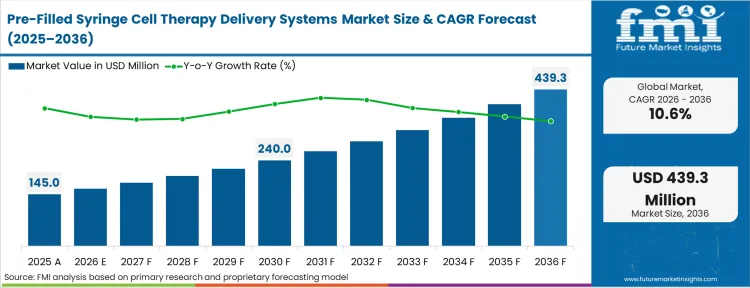
The Pre-Filled Syringe Cell Therapy Delivery Systems Market was valued at USD 145.0 million in 2025. The sector is expected to cross USD 160.4 million in 2026 at a CAGR of 10.60% during the forecast period. With steady investment, the market is projected to reach USD 424.4 million by 2036 as developers are moving away from traditional vial‑and‑transfer workflows in hospitals and adopting simpler, point‑of‑care delivery methods that help maintain healthier cells.
Biopharmaceutical procurement directors are being forced to qualify living-cell-specific enclosures rather than repurposing standard prefilled syringes drug molecules containers. The stakes for delaying this transition involve severe batch rejection rates at the clinical site, as traditional materials compromise sensitive cellular payloads through subtle chemical interactions. A critical vulnerability remains in standard silicone lubrication, which induces protein aggregation and cell death over extended storage. Facilities that integrate specialized polymer based prefilled syringe formats secure a vital workflow advantage by eliminating the final sterile transfer step entirely, ensuring maximum therapeutic dosage reaches the patient.
Before growth becomes self-reinforcing, the validation of cryogenic freezing directly within the primary delivery vehicle must be achieved universally. Primary packaging manufacturers trigger this inflection by developing closure integrities that withstand -196°C environments without compromising the sterile barrier upon thawing. Once this gate is passed, the operational friction of onsite thawing and re-drawing is removed entirely.
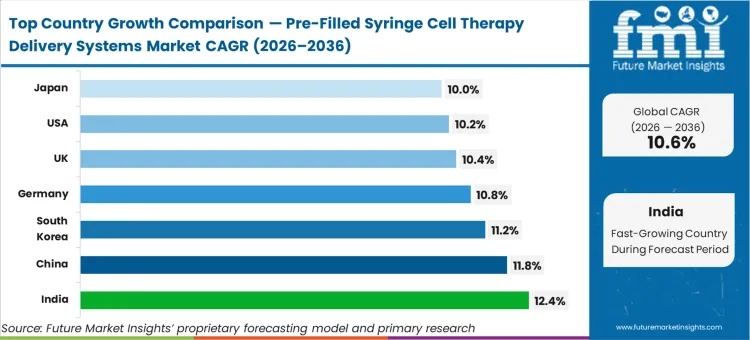
India is expected to advance at 12.4%, followed by China expanding at 11.8%, and South Korea at 11.2%. Germany is poised to record 10.8%, while the United Kingdom tracks at 10.4%, the United States grows at 10.2%, and Japan registers a 10.0% trajectory. The divergence across this range reflects the varying speeds at which decentralized cellular manufacturing hubs mandate error-proof, integrated delivery systems for their rapidly expanding domestic clinic networks.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 160.4 million |
| Industry Value (2036) | USD 424.4 million |
| CAGR (2026-2036) | 10.60% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
The Pre-Filled Syringe Cell Therapy Delivery Systems Market encompasses primary packaging components specifically engineered to store, transport, and administer live-cell therapeutics without compromising cellular viability. Unlike traditional biologics containers, these systems are fundamentally defined by their inert material properties, advanced closure integrity under extreme cryogenic conditions, and complete elimination of silicone oils or heavy metal extractables that are toxic to living cellular suspensions.
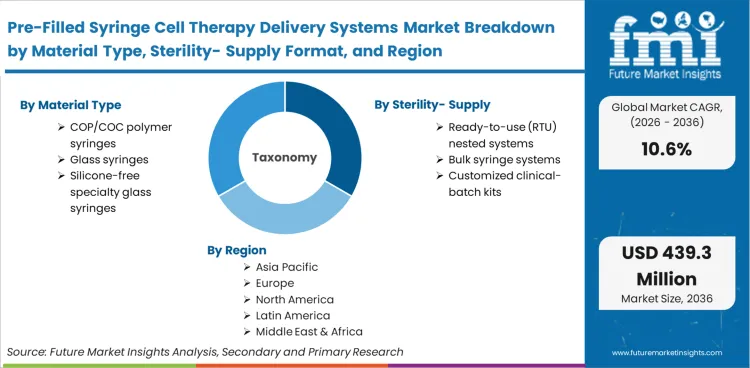
Scope incorporates highly specialized delivery architectures designed explicitly for cellular payloads, including cyclic olefin polymers cop polymer syringes, silicone-free specialty glass formats, and cryogenically stable elastomer closures. The market covers all integrated components that maintain direct contact with the therapeutic agent from the point of final fill-finish through bedside administration, including specialized tip caps, integrated needle mechanisms, and nested ready-to-use tub configurations required by automated aseptic filling lines.
Standard borosilicate glass containers designed for traditional small-molecule or non-cellular biologic applications are explicitly excluded, as their tungsten residuals and required silicone lubrication render them lethal to live cell therapies. Secondary packaging, thermal shipping shippers, and standalone administration sets not integrated into the primary syringe body are also excluded, as they do not address the primary material contact challenge defining this sector.
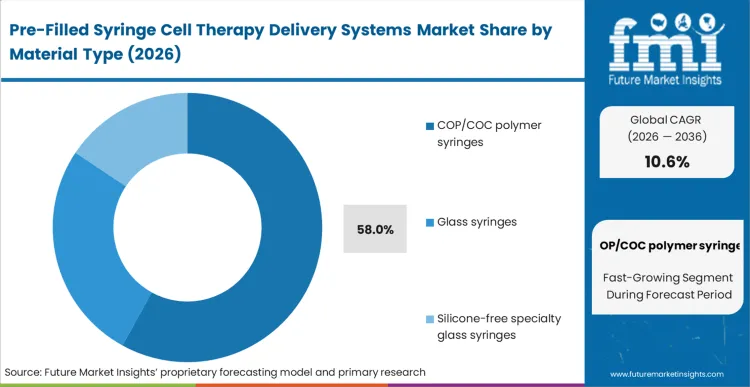
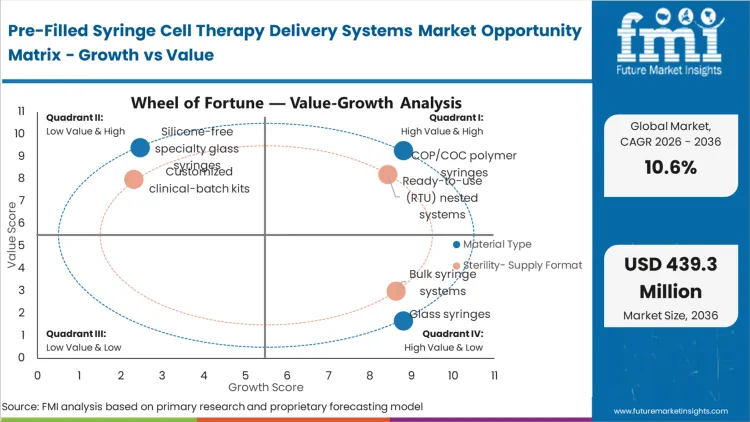
Legacy glass architectures fail to deliver the resilience required when subjecting primary containers to liquid nitrogen freezing protocols. The reason COP/COC polymer syringes command 58.0% share in 2026 stems from this operational reality: biopharmaceutical developers cannot accept the risk of catastrophic container breakage at the clinical site.
According to FMI's estimates, the inherent inertness of cyclic olefin polymers eliminates the tungsten residuals and pH shifts historically associated with glass forming processes. This material transition allows formulation scientists to stabilize sensitive cellular payloads without overcompensating for container-induced degradation. A buyer who delays migrating away from legacy bfs syringes or standard glass alternatives faces unacceptable clinical trial variances caused directly by packaging interference.
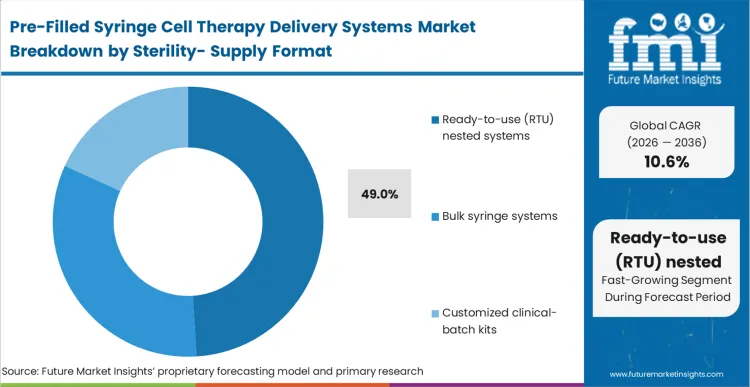
Facility managers are currently being forced to decide whether to maintain expensive, footprint-heavy washing and depyrogenation lines or outsource that burden entirely to component suppliers. Capturing a dominant 49.0% share, Ready-to-use (RTU) nested systems represent the outcome of this decision matrix. By receiving syringes and injectable drugs packaging pre-sterilized and arrayed in standard tubs, manufacturing suites can feed components directly into aseptic isolators.
As per FMI's projection, this removes the contamination risk burden upstream and allows commercial facilities to maximize their core therapeutic processing footprint. Operations directors who fail to integrate RTU systems throttle their own fill-finish throughput, permanently capping their commercial output regardless of their upstream bioreactor capacity.
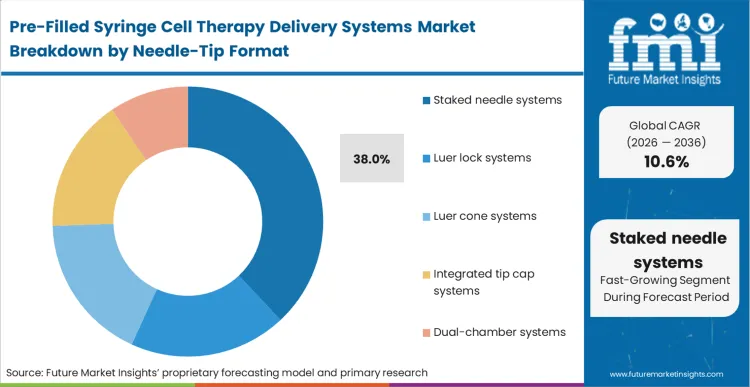
When clinicians must manually attach needles to highly concentrated, extremely expensive cell therapy doses, the risk of dead-volume waste and accidental contamination spikes dramatically. Staked needle systems resolve this critical vulnerability, explaining their 38.0% position in the market structure.
FMI analysts opine that integrating the cannula directly into the syringe body during manufacturing eliminates the luer hub, a notoriously problematic zone where living cells can sheer or aggregate during injection. This architectural choice directly preserves the functional cell count delivered into the patient's bloodstream. Therapeutic brands that overlook this dynamic and specify modular biopharmaceuticals packaging inadvertently reduce their delivered therapeutic dose, risking compromised clinical outcomes at the bedside.
The entire logistical framework of cellular medicine relies on extreme temperature control to halt cellular metabolism during transit. Holding a 41.0% share, deep-cold and cryogenic-compatible systems align precisely with the baseline supply chain requirements of unmodified cell suspensions.
Based on FMI's assessment, primary packaging suppliers who master this segment dominate the commercial contracts because they solve the most severe engineering constraint in the entire workflow. The ability to maintain complete closure integrity down to -196°C without compromising the elastomer plunger seal determines the viability of global distribution. Procurement leaders relying on standard refrigerated injectable drug delivery components for advanced cell lines face inevitable clinical failures when temperature excursions breach the container's structural limits.
The absolute necessity to guarantee vein-to-vein cellular viability compels clinical supply chain directors to adopt specialized delivery systems. Traditional vial-to-syringe transfer protocols introduce catastrophic risks of particulate contamination, cell shearing, and dosing inaccuracies that compromise multimillion-dollar therapeutic regimens. By specifying pre-filled architectures, procurement heads eliminate the bedside handling variables entirely, securing the integrity of the dose. This moves the responsibility of final preparation from stressed hospital pharmacies directly into the highly controlled environments of GMP manufacturing suites.
The primary operational friction slowing mass adoption involves the protracted and complex material qualification cycles required by regulatory bodies. Living cell therapies exhibit profound sensitivities to subtle leachable compounds that small-molecule drugs easily tolerate, meaning every new packaging format must undergo rigorous compatibility testing that often stretches for years. This creates an immense switching cost. While advanced drug delivery technology providers offer accelerated predictive modeling data, therapeutic developers remain fundamentally constrained by the timeline required to generate real-time stability data before regulatory submission.
Opportunities in the Pre-Filled Syringe Cell Therapy Delivery Systems Market
The regional assessment divides the Pre-Filled Syringe Cell Therapy Delivery Systems Market into North America, Europe, Asia Pacific, and additional key regions spanning more than 40 countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 12.4% |
| China | 11.8% |
| South Korea | 11.2% |
| Germany | 10.8% |
| United Kingdom | 10.4% |
| United States | 10.2% |
| Japan | 10.0% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
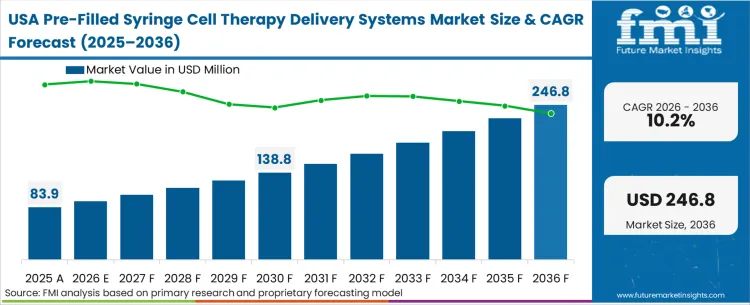
The intense concentration of advanced clinical stage cell therapy pipelines in this region dictates a procurement environment strictly focused on rapid scalability and risk mitigation. In FMI's view, North American therapeutic developers prioritize engaging packaging partners during Phase I trials to lock down material compatibility early, effectively shutting out component suppliers who attempt to enter during late-stage commercialization. This dynamic forces packaging manufacturers to invest heavily in early-phase engineering support rather than competing purely on commercial scale pricing.
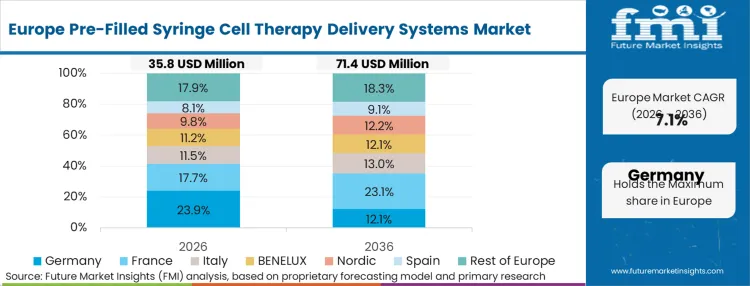
Stringent regulatory frameworks governing particulate contamination and extractable profiles shape adoption patterns fundamentally differently across this region. European medicine agencies heavily penalize any variations in therapeutic efficacy linked to primary packaging interactions, driving an aggressive switch from standard borosilicate platforms. According to FMI's estimates, this policy-led environment forces local contract manufacturing organizations to overhaul their filling architectures to accommodate specialized, silicone-free polymer systems well ahead of global mandates.
Economic cost considerations combined with significant state-driven capital infusion in biomanufacturing infrastructure drive decisions throughout this highly dynamic geography. Asian therapeutic developers are rapidly scaling allogeneic (off-the-shelf) cell therapies, which demand massive volumes of standardized, highly reliable primary packaging. As per FMI's projection, the sheer scale of production planned across the region forces a transition from custom clinical batches to fully automated, high-throughput nested syringe formats to maintain unit economics.
FMI's report includes extensive coverage of additional countries not detailed in these bullets. Across these broader emerging markets, a clear pattern emerges where the lack of established legacy biological manufacturing infrastructure accelerates the adoption of advanced, integrated polymer systems, bypassing glass entirely.
The competitive landscape for cell therapy delivery systems is intensely concentrated because the validation barrier to enter this space requires years of specialized material science development. Leading packaging manufacturers like Becton, Dickinson and Company, SCHOTT Pharma, and Gerresheimer hold commanding positions precisely because they own the proprietary compounding formulas for cryogenically stable polymers and silicone-free elastomer coatings. Biopharmaceutical buyers do not select vendors based on unit pricing; they filter exclusively on the availability of robust, pre-compiled Drug Master Files (DMFs) and proven extractable/leachable data that can survive regulatory scrutiny.
Incumbents like Stevanato Group and West Pharmaceutical Services possess a distinct advantage rooted in their deep integration with the world's leading fill-finish equipment manufacturers. This alignment ensures their nested tub formats run flawlessly on high-speed robotic isolators. Challengers attempting to disrupt this space must build comprehensive analytical testing capabilities to prove their novel materials do not alter cellular phenotypes under deep cold. Creating a technically superior auto injectors component is insufficient; vendors must provide the exhaustive scientific documentation proving the container's absolute biological inertness.
As commercial cell therapy volumes scale toward 2036, the tension between buyer preferences for dual-sourcing and the reality of vendor lock-in will define procurement strategies. Large therapeutic developers desperately want to qualify multiple suppliers to mitigate supply chain risks, but the immense cost of validating a second primary packaging system often makes this impossible. Consequently, the market is poised to become even more concentrated, with top-tier vendors absorbing smaller specialized elastomer and polymer firms to offer complete, fully integrated containment ecosystems that lock buyers in from Phase I through commercialization.
| Metric | Value |
|---|---|
| Quantitative Units | USD 160.4 million to USD 424.4 million, at a CAGR of 10.60% |
| Market Definition | The Pre-Filled Syringe Cell Therapy Delivery Systems Market encompasses primary packaging components specifically engineered to store, transport, and administer live-cell therapeutics without compromising cellular viability at cryogenic temperatures. |
| Material Type Segmentation | COP/COC polymer syringes, Glass syringes, Silicone-free specialty glass syringes |
| Sterility / Supply Format Segmentation | Ready-to-use (RTU) nested systems, Bulk syringe systems, Customized clinical-batch kits |
| Needle / Tip Format Segmentation | Staked needle systems, Luer lock systems, Luer cone systems, Integrated tip cap systems, Dual-chamber systems |
| Temperature / Stability Profile Segmentation | Deep-cold / cryogenic-compatible systems, Refrigerated systems, Ambient / short-hold systems |
| Regions Covered | North America, Europe, Asia Pacific |
| Countries Covered | United States, Germany, United Kingdom, China, Japan, South Korea, India, and 40 plus countries |
| Key Companies Profiled | Becton, Dickinson and Company, SCHOTT Pharma AG & Co. KGaA, Gerresheimer AG, Stevanato Group S.p.A., West Pharmaceutical Services, Inc., Terumo Corporation, Nipro Corporation |
| Forecast Period | 2026 to 2036 |
| Approach | Procurement directors and packaging engineers were interviewed to establish material qualification timelines. The data baseline anchors to commercialized cell therapy doses and active clinical trial pipelines. Forecasts were cross-validated against capacity expansion metrics from sterile fill-finish equipment manufacturers. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
How large is the Pre-Filled Syringe Cell Therapy Delivery Systems Market in 2026?
The market is valued at USD 160.4 million in 2026. This figure reflects the immediate capital being deployed by clinical developers to transition away from legacy hospital-based transfer protocols toward highly controlled, point-of-care delivery systems.
What will it be valued at by 2036?
The sector is projected to reach USD 424.4 million by 2036. This expansion signals the universal integration of specialized living-cell enclosures as commercial cell therapy pipelines mature and demand high-throughput, automated fill-finish architectures.
What CAGR is projected?
A compound annual growth rate of 10.60% is forecast from 2026 to 2036. This steady pace tracks the rigorous, multi-year material qualification cycles required by regulatory bodies to validate new primary packaging, rather than a rapid consumer-style adoption curve.
Which Material Type segment leads?
COP/COC polymer syringes hold a dominant 58.0% share. Biopharmaceutical buyers select these materials because they eliminate the catastrophic breakage risks associated with glass at cryogenic temperatures and remove tungsten residuals that threaten delicate cellular suspensions.
Which Sterility / Supply Format segment leads?
Ready-to-use (RTU) nested systems capture a 49.0% share. Facility managers prioritize these formats because they arrive pre-sterilized, allowing commercial manufacturing suites to bypass expensive onsite washing equipment and feed directly into automated robotic isolators.
Which Needle / Tip Format segment leads?
Staked needle systems represent 38.0% of the market. Integrating the cannula directly into the syringe body prevents the mechanical sheer forces and dead-volume waste typically seen at modular luer hubs, thereby maximizing the viable cell dose delivered to the patient.
What drives rapid growth?
The absolute necessity to guarantee vein-to-vein cellular viability forces clinical operations to adopt these pre-filled systems. By eliminating the manual bedside transfer steps that introduce particulate contamination and dosing errors, therapeutic developers secure the integrity of multimillion-dollar treatments.
What is the primary restraint?
The absolute necessity to guarantee vein-to-vein cellular viability forces clinical operations to adopt these pre-filled systems. By eliminating the manual bedside transfer steps that introduce particulate contamination and dosing errors, therapeutic developers secure the integrity of multimillion-dollar treatments.
What is the primary restraint?
The intense regulatory burden of extractable and leachable testing creates massive friction. Packaging engineers must generate years of real-time stability data to prove novel materials do not alter cellular phenotypes under deep cold, severely delaying the commercial implementation of new formats.
Which country grows fastest?
India advances at a leading 12.4% CAGR, outpacing China's 11.8%. While China focuses on massive state-backed scale, India's trajectory is driven by the rapid decentralization of its clinical networks, requiring highly standardized, error-proof delivery systems that can function across varied domestic logistical conditions.
How do European extractable regulations impact vendor selection?
European medicine agencies maintain zero-tolerance policies for container-induced cellular degradation. This forces regional CMOs to bypass legacy glass platforms entirely and mandate fully silicone-free polymer systems well ahead of global compliance deadlines.
Why is silicone lubrication being eliminated from these systems?
Standard silicone oil, used to ensure smooth plunger glide, actively induces protein aggregation and subsequent cell death over extended storage holds. Eliminating it forces material science firms to develop advanced fluoropolymer coatings that maintain functionality without toxic fluid interference.
What role do Drug Master Files (DMFs) play in competitive dynamics?
Buyers filter out inexperienced vendors explicitly based on the lack of comprehensive DMFs. A supplier that provides robust, pre-compiled analytical testing data for cryogenic conditions radically shortens the therapeutic developer's time-to-market, locking in long-term master supply agreements.
Why do buyers struggle to dual-source in this market?
Therapeutic developers desire multiple suppliers to mitigate supply chain risks, but the immense financial and chronological cost of running full stability validation on a second primary packaging format effectively binds them to their initial Phase I vendor selection.
How does cryogenic storage (-196°C) alter component engineering?
Standard rubber plungers shatter and lose closure integrity when exposed to liquid nitrogen environments. Suppliers must formulate specialized low-temperature elastomers that contract predictably with the polymer barrel, ensuring the sterile barrier remains uncompromised upon rapid bedside thawing.
Why are RTU nested formats critical for commercial scalability?
As cell therapies move from bespoke clinical batches to commercial volumes, manual filling becomes impossible. RTU tubs are dimensionally standardized to interface flawlessly with automated isolator tooling, removing the filling bottleneck at the contract manufacturing level.
How does the United States market dictate procurement timing?
The intense venture-backed competition in the United States cell therapy sector means clinical trial delays are financially ruinous. Consequently, United States procurement directors must lock down guaranteed supply allocations of validated polymer enclosures during Phase I, preventing late-entry suppliers from capturing share.
What is the operational consequence of using legacy vial-and-transfer methods?
Hospitals attempting to extract cellular suspensions from standard vials into administration syringes routinely document up to a 20% loss in viable cell count due to mechanical sheer and container dead-volume, directly degrading the patient's therapeutic outcome.
How do staked needles protect delicate cellular membranes?
By eliminating the internal ridges and turbulent flow zones inherent in modular attachable hubs, staked architectures maintain a smooth, continuous fluid path. This prevents cells from aggregating or bursting as they are expressed from the syringe barrel into the administration line.
What advantage do incumbents have over new entrants?
Established packaging giants have spent decades integrating their dimensional specifications with the world's leading sterile filling equipment manufacturers. A new entrant cannot simply produce a viable syringe; they must prove it will run without jamming on the exact robotic lines utilized by global CMOs.
Why does Japan exhibit a slower 10.0% CAGR compared to its neighbors?
Japanese regulatory authorities require incredibly prolonged, localized material validation cycles before approving novel primary packaging. This cautious framework suppresses immediate volume spikes but ensures long-term operational stability free from container-related clinical trial failures.
How does the shift to allogeneic (off-the-shelf) therapies change the packaging demand?
Autologous therapies require single-batch, bespoke handling, whereas allogeneic treatments are manufactured in large, centralized runs for broad distribution. This change mandates the adoption of high-throughput, automated fill-finish injectable drugs market infrastructure, drastically accelerating the consumption of standardized nested syringe tubs.
What is the stakes for a biomanufacturer failing to secure cryogenic closure data?
Without exhaustive proof that the sterile barrier holds during both freezing and thawing cycles, regulatory agencies will refuse to approve the investigational new drug (IND) application. The biomanufacturer is then forced to restart multi-year stability trials, potentially losing their commercial viability entirely.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.