Demand for Antibody-mediated Rejection Prevention in USA
Demand for Antibody-mediated Rejection Prevention in USA Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Demand for Antibody-mediated Rejection Prevention in USA 2026 to 2036
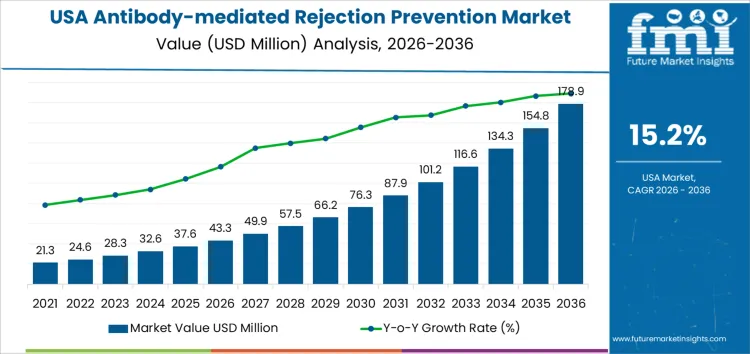
Demand for antibody-mediated rejection prevention in USA is valued at USD 43.3 million in 2026 and is expected to reach USD 178.9 million by 2036, reflecting a CAGR of 15.2%. Demand expands due to rising solid organ transplant volumes, improved post-transplant survival rates, and increasing clinical focus on antibody-mediated rejection as a key cause of graft failure. Advances in immunology diagnostics and early risk stratification strengthen adoption of targeted preventive therapies.
Quick Stats for Demand for Antibody-mediated Rejection Prevention in USA
- USA Antibody-mediated Rejection Prevention Sales Value (2026): USD 43.3 million
- USA Antibody-mediated Rejection Prevention Forecast Value (2036): USD 178.9 million
- USA Antibody-mediated Rejection Prevention Forecast CAGR (2026 to 2036): 15.2%
- Leading Treatment Segment in USA: Monoclonal Antibodies
- Fastest-Growing Region in USA: West USA
- Key Growth Regions in USA: West USA, South USA, Northeast USA, Midwest USA
- Top Players in USA: Alexion (AstraZeneca); Bristol-Myers Squibb; Genentech (Roche); Novartis; Sanofi Genzyme
Monoclonal antibodies lead treatment-segment usage because these therapies enable precise modulation of immune pathways involved in donor-specific antibody activity. Clinicians prefer monoclonal antibody-based regimens for their specificity, predictable pharmacokinetics, and compatibility with combination immunosuppressive protocols. Expanding clinical evidence and guideline inclusion support broader use across kidney, heart, and lung transplant settings.
West USA represents the fastest-growing region due to high transplant center density, advanced clinical research activity, and early adoption of novel biologics. West USA, South USA, Northeast USA, and Midwest USA together account for core demand driven by established transplant programs and referral networks. Alexion (AstraZeneca), Bristol-Myers Squibb, Genentech (Roche), Novartis, and Sanofi Genzyme anchor competitive activity through biologics pipelines, transplant-focused clinical programs, and commercialization strategies aligned with evolving graft preservation and long-term transplant outcome requirements across the United States.
USA Antibody-mediated Rejection Prevention Key Takeaways
| Metric | Value |
|---|---|
| USA Antibody-mediated Rejection Prevention Sales Value (2026) | USD 43.3 million |
| USA Antibody-mediated Rejection Prevention Forecast Value (2036) | USD 178.9 million |
| USA Antibody-mediated Rejection Prevention Forecast CAGR (2026 to 2036) | 15.2% |
Why is the Demand for Antibody-mediated Rejection Prevention in USA Growing?
Demand for antibody-mediated rejection prevention in the United States grows due to rising transplant volumes and improved long term graft survival expectations. Kidney, heart, lung, and liver transplant programs expand as surgical outcomes and post-transplant care improve. Increased use of sensitized donors and recipients elevates risk of antibody mediated immune responses, requiring proactive prevention strategies. Aging recipient populations and higher prevalence of comorbidities increase immunologic complexity after transplantation. Clinical protocols emphasize early identification of donor specific antibodies to reduce graft injury and failure rates.
Advances in immunology improve understanding of humoral rejection mechanisms, supporting targeted prevention approaches. Transplant centers adopt refined immunosuppression regimens and antibody monitoring to maintain graft function. Growth in living donor transplants increases need for individualized rejection risk management. Regulatory and payer focus on outcome based care supports interventions that reduce rehospitalization and graft loss. Expansion of transplant registries and data analytics improves risk stratification and protocol standardization across centers. Long term follows up programs increase utilization of preventive therapies throughout post-transplant care cycles. Research activity in biologics and immune modulation strengthens clinical adoption of antibody mediated rejection prevention strategies across the healthcare system.
How Are the Segments Classified in the Demand for Antibody-Mediated Rejection Prevention in USA?
Demand for antibody-mediated rejection prevention in USA is shaped by solid organ transplant volumes, long-term graft survival priorities, and standardized immunosuppression protocols. Clinicians evaluate therapeutic efficacy, safety profiles, timing of intervention, and compatibility with transplant regimens. Adoption patterns reflect concentration within specialized transplant centers and hospitals managing complex post-transplant care under established clinical and regulatory frameworks.
Which Treatment Modality Accounts for the Largest Share in Antibody-Mediated Rejection Prevention in USA?
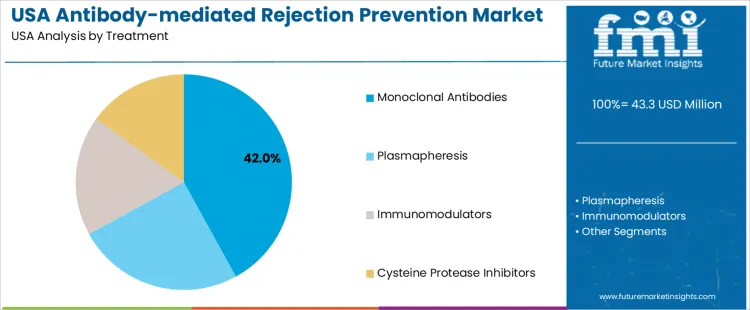
Monoclonal antibodies hold 42.0%, accounting for the largest treatment share in USA demand. These therapies target specific immune pathways responsible for antibody-mediated graft injury, supporting controlled immunosuppression and improved graft outcomes. Plasmapheresis holds 25.0%, applied to reduce circulating donor-specific antibodies during acute rejection episodes. Immunomodulators account for 18.0%, supporting adjunct immune regulation in combination protocols. Cysteine protease inhibitors hold 15.0%, serving targeted therapeutic roles in selected clinical scenarios. Treatment distribution reflects reliance on biologic therapies for precise immune pathway control.
Key Points
- Monoclonal antibodies lead due to targeted immune suppression.
- Plasmapheresis supports antibody reduction in acute settings.
- Adjunct therapies complement primary treatment protocols.
Which End-User Setting Drives Demand for Antibody-Mediated Rejection Prevention in USA?
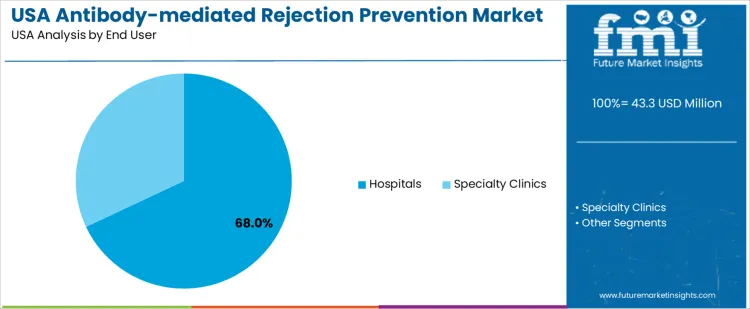
Hospitals hold 68.0%, driving demand for antibody-mediated rejection prevention in USA. Transplant hospitals provide specialized immunology expertise, monitoring infrastructure, and intensive care capabilities required for rejection management. These settings coordinate multidisciplinary transplant teams overseeing diagnosis, treatment selection, and follow-up. Specialty clinics hold 32.0%, focusing on post-transplant monitoring and outpatient management under hospital-linked care pathways. End-user distribution reflects centralization of rejection prevention within hospital-based transplant programs.
Key Points
- Hospitals dominate due to transplant and monitoring capabilities.
- Multidisciplinary teams support complex immune management.
- Specialty clinics manage structured post-transplant follow-up.
What are the Key Dynamics in the Antibody-mediated Rejection Prevention Industry?
Demand increases as transplant centers, immunology specialists, and multidisciplinary care teams in USA adopt therapies and protocols to prevent antibody-mediated rejection (AMR) following solid organ transplantation. AMR prevention supports long-term graft survival in kidney, heart, lung, and liver transplant recipients. Adoption aligns with high transplant volumes, expansive immunosuppressive regimens, and structured surveillance programs across tertiary care hospitals and transplant networks.
How do transplant activity levels and clinical practice standards drive demand in USA?
USA performs large numbers of organ transplants annually, with kidney and heart transplants representing significant procedural volumes. Clinicians implement pre-transplant immunological risk assessment including donor-specific antibody screening to inform prophylactic strategies. Use of targeted immunosuppressants, plasmapheresis, intravenous immunoglobulin, and complement inhibitors reflects a shift toward personalized AMR prevention protocols. Transplant centers incorporate routine monitoring of antibody profiles and early intervention pathways to reduce acute and chronic rejection risk. Multidisciplinary teams integrate surgical care, nephrology, and immunology to optimize prevention and long-term graft function. Evidence-based guidelines from transplant societies influence regimen selection and standardization across programs.
How do reimbursement factors, therapy costs, and clinical evidence requirements influence demand stability in USA?
Immunosuppressive and biologic therapies used for AMR prevention involve high cost, prompting careful consideration by transplant programs and payers. Medicare coverage for end-stage organ failure and post-transplant care influences therapy utilization patterns. Clinical evidence demonstrating superiority of specific regimens affects formulary decisions and institutional protocols. Variation in patient immunological risk profiles requires tailored prevention approaches, adding complexity to protocol selection. Specialist training and familiarity with AMR strategies influence adoption rates. Demand remains strong in high-volume transplant centers where graft survival outcomes justify investment, while broader access depends on cost-effectiveness data, payer alignment, and continued refinement of prevention standards within USA transplant practice.
How Is Demand for Antibody-mediated Rejection Prevention in the USA Evolving by Region?
Demand for antibody-mediated rejection prevention in the USA is rising due to increasing solid organ transplants, improved diagnostic sensitivity, and protocol-driven immunology management. West USA leads with a 17.5% CAGR, supported by high transplant volumes and advanced desensitization programs. South USA follows at 15.7%, driven by expanding transplant centers and population growth. Northeast USA records a 14.0% CAGR, shaped by academic transplant programs and evidence-based immunosuppression strategies. Midwest USA posts 12.2%, reflecting integrated health systems and standardized post-transplant care. Regional variation reflects transplant density, access to specialized immunology services, and adoption of antibody monitoring protocols across the USA.
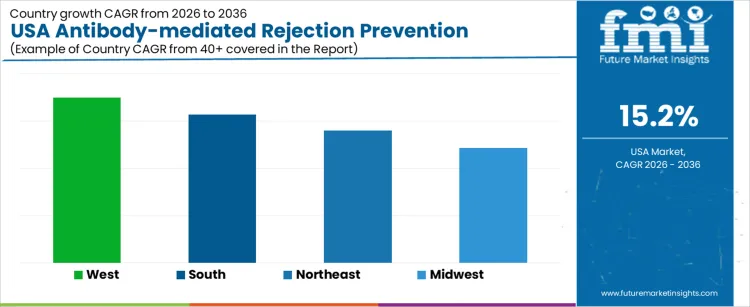
| Region | CAGR (2026 to 2036) |
|---|---|
| West USA | 17.5% |
| South USA | 15.7% |
| Northeast USA | 14.0% |
| Midwest USA | 12.2% |
What clinical and transplant ecosystem factors drive growth in West USA?
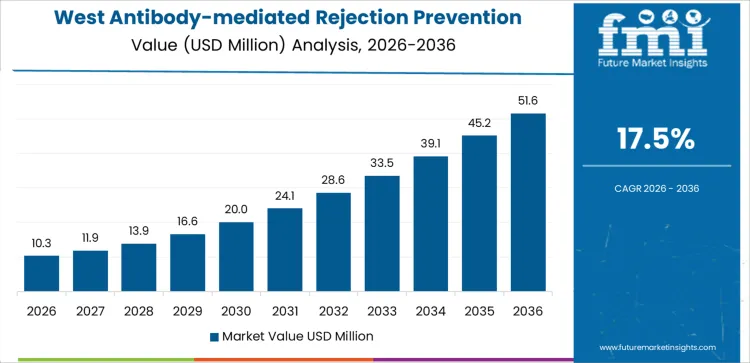
West USA drives demand through concentration of high-volume transplant centers, immunology expertise, and early adoption of advanced rejection prevention protocols. Region’s CAGR of 17.5% reflects extensive use of therapies targeting donor-specific antibodies following kidney, heart, and lung transplants. Centers emphasize early detection through sensitive antibody assays and proactive intervention strategies. Multidisciplinary transplant teams integrate plasmapheresis, immunomodulatory agents, and close monitoring into care pathways. Strong clinical trial participation accelerates adoption of emerging prevention approaches. Demand favors evidence-supported regimens with predictable outcomes. Growth remains protocol-driven and innovation-aligned, supported by complex case management and referral inflows from surrounding states.
- High transplant center concentration
- Early adoption of antibody monitoring
- Multidisciplinary rejection prevention protocols
- Strong clinical trial participation
Why is antibody-mediated rejection prevention expanding rapidly in South USA?
South USA demand is shaped by rising transplant procedure volumes, demographic expansion, and improved access to specialty care. Region’s CAGR of 15.7% reflects increased focus on preventing post-transplant complications in kidney and liver recipients. Health systems expand transplant programs and standardize immunosuppression pathways to manage growing patient cohorts. Clinicians prioritize therapies that reduce readmissions and graft loss risk. Regional centers invest in laboratory capabilities for antibody detection and longitudinal monitoring. Demand growth remains volume-driven, aligned with expanding transplant infrastructure and emphasis on long-term graft survival across diverse patient populations.
- Growing transplant procedure volumes
- Expansion of regional transplant programs
- Focus on graft survival and readmission reduction
- Improved antibody testing infrastructure
How do academic transplant programs influence demand patterns in Northeast USA?
Northeast USA demand reflects leadership of academic medical centers and structured transplant research programs. Region’s CAGR of 14.0% is supported by evidence-based adoption of antibody-mediated rejection prevention strategies. Centers integrate detailed risk stratification, antibody profiling, and protocolized treatment escalation. Physicians emphasize long-term outcomes, safety monitoring, and guideline alignment. Clinical education and research inform consistent practice across affiliated hospitals. Demand favors therapies with strong clinical validation and reimbursement clarity. Growth aligns with protocol refinement and dissemination rather than rapid expansion of transplant volumes.
- Academic-led transplant care models
- Evidence-based protocol development
- Advanced antibody risk stratification
- Guideline-aligned treatment adoption
What system-level factors shape measured growth in Midwest USA?
Midwest USA demand is anchored in integrated health systems, centralized transplant services, and cost-managed care delivery. Region’s CAGR of 12.2% reflects steady adoption of antibody-mediated rejection prevention within standardized post-transplant pathways. Providers emphasize consistency, formulary management, and predictable clinical outcomes. Antibody monitoring is incorporated selectively based on patient risk profiles. Investment decisions align with long-term care planning rather than aggressive protocol expansion. Growth remains disciplined and system-driven, aligned with maintaining graft function and optimizing resource utilization across regional transplant networks.
- Integrated transplant service networks
- Standardized post-transplant protocols
- Selective antibody monitoring strategies
- Cost-managed and outcome-focused adoption
What is the competitive landscape of demand for antibody-mediated rejection prevention in the USA?
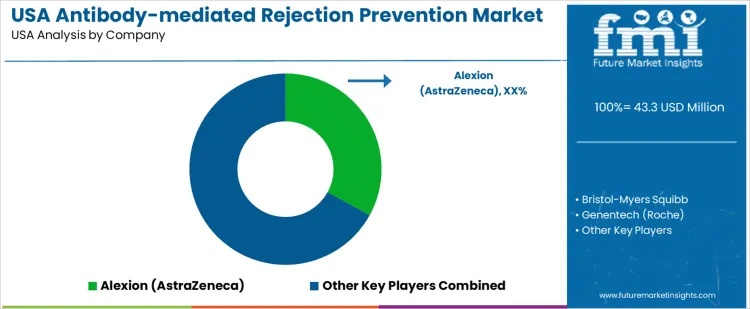
Demand for antibody-mediated rejection (AMR) prevention in the USA is driven by the volume of organ transplants, particularly kidney, heart, and lung procedures, and the need to maintain graft survival in sensitized patients. Clinical use focuses on immunomodulatory therapies, desensitization protocols, and biologic agents that target donor-specific antibodies and complement pathways. Buyers evaluate clinical efficacy, safety profiles, dosing regimens, and compatibility with existing immunosuppressive protocols. Procurement teams prioritize therapies with established clinical evidence, FDA approval for transplant indications or strong off-label support documented in USA practice guidelines, and reliable supply chains capable of supporting hospital pharmacy systems. Trend in the USA market reflects increased emphasis on personalized immunosuppression, combined biologic regimens, and preventative strategies in high-risk transplant populations.
Alexion, part of AstraZeneca, holds a leading position in the USA with complement-targeting therapies used to manage antibody-mediated injury and PAM risk, supported by clinical trial data and broad hospital formulary inclusion. Bristol-Myers Squibb participates through calcineurin inhibitors and adjunctive biologic agents that modulate immune response in transplant protocols. Genentech (Roche) contributes monoclonal antibody therapies that influence B-cell activity and antibody production used in desensitization and rejection prevention strategies. Novartis supports demand with mTOR inhibitors and biologics integrated into transplant immunosuppression regimens favored by USA transplant centers. Sanofi Genzyme maintains visibility with immunomodulatory agents deployed in select AMR prevention approaches. Competitive positioning in the USA reflects regulatory alignment, clinical evidence strength, physician familiarity, and integration with comprehensive transplant care teams.
Key Players in USA Antibody-mediated Rejection Prevention Demand
- Alexion (AstraZeneca)
- Bristol-Myers Squibb
- Genentech (Roche)
- Novartis
- Sanofi Genzyme
Scope of the Report
| Items | Details |
|---|---|
| Quantitative Units | USD million |
| Treatment | Monoclonal Antibodies; Plasmapheresis; Immunomodulators; Cysteine Protease Inhibitors |
| End User | Hospitals; Specialty Clinics |
| Regions Covered | West USA; South USA; Northeast USA; Midwest USA |
| Key Companies Profiled | Alexion (AstraZeneca); Bristol-Myers Squibb; Genentech (Roche); Novartis; Sanofi Genzyme |
| Additional Attributes | Demand is driven by rising solid organ transplant volumes, improved graft survival protocols, and wider adoption of targeted biologics. Hospitals dominate usage due to transplant infrastructure. Growth depends on clinical outcomes, immunosuppression guidelines, pricing of biologics, and availability of specialized transplant care centers across regions. |
USA Antibody-mediated Rejection Prevention Demand by Segments
Treatment:
- Monoclonal Antibodies
- Plasmapheresis
- Immunomodulators
- Cysteine Protease Inhibitors
End User:
- Hospitals
- Specialty Clinics
Region:
- West USA
- South USA
- Northeast USA
- Midwest USA
Bibliography
- United States Department of Health and Human Services. (2023). Organ transplant immunosuppression guidelines including antibody-mediated rejection prevention protocols. HHS Clinical Practice Guidance Publications.
- American Society of Transplantation. (2023). Consensus recommendations on diagnosis, prevention, and management of antibody-mediated rejection in organ transplantation. AST Clinical Practice Guidelines.
- National Kidney Foundation. (2023). Kidney transplant care and rejection prevention standards including antibody-mediated protocols. NKF Clinical Practice Publications.
- Centers for Medicare & Medicaid Services. (2023). Coverage and quality measures for transplant immunosuppressive therapy and antibody-mediated rejection prophylaxis. CMS Regulatory and Policy Publications.
Frequently Asked Questions
How big is the demand for antibody-mediated rejection prevention in USA in 2026?
The demand for antibody-mediated rejection prevention in USA is estimated to be valued at USD 43.3 million in 2026.
What will be the size of antibody-mediated rejection prevention in USA in 2036?
The market size for the antibody-mediated rejection prevention in USA is projected to reach USD 178.9 million by 2036.
How much will be the demand for antibody-mediated rejection prevention in USA growth between 2026 and 2036?
The demand for antibody-mediated rejection prevention in USA is expected to grow at a 15.2% CAGR between 2026 and 2036.
What are the key product types in the antibody-mediated rejection prevention in USA?
The key product types in antibody-mediated rejection prevention in USA are monoclonal antibodies, plasmapheresis, immunomodulators and cysteine protease inhibitors.
Which end user segment is expected to contribute significant share in the antibody-mediated rejection prevention in USA in 2026?
In terms of end user, hospitals segment is expected to command 68.0% share in the antibody-mediated rejection prevention in USA in 2026.
Table of Content
- Executive Summary
- USA Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- USA Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- USA Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- USA Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Treatment
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Treatment , 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Treatment , 2025 to 2035
- Monoclonal Antibodies
- Plasmapheresis
- Immunomodulators
- Cysteine Protease Inhibitors
- Monoclonal Antibodies
- Y to o to Y Growth Trend Analysis By Treatment , 2020 to 2024
- Absolute $ Opportunity Analysis By Treatment , 2025 to 2035
- USA Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2025 to 2035
- Hospitals
- Specialty Clinics
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2020 to 2024
- Absolute $ Opportunity Analysis By End User, 2025 to 2035
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Treatment
- By End User
- Competition Analysis
- Competition Deep Dive
- Alexion (AstraZeneca)
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Bristol-Myers Squibb
- Genentech (Roche)
- Novartis
- Sanofi Genzyme
- Alexion (AstraZeneca)
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: USA Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: USA Market Value (USD Million) Forecast by Prodct , 2021 to 2036
- Table 3: USA Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 4: USA Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: USA Market Value (USD Million) Forecast by Prodct , 2021 to 2036
- Table 6: USA Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: USA Market Pricing Analysis
- Figure 2: USA Market Value (USD Million) Forecast 2021-2036
- Figure 3: USA Market Value Share and BPS Analysis by Prodct , 2026 and 2036
- Figure 4: USA Market Y-o-Y Growth Comparison by Prodct , 2026-2036
- Figure 5: USA Market Attractiveness Analysis by Prodct
- Figure 6: USA Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 7: USA Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 8: USA Market Attractiveness Analysis by End User
- Figure 9: USA Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: USA Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: USA Market Attractiveness Analysis by Region
- Figure 12: USA Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: USA Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 14: USA Market Value Share and BPS Analysis by Prodct , 2026 and 2036
- Figure 15: USA Market Y-o-Y Growth Comparison by Prodct , 2026-2036
- Figure 16: USA Market Attractiveness Analysis by Prodct
- Figure 17: USA Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 18: USA Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 19: USA Market Attractiveness Analysis by End User
- Figure 20: USA Market - Tier Structure Analysis
- Figure 21: USA Market - Company Share Analysis

