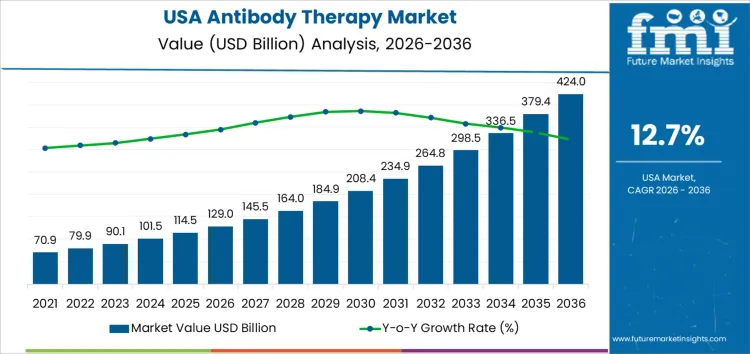
Demand for antibody therapy in USA is valued at USD 129.0 billion in 2026 and is projected to reach USD 424.0 billion by 2036, reflecting a CAGR of 12.7%. Demand accelerates due to rising incidence of oncology, autoimmune, and chronic inflammatory conditions, alongside expanding biologics pipelines and favorable reimbursement coverage. Continued clinical success and broader indication approvals reinforce sustained adoption across care settings.
Monoclonal antibodies lead therapy-type usage because high target specificity, strong efficacy profiles, and manageable safety outcomes support wide clinical acceptance. Advancements in antibody engineering, including bispecific formats and optimized Fc regions, improve therapeutic performance and dosing efficiency. Combination regimens and earlier-line use further expand treatment penetration.
West USA, South USA, Northeast USA, and Midwest USA represent key growth regions supported by dense hospital networks, academic research centers, and biopharmaceutical manufacturing hubs. Genentech (Roche), AbbVie, Bristol-Myers Squibb, Amgen, and Merck & Co. anchor competitive activity through robust pipelines, large-scale manufacturing capacity, and commercialization capabilities aligned with evolving clinical standards and precision medicine strategies across the United States.
| Metric | Value |
|---|---|
| USA Antibody Therapy Sales Value (2026) | USD 129.0 billion |
| USA Antibody Therapy Forecast Value (2036) | USD 424.0 billion |
| USA Antibody Therapy Forecast CAGR (2026 to 2036) | 12.7% |
Demand for antibody therapy in the United States grows due to rising prevalence of cancer, autoimmune disorders, and chronic inflammatory diseases requiring targeted treatment approaches. Aging population trends increase incidence of oncology indications, rheumatoid arthritis, inflammatory bowel disease, and ophthalmic conditions that rely on biologic intervention. Clinical practice increasingly emphasizes precision medicine, where monoclonal antibodies offer high specificity and predictable therapeutic response. Expansion of biologics pipelines among domestic pharmaceutical companies increases availability of approved therapies across multiple disease areas. Favorable reimbursement structures within public and private insurance systems support patient access to advanced biologic treatments.
Hospital networks and specialty clinics expand infusion and specialty pharmacy infrastructure to manage sustained treatment volumes. Advances in antibody engineering improve safety profiles, dosing schedules, and delivery formats, which supports broader clinical adoption. Growth in companion diagnostics enhances patient selection accuracy, increasing initiation rates for antibody based regimens. Manufacturing capacity expansion within the country strengthens supply reliability and reduces production risk. Ongoing clinical research activity reinforces physician familiarity with antibody therapies, supporting continued integration into standard treatment protocols across the healthcare system.
Demand for antibody therapy in USA is shaped by high oncology treatment volumes, expansion of biologics-based care, and structured reimbursement frameworks. Providers evaluate therapeutic efficacy, safety monitoring requirements, administration complexity, and compatibility with institutional treatment protocols. Adoption patterns reflect concentration within hospital-centered care delivery models, supported by specialist supervision, infusion infrastructure, and alignment with FDA-approved biologic treatment pathways.
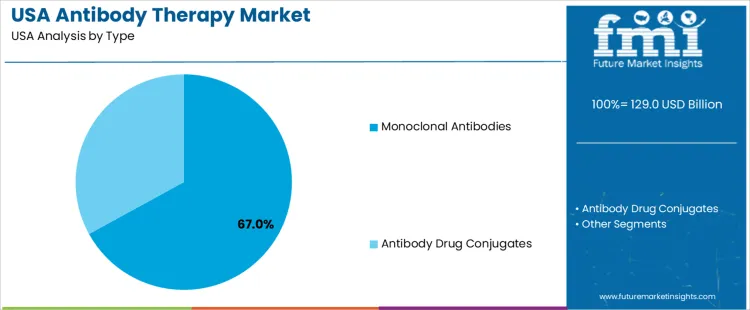
Monoclonal antibodies hold 67.0%, representing the leading therapy type in USA demand. These therapies provide targeted mechanisms of action, predictable pharmacokinetics, and extensive clinical validation across oncology, immunology, and inflammatory diseases. Clinical familiarity and broad label indications support routine integration into standard treatment regimens. Antibody drug conjugates hold 33.0%, supporting targeted cytotoxic delivery for specific cancer indications. Type distribution reflects stronger reliance on monoclonal antibodies due to wider clinical adoption, longer usage history, and established reimbursement positioning within USA healthcare systems.
Key Points
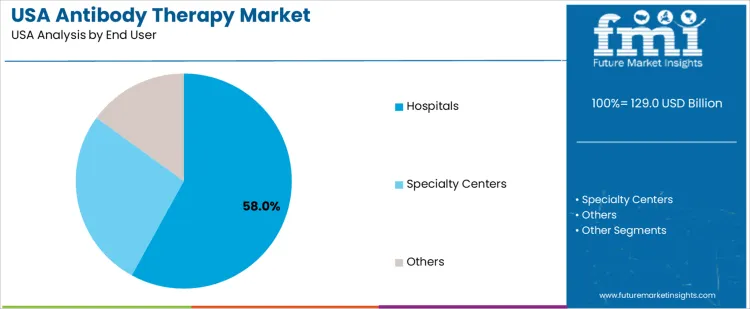
Hospitals hold 58.0%, driving antibody therapy demand in USA. These facilities provide infusion infrastructure, specialist oversight, and adverse-event monitoring required for biologic treatments. Oncology and immunology departments manage complex dosing schedules and reimbursement documentation. Specialty centers hold 27.0%, focusing on disease-specific biologic delivery and advanced treatment protocols. Other end users hold 15.0%, including outpatient clinics and research-linked facilities. End-user distribution reflects centralization of biologic therapy administration within hospital-based care environments.
Key Points
Demand rises as clinicians and healthcare systems in USA expand use of antibody therapies to treat cancer, autoimmune disorders, infectious diseases, and rare conditions. Antibody-based medicines support targeted intervention with improved efficacy and safety profiles compared with conventional treatments. Adoption aligns with advanced clinical infrastructure, widespread diagnostic access, and insurance coverage through Medicare, Medicaid, and private payers. Usage concentrates in oncology centers, specialty clinics, and hospital systems across major metropolitan regions.
USA experiences high incidence of cancers, rheumatoid arthritis, multiple sclerosis, and inflammatory conditions where antibody therapies often form standard care. Oncology specialists rely on monoclonal antibody regimens for targeted tumor antigen binding, immune modulation, and antibody–drug conjugate delivery. Rheumatologists prescribe biologic antibodies for chronic autoimmune disease management. Infectious disease programs use monoclonal antibodies for specific viral and toxin neutralization. High diagnostic screening and imaging rates enable early intervention planning that incorporates antibody therapeutics. Large treatment centers and integrated health systems sustain high procedural volumes and therapy utilization across diverse patient populations.
Antibody therapies are among the highest cost medicines, prompting detailed payer evaluations of cost effectiveness and formulary placement. US Food and Drug Administration approval and postmarked surveillance require extensive clinical evidence and long development timelines. Biologics pricing variability and Medicare reimbursement policies influence therapy uptake decisions at hospitals and health systems. Private insurers negotiate coverage terms that affect patient access and out-of-pocket costs. Biosimilars introduce competitive pricing in established indications, moderating total spend. Economic evaluation and health technology assessment shape formulary decisions. Demand remains robust where clinical benefit is established, while long-term growth depends on pricing transparency, reimbursement policy evolution, and balanced access strategies within USA healthcare delivery.
Demand for antibody therapy in the USA is expanding due to oncology incidence growth, broader autoimmune indications, and steady integration of biologics into standard care pathways. West USA leads with a 14.6% CAGR, supported by innovation hubs and early adoption of novel antibodies. South USA follows at 13.1%, driven by expanding treatment access, infusion capacity, and population growth. Northeast USA records an 11.7% CAGR, shaped by academic medicine, clinical trials, and guideline-driven uptake. Midwest USA posts 10.2%, reflecting structured hospital networks and disciplined reimbursement management. Regional differences reflect care delivery models, trial exposure, infusion infrastructure, and pace of adoption within regulated clinical settings.
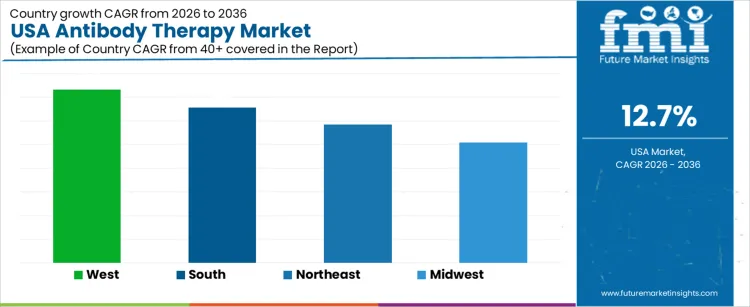
| Region | CAGR (2026 to 2036) |
|---|---|
| West USA | 14.6% |
| South USA | 13.1% |
| Northeast USA | 11.7% |
| Midwest USA | 10.2% |
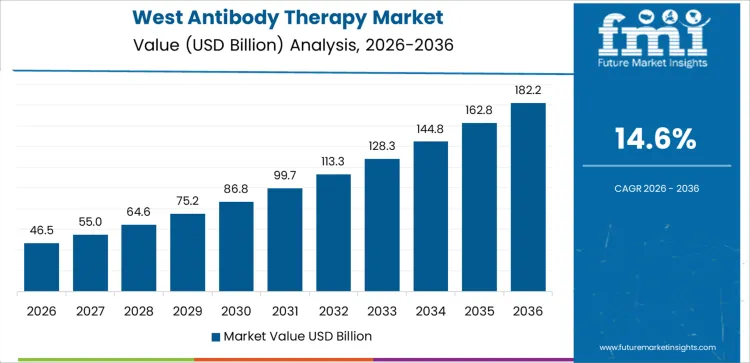
West USA drives demand through concentration of oncology centers, biotech development, and early clinical adoption. Region’s CAGR of 14.6% reflects rapid uptake of monoclonal antibodies across cancer, immunology, and rare disease indications. Academic hospitals and integrated health systems adopt newly approved therapies quickly following FDA clearance. Infusion centers expand capacity to manage outpatient biologic administration efficiently. Precision diagnostics and biomarker testing support targeted prescribing. Providers emphasize outcomes tracking, pharmacovigilance, and protocol optimization. Demand favors therapies with differentiated mechanisms and strong real-world evidence. Growth remains innovation-led, aligned with pipeline maturity and proximity to clinical research activity.
South USA demand is shaped by population growth, expanding healthcare access, and scaling infusion services. Region’s CAGR of 13.1% reflects rising use of antibody therapies for oncology, inflammatory diseases, and chronic conditions. Health systems invest in infusion clinics to manage increasing patient volumes. Community hospitals and specialty practices broaden biologic offerings under standardized protocols. Payer coverage expansion supports treatment continuity. Providers prioritize therapies with established safety profiles and predictable reimbursement. Demand growth remains volume-driven, supported by demographic trends and broader access to specialty care across urban and suburban markets.
Northeast USA demand reflects academic medicine leadership, clinical trial density, and evidence-based treatment adoption. Region’s CAGR of 11.7% is supported by strong presence of teaching hospitals and research institutions. Physicians gain early exposure to novel antibodies through trials and compassionate use programs. Treatment decisions emphasize guideline alignment, outcomes data, and long-term safety monitoring. Infusion services operate within tightly managed hospital systems. Demand favors therapies with companion diagnostics and defined patient selection criteria. Growth aligns with clinical validation and protocol updates rather than rapid volume expansion.
Midwest USA demand is anchored in integrated health systems, cost management, and consistent care delivery. Region’s CAGR of 10.2% reflects steady adoption of antibody therapies within approved indications. Providers focus on treatment efficiency, patient adherence, and reimbursement alignment. Hospital networks standardize formularies to manage biologic costs and ensure supply reliability. Infusion capacity expands cautiously, matched to stable patient volumes. Demand growth remains disciplined and system-driven, aligned with chronic disease management and long-term care planning rather than rapid therapeutic expansion.
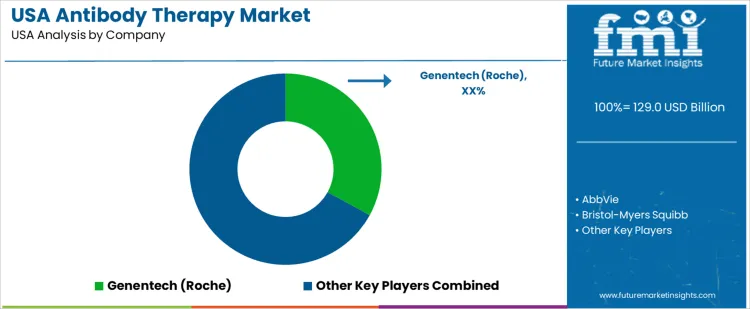
Demand for antibody therapy in the USA is driven by oncology treatment protocols, autoimmune disease management, and expansion of targeted biologic use across multiple therapeutic areas. Adoption spans monoclonal antibodies, antibody-drug conjugates, and bispecific formats administered in hospital and outpatient settings requiring robust clinical evidence and regulatory approval under FDA frameworks. Buyers evaluate clinical efficacy, safety profiles, dosing regimens, and formulation stability compatible with USA treatment standards. Procurement teams prioritize suppliers with strong domestic R&D, local regulatory expertise, and established distribution and cold-chain infrastructure. Trend in the USA market reflects increasing integration of antibody therapies into standard cancer care pathways and growth in immunology indications.
Genentech, a member of the Roche Group, holds a leading position in the USA with a broad portfolio of FDA-approved antibody therapies used across oncology and autoimmune indications and supported by extensive clinical and commercial infrastructure. AbbVie participates with antibody-based treatments for immunology and oncology, backed by strong physician engagement and payer coverage. Bristol-Myers Squibb maintains visibility with monoclonal antibodies and antibody-drug conjugates integrated into cancer treatment regimens. Amgen supports demand with antibody products targeting inflammatory and oncologic disorders widely prescribed within USA clinical practice. Merck & Co. contributes with antibody therapies used in immuno-oncology protocols and combination regimens. Competitive positioning in the USA reflects regulatory alignment, clinical evidence strength, physician familiarity, and comprehensive support services enhancing patient access and outcomes.
| Items | Details |
|---|---|
| Quantitative Units | USD billion |
| Type | Monoclonal Antibodies; Antibody Drug Conjugates |
| End User | Hospitals; Specialty Centers; Other Healthcare Facilities |
| Regions Covered | West USA; South USA; Northeast USA; Midwest USA |
| Key Companies Profiled | Genentech (Roche); AbbVie; Bristol-Myers Squibb; Amgen; Merck & Co. |
| Additional Attributes | Demand is driven by oncology, autoimmune, and inflammatory disease treatment protocols. Monoclonal antibodies account for majority clinical utilization, while antibody drug conjugates expand in targeted cancer therapy. Hospital-based administration dominates, supported by specialty infusion centers and expanding reimbursement coverage for biologic therapies. |
How big is the demand for antibody therapy in USA in 2026?
The demand for antibody therapy in USA is estimated to be valued at USD 129.0 billion in 2026.
What will be the size of antibody therapy in USA in 2036?
The market size for the antibody therapy in USA is projected to reach USD 424.0 billion by 2036.
How much will be the demand for antibody therapy in USA growth between 2026 and 2036?
The demand for antibody therapy in USA is expected to grow at a 12.7% CAGR between 2026 and 2036.
What are the key product types in the antibody therapy in USA?
The key product types in antibody therapy in USA are monoclonal antibodies and antibody drug conjugates.
Which end user segment is expected to contribute significant share in the antibody therapy in USA in 2026?
In terms of end user, hospitals segment is expected to command 58.0% share in the antibody therapy in USA in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.