Antibody Conjugation Kits Market
Antibody Conjugation Kits Market Analysis Size, Share, and Forecast Outlook 2026 to 2036
Antibody Conjugation Kits Market Forecast and Outlook 2026 to 2036
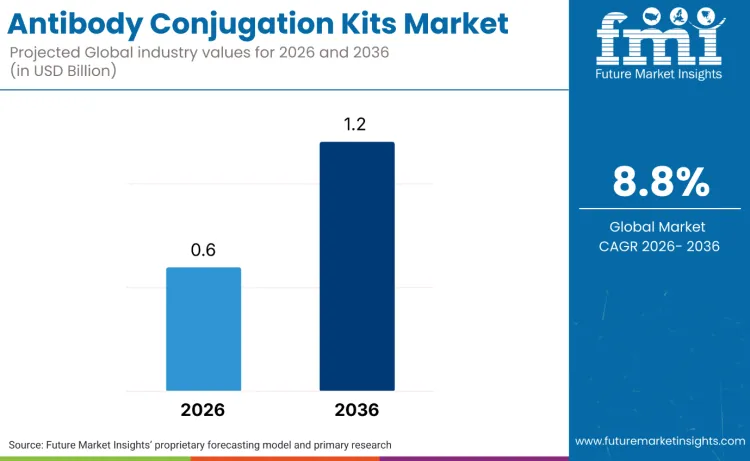
The global antibody conjugation kits market is forecasted to reach USD 0.6 billion in 2026 and expand to USD 1.2 billion by 2036, which shows a CAGR of 8.8%. As per FMI opinion, the antibody conjugation kits market is witnessing a significant transition from conventional drug discovery to more precise, targeted therapies. The shift is fueled by the increasing adoption of antibody-drug conjugates (ADCs) in oncology and immunotherapy, where precision targeting of cancer cells is becoming a cornerstone of treatment. This evolution is supported by advances in conjugation technologies that enhance efficacy while minimizing off-target effects. The focus is now on optimizing conjugation methods, improving payload stability, and ensuring better biocompatibility for broader therapeutic applications.
“With Seagen’s proprietary, world-leading Antibody-Drug Conjugate (ADC) technology, together with the scale and strength of Pfizer's capabilities and expertise, we are poised to change the cancer treatment paradigm. We believe oncology will be a significant growth driver for Pfizer and contribute meaningfully to the achievement of our near- and long-term financial goals.” - Dr. Albert Bourla, Pfizer Chairman and Chief Executive Officer
Following the acquisition of Seagen by Pfizer in 2023, the above statement was released. Companies such as AbbVie have also a strategic focus on enhancing ADC technology. In 2025, AbbVie expanded its ADC pipeline to include several promising oncology therapeutics. By strengthening collaborations with contract development and manufacturing organizations (CDMOs), AbbVie is positioning itself to lead in the ADC space, ensuring the seamless progression from clinical trials to commercial-scale manufacturing.
The market’s growth is owing to the need for high-throughput, scalable conjugation processes and the development of kits that offer high-quality, reproducible results. The growing focus on personalized medicine and biologics is pushing for antibody conjugation kits that enable precision drug formulation and clinical efficiency. In October 2024, Merck & Co., Inc, a leading science and technology company invested USD 82 million for the expansion of ADC manufacturing for novel cancer therapies. Major players in the field, such as GenScript, Merck, and Thermo Fisher, are prioritizing innovations that streamline conjugation workflows, enhance product quality, and integrate with broader biologics manufacturing processes.
Summary of the Antibody Conjugation Kits Market
What Is the Growth Outlook on the Antibody Conjugation Kits Market as per Future Market Insights Projection?
Future Market Insights projects the antibody conjugation kits market to grow at a CAGR of 8.8% from 2026 to 2036, expanding from USD 0.6 billion in 2026 to USD 1.2 billion by 2036. This outlook indicates that antibody conjugation is becoming an embedded step within biologics development workflows rather than an optional laboratory activity. Demand is increasingly centered on solutions that deliver controlled molecular outcomes, repeatable performance, and alignment with regulatory and documentation expectations across research and targeted therapy programs.
Evidence:
- FMI bottom-up revenue estimation mapped across conjugation formats, application areas, and end-user categories
- Continued expansion of antibody-drug conjugate pipelines, increasing reliance on consistent drug-to-antibody ratios and verifiable conjugation performance
- Rising preference for benchtop conjugation platforms capable of mirroring industrial precision during discovery and preclinical stages
How Do FMI Analysts Perceive the Antibody Conjugation Kits Market to Evolve?
FMI analysts view the market moving away from random chemical modification toward engineered conjugation systems that allow controlled attachment and consistent molecular placement. Antibody conjugation is being repositioned as a validation-ready workflow supporting performance accountability across discovery, preclinical evaluation, and targeted therapy development. Competitive advantage is increasingly defined by reliability of outcomes, readiness for automated environments, and minimized experimental variability across laboratories.
Evidence:
- Gradual substitution of lysine-targeted chemistries with enzymatic and bioorthogonal click-based approaches
- Research teams prioritizing uniform conjugates that meet rising reproducibility and documentation expectations
- Growing integration of conjugation chemistries with automated screening and biologics development platforms
Which Segments Lead Demand in the Antibody Conjugation Kits Market?
NHS-ester dye labeling kits lead by product type with a 26.0% share, while biopharma R&D represents the largest end-user segment with a 34.5% share. Leadership reflects where speed, scalability, and operational familiarity remain most monetizable, particularly in high-throughput flow cytometry and standardized assay development environments.
Evidence:
- FMI segment-level revenue modeling calibrated to research throughput and workflow intensity
- Continued reliance on rapid, robust amide-bond chemistry for routine analytical applications
- Strong uptake in biopharma R&D environments prioritizing internal reagent customization
Which Regions Are Positioned as Key Growth Engines for the Market?
China, India, Brazil, the United States, and Germany emerge as primary growth contributors, supported by ongoing expansion of biologics research capabilities, stronger capital allocation toward precision medicine, and wider deployment of advanced analytical systems. Market momentum in these countries is shaped by upgrades in research capability and workflow sophistication rather than higher reagent throughput alone.
Evidence:
- FMI country-level demand modeling reflecting research infrastructure build-out and analytical platform penetration
- Public and private programs accelerating biologics research and laboratory modernization
- Increasing reliance on precision conjugation tools within targeted therapy development pipelines
What Is the Definition of the Antibody Conjugation Kits Market?
The antibody conjugation kits market covers analytical labeling systems developed to support controlled antibody modification within research and laboratory settings. These platforms enable assessment of conjugation efficiency, precise attachment positioning, and functional performance across applications such as flow cytometry, immunoassays, imaging workflows, and preclinical antibody-drug conjugate studies.
Evidence:
- FMI market classification based on conjugation approach and intended analytical use
- Explicit separation between laboratory-grade conjugation systems and finished therapeutic products
- Consistency with established research workflows and documented analytical applications
What Are Globally Unique Trends Shaping the Antibody Conjugation Kits Market?
Globally distinctive trends shaping the market include faster uptake of site-controlled conjugation approaches, broader deployment across complex multi-modal constructs, and rising demand for workflows compatible with automated research environments. Market direction is further influenced by the use of AI-enabled protein design and predictive conjugation tools, which shorten experimental cycles and support distributed research execution.
Evidence:
- Increasing adoption of enzymatic and bioorthogonal chemistries for controlled conjugation
- Broadening application scope beyond fluorescent tags into ADCs, radiopharmaceuticals, and oligonucleotide-linked constructs
- Growing preference for conjugation kits designed to integrate with high-throughput and automated discovery platforms
Antibody Conjugation Kits Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 0.6 billion |
| Market Forecast Value (2036) | USD 1.2 billion |
| Forecast CAGR (2026 to 2036) | 8.8% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research.
How Does Precision Oncology Policy Drive Demand for Antibody Conjugation Kits?
The acceleration of precision oncology frameworks is significantly reshaping antibody conjugation workflows. Regulatory focus on Quality-by-Design (QbD), traceable drug-to-antibody ratios, and reproducible molecular positioning is reducing tolerance for stochastic conjugation methods, shifting demand toward engineered, validation-ready conjugation kits embedded within biologics development pipelines. For instance, Thermo Fisher Scientific Inc. offers site-specific antibody labeling kits (such as SiteClick Duet and Quartet) that provide reproducible, position-controlled conjugation chemistry away from antigen binding sites, enabling precise modification outcomes and consistent degree of labeling across experiments. This policy and regulatory alignment places antibody conjugation at the center of compliant ADC discovery and preclinical evaluation rather than as a discretionary laboratory step.
How is the Antibody Conjugation Kits Market Segmented?
The antibody conjugation kits market has been segmented based on product type, application, end user, and region. In terms of product type, the market is divided into NHS-ester dye labeling kits, click chemistry labeling kits, site-specific conjugation kits, biotinylation/streptavidin kits, enzyme (HRP/AP) conjugation kits, and nanoparticle (gold/latex) conjugation kits. By application, the market is divided into flow cytometry reagents, immunofluorescence/imaging, ELISA and plate-based immunoassays, lateral flow/rapid tests, and ADC research (preclinical tools). By end user, the market is segmented into biopharma R&D, academic and core facilities, diagnostic developers (IVD), CROs/assay developers, and OEM reagent manufacturers. Regionally, the market is divided into North America, Europe, Asia Pacific, Latin America, and Middle East & Africa.
Why Do NHS-ester Dye Labeling Kits Remain Dominant?

NHS-ester dye labeling kits continue to lead the market, accounting for 26.0% of the share, driven by their widespread use in flow cytometry applications, automated workflows, and standardized research applications. This product type offers the efficiency needed for large-scale antibody labeling experiments and high-volume sample processing, making it ideal for academic, pharmaceutical, and industrial laboratories. Revvity markets fluorescent labeling products such as IVISense™ 680 fluorescent dye kits in NHS-ester formats for protein and antibody conjugation.
From a process efficiency standpoint, NHS-ester reactions are preferred for their speed and stability. The conjugation completes within minutes to a few hours under physiological or slightly alkaline conditions, resulting in a robust and irreversible amide bond. This chemical stability is critical for downstream applications such as flow cytometry, immunofluorescence, and high-resolution imaging, where the integrity of the dye-antibody bond directly impacts signal accuracy and background noise.
How Do Application Requirements Influence Antibody Conjugation Kits Selection?
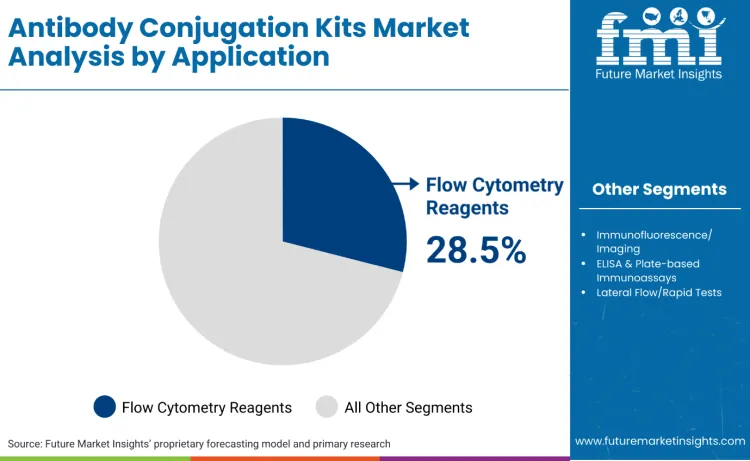
Flow cytometry reagents stands as the dominant segment, commanding 28.5% of the market share. The widespread adoption of antibody conjugation kits for flow cytometry reagents is driven by the urgent need for laboratory agility and the increasing complexity of multi-parametric analysis. Thermo Fisher’s DyLight™ and Pierce™ NHS-ester antibody labeling kits are widely used to create custom fluorescent antibodies for flow cytometry. In the current research environment, the ability to customize an optical panel is a strategic necessity. Commercial pre-conjugated antibodies often fail to meet the specific requirements of niche targets or unique host species. Conjugation kits empower researchers to bypass these supply chain constraints, allowing for the rapid creation of bespoke reagents that align perfectly with the laser configurations of their specific flow cytometers.
Efficiency and signal optimization serve as the primary technical catalysts. Modern flow cytometry often requires the simultaneous detection of twenty or more parameters, which necessitates precise control over the fluorophore-to-protein ratio. From a cost-management perspective, these kits offer a superior return on investment compared to the procurement of multiple pre-labeled antibodies. They allow for the bulk purchase of unconjugated primary antibodies, which can then be partitioned and labeled with various dyes as needed. This flexibility reduces waste and inventory overhead while accelerating the transition from experimental design to data acquisition. Ultimately, the democratization of labeling technology via these kits has transformed flow cytometry from a rigid platform into a highly iterative and scalable analytical tool.
How Are Engineered Conjugation Platforms Replacing One-Off Chemistry?
The antibody conjugation kits market is moving decisively away from stochastic chemical labeling toward engineered, validation-ready platforms embedded within biologics development workflows. As antibody-drug conjugates advance from experimental programs into regulated pipelines, tolerance for variability in drug-to-antibody ratios and molecular positioning is rapidly declining. Kit suppliers that can deliver site-controlled attachment, reproducible outcomes, and documentation-aligned performance are increasingly preferred by biopharma R&D teams. This shift is turning conjugation from a discretionary bench step into a standardized workflow layer supporting traceability across discovery, preclinical evaluation, and translational research.
Why Is Precision Oncology Policy Reshaping Kit Design and Adoption?
Regulatory emphasis on Quality-by-Design, molecular traceability, and reproducibility is accelerating demand for site-specific and bioorthogonal conjugation chemistries. Oncology-focused pipelines now require conjugation systems that minimize heterogeneity and align with downstream analytical and regulatory expectations. Following Pfizer’s acquisition of Seagen in 2023, leadership statements underscored the strategic role of controlled ADC technologies in reshaping cancer treatment models. Similar investments by AbbVie and Merck into ADC manufacturing capacity signal that conjugation precision is becoming a gating factor for clinical scalability rather than a secondary optimization step.
How Is Automation Readiness Redefining Competitive Advantage?
Laboratories are increasingly prioritizing conjugation kits that integrate seamlessly with automated screening, high-throughput analytics, and benchtop workflows that mirror industrial precision. NHS-ester systems remain dominant where speed and familiarity matter, particularly in flow cytometry, while enzymatic and click-based platforms are gaining share in environments demanding uniformity and validation readiness. Market leaders are differentiating through workflow compatibility, ambient stability, and reduced experimental iteration rather than reagent novelty alone.
What Structural Shift Will Define Long-Term Market Leadership?
From 2026 to 2036, competitive advantage will favor suppliers that treat antibody conjugation as a scalable platform rather than a consumable chemistry product. Players capable of maintaining consistent performance across the USA, Germany, China, India, and Japan while supporting multi-modal constructs such as ADCs, radiopharmaceuticals, and oligonucleotide conjugates are positioned to capture durable value. Antibody conjugation is transitioning into a performance-critical delivery framework that anchors accountability, reproducibility, and regulatory alignment across modern biologics development.
How Is the Antibody Conjugation Kits Market Evolving Globally?
Global demand for antibody conjugation kit products is increasing as researchers expand analytical capabilities while addressing precision conjugation, kinetic characterization, and mechanism validation application requirements. Growth demonstrates rising use of automated analytical platforms, target-specific conjugation positioning, and efficiency-optimized detection specifications across pharmaceutical institutions, specialized research facilities, and precision medicine development. Product selection focuses on conjugation efficiency, analytical throughput, and platform performance under various research scenarios. China records 15.3% CAGR, India records 13.7% CAGR, Brazil records 12.1% CAGR, USA records 9.1% CAGR, and Germany records 5.7% CAGR. Adoption remains driven by precision analytical requirements and conjugation capabilities rather than volume expansion alone.
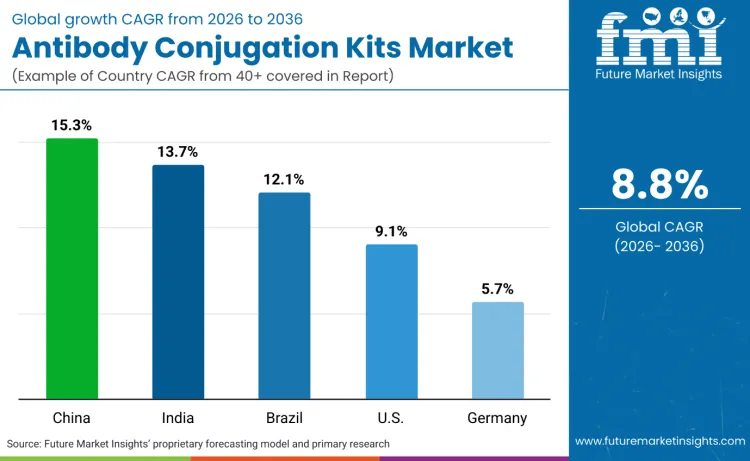
| Country | CAGR (2026-2036) |
|---|---|
| China | 15.3% |
| India | 13.7% |
| Brazil | 12.1% |
| USA | 9.1% |
| Germany | 5.7% |
China: How is the Rising Pharmaceutical Development Infrastructure Driving Advanced Conjugation Adoption?
The primary driver in China is the significant growth of domestic pharmaceutical development capabilities and precision medicine research institutions. With the 2025 expansion of biotechnology research facilities, Chinese metropolitan areas are now experiencing millions of new analytical platform implementations, making traditional conjugation methods insufficient. A key development is the "Precision Medicine Initiative", which has promoted advanced analytical platform adoption for domestic pharmaceutical development to ensure research capabilities and conjugation innovation. Driven by this national scale of analytical capability expansion, the Chinese antibody conjugation kits market is set to grow at a 15.3% CAGR during the study period, led by massive demand in pharmaceutical development and research sectors.
As per FMI insights, the next five years are set to witness the integration of AI-powered discovery informatics with high-throughput conjugation hardware. As China targets "manufacturing sovereignty" in biologics, there is a growing demand for specialized conjugation kits that support complex modalities like antibody-drug conjugates and targeted therapy characterization. The push for decentralized healthcare will lead to a shift toward thermostable, point-of-care conjugated diagnostics, bypassing traditional cold-chain constraints in lower-tier cities.
India: Is the "Research Excellence" Movement Elevating Conjugation Standards?
In India, the market is dominated by the evolution toward "Research Excellence" and precision-positioned analytical categories. A major driver is the recent 2025 Research Development and Innovation Scheme, which allows Indian research institutions to position advanced analytical platforms alongside international research capabilities from a single quality framework. This "precision-first" approach is essential for Indian researchers currently competing with international research standards in domestic pharmaceutical development applications. Reflecting this maturity, the Indian antibody conjugation kits market is projected to grow at a 13.7% CAGR during the study period.
The rise of biosimilars and biologics manufacturing, supported by expanded infrastructure, will necessitate high-precision conjugation kits for real-time process monitoring and quality control over the next coming years. Second, India's growing role as a global hub for drug discovery outsourcing creates a massive opening for high-throughput conjugation services tailored to lead optimization. Finally, there is a burgeoning demand for decentralized diagnostic kits that use stabilized conjugated antibodies, addressing the healthcare needs of tier-2 and tier-3 cities while bypassing cold-chain logistics.
Brazil: Will Expanding Biotechnology Sector Accelerate Platform Integration?
Brazil's antibody conjugation kits market is uniquely shaped by biotechnology sector expansion and research infrastructure development. The Brazilian antibody conjugation kits market is expected to grow at a 12.1% CAGR during the study period, as research-focused organizations invest in validated analytical and precision conjugation categories.
Over the next five years, the market will shift toward high-growth niches like bioprospecting informatics and decentralized diagnostic kits. As domestic pharmaceutical leaders transition from generic drug production to complex biologics and biosimilars, there is an immediate opportunity to provide high-precision validation assays for protein characterization. Furthermore, the integration of AI-driven screening into Brazil's expansive natural genetic library will likely redefine discovery timelines.
Germany: Is the "Analytical Innovation" Strategy Revitalizing Research Capabilities?
In Germany, the market is being revitalized by the "Analytical Innovation" initiative, which integrates advanced conjugation methods and automated workflow integration into research platform positioning. A major development is research institutions and pharmaceutical companies upgrading to precision-positioned analytical platforms that feature "innovation-heritage excellence" to compete against standard analytical methods in domestic research applications. As German institutions prioritize analytical innovation and research excellence, the German antibody conjugation kits market is set to grow at a 5.7% CAGR during the study period, focusing heavily on precision and analytical capability enhancement.
The market is set to witness the integration of digital health and companion diagnostics over the next five years. As the German government streamlines regulatory barriers for in vitro diagnostics (IVDs) through 2026, there is a significant opening for firms to develop AI-integrated conjugation assays that support personalized medicine and rare disease therapies. Additionally, the mandate for "Green Labs" is creating a surge in demand for environmentally friendly, solvent-free conjugation processes, offering a high-margin niche for providers of automated, eco-friendly conjugation platforms.
How is the USA Market Maturing Around Reimbursement?
The USA antibody conjugation kits market is set to grow at a 9.1% CAGR during the study period, shaped by a reimbursement environment that increasingly rewards standardized, kit-based laboratory workflows over manual, high-labor conjugation techniques. The Centers for Medicare & Medicaid Services 2026 Physician Fee Schedule (CMS-1832-F) introduced an efficiency adjustment that pressures laboratories to protect margins under tighter conversion factors. As a result, labs are moving toward pre-validated conjugation kits that reduce technician time, minimize batch variability, and support reproducible assay preparation aligned with payer expectations for operational efficiency.
Regulatory stabilization further accelerates this transition. Following a brief legal interruption, the USA Food and Drug Administration September 2025 Final Rule (89 FR 37286) re-established the pathway for oversight of laboratory developed test components. This action compels kit manufacturers to supply clinical-grade validation data, allowing laboratories to meet the May 6, 2026 Stage 2 deadline for registration and listing of LDT activities.
Who are the Major Players Active in the Antibody Conjugation Kits Market?
The competitive landscape for the antibody conjugation kits market is defined by a strategic transition toward AI-guided conjugation optimization and the integration of ambient-stable reagents to reduce cold-chain dependency. The market leaders are aggressively expanding their vertical footprints to defend against specialized disruptors. Thermo Fisher Scientific reported revenue bolstered by the integration of Olink to fuse proteomics data with their dominant antibody labeling portfolio. Simultaneously, Merck KGaA announced an expansion of its ADC manufacturing capabilities in St. Louis, focusing on high‑potency API production and continuous flow manufacturing. This strategy moves the value proposition from simple benchtop kits to scalable, clinical-grade conjugation platforms. Danaher Corporation, through its Cytiva and Abcam brands, is leveraging its broad market penetration in monoclonal antibody production to standardize automation-ready conjugation protocols across the drug development lifecycle.
Future Market Insights is of the opinion that antibody conjugation kit suppliers that treat conjugation as a standardized, validation-ready platform rather than a one-off chemistry step will hold a durable advantage over the forecast period. Players that can deliver repeatable molecular positioning, controlled drug-to-antibody ratios, and documentation-aligned performance across discovery, preclinical, and translational workflows, while maintaining consistency across the USA, Germany, China, India, and Japan, are best positioned to lead long-term value creation.
Key Industry Developments:
- In November 2025, Revvity, Inc. announced to acquire ACD/Labs for the expansion of its manufacturing portfolio. This deal is set to help Revvity Signals’ solutions in scientific innovations.
- In August 2024, Danaher finalized the landmark USD 5.1 billion acquisition of Abcam (completed mid-2024), integrating one of the world's largest libraries of validated antibodies and conjugation expertise into its Life Sciences segment.
Key Players in the Antibody Conjugation Kits Market
- Thermo Fisher Scientific Inc.
- Revvity, Inc.
- Danaher Corporation
- Merck KGaA, Darmstadt, Germany
- Maravai LifeSciences Holdings, Inc.
- Bio-Rad Laboratories, Inc.
- PerkinElmer, Inc.
- Cell Signaling Technology, Inc.
- BioVision, Inc.
- Promega Corporation
Market Definition
The antibody conjugation kits market refers to the global research and analytical applications of biochemical labeling platforms designed to measure conjugation efficiency, antibody modification, and functional characteristics. It includes both precision and standard conjugation offerings, with segments that cater to various research applications such as flow cytometry, immunoassays, and biomarker characterization. The market size is measured in USD billion and analyzed over the 2026 to 2036 period.
The antibody conjugation kits market includes product types such as NHS-ester labeling kits, site-specific conjugation kits, biotinylation systems, and specialized conjugation platforms, utilized through both research institutions and pharmaceutical development applications. The market is expanding due to growing interest in precision medicine development, analytical platform innovations, and evolving research requirements toward higher-precision and more specialized antibody modification capabilities.
Market Inclusion
Included in the antibody conjugation kits market scope are analytical products categorized by type (NHS-ester, site-specific, biotinylation, and specialized conjugation), with segmentation based on applications (flow cytometry, immunofluorescence, immunoassays), research focus (pharmaceutical, academic, diagnostic), and analytical requirements (precision, throughput, automation compatibility). The scope includes conjugation platforms with a focus on analytical precision, research capabilities, and conjugation innovation, particularly those marketed through specialized suppliers, research institutions, and pharmaceutical development facilities.
Geographically, the market encompasses key regions such as North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa, with country-level analysis for significant markets like China, India, Brazil, USA, and Germany.
Market Exclusion
Excluded from the antibody conjugation kits market scope are non-conjugation detection platforms and analytical methods not designed for antibody modification, such as protein quantification assays, cell viability measurements, and nucleic acid detection platforms. Additionally, products not intended for research or pharmaceutical applications are not included.
The market excludes non-analytical conjugation products, as well as conjugation-related products that are not marketed as analytical platforms, such as therapeutic antibody conjugates or antibody-based therapeutic products. Furthermore, products with non-compliant analytical specifications or those that do not meet regional regulatory standards for research applications are outside the market's defined scope.
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Product Type | NHS-ester Dye Labeling Kits; Click Chemistry Labeling Kits; Site-specific Conjugation Kits; Biotinylation/Streptavidin Kits; Enzyme (HRP/AP) Conjugation Kits; Nanoparticle (Gold/Latex) Conjugation Kits |
| Application | Flow Cytometry Reagents; Immunofluorescence/Imaging; ELISA & Plate-based Immunoassays; Lateral Flow/Rapid Tests; ADC Research (Preclinical Tools) |
| End User | Biopharma R&D; Academic & Core Facilities; Diagnostic Developers (IVD); CROs/Assay Developers; OEM Reagent Manufacturers |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Countries Covered | China, India, Brazil, USA, Germany, and 40+ countries |
| Key Companies Profiled | Thermo Fisher Scientific; Revvity; Danaher; Merck KGaA; Maravai LifeSciences |
| Additional Attributes | Dollar sales by product type, application, and end user; performance in clinical applications; product quality improvement, product complexity enhancement; impact on brand positioning, order fulfilment, and market differentiation during formulation processes; procurement dynamics driven by premium positioning, product innovation programs, and long-term brand partnerships. |
Source: FMI historical analysis and forecast data.
Antibody Conjugation Kits Market by Segment
Product Type:
- NHS-ester Dye Labeling Kits
- Click Chemistry Labeling Kits
- Site-specific Conjugation Kits
- Biotinylation/Streptavidin Kits
- Enzyme (HRP/AP) Conjugation Kits
- Nanoparticle (Gold/Latex) Conjugation Kits
Application:
- Flow Cytometry Reagents
- Immunofluorescence/Imaging
- ELISA & Plate-based Immunoassays
- Lateral Flow/Rapid Tests
- ADC Research (Preclinical Tools)
End User:
- Biopharma R&D
- Academic & Core Facilities
- Diagnostic Developers (IVD)
- CROs/Assay Developers
- OEM Reagent Manufacturers
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- Thailand
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- Netherlands
- Rest of Europe
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Colombia
- Argentina
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- International Bioconjugation Research Association. (2023). Conjugation Standards for Antibody Labeling Kits: Analytical Guidelines. IBRA.
- Global Biochemical Research Council. (2023). Platform Specifications for Conjugation Products: Precision and Validation Requirements. GBRC.
- Pharmaceutical Research Institute. (2024). PRI Guidelines for Analytical Positioning in Antibody Conjugation (updated precision specifications for research and pharmaceutical applications). PRI.
- Martinez, D. L., et al. (2023). Precision positioning effects of analytical conjugation platforms on research capability development: A comprehensive analysis of conjugation efficiency and research analytical applications. Journal of Analytical Research Methods, 28, 145-162.
- Thompson, R. K., et al. (2023). Sixth annual antibody conjugation kits market report: Special focus on analytical innovation and precision positioning in global research applications. International Analytical Industry Review, 31, 89-106.
Frequently Asked Questions
How big is the antibody conjugation kits market in 2026?
The global antibody conjugation kits market is estimated to be valued at USD 0.6 billion in 2026.
What will be the size of the antibody conjugation kits market in 2036?
The market size for the antibody conjugation kits market is projected to reach USD 1.2 billion by 2036.
How much will the antibody conjugation kits market grow between 2026 and 2036?
The antibody conjugation kits market is expected to grow at a 8.8% CAGR between 2026 and 2036.
What are the key product types in the antibody conjugation kits market?
The key product types in the antibody conjugation kits market include NHS-ester dye labeling kits, site-specific conjugation kits, biotinylation/streptavidin kits, and click chemistry labeling kits.
Which end user will contribute a significant share in the antibody conjugation kits market in 2026?
In terms of end user, the biopharma R&D segment is set to command a 34.5% share in the antibody conjugation kits market in 2026.
Table of Content
- Executive Summary
- Global Market Overview
- Key Insights
- Market Forecast
- Strategic Recommendations
- Market Overview
- Market Definition
- Market Taxonomy
- Scope and Limitations
- Research Methodology
- Research Approach
- Data Sources
- Analytical Framework
- Market Dynamics
- Drivers
- Restraints
- Opportunities
- Trends
- Market Sizing and Forecast
- Market Value (2026 to 2036)
- Y-o-Y Growth Analysis
- Market Share by Product Type
- Market Share by Application
- Market Share by End User
- Market Segmentation Analysis
- Product Type
- NHS-ester Dye Labeling Kits
- Click Chemistry Labeling Kits
- Site-specific Conjugation Kits
- Biotinylation/Streptavidin Kits
- Enzyme (HRP/AP) Conjugation Kits
- Nanoparticle (Gold/Latex) Conjugation Kits
- Application
- Flow Cytometry Reagents
- Immunofluorescence/Imaging
- ELISA & Plate-based Immunoassays
- Lateral Flow/Rapid Tests
- ADC Research (Preclinical Tools)
- End User
- Biopharma R&D
- Academic & Core Facilities
- Diagnostic Developers (IVD)
- CROs/Assay Developers
- OEM Reagent Manufacturers
- Product Type
- Regional Market Analysis
- Asia Pacific
- North America
- Europe
- Latin America
- Middle East & Africa
- Competitive Landscape
- Market Share of Key Players
- Competitive Benchmarking
- Key Player Strategies
- Key Takeaways
- Market Summary
- Future Trends and Forecast
- Strategic Recommendations
List of Tables
- Global Antibody Conjugation Kits Market Size (USD Billion), 2021-2036
- Antibody Conjugation Kits Market Y-o-Y Growth (%), 2021-2036
- Global Market Share by Product Type, 2026
- Global Market Share by Application, 2026
- Global Market Share by End User, 2026
- Antibody Conjugation Kits Market Share by Region, 2026
- Market Share by Product Type (USD Billion), 2026-2036
- Market Share by End User (USD Billion), 2026-2036
- Key Player Market Share in the Antibody Conjugation Kits Market
- Antibody Conjugation Kits Market Absolute $ Opportunity (USD Billion), 2026-2036
- Antibody Conjugation Kits Market CAGR (%) by Region, 2026-2036
List of Figures
- Global Antibody Conjugation Kits Market Size (USD Billion), 2021-2036
- Antibody Conjugation Kits Market Y-o-Y Growth (%) from 2021-2036
- Antibody Conjugation Kits Market Share by Product Type (%) in 2026
- Antibody Conjugation Kits Market Share by Application (%) in 2026
- Global Antibody Conjugation Kits Market by Region (%) 2026-2036
- Global Antibody Conjugation Kits Market by End User
- Competitive Landscape of Key Antibody Conjugation Kits Market Players
- Market Trends in Antibody Conjugation Technologies
- Antibody Conjugation Kits Adoption by Region
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

