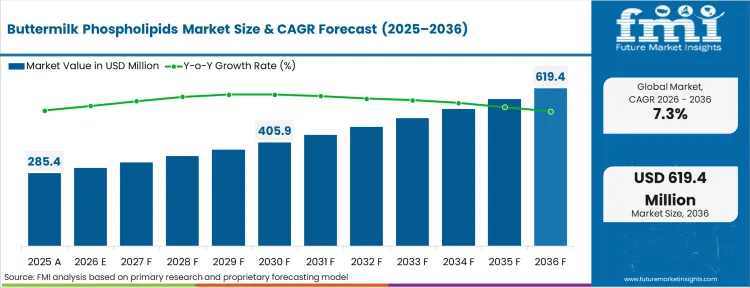
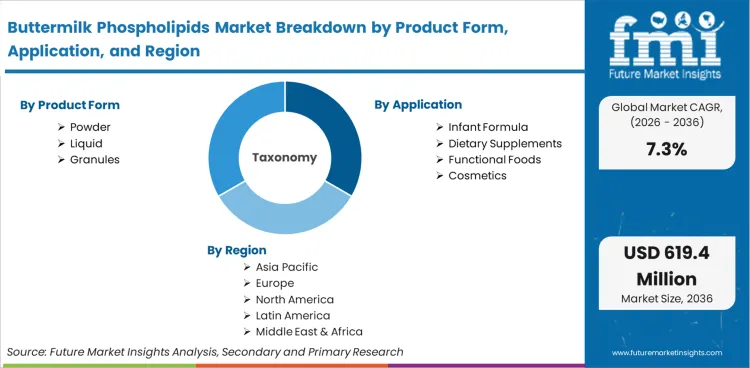
The buttermilk phospholipids market was valued at USD 285.4 million in 2025. The industry is expected to cross USD 306.2 million in 2026 at a CAGR of 7.3% during the forecast period. Continuous investment propels the valuation to USD 618.5 million through 2036 as early life nutrition formulators systematically replace plant-based lecithins with dairy-derived milk fat globule membrane fractions to achieve strict biomimicry with human breast milk profiles.
Procurement directors at tier-1 pediatric nutrition brands are currently facing a critical supply chain calculation. They must decide whether to secure long-term material contracts now or risk being completely priced out of the active ingredient pool. Delaying this qualification means ceding premium label claims regarding cognitive development to competitors who have already locked in pediatric nutrition supply lines. The reality is that buying these phospholipids means tying ingredient security directly to the global butter churning output, creating a volatile price environment that penalizes spot buyers.
Before growth becomes entirely self-reinforcing, the industry must cross a specific analytical standardization gate. Once regulatory bodies finalize harmonized testing methods for identifying distinct sphingomyelin and phosphatidylcholine ratios, major brands will be able to substantiate on-pack cognitive claims without fearing regional compliance audits. Analytical equipment manufacturers and dairy cooperatives are actively collaborating to close this testing gap.
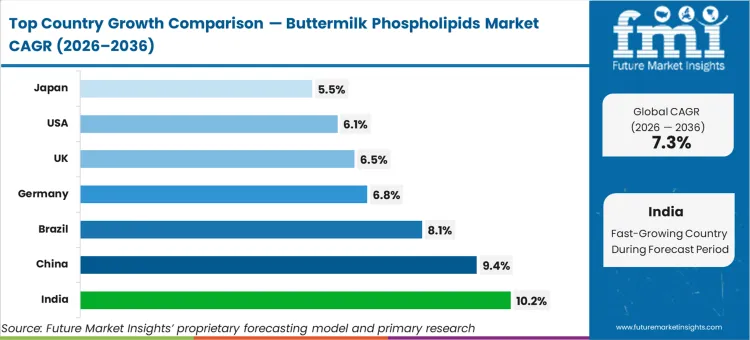
FMI expects India to expand at 10.2%, followed by China at 9.4%, Brazil at 8.1%, Germany at 6.8%, the United Kingdom at 6.5%, the United States at 6.1%, and Japan at 5.5%. The divergence across this range stems directly from the underlying consolidation of local dairy processing infrastructure, allowing emerging regions to finally capture and dry buttermilk streams that were historically lost to unorganized liquid milk distribution.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 306.2 million |
| Industry Value (2036) | USD 618.5 million |
| CAGR (2026-2036) | 7.3% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
The buttermilk phospholipids market encompasses the extraction, purification, and commercialization of complex polar lipid fractions derived specifically from the aqueous phase of butter churning. It is functionally distinct from general soy, sunflower, or egg lecithins because dairy phospholipids contain unique sphingolipids and a lipid membrane architecture that closely mirrors human physiological structures. This specific biological origin defines its value and its processing constraints.
This market includes all concentrated phospholipid ingredients sourced directly from sweet cream buttermilk, cultured buttermilk, or butter serum. It covers liquid concentrates, granulated extracts, and spray-dried powders utilized in commercial formulations. The scope integrates specialized fractions enriched for milk fat globule membrane components deployed specifically across the specialty food ingredients sector for targeted nutritional interventions.
The market explicitly excludes plant-derived lecithins, egg yolk phospholipids, and synthetic lipid structures. It also excludes raw, unconcentrated buttermilk intended for standard beverage consumption or basic baking applications. These exclusions are applied because unconcentrated buttermilk does not meet the functional dosage requirements for fortification, and non-dairy phospholipids cannot support the specific sphingolipid label claims that define this commercial category.
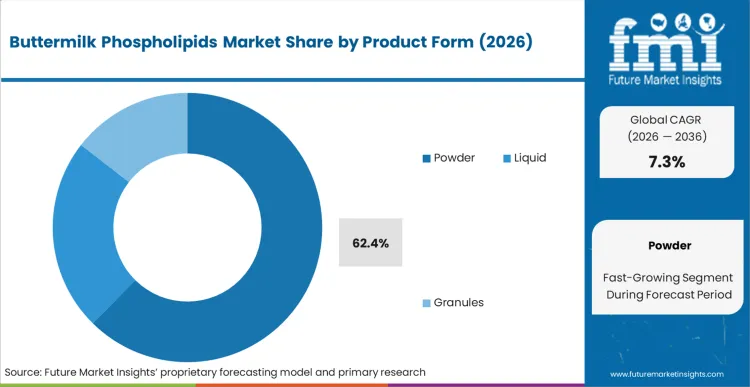
The reason powder holds a dominant 62.4% share is entirely rooted in lipid oxidation mechanics, not simply mixing convenience. According to FMI's estimates, preserving the delicate double bonds in milk-derived phospholipids requires stringent moisture control that liquid concentrates cannot economically maintain across global transit routes. Plant managers formulating premium infant nutrition require an ingredient that can survive two years of ambient shelf life without developing off-flavors that cause product rejection. Liquid formats mandate expensive cold-chain logistics and shorter production runs, eroding the margin profile for high-volume manufacturers. Brands that attempt to cut costs by utilizing liquid fractions in dry-blend facilities face catastrophic fat oxidation failures in the final consumer product.
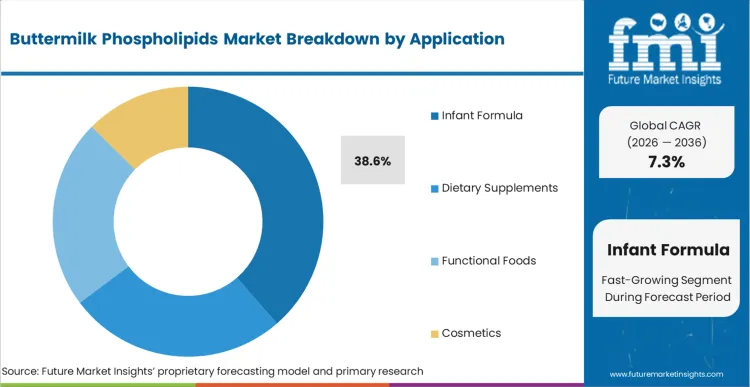
Pediatric nutrition brands are currently being forced to decide whether their next-generation products will feature standard plant lecithins or biologically accurate milk fat globule membranes. According to FMI's estimates, the infant formula holds a dominant 38.6% share. Infant formula dictates the market direction because it represents the only sector where regulatory bodies explicitly recognize the physiological necessity of specific dietary lipid structures for cognitive development. FMI analysts opine that formulators are aggressively rewriting recipes to include these dairy fractions, transitioning them from a premium niche additive to a baseline requirement for tier-1 product qualification. Brands relying entirely on legacy plant-based emulsifiers face an unbridgeable commercial gap when competing against products bearing clinically substantiated neurological development claims.
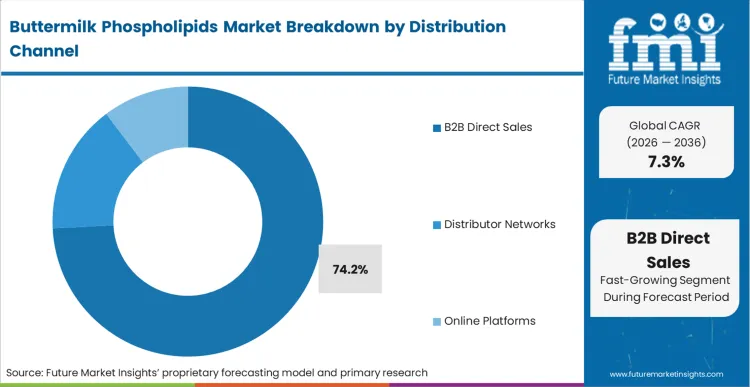
The commercial outcome of utilizing advanced dairy lipids forces formulators into highly collaborative relationships with primary extractors, explaining why B2B direct sales completely dominate the distribution architecture. These are not standardized commodity ingredients that can be purchased through fragmented broker networks based on a generic specification sheet. As per FMI's projection, the highly variable nature of raw buttermilk requires dairy processors to custom-calibrate their membrane filtration to meet the exact phospholipid ratios demanded by individual pharmaceutical and nutritional clients. According to FMI's estimates, the B2B direct sales holds a dominant 74.2% share. Relying on third-party distributors introduces unacceptable variability into the supply chain, severing the vital technical feedback loop between the extraction plant and the final product formulation laboratory.
The pressure to replicate human physiological profiles precisely requires early life nutrition brands to upgrade their lipid sourcing strategies. Plant-based lecithins fulfill the basic emulsification requirement but fail to deliver the distinct neurological building blocks found exclusively in mammalian milk fat. This forces formulation directors to actively seek out and qualify dairy-derived alternatives, regardless of the higher base cost. The commercial stakes of delaying this transition are severe, as parents and healthcare providers increasingly base purchasing decisions on the presence of advanced cognitive support ingredients, penalizing legacy formulations.
The primary operational obstacle restricting market expansion is the absolute physical limitation of raw material supply. Buttermilk is exclusively a by-product of butter churning, meaning the maximum theoretical yield of dairy phospholipids is permanently capped by global butter consumption rates. This friction cannot be solved by simply building more extraction facilities. Processors are currently attempting to mitigate this by developing specialized extraction techniques to pull polar lipids directly from broader whey streams, but these alternative methods currently yield lower concentrations and require significantly higher energy inputs to achieve commercial purity.
Opportunities in the Buttermilk Phospholipids Market
Based on the regional analysis, the Buttermilk Phospholipids market is segmented into North America, Latin America, Western Europe, Eastern Europe, Asia Pacific, and Middle East & Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 10.2% |
| China | 9.4% |
| Brazil | 8.1% |
| Germany | 6.8% |
| United Kingdom | 6.5% |
| United States | 6.1% |
| Japan | 5.5% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
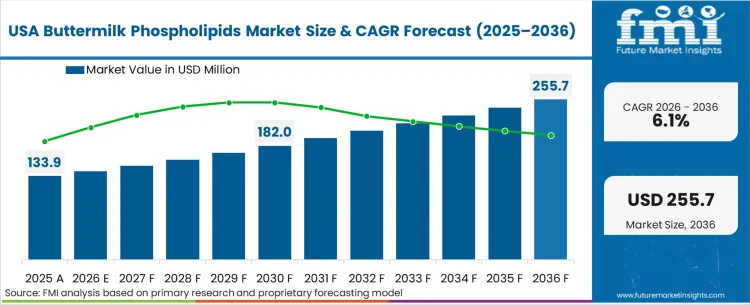
The regulatory framework surrounding pediatric nutrition specifically shapes how buyers operate in the North American landscape. The stringent GRAS notification process and FDA infant formula guidelines obligate manufacturers to produce overwhelming clinical evidence before integrating new lipid structures into commercial product lines. Based on FMI's assessment, this compliance burden slows the initial speed of formulation updates but creates a massive competitive moat for the specialized dairy processors who successfully navigate the qualification protocols. Procurement teams in this region prioritize unassailable quality documentation over raw material pricing, effectively locking out low-tier international suppliers who cannot maintain rigorous audit trails.
FMI's report includes extensive analysis of the Canadian and Mexican dairy extraction sectors. These adjacent markets are rapidly upgrading their membrane filtration capabilities to serve as secondary supply nodes for US-based pediatric brands seeking geographic supply chain redundancy.
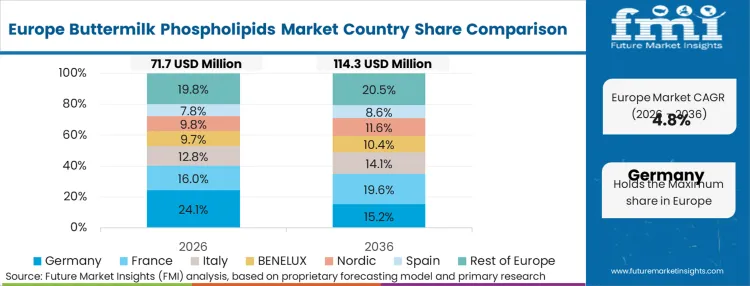
The density and maturity of Western Europe's cooperative dairy infrastructure completely define this region's operational advantage. Unlike markets that must build extraction facilities from scratch, European processors are leveraging decades of installed ultrafiltration capacity, adapting existing milk protein concentrate lines to isolate high-value polar lipids. In FMI's view, this infrastructure density allows European suppliers to dominate global export volumes for premium infant formula ingredients. Operations heads here focus entirely on optimizing yield from existing buttermilk streams rather than securing new raw milk sources.
FMI's report includes detailed tracking of specialized lipid extraction projects across France, Denmark, and the Netherlands. These nations represent the technical epicenter for dairy fractionation, consistently dictating the global baseline for ingredient purity and functional performance.
Cost structures and the availability of uncommitted capital define the adoption trajectory across the Asia Pacific region. Historic reliance on imported premium infant formula is driving massive domestic investments to replicate Western formulation standards utilizing local dairy resources. According to FMI's estimates, the ability to fund large-scale membrane filtration installations determines which local players can upgrade from generic milk powders to high-margin functional ingredients. Procurement practices here are intensely focused on securing technical partnerships with established Western equipment manufacturers to accelerate domestic production capabilities.
FMI's report includes analysis of the rapidly evolving dairy processing landscapes in Oceania and Southeast Asia. These sub-regions are strategically pivoting their export focus away from bulk commodity powders toward specialized functional lipid fractions.
The buttermilk phospholipids market operates under severe concentration because the barrier to entry extends far beyond basic capital expenditure. Establishing a viable commercial presence requires deep physical integration with massive butter churning operations, effectively locking out standalone ingredient manufacturers who lack direct access to raw dairy streams. Leading companies like Arla Foods Ingredients and Fonterra dominate because they control the entire custody chain, from the raw milk collection to the final membrane filtration. Buyers utilize this exact level of supply chain control as their primary variable for distinguishing qualified long-term partners from opportunistic spot vendors.
To challenge the established incumbents, new entrants must build sophisticated, multi-stage fractionation capabilities that can reliably separate specific sphingomyelin ratios without degrading the delicate lipid structures. Incumbents maintain their advantage because they have spent decades optimizing the thermal and mechanical parameters of their dairy separation equipment, a advantage that protects them from sudden price undercutting. A challenger cannot simply purchase standard filtration membranes: they must develop the proprietary engineering knowledge to handle highly viscous, lipid-rich dairy retentates without constantly fouling their processing lines.
Major pediatric brands actively resist this vendor concentration by aggressively funding clinical trials on alternative lipid sources and funding capacity expansions at tier-2 cooperative dairies. The tension between the buyers' need for diversified, low-cost supply and the dominant processors' incentive to limit capacity and maintain premium pricing will define the commercial landscape through 2036. FMI anticipates the market will remain highly consolidated at the extraction level, forcing buyers to secure volume through long-term strategic joint ventures rather than traditional transactional purchasing.
| Metric | Value |
|---|---|
| Quantitative Units | USD 306.2 million to USD 618.5 million, at a CAGR of 7.3% |
| Market Definition | The market covers concentrated polar lipid fractions extracted from butter processing byproducts. It focuses strictly on biologically active dairy components utilized for advanced nutritional and functional fortification. |
| Product Form Segmentation | Powder, Liquid, Granules |
| Application Segmentation | Infant Formula, Dietary Supplements, Functional Foods, Cosmetics |
| Distribution Channel Segmentation | B2B Direct Sales, Distributor Networks, Online Platforms |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, Asia Pacific, Middle East and Africa |
| Countries Covered | United States, Germany, China, Japan, United Kingdom, India, Brazil, and 40 plus countries |
| Key Companies Profiled | Arla Foods Ingredients Group P/S, Fonterra Co-operative Group Limited, Lactalis Ingredients, Valio Ltd., Royal FrieslandCampina N.V., Glanbia Nutritionals, Agropur Dairy Cooperative, Epi Ingredients |
| Forecast Period | 2026 to 2036 |
| Approach | Primary interviews targeted procurement heads at pediatric nutrition firms and dairy process engineers. The volume baseline anchored to global butter production ratios and ultrafiltration installation capacities. Forecasts were cross-validated against specialized infant formula import records. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the buttermilk phospholipids market in 2026?
The industry is expected to cross USD 306.2 million in 2026. This figure signals the baseline volume required to support the current transition of tier-1 pediatric nutrition brands moving away from plant-based lecithins toward accurate human milk biomimicry.
What will it be valued at by 2036?
Sustained investment propels the valuation to USD 618.5 million by 2036. This doubling reflects the aggressive scaling of membrane filtration infrastructure at cooperative dairies attempting to capture value from previously discarded buttermilk streams.
What CAGR is projected?
The market is set to expand at a 7.3% CAGR. This rate is strictly constrained by the global output of butter churning facilities, which effectively caps the maximum theoretical volume of extractable raw material regardless of downstream demand.
Which product form segment leads?
The powder segment dominates the market structure. Powdering locks the highly oxidative lipid fractions inside a protective matrix, allowing formulators to achieve the extended ambient shelf life required to survive global infant formula distribution networks.
Which Application segment leads?
Infant formula holds the primary volume share. Formulators in this sector use dairy phospholipids to deliver the precise sphingomyelin architecture required to substantiate premium on-pack cognitive development claims that soy alternatives cannot legally support.
Which Distribution Channel segment leads?
B2B direct sales completely dominate the supply architecture. The technical complexity of custom-calibrating lipid ratios requires a direct, unbroken feedback loop between the dairy processing engineer and the final pharmaceutical formulation laboratory.
What drives rapid growth?
The overwhelming regulatory and consumer push for precise physiological biomimicry in early life nutrition actively forces pediatric brands to abandon legacy plant emulsifiers in favor of biologically accurate milk fat globule membranes.
What is the primary restraint?
The physical reliance on butter manufacturing creates a rigid supply ceiling. Because these specific lipids are solely derived from the aqueous phase of churning, production volumes are inextricably tied to global milk fat demand rather than specialized nutritional requirements.
Which country grows fastest?
India expands at 10.2%, outpacing China's 9.4%. The rapid consolidation of India's historically fragmented dairy sector finally provides the centralized volume scale necessary to install commercial ultrafiltration equipment, whereas China is already modernizing its established infrastructure.
How do specific regional compliance frameworks shape the North American landscape?
The intense FDA GRAS notification process forces procurement teams to prioritize unassailable documentation trails over raw ingredient pricing. This regulatory burden creates a massive operational moat for established European cooperative dairies that already possess the required clinical validation archives.
Why is direct whey extraction critical to the trajectory of this market?
Processors are adapting membrane technology to pull polar lipids directly from cheese whey, attempting to decouple their premium ingredient output from stagnant global butter churning volumes and solve the industry's most severe supply bottleneck.
How do large pediatric brands resist vendor consolidation at the extraction level?
Global formula manufacturers actively combat the leverage held by dominant dairy cooperatives like Arla and Fonterra by funding independent clinical trials on alternative lipid sources and underwriting the capital expenditure required to install filtration membranes at tier-2 regional dairies.
Why do cosmetic formulators utilize these dairy lipids?
The inherent complexity of milk fat globule membranes provides powerful natural emulsification properties, allowing clean-label personal care brands to replace synthetic stabilizers while maintaining optimal topical texture.
What specific analytical challenge must the industry overcome?
Current growth is hindered by the lack of globally harmonized testing methods for identifying distinct sphingomyelin ratios. Standardizing these analytical protocols is the gate required for brands to confidently assert neurological claims without failing regional compliance audits.
Why does Germany act as the primary export hub for these ingredients?
Germany's highly optimized, high-volume butter production network generates massive, consistent pools of raw sweet cream buttermilk. Local process engineers focus entirely on extracting maximum commercial margin from this existing waste stream rather than sourcing new milk supplies.
How does the Japanese market diverge from the global application standard?
Unlike Western markets dominated by pediatric formulation, Japan's aging demographic forces buyers to funnel these premium lipids into specialized functional foods targeting adult cognitive preservation, indicating where the global market application balance will eventually shift.
What does the absolute volume dominance in B2B Direct Sales indicate about product standardization?
It confirms that these are highly customized active ingredients, not interchangeable commodities. Buyers refuse to source through fragmented broker networks because third-party distributors disrupt the chain-of-custody documentation required for stringent pediatric safety compliance.
Why cannot liquid phospholipid concentrates capture greater market share?
Liquid formats mandate expensive, continuous cold-chain logistics and shorter factory production runs. The margin erosion caused by preventing rapid lipid oxidation in liquid state makes the format commercially unviable for high-volume global formulators.
What role does the United Kingdom play in the market's expansion?
The UK provides the critical clinical validation engine for the industry. Its advanced pediatric research network continuously generates the peer-reviewed data that global medical marketing teams require to justify the massive price premiums of MFGM-enhanced formula.
Why do formulators face a penalty for delaying the transition to dairy lipids?
Brands that cling to legacy soy lecithins are being systematically removed from preferred pediatric healthcare recommendation lists. The penalty is not regulatory; it is a permanent loss of retail shelf positioning to competitors who can legally advertise cognitive and immune system biomimicry.
Why do formulators face a penalty for delaying the transition to dairy lipids?
Brands that cling to legacy soy lecithins are being systematically removed from preferred pediatric healthcare recommendation lists. The penalty is not regulatory; it is a permanent loss of retail shelf positioning to competitors who can legally advertise cognitive and immune system biomimicry.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.