Demand for automated cell therapy processing systems in Japan is valued at USD 101.0 billion in 2026 and is forecasted to reach USD 319.2 billion by 2036, reflecting a CAGR of 12.2%. Demand expands through rapid advancement of regenerative medicine, supportive regulatory pathways, and increasing clinical translation of cell-based therapies. Emphasis on process standardization, reproducibility, and contamination risk reduction accelerates adoption of automated platforms across research institutes, biopharma manufacturing, and hospital-based cell processing units.
Stem cell therapy systems lead type usage because these platforms enable scalable, closed-system processing for autologous and allogeneic workflows. Automation supports precise cell isolation, expansion, washing, and formulation while maintaining product consistency and compliance with quality requirements. Integration of digital monitoring, single-use consumables, and modular designs improves throughput and reduces operator dependency.

Kyushu & Okinawa, Kanto, Kansai, Chubu, and Tohoku represent key growth regions driven by concentration of academic medical centers, regenerative medicine hubs, and biopharmaceutical manufacturing facilities. Terumo Corporation, Cytiva, Lonza, Miltenyi Biotec, and Takara Bio anchor competitive activity through advanced automation technologies, localized technical support, and end-to-end solutions aligned with Japan’s regenerative medicine framework and clinical manufacturing standards.
| Metric | Value |
|---|---|
| Japan Automated Cell Therapy Processing Systems Sales Value (2026) | USD 101.0 billion |
| Japan Automated Cell Therapy Processing Systems Forecast Value (2036) | USD 319.2 billion |
| Japan Automated Cell Therapy Processing Systems Forecast CAGR (2026-2036) | 12.2% |
Demand for automated cell therapy processing systems in Japan grows due to expansion of regenerative medicine, oncology research, and advanced therapeutic manufacturing. Japan maintains an early approval framework for regenerative therapies, which accelerates clinical translation and increases production requirements. Hospitals and research centers adopt automation to support consistent cell handling, reduced contamination risk, and reproducible outcomes across patient specific therapies. Aging population trends raise incidence of cancer and degenerative diseases, which sustains development of cell based treatments requiring precise processing control. Automation supports closed system workflows aligned with stringent quality and sterility expectations within Japanese healthcare facilities.
Pharmaceutical and biotechnology firms invest in scalable platforms to manage autologous and allogeneic therapies under limited cleanroom space. Workforce constraints within skilled laboratory roles increase reliance on systems that reduce manual intervention and operator variability. Integration with digital monitoring and batch recording improves compliance with domestic regulatory documentation standards. Academic institutions expand translational research programs that require standardized processing across multicenter studies. Government supported innovation funding encourages adoption of advanced manufacturing technologies that strengthen domestic cell therapy infrastructure and long term production capability.
Demand for automated cell therapy processing systems in Japan is shaped by regenerative medicine leadership, standardized manufacturing requirements, and strict quality control frameworks. Buyers evaluate process automation, contamination risk reduction, scalability, and compliance with Japanese regulatory pathways for cell-based therapies. Adoption patterns reflect strong alignment with research-driven development models, hospital-linked manufacturing, and gradual transition toward controlled commercial production environments.
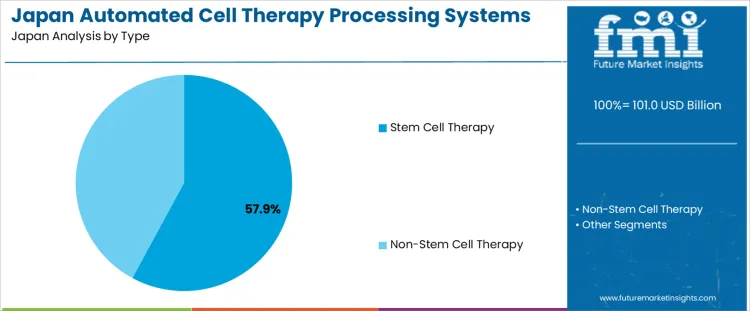
Stem cell therapy systems hold 57.9%, making them the leading type segment in Japan. Japan’s regulatory framework for regenerative medicine supports accelerated development and clinical translation of stem cell therapies. Automated systems are used to standardize cell isolation, expansion, and processing while minimizing operator variability. These systems support consistent quality across autologous and allogeneic workflows in academic hospitals and specialized research centers. Non-stem cell therapy systems hold 42.1%, supporting immune cell therapies and other advanced treatments. Type distribution reflects Japan’s strategic focus on stem cell research leadership and controlled scale-up under validated manufacturing conditions.
Key Points:
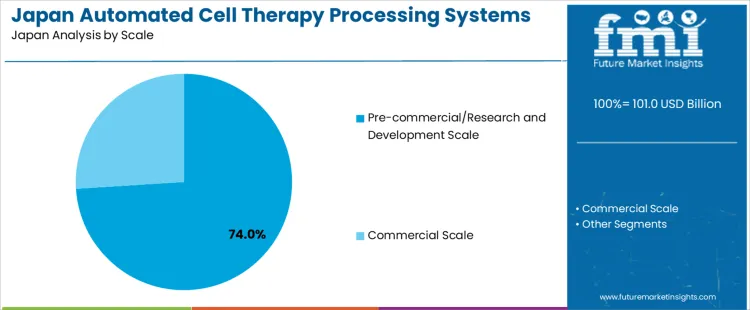
Pre-commercial and research and development scale holds 74.0%, making it the leading scale segment in Japan. Automated processing systems are widely deployed in pilot facilities, university hospitals, and translational research centers. These environments require flexible platforms supporting protocol optimization, small-batch processing, and frequent process adjustments. Commercial scale holds 26.0%, reflecting limited but growing transition toward standardized production for approved therapies. Scale distribution reflects Japan’s emphasis on research-led innovation, early clinical validation, and cautious progression toward large-scale commercialization within regulated regenerative medicine pathways.
Key Points:
Demand increases as Japan expands regenerative medicine, cell therapy clinical trials, and commercial manufacturing under a structured regulatory framework. Automated processing systems support cell isolation, expansion, formulation, and closed handling to improve consistency and reduce contamination risk. Adoption aligns with aging demographics, advanced hospital infrastructure, and national focus on regenerative therapies. Usage concentrates in academic hospitals, contract development and manufacturing organizations, and biotechnology firms across major life science clusters.
Japan promotes regenerative medicine through dedicated legislation enabling conditional and time-limited approval pathways. Hospitals and research institutes increase use of automated systems to standardize autologous and allogeneic cell processing workflows. Automation reduces operator variability, supporting reproducibility required for clinical-grade manufacturing. Academic medical centers adopt closed systems to meet aseptic processing expectations. Biotechnology firms developing cell therapies value scalability and digital traceability embedded in automated platforms. Government-supported research programs and translational hubs encourage equipment investment. Demand strengthens where automation supports faster clinical translation and alignment with Japan’s emphasis on precision, quality, and patient safety in advanced therapies.
Automated cell therapy systems require significant capital investment, limiting adoption among smaller laboratories. Specialized training is necessary to operate and validate automated platforms, affecting deployment speed. PMDA regulatory expectations require extensive documentation, process validation, and change control, increasing implementation effort. Facility space constraints in urban hospitals complicate system integration. Customization needs vary by therapy type, reducing standardization benefits in early-stage programs. Reimbursement uncertainty for cell therapies influences long-term planning. Demand remains stable in well-funded institutions and commercial programs, while broader expansion depends on cost reduction, workforce development, and continued regulatory support for regenerative medicine in Japan.
Demand for automated cell therapy processing systems in Japan is accelerating due to regenerative medicine expansion, aging demographics, and investment in advanced bio manufacturing. Kyushu and Okinawa lead with a 15.2% CAGR, supported by cell therapy production sites and port-linked logistics. Kanto follows at 14.0%, driven by academic hospitals, biopharma headquarters, and clinical trial intensity. Kansai records a 12.3% CAGR, shaped by translational research and hospital-based manufacturing. Chubu posts 10.8%, reflecting manufacturing discipline and process automation needs. Tohoku shows 9.5%, supported by regional medical programs. Rest of Japan records 9.0%, reflecting evaluation-stage adoption. Regional variation reflects clinical concentration, manufacturing readiness, and regulatory engagement across Japan.
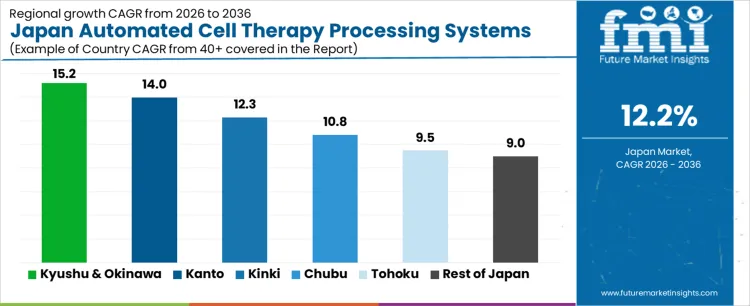
| Region | CAGR (2026-2036) |
|---|---|
| Kyushu & Okinawa | 15.2% |
| Kanto | 14.0% |
| Kansai | 12.3% |
| Chubu | 10.8% |
| Tohoku | 9.5% |
| Rest of Japan | 9.0% |
Kyushu and Okinawa drive demand through growing cell therapy manufacturing capacity and export-oriented bioprocessing activity. Region’s CAGR of 15.2% reflects deployment of automated systems for cell expansion, washing, formulation, and closed-system processing. Facilities prioritize contamination control, reproducibility, and compliance with Japanese regenerative medicine regulations. Port proximity supports import of consumables and outbound logistics for clinical materials. Operators favor modular platforms enabling rapid scale adjustments and protocol standardization. Workforce availability supports operation of automated equipment with reduced manual handling. Demand remains production-led, aligned with commercial readiness and late-stage clinical programs rather than early research alone.
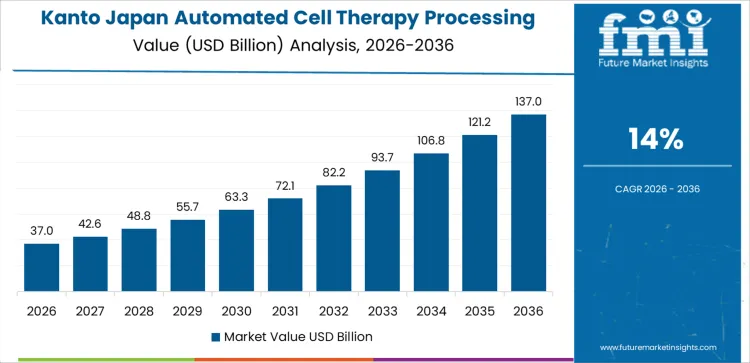
Kanto demand is shaped by concentration of academic medical centers, biopharma headquarters, and clinical trial sponsors. Region’s CAGR of 14.0% reflects extensive use of automated processing systems in hospital-based GMP suites and centralized manufacturing facilities. Institutions prioritize process consistency, electronic batch records, and integration with quality management systems. Clinical trial throughput requires reliable automation to reduce variability and labor dependence. Vendors support customization aligned with diverse cell therapy modalities. Demand growth aligns with translational research progression and clinical scale-up timelines rather than immediate mass production.
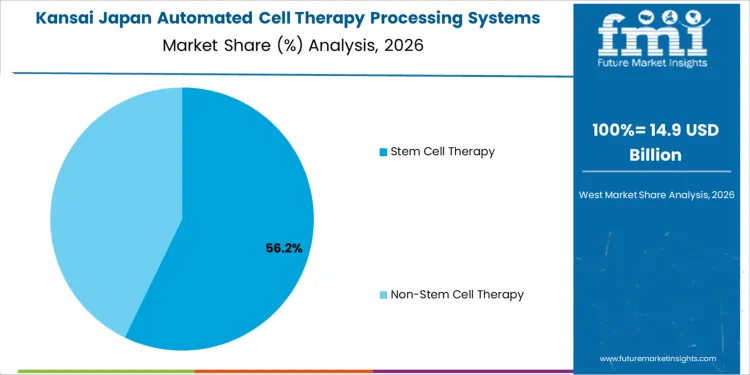
Kansai demand reflects strong translational research, specialty hospitals, and regional bioclusters. Region’s CAGR of 12.3% is supported by adoption of automated systems to transition therapies from research to clinical manufacturing. Providers focus on reducing operator variability and meeting stringent documentation standards. Automation supports parallel processing and protocol replication across sites. Procurement emphasizes validated platforms with local technical support. Demand remains structured and program-driven, aligned with therapy advancement milestones and collaborative research initiatives.
Chubu demand is anchored in manufacturing discipline and process engineering expertise. Region’s CAGR of 10.8% reflects use of automated systems to standardize cell processing workflows and improve yield consistency. Facilities emphasize equipment reliability, uptime, and compatibility with continuous improvement practices. Adoption supports pilot manufacturing and early commercial preparation. Investment decisions align with capital planning cycles and technology readiness assessments. Growth remains moderate and manufacturing-led.
Tohoku demand is influenced by regional medical centers, public research programs, and capacity building initiatives. Region’s CAGR of 9.5% reflects deployment of automated systems for controlled clinical production and research manufacturing. Institutions prioritize ease of operation, training efficiency, and compliance assurance. Systems selected support smaller batch sizes and flexible protocols. Demand growth remains cautious and capability-focused, aligned with regional healthcare development objectives.
Rest of Japan shows steady demand driven by feasibility studies, pilot installations, and incremental modernization. Region’s CAGR of 9.0% reflects adoption of automated systems for evaluation, training, and limited clinical production. Organizations prioritize cost assessment, workflow learning, and regulatory preparedness. Demand remains exploratory, aligned with future expansion planning rather than immediate scale-up.
Demand for automated cell therapy processing systems in Japan is driven by expansion of regenerative medicine, CAR-T programs, and government support for advanced cell-based therapies. Usage spans cell isolation, washing, expansion, formulation, and closed-system processing within hospitals, research institutes, and biopharmaceutical manufacturing sites. Buyers evaluate system sterility, closed-loop automation, scalability from clinical to commercial volumes, and compatibility with Japanese GMP and PMDA regulatory expectations. Procurement teams prioritize suppliers offering validated workflows, local technical support, and integration with existing cleanroom and bioprocess infrastructure. Trend in the Japan market reflects growth of autologous therapies, increasing clinical trial activity, and strong emphasis on process standardization to ensure reproducibility and patient safety.
Terumo Corporation maintains a leading position through domestically developed automated cell processing platforms widely adopted in Japanese hospitals and cell therapy centers. Systems align with closed processing requirements and clinical workflow needs across regenerative medicine applications. Cytiva Japan participates with automated cell processing and bioreactor systems used in translational research and commercial manufacturing environments. Lonza Japan maintains visibility through cell therapy manufacturing platforms and automated solutions supporting scale-up and process control. Miltenyi Biotec Japan supplies automated cell separation and processing systems used extensively in research and early clinical production. Takara Bio contributes with cell processing instruments and reagents aligned with Japan’s academic and clinical research ecosystem. Competitive positioning in Japan reflects regulatory alignment, system reliability, automation depth, and strong domestic service capability supporting sensitive cell therapy workflows.
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Type | Stem Cell Therapy; Non-Stem Cell Therapy |
| Scale | Pre-commercial / Research and Development Scale; Commercial Scale |
| Regions Covered | Kyushu & Okinawa; Kanto; Kansai; Chubu; Tohoku; Rest of Japan |
| Key Companies Profiled | Terumo Corporation; Cytiva; Lonza; Miltenyi Biotec; Takara Bio |
| Additional Attributes | Dollar sales by therapy type and processing scale; regional CAGR trends across Kanto and Kansai; rising adoption in commercial-scale manufacturing; integration of closed and automated systems; increasing demand from regenerative medicine programs; regulatory-driven standardization; growing investments in cell therapy infrastructure by pharmaceutical and biotechnology companies. |
How big is the demand for automated cell therapy processing systems in Japan in 2026?
The demand for automated cell therapy processing systems in Japan is estimated to be valued at USD 101.0 billion in 2026.
What will be the size of automated cell therapy processing systems in Japan in 2036?
The market size for the automated cell therapy processing systems in Japan is projected to reach USD 319.2 billion by 2036.
How much will be the demand for automated cell therapy processing systems in Japan growth between 2026 and 2036
The demand for automated cell therapy processing systems in Japan is expected to grow at a 12.2% CAGR between 2026 and 2036.
What are the key product types in the automated cell therapy processing systems in Japan?
The key product types in automated cell therapy processing systems in Japan are stem cell therapy and non-stem cell therapy.
Which scale segment is expected to contribute significant share in the automated cell therapy processing systems in Japan in 2026?
In terms of scale, pre-commercial/research and development scale segment is expected to command 74.0% share in the automated cell therapy processing systems in Japan in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.