The antibody quantification market is projected to grow from USD 7.8 billion in 2026 to USD 17.0 billion by 2036, registering a CAGR of 8.10%. Capital allocation across the forecast period is anchored in platform longevity and revenue recurrence rather than short-term shipment growth. Investors favor technologies embedded deeply within biologics development and manufacturing workflows, where assay continuity is essential. High regulatory scrutiny increases the value of validated systems, reinforcing selective investment in suppliers with established performance records.
Return generation is supported by steady consumables demand, method revalidation cycles, and long-term service agreements. Capital is deployed cautiously toward automation, multiplexing capability, and data integrity enhancements that improve laboratory efficiency without disrupting validated processes. Expansion strategies prioritize customer retention and lifecycle value over aggressive geographic scaling. As a result, market growth reflects disciplined capital deployment aligned with predictable usage patterns and defensible margins rather than rapid volume-led expansion.
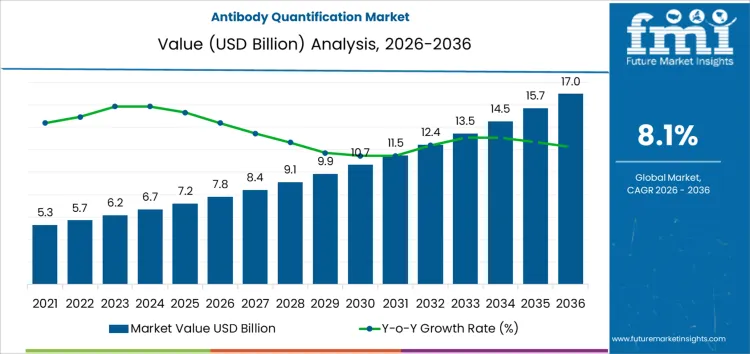
| Metric | Value |
|---|---|
| Antibody Quantification Market Value (2026) | USD 7.8 Billion |
| Antibody Quantification Market Forecast Value (2036) | USD 17.0 Billion |
| Antibody Quantification Market Forecast CAGR 2026 to 2036 | 8.10% |
Growth of the antibody quantification market is driven by expanding biopharmaceutical research and rising production of monoclonal antibodies for therapeutic and diagnostic applications. Antibody quantification is a critical step in drug discovery, process development, and quality control, ensuring accurate measurement of antibody concentration, purity, and potency. Increasing investment in biologics, biosimilars, and antibody-based therapies is generating sustained demand for reliable quantification assays and analytical platforms across research laboratories and manufacturing facilities.
Advancements in analytical technologies are further strengthening market growth. Improved immunoassays, spectrophotometric techniques, chromatography-based methods, and automated platforms are enabling higher accuracy, reproducibility, and throughput. Automation and integration with laboratory information systems are improving workflow efficiency and reducing variability in large-scale testing environments. These capabilities are particularly important for contract research organizations and biomanufacturers managing high sample volumes under strict regulatory requirements.
Regulatory emphasis on data accuracy, validation, and quality assurance is also contributing to market expansion. Stringent guidelines governing biologics development and manufacturing require validated analytical methods throughout the product lifecycle. Growing adoption of personalized medicine, vaccine development, and infectious disease research is increasing the scope of antibody testing applications. Together, biologics pipeline expansion, technology improvements, and regulatory compliance requirements are supporting robust growth of the antibody quantification market over the forecast period.
The antibody quantification market is segmented by product type and assay type, with clear dominance in specific categories. By product type, primary antibodies represent the leading segment, supported by their direct role in target detection and quantification workflows. Other product types in the antibody quantification market include secondary antibodies and conjugated antibodies, which support signal amplification and assay versatility. By assay type, ELISA and immunoassays hold the dominant position, reflecting widespread use across research, diagnostics, and biopharmaceutical quality control. Additional assay types such as western blotting, flow cytometry, protein microarrays, mass spectrometry-based assays, and other quantification methods contribute to diversified analytical demand.
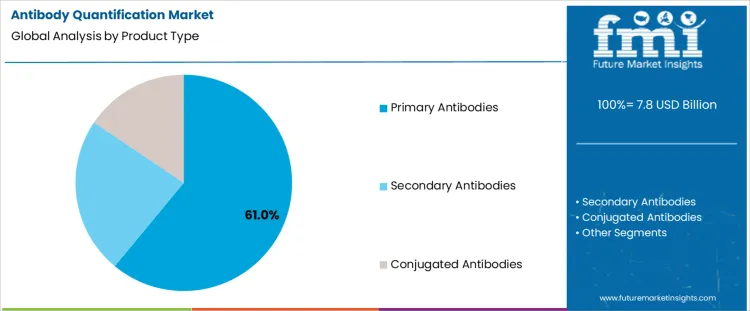
Primary antibodies lead the antibody quantification market by product type with 61% share because they bind directly to specific antigens, enabling accurate measurement of protein concentration and expression levels. Research laboratories and biopharmaceutical companies rely on primary antibodies for target specificity in immunoassays and analytical workflows. Their use is critical in drug discovery, biomarker validation, and therapeutic antibody development. Consistent performance across multiple assay platforms supports repeatability and data reliability. Growth in proteomics research and cell signaling studies increases demand for high-quality primary antibodies. These functional and application-driven factors explain why primary antibodies remain the most widely used product type in antibody quantification activities.
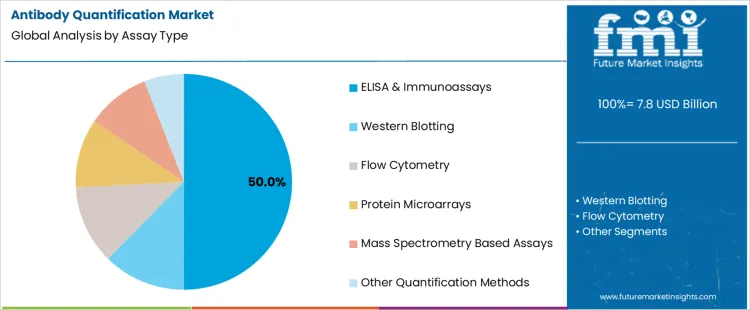
ELISA and immunoassays dominate the antibody quantification market by assay type with 50% share because they provide high sensitivity, scalability, and reproducibility for protein measurement. These assays are widely applied in research laboratories, clinical diagnostics, and biopharmaceutical manufacturing environments. Standardized protocols enable consistent antibody quantification across large sample volumes. Compatibility with automation supports high-throughput screening and quality control testing. ELISA-based formats allow quantitative analysis with minimal instrumentation complexity. Expansion of immunology research, vaccine development, and biologics production increases reliance on these assays. These advantages explain why ELISA and immunoassays remain the preferred quantification approach.
The antibody quantification market is driven by increasing demand for accurate and high-throughput measurement of antibody concentrations in research, clinical diagnostics, and biologics manufacturing. Growth in therapeutic monoclonal antibodies, biosimilars, and immuno-based vaccines is expanding the need for precise quantification tools. Regulatory requirements for quality control and batch release testing are reinforcing adoption of standardized assays. Platforms such as ELISA, flow cytometry, biosensors, and label-free technologies are widely used for sensitivity and specificity. Investment in automation and data analytics is enhancing throughput and reproducibility. Cost of assays, throughput needs, and integration with upstream/downstream workflows remain key factors influencing market choices.
Challenges in the antibody quantification market include high costs of advanced quantification platforms and consumables, which can limit adoption among smaller laboratories and academic institutions. Complex assay protocols and need for technical expertise increase hands-on time and training requirements. Variability between platforms such as ELISA vs. label-free biosensors can complicate standardization and cross-study comparison. Interference from sample matrices (serum, plasma) and low-abundance antibodies can affect assay sensitivity. Regulatory scrutiny and validation demands for clinical and biopharmaceutical applications require rigorous documentation and quality control, extending time to implementation. These constraints collectively temper market growth and technology uptake.
Innovation and technology trends are reshaping the antibody quantification market toward higher sensitivity, automation, and data integration. Next-generation platforms incorporating microfluidics and multiplex detection enable simultaneous quantification of multiple antibody isotypes with minimal sample volume. Label-free technologies, such as surface plasmon resonance and bio-layer interferometry, offer real-time binding measurements and kinetic profiles. Integration with automated liquid handling systems accelerates throughput and reduces human error. Cloud-based analytics and digital dashboards facilitate real-time data visualization and compliance tracking. Demand for point-of-care quantification solutions is driving development of portable, low-complexity devices adaptable to clinical and field environments.
Demand for antibody quantification solutions varies across countries depending on biopharmaceutical research intensity, biologics manufacturing scale, and clinical testing volumes. North America and Asia Pacific remain the primary growth regions due to strong pipelines for monoclonal antibodies, vaccines, and biosimilars. The United States leads growth with a CAGR of 8.9% during 2026 to 2036, supported by advanced research infrastructure and regulatory-driven quality control requirements.
China and India follow with CAGRs of 8.6% and 8.3%, respectively, driven by expanding domestic biologics manufacturing and contract research activity. Germany and the UK record stable growth at 7.8% and 7.5%, reflecting mature but innovation-driven laboratory environments. Country-level expansion is shaped by assay standardization, laboratory automation, and regulatory compliance requirements.
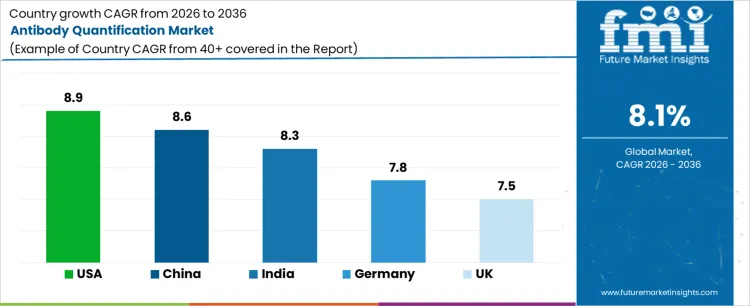
| Country | CAGR (%) |
|---|---|
| USA | 8.9% |
| China | 8.6% |
| India | 8.3% |
| Germany | 7.8% |
| UK | 7.5% |
Demand for antibody quantification solutions in the United States, expanding at a CAGR of 8.9% during 2026 to 2036, is driven by extensive biopharmaceutical research and large-scale biologics manufacturing activity. The antibody quantification market in the United States benefits from widespread use of immunoassays across monoclonal antibody discovery, process development, and batch release testing. Pharmaceutical companies rely on accurate antibody concentration measurement to meet regulatory documentation and quality assurance requirements.
High volumes of biosimilar development and late-stage clinical trials increase routine assay usage. Contract research organizations and contract development and manufacturing organizations contribute steady demand through outsourced analytical testing. Academic research institutions further support baseline consumption through immunology and oncology research programs. Replacement demand remains strong as laboratories upgrade legacy ELISA platforms to automated and higher-throughput systems. These combined factors support sustained, value-driven expansion across the United States.
Sales of antibody quantification systems in China, growing at a CAGR of 8.6% from 2026 to 2036, are supported by rapid expansion of domestic biopharmaceutical manufacturing and clinical research capacity. The antibody quantification market in China is driven by rising monoclonal antibody pipelines, vaccine development programs, and biosimilar commercialization. Pharmaceutical manufacturers are strengthening analytical capabilities to improve batch consistency and validation readiness. Growth of contract development and manufacturing organizations increases demand for standardized antibody assays. Government-backed biotechnology initiatives continue to expand laboratory infrastructure nationwide. Academic research institutes contribute incremental demand through infectious disease and immunology research. Replacement demand is emerging as laboratories shift toward automated and higher-sensitivity quantification platforms. Procurement behavior increasingly prioritizes scalability, cost efficiency, and compliance alignment. These dynamics position China as a high-growth and strategically important country.
Demand for antibody quantification technologies in India, expanding at a CAGR of 8.3% during 2026 to 2036, is driven by strong growth in biosimilar development, vaccine production, and clinical research services. The antibody quantification market in India benefits from rising investments in pharmaceutical research facilities and expanding contract research organizations. Laboratories require reliable antibody measurement methods to meet international regulatory expectations for export-oriented biologics. Vaccine research and diagnostic assay development generate recurring testing requirements. Academic institutions add incremental demand through publicly funded life sciences research. Cost-sensitive procurement favors versatile platforms capable of supporting multiple applications. Replacement demand is increasing as laboratories move from manual or semi-quantitative techniques to standardized immunoassay systems. Availability of skilled scientific talent and improving laboratory infrastructure further support adoption. These factors collectively sustain steady and broad-based growth across India.
Demand for antibody quantification solutions in Germany, growing at a CAGR of 7.8% over the forecast period, is sustained by a strong pharmaceutical manufacturing base and advanced biotechnology research ecosystem. The antibody quantification market in Germany is shaped by emphasis on analytical precision, process validation, and regulatory compliance. Pharmaceutical manufacturers prioritize reproducible and well-documented assay methods across biologics development workflows. Research institutes and industrial laboratories support consistent assay utilization. Replacement demand arises as laboratories upgrade platforms to meet evolving analytical standards. Procurement decisions focus on reliability, accuracy, and long-term operational stability. Limited expansion in laboratory numbers is offset by high-value analytical investments. Collaboration between academia and industry further strengthens demand. These structural characteristics enable Germany to maintain stable, technology-driven growth in antibody quantification adoption.
Sales of antibody quantification systems in the United Kingdom, expanding at a CAGR of 7.5% during 2026 to 2036, reflect sustained strength in academic research and biopharmaceutical innovation. The antibody quantification market in the UK benefits from active life sciences clusters and clinical research programs. Laboratories emphasize assay accuracy and reproducibility to support biologics development and diagnostics. Research funding stability supports consistent assay usage across universities and research institutes. Replacement of aging analytical equipment drives recurring procurement. Contract research activity and vaccine development further strengthen demand. Procurement decisions prioritize compliance readiness and ease of integration into existing workflows. These factors collectively support steady, research-led expansion across the United Kingdom.
Competition in the antibody quantification market centers on validated assay platforms, sensitivity claims, and range of supported formats as presented in official product literature. Thermo Fisher Scientific positions its quantification solutions with brochures detailing multimodal platforms including spectrophotometric, fluorometric, and immunoassay-based assays with broad dynamic ranges. Its materials emphasize compatibility with standard workflows and integration with laboratory informatics. Abcam Plc competes by offering technically validated antibody quantification kits with datasheets that specify detection limits, linear range, and recommended use cases across immunoassays and protein analytics. Bio-Rad Laboratories differentiates through high-sensitivity ELISA kits and multiplex panels, with official literature focusing on reproducibility, lot-to-lot consistency, and quality control parameters.
Strategic positioning is reinforced through platform breadth and cross-technology support. Merck KGaA markets antibody quantification reagents and instrument packages with brochures highlighting proprietary chemistries and performance benchmarks for colorimetric, chemiluminescent, and fluorescence-based assays. Cell Signaling Technology emphasizes its rigorously validated antibody quantification kits with documentation on specificity and recommended protocols. BD (Becton Dickinson) competes by integrating flow cytometry-compatible quantification reagents with system-level performance claims in its product guides. PerkinElmer Inc. positions its detection platforms with brochures that detail low-level quantification capability and software-assisted data analysis tools.
High-performance analytics define several competitors’ strategies. Agilent Technologies offers antibody quantification through advanced spectroscopic and microfluidics solutions, with official materials emphasizing precision, minimal sample requirements, and automation readiness. Roche Diagnostics competes with immunoassay systems backed by datasheets that outline sensitivity, throughput, and regulatory compliance for clinical and research settings. Santa Cruz Biotechnology and GeneTex, Inc. focus on validated ELISA and immunodetection kits, with brochures specifying antibody pairing recommendations, dynamic ranges, and application notes. Across all players, competition is rooted in brochure-verified performance metrics, detection sensitivity, workflow compatibility, and documented user support rather than commodity pricing.
| Attribute | Description |
|---|---|
| Quantitative Unit | USD Billion |
| Product Type | Primary Antibodies, Secondary Antibodies, Conjugated Antibodies |
| Assay Type | ELISA & Immunoassays, Western Blotting, Flow Cytometry, Protein Microarrays, Mass Spectrometry Based Assays, Other Quantification Methods |
| Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Rest of Asia Pacific, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, Rest of Europe, United States, Canada, Mexico, Brazil, Chile, Rest of Latin America, Kingdom of Saudi Arabia, Other GCC Countries, Turkey, South Africa, Other African Union, Rest of Middle East & Africa |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Key Companies Profiled | Thermo Fisher Scientific, Abcam Plc, Bio-Rad Laboratories, Merck KGaA, Cell Signaling Technology, BD (Becton Dickinson), PerkinElmer Inc., Agilent Technologies, Roche Diagnostics, Santa Cruz Biotechnology, GeneTex, Inc. |
| Additional Attributes | Dollar sales by product type and assay method are evaluated across research, clinical, and biopharmaceutical laboratories. The report includes country-level demand assessment, growth projections for 2026 to 2036, analysis of assay sensitivity and reproducibility trends, evaluation of automation and multiplexing adoption, competitive benchmarking of suppliers, and assessment of procurement patterns in life sciences research workflows. |
How big is the antibody quantification market in 2026?
The global antibody quantification market is estimated to be valued at USD 7.8 billion in 2026.
What will be the size of antibody quantification market in 2036?
The market size for the antibody quantification market is projected to reach USD 17.0 billion by 2036.
How much will be the antibody quantification market growth between 2026 and 2036?
The antibody quantification market is expected to grow at a 8.1% CAGR between 2026 and 2036.
What are the key product types in the antibody quantification market?
The key product types in antibody quantification market are primary antibodies, secondary antibodies and conjugated antibodies.
Which assay type segment to contribute significant share in the antibody quantification market in 2026?
In terms of assay type, elisa & immunoassays segment to command 50.0% share in the antibody quantification market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.