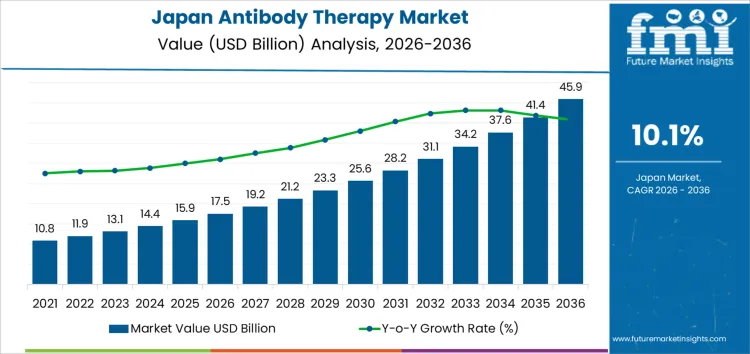
Demand for antibody therapy in Japan is valued at USD 17.5 billion in 2026 and is forecasted to reach USD 45.9 billion by 2036, reflecting a CAGR of 10.1%. Demand accelerates due to rising prevalence of oncology, autoimmune, and inflammatory diseases, alongside strong clinical outcomes associated with targeted biologics. Supportive reimbursement pathways, expedited approvals, and continued innovation in biologic manufacturing reinforce sustained expansion.
Monoclonal antibodies lead therapy-type usage because high specificity, favorable efficacy profiles, and expanding indications improve clinical adoption across oncology and immunology. Clinicians favor next-generation mAbs with enhanced binding affinity, reduced immunogenicity, and optimized dosing regimens. Progress in antibody engineering and combination therapies strengthens therapeutic value across treatment lines.
Kyushu & Okinawa, Kanto, Kansai, Chubu, and Tohoku represent key growth regions driven by concentration of tertiary hospitals, oncology centers, and clinical research institutions. Takeda Pharmaceutical Company, Astellas Pharma, Chugai Pharmaceutical, Daiichi Sankyo, and Ono Pharmaceutical anchor competitive activity through robust pipelines, domestic clinical development, and partnerships aligned with Japan’s regulatory standards and precision medicine priorities.
| Metric | Value |
|---|---|
| Japan Antibody Therapy Sales Value (2026) | USD 17.5 billion |
| Japan Antibody Therapy Forecast Value (2036) | USD 45.9 billion |
| Japan Antibody Therapy Forecast CAGR (2026-2036) | 10.1% |
Demand for antibody therapy in Japan grows due to rising prevalence of cancer, autoimmune disorders, and chronic inflammatory diseases that require targeted treatment approaches. Aging population trends increase incidence of conditions such as rheumatoid arthritis, oncology indications, and ophthalmic disorders, which sustain long term therapy utilization. Clinical practice emphasizes precision medicine, and monoclonal antibodies provide specificity that aligns with treatment optimization goals. Expansion of biologics pipelines within domestic pharmaceutical firms increases availability of approved therapies across multiple indications. National health insurance coverage supports patient access to advanced biologic treatments within hospital and outpatient settings.
Academic medical centers conduct extensive clinical research, which strengthens physician familiarity and adoption of antibody based regimens. Advances in antibody engineering improve efficacy, safety profiles, and dosing convenience, supporting broader clinical application. Growth in companion diagnostics improves patient selection accuracy, which increases treatment initiation rates. Manufacturing capacity expansion within Japan improves supply stability and reduces reliance on imports. Regulatory pathways for biologics remain well defined, supporting timely approvals and lifecycle management. Hospital infusion infrastructure and specialty pharmacies expand service capacity, sustaining continued uptake across therapeutic areas.
Demand for antibody therapy in Japan is shaped by aging population dynamics, structured oncology care pathways, and regulatory support for biologics innovation. Buyers assess clinical efficacy, safety profiles, administration protocols, and compatibility with hospital-based treatment models. Adoption patterns reflect concentration within institutional care settings, supported by specialist oversight, reimbursement frameworks, and standardized treatment guidelines across Japanese healthcare systems.
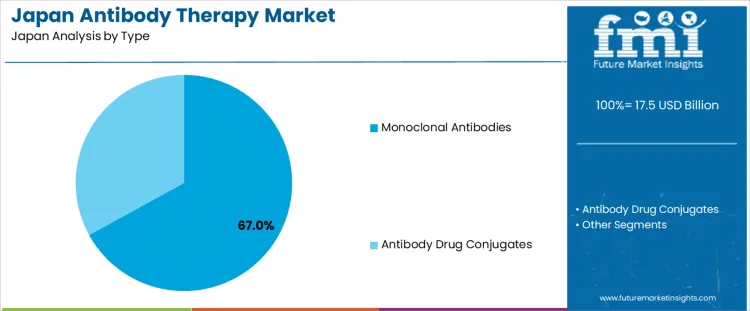
Monoclonal antibodies hold 67.0%, making them the leading therapy-type segment in Japan. These therapies provide targeted action, predictable pharmacokinetics, and established clinical evidence across oncology, autoimmune, and inflammatory indications. Japanese hospitals rely on monoclonal antibodies due to well-defined treatment protocols and monitoring familiarity. Antibody drug conjugates hold 33.0%, supporting targeted cytotoxic delivery with reduced systemic exposure. These therapies are used in specific oncology indications requiring precision targeting. Type distribution reflects stronger reliance on monoclonal antibodies due to broader approvals, longer clinical history, and integration into standard treatment regimens.
Key Points:
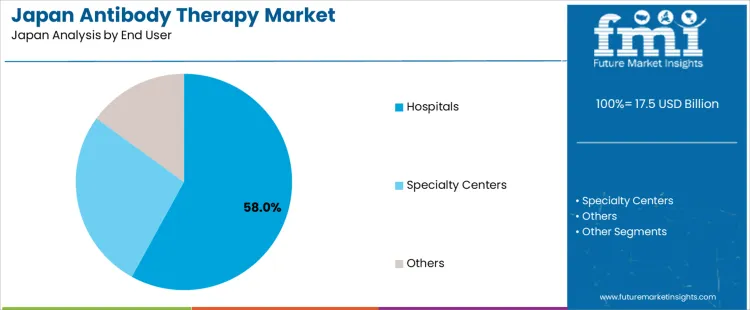
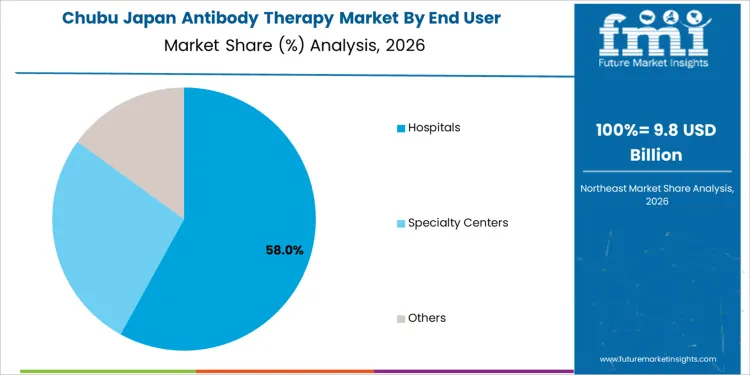
Hospitals hold 58.0%, making them the leading end-user segment in Japan. Antibody therapies require controlled administration, cold-chain handling, and specialist supervision, favoring hospital-based delivery. Oncology and immunology departments manage infusion protocols and adverse event monitoring. Specialty centers hold 27.0%, focusing on disease-specific treatment and advanced care delivery. Other end users hold 15.0%, including outpatient and research-linked settings. End-user distribution reflects concentration of biologic therapy administration within hospitals aligned with Japan’s centralized care and reimbursement structure.
Key Points:
Demand increases as Japan expands use of biologic medicines to address cancer, autoimmune disorders, and chronic diseases associated with an aging population. Antibody therapies play a central role in hospital-based treatment and specialty care. Adoption aligns with advanced clinical infrastructure, national insurance coverage, and strong domestic pharmaceutical R and D capability. Usage concentrates in oncology centers, university hospitals, and large healthcare networks across major prefectures.
Japan’s rapidly aging population increases incidence of cancers, rheumatoid arthritis, inflammatory bowel disease, and ophthalmic conditions treated with monoclonal antibodies. Oncologists rely on targeted antibody therapies to improve survival outcomes and reduce systemic toxicity compared with traditional chemotherapy. Hospitals adopt antibody drugs within standardized treatment guidelines supported by specialist societies. High diagnostic accuracy enables patient stratification suitable for biologic treatment. Domestic pharmaceutical companies and research institutes contribute to antibody development and lifecycle management. National Health Insurance reimbursement ensures broad patient access once therapies are listed. Demand remains strong where clinical efficacy and guideline inclusion support routine use across tertiary and regional hospitals.
Japan applies strict drug price revisions under the national reimbursement system, affecting revenue predictability for antibody therapies. PMDA review processes require extensive clinical and quality data, extending approval timelines for new indications. High manufacturing costs and complex biologics supply chains influence pricing strategy and availability. Biosimilar entry introduces competitive pressure in mature indications. Hospitals manage usage through formulary controls and cost-effectiveness considerations. Capacity constraints in biologics manufacturing affect supply planning. Demand remains stable due to medical necessity, while growth depends on innovation, reimbursement sustainability, and balance between originator and biosimilar adoption within Japan’s regulated healthcare system.
Demand for antibody therapy in Japan is expanding due to rising oncology incidence, autoimmune disease prevalence, and adoption of targeted biologics in clinical practice. Kyushu and Okinawa lead with a 12.6% CAGR, supported by regional cancer treatment centers and biologics administration capacity. Kanto follows at 11.6%, driven by academic hospitals, clinical trial activity, and early adoption of novel antibodies. Kansai records a 10.2% CAGR, shaped by specialty hospitals and standardized treatment protocols. Chubu posts 8.9%, reflecting structured hospital networks and reimbursement-driven uptake. Tohoku shows 7.8%, supported by regional access programs. Rest of Japan records 7.4%, reflecting steady therapy continuation. Regional variation reflects healthcare infrastructure density, trial participation, and treatment access across Japan.
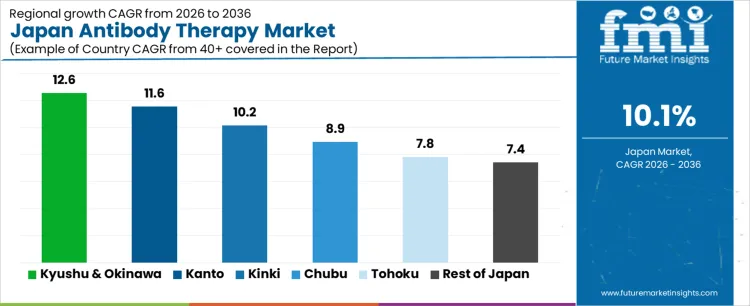
| Region | CAGR (2026 to 2036) |
|---|---|
| Kyushu & Okinawa | 12.6% |
| Kanto | 11.6% |
| Kansai | 10.2% |
| Chubu | 8.9% |
| Tohoku | 7.8% |
| Rest of Japan | 7.4% |
Kyushu and Okinawa drive demand through expanding oncology services, specialty hospitals, and regional biologics infusion capacity. Region’s CAGR of 12.6% reflects increasing use of monoclonal antibodies in cancer, inflammatory diseases, and rare disorders. Hospitals emphasize precision therapy selection aligned with diagnostic testing and treatment guidelines. Infusion centers expand capacity to support outpatient administration and continuity of care. Regional healthcare programs support access to advanced biologics outside major metropolitan areas. Demand favors established antibody therapies with strong clinical evidence and reimbursement clarity. Growth remains patient-volume driven, supported by rising diagnosis rates and structured treatment pathways.
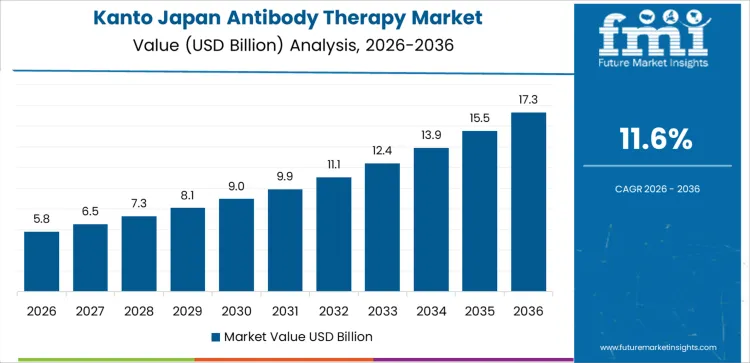
Kanto demand is shaped by concentration of academic medical centers, pharmaceutical research activity, and clinical trial participation. Region’s CAGR of 11.6% reflects early adoption of next-generation antibody therapies in oncology, immunology, and hematology. Teaching hospitals integrate novel antibodies into treatment protocols following regulatory approval. Clinical trials accelerate physician familiarity and post-approval uptake. Demand emphasizes therapies with companion diagnostics and personalized treatment strategies. Hospitals prioritize supply reliability, pharmacovigilance, and outcome monitoring. Growth aligns with innovation pipelines and trial-driven exposure rather than population growth alone.
Kansai demand reflects balanced uptake across oncology centers, specialty clinics, and tertiary hospitals. Region’s CAGR of 10.2% is supported by standardized use of antibody therapies for cancer, rheumatoid conditions, and chronic inflammatory diseases. Providers focus on treatment consistency, protocol adherence, and patient safety. Infusion services operate within established hospital frameworks. Demand growth remains steady, aligned with guideline updates and expansion of approved indications rather than experimental use. Procurement emphasizes reimbursement compatibility and clinical familiarity.
Chubu demand is anchored in structured hospital networks and regional healthcare delivery systems. Region’s CAGR of 8.9% reflects continued adoption of antibody therapies for approved indications within controlled clinical settings. Providers emphasize treatment efficiency, patient throughput, and cost management under reimbursement frameworks. Demand remains linked to stable patient volumes and disease prevalence. Uptake focuses on widely established monoclonal antibodies rather than rapid adoption of newly launched products. Growth remains moderate and system-driven.
Tohoku demand is influenced by regional healthcare access initiatives and aging population needs. Region’s CAGR of 7.8% reflects steady use of antibody therapies in community hospitals and regional medical centers. Providers prioritize accessibility, treatment continuity, and patient monitoring. Infusion services remain centralized, supporting controlled administration. Demand growth remains cautious and necessity-driven, aligned with public healthcare planning and disease management programs rather than innovation-led expansion.
Rest of Japan shows stable demand driven by continuation of existing antibody treatment regimens and replacement prescribing. Region’s CAGR of 7.4% reflects maintenance of therapy volumes across smaller hospitals and clinics. Providers emphasize cost control, availability, and adherence to established protocols. Adoption of new antibody therapies remains gradual. Growth aligns with ongoing disease management rather than expansion of therapeutic scope.
Demand for antibody therapy in Japan is driven by aging demographics, oncology treatment protocols, autoimmune disease management, and infectious disease applications. Usage spans monoclonal antibodies, antibody-drug conjugates, and bispecific formats administered in hospital and clinic settings requiring rigorous clinical data and regulatory approval under Japan’s PMDA framework. Buyers evaluate clinical efficacy, safety profiles, dosing schedules, and formulation stability compatible with Japanese treatment standards. Procurement teams prioritize suppliers with local regulatory expertise, domestic trial data, and established distribution and cold-chain infrastructure. Trend in the Japan market reflects increased adoption of targeted biologics, expansion of indications for immuno-oncology, and integration of antibody therapies into standard treatment regimens.
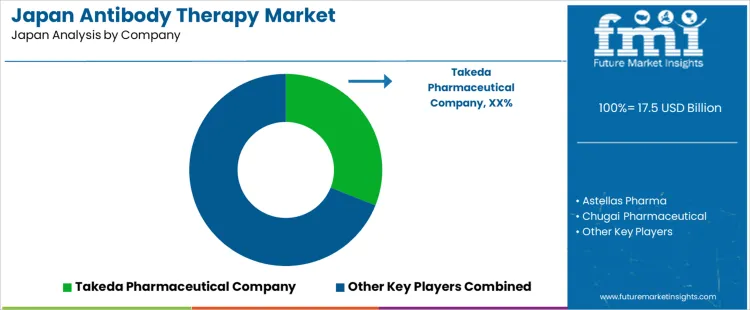
Takeda Pharmaceutical Company holds a leading position in Japan through development and commercialization of antibody therapies targeting oncology and autoimmune conditions supported by domestic clinical programs and PMDA approvals. Astellas Pharma participates with monoclonal antibody products for cancer and inflammatory diseases distributed broadly across Japanese medical institutions. Chugai Pharmaceutical, part of the Roche Group, maintains visibility through its portfolio of antibody therapies including joint development and local marketing collaborations. Daiichi Sankyo supports demand with antibody-drug conjugates and targeted biologics approved for specialized indications in Japan. Ono Pharmaceutical contributes with immune-modulating antibody therapies used in oncology and autoimmune care settings. Competitive positioning in Japan reflects regulatory alignment, clinical evidence strength, physician familiarity, and comprehensive support services that enhance therapy adoption and patient outcomes.
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Type | Monoclonal Antibodies; Antibody Drug Conjugates |
| End User | Hospitals; Specialty Centers; Others |
| Regions Covered | Kanto; Kansai; Chubu; Kyushu & Okinawa; Tohoku; Rest of Japan |
| Key Companies Profiled | Takeda Pharmaceutical Company; Astellas Pharma; Chugai Pharmaceutical; Daiichi Sankyo; Ono Pharmaceutical |
| Additional Attributes | Demand driven by rising oncology and autoimmune disease prevalence, strong biologics R&D pipelines, and regulatory support for advanced therapeutics. Hospitals remain primary administration centers, while specialty centers expand usage for targeted oncology care. Growth reflects Japan’s focus on precision medicine, aging population needs, and domestic innovation in antibody engineering. |
How big is the demand for antibody therapy in Japan in 2026?
The demand for antibody therapy in Japan is estimated to be valued at USD 17.5 billion in 2026.
What will be the size of antibody therapy in Japan in 2036?
The market size for the antibody therapy in Japan is projected to reach USD 45.9 billion by 2036.
How much will be the demand for antibody therapy in Japan growth between 2026 and 2036?
The demand for antibody therapy in Japan is expected to grow at a 10.1% CAGR between 2026 and 2036.
What are the key end user types in the antibody therapy in Japan?
The key end-user types in antibody therapy in Japan are hospitals, specialty centers, and others
Which end user segment is expected to contribute significant share in the antibody therapy in Japan in 2026?
In terms of end user, hospitals segment is expected to command 58.0% share in the antibody therapy in Japan in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.