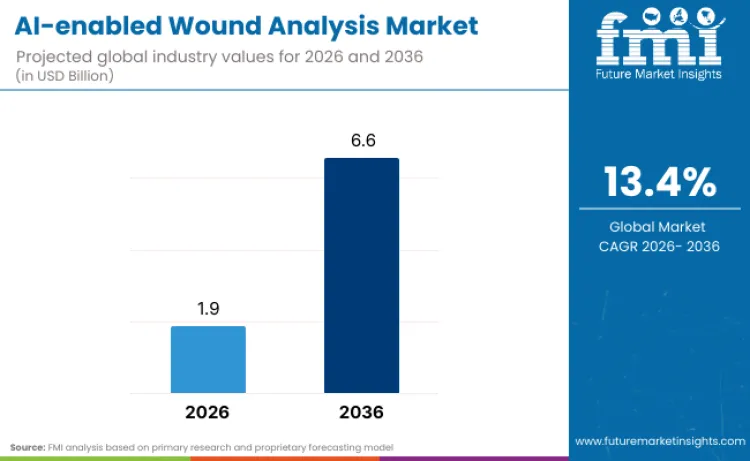
The AI-enabled wound analysis market is on a trajectory to surge from USD 1.9 billion in 2026 toward USD 6.6 billion by 2036, driven by 13.4% CAGR. Based on the analysis by Future Market Insights, growth in the AI-enabled wound analysis industry stems from expanding patient loads and clinician burnout which are forcing healthcare systems to adopt digital tools. These guidelines show that digital management systems significantly reduce healthcare resource use while improving assessment accuracy. As per FMI's projection, regulatory support is converting experimental pilots into standard clinical protocols. Neil Sharma, CCO of Swift Medical, noted in January 2025 that serving one million patients is a testament to provider dedication using peer-reviewed technologies.
‘This milestone is a testament to the relentless dedication of our healthcare provider partners who are leveraging peer-reviewed, scalable, and widely adopted technologies like Swift Skin & Wound to drive meaningful improvements in patient outcomes.’ - Neil Sharma, Chief Commercial Officer, Swift Medical
Diabetic foot ulcers represent 24.8% of industry value, driving manufacturers to prioritize preventative solutions over passive measurement. Molnlycke Health Care validated this high-growth niche by investing USD 8 million in Siren during January 2025. This capital injection aims to scale smart textile technologies that detect temperature changes before ulcers form, addressing a massive unmet need.
Manufacturers are shifting strategies from simple wound imaging to predictive analytics that prevent costly hospitalizations and improve long-term patient mobility.
Summary of the AI enabled Wound Analysis Market
What Is Growth Outlook for AI‑enabled Wound Analysis Market as per Future Market Insights Projection?
Future Market Insights projects the AI‑enabled wound analysis market to grow at a CAGR of 13.4% from 2026 to 2036, rising from USD 1.9 billion in 2026 to USD 6.6 billion by 2036.
FMI Research Approach: Based on FMI’s proprietary bottom‑up forecasting model that incorporates revenue estimation, historical adoption benchmarking across chronic wound technologies, primary interviews with digital health vendors and wound care providers, and scenario analysis aligned with regulatory adoption timelines.
How Do FMI Analysts Perceive the AI‑enabled Wound Analysis Market to Evolve?
FMI analysts perceive the market shifting from basic wound imaging toward predictive, preventive, and reimbursement‑aligned AI platforms built directly into routine clinical workflows.
FMI Research Approach: Supported by comparative analysis of pilot deployments versus reimbursed platforms, adoption patterns across hospital and home healthcare settings, FDA‑issued AI lifecycle management requirements, and WHO digital clinical decision support guidance.
Which Country Holds the Largest Share in the Global AI‑enabled Wound Analysis Market?
The United States holds the largest share of global market value.
FMI Research Approach: Based on FMI’s country‑level revenue modeling by care setting, value‑based care reimbursement frameworks and readmission penalties, FDA clearance records for AI‑enabled wound analysis software, and commercial deployment disclosures from major USA hospital systems.
How Large Will the AI‑enabled Wound Analysis Market Be by 2036?
The AI-enabled wound analysis market is estimated to reach USD 6.6 billion by 2036.
FMI Research Approach: Derived from FMI’s long‑term revenue forecast using product and application mix, scaling assumptions for smartphone‑based applications and remote monitoring, exclusion of traditional wound care products and clinical services, and cross‑validation against digital diagnostics growth curves.
What Is Definition of the AI‑enabled Wound Analysis Market?
The market consists of AI‑enabled software platforms and technology‑driven systems used to assess, measure, monitor, and analyze wounds in both clinical and remote settings.
FMI Research Approach: Based on FMI’s taxonomy, including validation of product scope across apps, platforms, and imaging systems; alignment with software‑as‑a‑medical‑device classifications; and exclusion of non‑AI dressings and manual clinical services.
What Are Globally Unique Trends Shaping the AI‑enabled Wound Analysis Market?
Key global trends include mobile‑first deployment, regulatory standardization of AI lifecycle management, and integration of AI analytics with physical wound care products.
FMI Research Approach: Evidenced by interoperability mandates, smartphone penetration across public health systems, AI governance guidance issued by global health authorities, and manufacturer investments in smart textiles and sensor‑embedded dressings.
AI-enabled Wound Analysis Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 1.9 billion |
| Industry Value (2036) | USD 6.6 billion |
| CAGR (2026-2036) | 13.4% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Why Demand for AI-enabled Wound Analysis Market is Growing?
Increasing economic burdens associated with chronic wounds are fundamentally reshaping procurement. FMI analysts opine that this financial pressure is forcing payers to reimburse automated measurement tools that prove cost-efficiency. Clinically, Swift Medical solidified adoption arguments with a January 2025 study showing its AI technology is 39% more accurate in measuring wounds across diverse skin tones compared to traditional methods. Such data confirms that automated analysis is evolving from a clinical luxury into an essential cost-containment mechanism.
How is the AI-enabled Wound Analysis Market Segmented?
The AI-enabled wound analysis market is segmented by product type, application, and end user, with a decisive shift toward mobile-first and condition-specific solutions. Smartphone-based apps command a 22.45% share, democratizing access to clinical-grade measurement, while hospital end-users retain a 28.4% share due to complex inpatient needs. Diabetic foot ulcer applications also capture 24.8% revenue share, driving investments into specialized detection hardware rather than generic imaging tools.
Why do Smartphone-based Apps hold a Dominant Share?
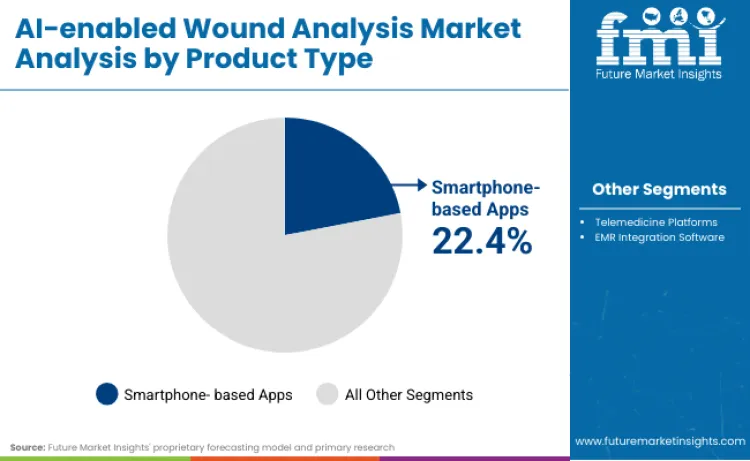
Smartphone-based applications hold a dominant 22.45% share as they leverage existing hardware to deliver clinical-grade wound measurement at a fraction of legacy costs. Ubiquity of high-resolution mobile cameras allows software providers to deploy updates remotely, bypassing lengthy hardware replacement cycles. NHS Digital Wound Care Standards, fully implemented by September 2025, mandate interoperable digital assessments, effectively making smartphone apps the standard of care for district nurses. Swift Medical utilized this exact delivery model to serve over 1 million patients by early 2025. Scalability inherent in app-based models allows rapid penetration into home healthcare settings, outperforming bulky proprietary consoles.
Why does the Diabetic Foot Ulcer Segment Lead in Clinical Application?
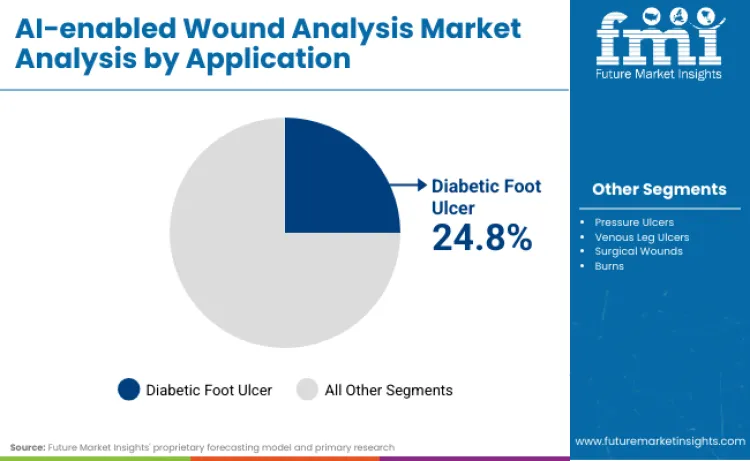
Diabetic foot ulcers capture 24.8% of market value, driven by rising diabetes prevalence and catastrophic costs associated with amputation. Prevention focuses on early detection of inflammation, capability traditional visual exams lack. Mölnlycke Health Care’s January 2025 investment in Siren,a maker of temperature-sensing smart socks,targets this exact segment, aiming to reduce ulcer risk by 68%. Smart textiles bridge the gap between continuous monitoring and clinical intervention. Compelling return-on-investment data for payers, who save thousands per prevented ulcer, ensures this segment commands premium pricing and sustained growth through 2036.
How is Regulatory Standardization Altering Entry Barriers?
Regulators are moving from "hands-off" to defining strict lifecycle management for AI medical devices. FDA issued 'Artificial Intelligence-Enabled Device Software Functions' draft guidance in January 2025, creating specific requirements for modification protocols. This raises entry barriers, favoring established players with auditable data pipelines over small startups. The AI-enabled wound analysis market is shifting toward a consolidated landscape where only those capable of navigating complex compliance frameworks can survive.
How is the Integration of AI into Traditional Dressings Evolving?
Industry leaders are embedding sensors into physical products to create "smart" therapeutics. Mölnlycke Health Care invested USD 8 million in Siren in January 2025 to integrate temperature-sensing technology with interactive wound dressing portfolios. ‘Our strategic investment in Siren reflects our commitment to bringing the benefits of digital health to chronic wound care and pioneering innovative digital solutions that revolutionise healthcare delivery.’ - Zlatko Rihter, CEO, Mölnlycke Health Care This convergence bridges the gap between textiles and digital monitoring. Such hybrid products represent the future of the sector, transforming passive bandages into active diagnostic tools.
What impact are spin-offs having on medtech agility?
Conglomerates are shedding health units to unlock value. 3M completed the spin-off of its healthcare business as Solventum in April 2024, creating a dedicated entity for health IT. This separation allows Solventum to aggressively pursue AI partnerships without capital constraints of a diversified industrial conglomerate. FMI observes that these focused entities are now moving faster to acquire AI capabilities, intensifying competition for software startups.
How Will AI-enabled Wound Analysis Market Expansion Unfold Across Key Global Regions?
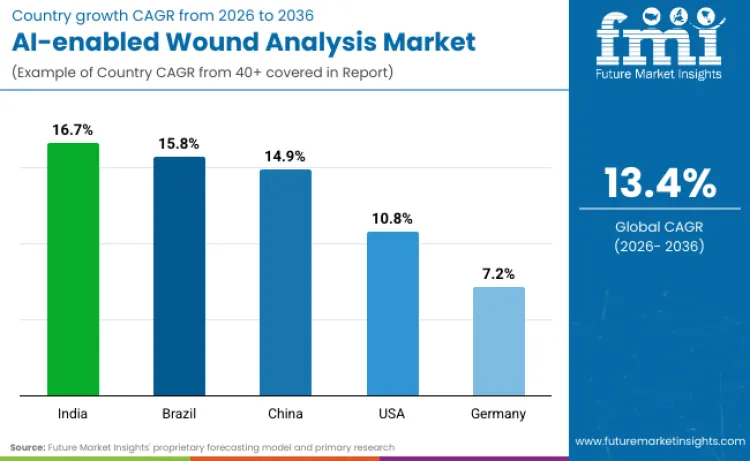
Global advanced wound care market reflect a fundamental divergence between developed and emerging economies, with the former battling reimbursement constraints while the latter race to expand access for rapidly aging populations. Emerging markets like India and Brazil are outpacing Western counterparts, recording double-digit CAGRs compared to Germany's steady 7.2%. FMI analysis suggests that this divergence is driven by 'leapfrog' adoption of mobile-first diagnostic tools in resource-constrained settings, contrasting with legacy infrastructure integration required in Europe. Resource-poor regions utilize AI to bridge specialist shortages, while developed nations focus on efficiency.
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 16.7% |
| Brazil | 15.8% |
| China | 14.9% |
| USA | 10.8% |
| Germany | 7.2% |
Source: Future Market Insights (FMI) historical analysis and forecast data.
How is Digital Infrastructure Catalyzing Growth in India?
Demand for AI-enabled wound analysis in India is poised to register a commanding 16.7% CAGR, catalyzed by dual engines of rising chronic disease prevalence and rapid digital infrastructure upgrades. Indian wound care sector value reached USD 173.6 million in 2024, driven heavily by diabetic foot ulcer cases. Apollo Hospitals validated this shift by allocating substantial budget to AI tools as of March 2025, specifically to automate documentation. Government support through Ayushman Bharat Digital Mission emphasizes interoperable health records, creating a fertile ecosystem for app-based wound registries. ‘Our objective is to free up two to three hours a day for doctors and nurses through the use of AI.’ - Sangita Reddy, Joint Managing Director, Apollo Hospitals This operational efficiency is critical for managing India's high patient-to-doctor ratios. Consequently, India is positioned as a high-volume growth engine, expected to rapidly operationalize mobile-AI solutions to bridge the specialist gap by 2036.
Why is Government Funding Pivotal for Brazil?
State-sponsored infrastructure is the primary force propelling the Brazil market, which is anticipated to expand at a 15.8% CAGR through 2036. Anvisa updated its regulatory agenda for 2024-2025 to streamline approval of 'Software as a Medical Device' (SaMD), reducing regulatory friction. Such clear state sponsorship guarantees funding for public health AI integrations. With regulatory friction decreasing and state funding increasing, Brazil is set to become the premier Latin American hub for regulated AI-medical device commercialization.
What Localization Strategies are driving China?
Local manufacturing mandates are reshaping the landscape in China, where the market is projected to grow at a 14.9% CAGR. Regulatory oversight is concurrently tightening, with NMPA issuing specific guidelines for AI medical device usability to ensure entrants meet strict quality standards. Physical capital commitment from global leaders validates the market's strategic importance. China is evolving from a passive import market to an active innovation hub, with domestic production expected to dominate local supply by 2036.
Why does Reimbursement Sophistication define the USA AI-enabled Wound Analysis Market?
Value-based care models that penalize hospital readmissions are the structural bedrock for sales in the United States, set to rise at a solid 10.8% CAGR. Hospitals are incentivized to deploy superior wound tracking to avoid penalties, creating an environment where Swift Medical could serve 1 million patients by January 2025. According to FMI's estimates, USA FDA January 2025 draft guidance on AI Lifecycle Management further stabilizes the sector by providing a clear roadmap for reimbursement-eligible devices. ‘At Swift Medical, our mission has always been to create the best outcomes for patients suffering from wounds by partnering with healthcare providers to Heal the World Faster and reduce unnecessary hospital visits.’ - Neil Sharma, Chief Commercial Officer, Swift Medical Scale of adoption confirms that AI wound care has moved from pilot phase to mass clinical utility. The USA will therefore remain the value leader, with market activity centered on high-margin, FDA-cleared predictive analytics platforms.
How is the DiGA Framework shaping Germany?
Germany stands apart as a reimbursement pioneer, with industry expansion projected at a 7.2% CAGR. Mölnlycke's strategic focus on Europe is evident in continued investment in digital ecosystems. Such unique reimbursement mechanism provides a stable revenue model for digital therapeutic developers. Germany serves as the proving ground for these apps, setting the standard for the wider EU market through 2036.
How is the Competitive Landscape in the AI-enabled Wound Analysis Market?
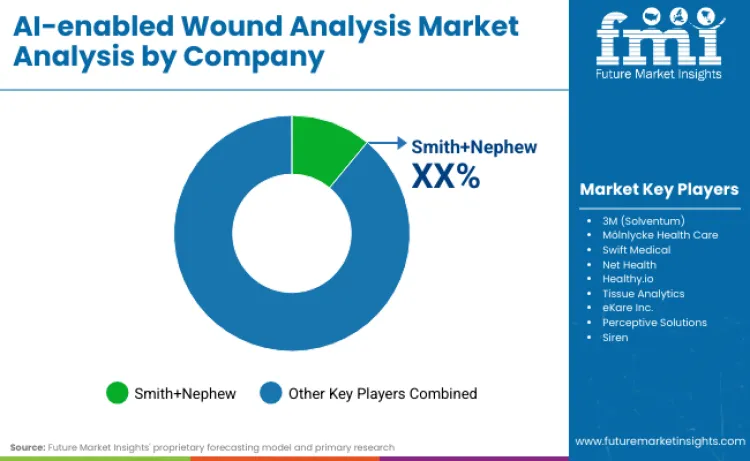
Smith+Nephew, holding an 18.4% market share, alongside 3M Solventum, are at the forefront, leveraging their extensive resources and seamless integration with traditional physical dressings. There is an increasing momentum in mergers and acquisitions as larger companies in the medical field actively search for innovative digital solutions to enhance their existing physical product offerings. A notable example of this shift is 3M's recent spin-off of Solventum in April 2024, which illustrates the movement towards more specialized business entities.
Top players control the market through early-mover advantages in wound debridement devices and established hospital networks that emerging software-only players cannot easily replicate without partnerships. Companies like Mölnlycke are leveraging early leads in prevention to set standards-of-care, while competitors explore smart textiles to establish differentiated positions. Mölnlycke’s investment in Siren during January 2025 for USD 8 million exemplifies this intensity. Zlatko Rihter, CEO of Mölnlycke, emphasizes this pivot toward pioneering innovative digital solutions that revolutionize healthcare delivery. This competitive frenzy ensures companies remain relevant in a sector rapidly moving away from analog management. Patent expirations for legacy hardware are forcing manufacturers to continuously innovate.
Recent Developments
- In January 2025, Mölnlycke Health Care announced a USD 8 million investment in Siren, a company specializing in temperature-sensing smart socks. This move marks a strategic shift from monitoring existing wounds to early detection of inflammation to prevent diabetic foot ulcers (DFUs) before they form.
- In January 2025, Swift Medical reported that it served over 1 million patients in 2024 using its AI-powered "Skin & Wound" platform.
Key Players in AI-enabled Wound Analysis Market
- Smith+Nephew
- 3M (Solventum)
- Mölnlycke Health Care
- Swift Medical
- Net Health
- Healthy.io
- Tissue Analytics
- eKare Inc.
- Perceptive Solutions
- Siren
Market Definition
The AI-enabled wound analysis market represents revenue generated from software platforms, applications, and technology-enabled systems that use artificial intelligence to assess, measure, monitor, and analyze wounds in clinical and remote care settings. As operationally defined in the article, the market measures commercially deployed AI-driven wound analysis solutions used to support clinical decision-making, documentation, and preventive care across chronic and acute wound types. Market sizing reflects expenditure on AI-enabled wound analysis products and platforms sold to healthcare providers and care organizations, analysed by product type, application, end user, and region, and expressed in USD billion.
The market includes AI-enabled wound analysis solutions explicitly segmented and quantified in the article, including smartphone-based apps, telemedicine platforms, EMR integration software, and AI-enabled imaging hardware. It covers applications across diabetic foot ulcers, pressure ulcers, venous leg ulcers, surgical wounds, and burns, with diabetic foot ulcers identified as the leading revenue-generating application. Revenue generated from deployment by hospitals, home healthcare agencies, long-term care facilities, and specialty clinics is included. The scope counts AI solutions used for wound measurement, predictive risk detection, digital documentation, remote patient monitoring, and integration with clinical workflows, including hybrid products combining smart textiles or dressings with digital analytics.
Market coverage spans North America, Europe, East Asia, South Asia, Latin America, and the Middle East & Africa. The market excludes revenues from traditional wound care products such as dressings, bandages, and topical therapies sold without embedded AI or digital analysis functionality. Clinical treatment services, hospital procedures, nursing labor, and professional care fees are not included. Sales of general-purpose medical imaging devices not positioned for AI-based wound analysis are excluded. Core electronic health record systems without AI wound-specific functionality are outside scope. Government healthcare spending, reimbursement payments, regulatory programs, and public funding initiatives referenced contextually are not counted as market revenue. Research-only AI tools, pilot studies, and non-commercial academic applications are also excluded unless deployed as commercial wound analysis solutions.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 1.9 billion |
| Product Type | Smartphone-based Apps, Telemedicine Platforms, EMR Integration Software, AI-enabled Imaging Hardware |
| Application | Diabetic Foot Ulcers, Pressure Ulcers, Venous Leg Ulcers, Surgical Wounds, Burns |
| End User | Hospitals, Home Healthcare Agencies, Long-term Care Facilities, Specialty Clinics |
| Regions Covered | North America, Europe, East Asia, South Asia, Latin America, Middle East & Africa |
| Countries Covered | United States, United Kingdom, Germany, India, China, Japan, Brazil and 40+ countries |
| Key Companies Profiled | Smith+Nephew, 3M (Solventum), Mölnlycke Health Care, Swift Medical, Net Health, Healthy.io, Tissue Analytics, eKare Inc., Perceptive Solutions, Siren |
| Additional Attributes | Dollar sales by product and application, regulatory-driven adoption analysis (FDA Lifecycle Guidance, DiGA framework, PBIA), AI integration in dressings assessment, remote patient monitoring evolution, cloud vs on-premise deployment evaluation, and competitive positioning analysis |
Source: FMI historical analysis and forecast data.
AI-enabled Wound Analysis Market by Segments
By Product Type:
- Smartphone-based Apps
- Telemedicine Platforms
- EMR Integration Software
- AI-enabled Imaging Hardware
By Application:
- Diabetic Foot Ulcers
- Pressure Ulcers
- Venous Leg Ulcers
- Surgical Wounds
- Burns
By End User:
- Hospitals
- Home Healthcare Agencies
- Long-term Care Facilities
- Specialty Clinics
By Region:
- North America
- Europe
- East Asia
- South Asia
- Latin America
- Middle East & Africa
Bibliography
- Health Technology Wales, & World Health Organization. (2024). Guidelines on digital wound management tools. HTW / WHO.
- Agência Nacional de Vigilância Sanitária. (2025, November). Anvisa modernization plan 2025: Medical device regulatory progress. Government of Brazil.
- Ministry of Science, Technology and Innovation. (2024, July). Brazilian artificial intelligence plan 2024-2028 (PBIA). Government of Brazil.
- NHS England. (2023, November). DAPB4086: Wound care information standard. National Health Service.
- Federal Institute for Drugs and Medical Devices. (2025, April). DiGA report 2024. BfArM, Government of Germany.
Frequently Asked Questions
What is the current global market size for AI-enabled wound analysis?
The global industry is valued at USD 1.9 billion in 2026, driven by increasing adoption of AI-enabled medical devices in chronic care.
What is the projected Compound Annual Growth Rate (CAGR) for the market over the next 10 years?
Revenue is projected to grow at 13.4% CAGR from 2026 to 2036.
Which application areas are the primary drivers of demand?
Diabetic foot ulcers drive highest value growth with 24.8% share, while smartphone apps maintain volume leadership.
What are the primary market risks or regulatory hurdles?
New FDA lifecycle management guidance (January 2025) creates stricter compliance barriers for software-as-a-medical-device entrants.
Who are the leading suppliers?
Smith+Nephew, 3M (Solventum), and Mölnlycke lead the market through extensive portfolios and recent investments in digital health.
Table of Content
- Executive Summary
- Market Introduction
- Market Definition
- Market Scope
- Market Taxonomy
- Research Methodology
- Study Framework
- Assumptions and Limitations
- Data Sources
- Market Estimation and Forecasting Approach
- Market Dynamics
- Drivers
- Restraints
- Opportunities
- Trends
- Global AI-enabled Wound Analysis Market Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global AI-enabled Wound Analysis Market Analysis by Product Type
- Global AI-enabled Wound Analysis Market Analysis by Application
- Global AI-enabled Wound Analysis Market Analysis by End User
- Global AI-enabled Wound Analysis Market Analysis by Region
- North America AI-enabled Wound Analysis Market Analysis
- United States
- Canada
- Mexico
- Latin America AI-enabled Wound Analysis Market Analysis
- Brazil
- Chile
- Rest of Latin America
- Western Europe AI-enabled Wound Analysis Market Analysis
- Germany
- United Kingdom
- France
- Italy
- Spain
- BENELUX
- Nordics
- Eastern Europe AI-enabled Wound Analysis Market Analysis
- Russia
- Poland
- Rest of Eastern Europe
- East Asia AI-enabled Wound Analysis Market Analysis
- China
- Japan
- South Korea
- South Asia & Pacific AI-enabled Wound Analysis Market Analysis
- India
- ASEAN
- Australia & New Zealand
- Middle East & Africa AI-enabled Wound Analysis Market Analysis
- GCC
- South Africa
- Rest of Middle East & Africa
- Competitive Landscape
- Assumptions and Acronyms
List of Tables
- Global AI-enabled Wound Analysis Market Size (USD Billion), 2021-2036
- Global AI-enabled Wound Analysis Market Y-o-Y Growth (%), 2021-2036
- Global AI-enabled Wound Analysis Market Absolute $ Opportunity (USD Billion), 2026-2036
- Global AI-enabled Wound Analysis Market Size by Product Type, 2021-2036
- Global AI-enabled Wound Analysis Market Size by Application, 2021-2036
- Global AI-enabled Wound Analysis Market Size by End User, 2021-2036
- Global AI-enabled Wound Analysis Market Size by Region, 2021-2036
- North America AI-enabled Wound Analysis Market Size by Country, 2021-2036
- Latin America AI-enabled Wound Analysis Market Size by Country, 2021-2036
- Western Europe AI-enabled Wound Analysis Market Size by Country, 2021-2036
- Eastern Europe AI-enabled Wound Analysis Market Size by Country, 2021-2036
- East Asia AI-enabled Wound Analysis Market Size by Country, 2021-2036
- South Asia & Pacific AI-enabled Wound Analysis Market Size by Country, 2021-2036
- Middle East & Africa AI-enabled Wound Analysis Market Size by Country, 2021-2036
- Global AI-enabled Wound Analysis Market Share (%) by Product Type, 2025
- Global AI-enabled Wound Analysis Market Share (%) by Application, 2025
- Global AI-enabled Wound Analysis Market Share (%) by End User, 2025
- Global AI-enabled Wound Analysis Market Share (%) by Region, 2025
List of Figures
- Global AI-enabled Wound Analysis Market Growth Framework
- AI-enabled Wound Analysis Market Value Chain Analysis
- AI-enabled Wound Analysis Market Technology Architecture Landscape
- AI-enabled Wound Analysis Market Opportunity Map
- AI-enabled Wound Analysis Market Scenario Forecast Model
- Global AI-enabled Wound Analysis Market Share by Product Type, 2025
- Global AI-enabled Wound Analysis Market Share by Application, 2025
- Global AI-enabled Wound Analysis Market Share by End User, 2025
- Global AI-enabled Wound Analysis Market Share by Region, 2025
- North America AI-enabled Wound Analysis Market Structure
- Latin America AI-enabled Wound Analysis Market Structure
- Western Europe AI-enabled Wound Analysis Market Structure
- Eastern Europe AI-enabled Wound Analysis Market Structure
- East Asia AI-enabled Wound Analysis Market Structure
- South Asia & Pacific AI-enabled Wound Analysis Market Structure
- Middle East & Africa AI-enabled Wound Analysis Market Structure
- AI-enabled Wound Analysis Market Competitive Landscape Mapping
- AI-enabled Wound Analysis Market Strategic Positioning Matrix
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

