Genomic Risk Stratification Testing Market
Genomic Risk Stratification Testing Market Size and Share Forecast Outlook 2026 to 2036
Genomic Risk Stratification Testing Market Forecast and Outlook 2026 to 2036
The genomic risk stratification testing market is likely to be valued at USD 4.2 billion in 2026 and reach USD 12.1 billion by 2036, at a CAGR of 11.2%. Growth reflects the shift of genomics from confirmatory diagnostics toward predictive decision support, where risk scores influence how aggressively clinicians intervene and how closely patients are monitored.
Testing demand is closely tied to clinical scenarios involving uncertainty, such as determining recurrence risk in early-stage cancer, identifying high-risk cardiovascular profiles beyond conventional factors, or prioritizing surveillance in inherited disease settings. Hospitals and diagnostic laboratories increasingly treat genomic risk scores as inputs to care planning rather than supplemental data, which anchors testing volumes to treatment pathways and long-term management strategies instead of episodic diagnostic events.
Quick Stats for Genomic Risk Stratification Testing Market
- Genomic Risk Stratification Testing Market Value (2026): USD 4.2 billion
- Genomic Risk Stratification Testing Market Forecast Value (2036): USD 12.1 billion
- Genomic Risk Stratification Testing Market Forecast CAGR 2026 to 2036: 11.2%
- Leading Test Kit Type by Utilization: NGS-based tests
- Fastest-Growing Countries: India, China, Brazil, USA, Germany
- Top Players in Global Demand: Illumina, Roche, Thermo Fisher Scientific, Qiagen, Agilent Technologies

Adoption patterns are shaped by integration depth within clinical workflows rather than simple availability of sequencing technology. Laboratories prioritize assays that translate complex variant data into standardized risk outputs usable by non-genetic specialists, reducing interpretation friction at the point of care. Health systems deploy risk stratification selectively where evidence demonstrates impact on outcomes or resource allocation, reinforcing demand in oncology, cardiology, and specialty clinics before broader population use.
Centralized sequencing models, coupled with automated bioinformatics pipelines, support scale while maintaining analytical consistency across sites. Reimbursement and guideline alignment further stabilize utilization by embedding genomic risk assessment into protocol-driven decision making. Market sustainability rests on the dependence of treatment intensity, surveillance frequency, and preventive strategies on validated genomic risk outputs, positioning these tests as structural components of modern clinical management rather than discretionary genomic add-ons.
Genomic Risk Stratification Testing Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 4.2 billion |
| Market Forecast Value (2036) | USD 12.1 billion |
| Forecast CAGR 2026 to 2036 | 11.2% |
Why is the Demand for Genomic Risk Stratification Testing Growing?
Demand for genomic risk stratification testing is increasing as clinicians and healthcare systems adopt genomic insights to refine prediction of disease progression and personalize prevention and treatment strategies. These tests analyze panels of genetic variants associated with elevated risk for conditions such as cancer, cardiovascular disease, and metabolic disorders, enabling providers to tailor screening intensity, therapeutic aggressiveness, and follow-up schedules based on quantified risk.
Oncology specialists use risk scores to inform decisions about adjuvant therapy and surveillance intervals, while cardiology teams integrate polygenic risk assessments with traditional risk calculators to identify individuals who may benefit from early intervention. Diagnostic laboratories and clinical genetic services invest in next generation sequencing, bioinformatics pipelines, and data interpretation tools to deliver consistent and clinically actionable results, and procurement teams evaluate test validity, clinical utility metrics, and integration with electronic health records to support workflow cohesion.
Growth in preventive care models and value-oriented reimbursement frameworks reinforces uptake of risk stratification testing because early identification of high-risk individuals can reduce downstream costs associated with advanced disease management. Population health programs incorporate genomic risk scores into risk adjustment and stratification strategies to allocate resources efficiently and measure outcomes across cohorts. Research initiatives expanding the catalog of validated risk loci and real-world evidence linking scores to clinical endpoints encourage broader adoption in primary and specialty care settings. Laboratory accreditation, standardized reporting formats, and clinician education initiatives further support reliable interpretation and use, contributing to sustained demand growth in the genomic risk stratification testing market.
How Is the Genomic Risk Stratification Testing Market Segmented?
Demand for genomic risk stratification testing is shaped by precision medicine adoption, preventive care emphasis, and expanding use of genomics in clinical decision-making. The sector is segmented by test kit type, application, and end user. Healthcare systems increasingly rely on genetic risk insights to guide therapy selection, screening intensity, and long-term disease management. Adoption reflects declining sequencing costs, improved bioinformatics, and integration of genomic data into care pathways. Test selection considers analytical depth, clinical validity, and compatibility with laboratory workflows. Segmentation illustrates how testing technology, clinical purpose, and service delivery setting influence utilization patterns and purchasing priorities.
Which Test Kit Type Accounts for the Highest Utilization in Genomic Risk Stratification?
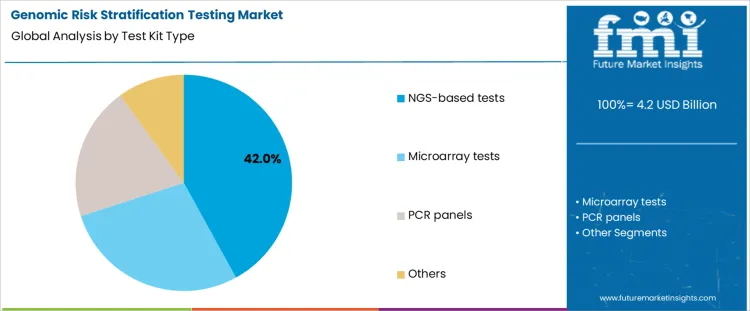
NGS-based tests register 42.0%, driven by their ability to analyze multiple genes and variants within a single assay. These tests support comprehensive risk profiling across oncology, cardiovascular, and inherited conditions. High data throughput enables detection of rare variants with clinical relevance. Microarray tests at 28.0% remain relevant for established panels requiring consistent coverage and lower computational demand. PCR panels at 20.0% address focused risk assessment where predefined markers are sufficient. Other test formats at 10.0% serve niche or emerging applications. Test kit segmentation reflects preference for platforms balancing breadth, depth, and scalability in clinical genomics.
Key Points
- NGS enables multi-gene analysis within a single workflow.
- Microarrays support standardized, cost-controlled testing.
- PCR panels address targeted risk assessment needs.
How Do Clinical Objectives Shape Application-Level Testing Demand?

Oncology risk profiling leads application demand at 46.0%, reflecting reliance on genomic insights for cancer predisposition and treatment planning. Genetic stratification supports early intervention strategies and personalized surveillance programs. Cardiovascular risk at 28.0% focuses on identifying inherited predispositions influencing prevention and lifestyle modification. Inherited disease risk at 26.0% addresses carrier screening and family-based risk assessment. Application segmentation highlights stronger utilization where genomic data directly informs clinical decisions, long-term monitoring strategies, and patient counseling pathways.
Key Points
- Oncology applications prioritize predictive and preventive insights.
- Cardiovascular testing supports proactive risk management.
- Inherited disease testing informs family-level decision-making.
Which End User Setting Drives Genomic Risk Testing Deployment?

Diagnostic laboratories account for 44.0% of testing deployment due to centralized sequencing infrastructure and bioinformatics capability. High sample throughput supports efficient processing and standardized reporting. Hospitals at 36.0% integrate testing into clinical pathways for diagnosis and treatment planning. Direct-to-provider services at 20.0% facilitate access for clinicians seeking genomic insights without in-house laboratory capacity. End-user segmentation reflects concentration where technical expertise, data interpretation capability, and integration with care delivery systems determine testing utilization.
Key Points
- Laboratories provide centralized sequencing and analysis capacity.
- Hospitals embed testing within clinical workflows.
- Provider-directed services expand access to genomic insights.
What are the Key Dynamics in the Genomic Risk Stratification Testing Market?
Demand for genomic risk stratification testing reflects clinical need to predict disease progression, recurrence probability, and treatment response using genomic signatures. Adoption concentrates in oncology, cardiology, and inherited disease management across hospital laboratories and specialized diagnostic providers. Global scope aligns with precision medicine integration into routine care pathways. Usage centers on multigene assays and algorithm-driven scores that support individualized risk assessment beyond traditional clinical factors.
How do clinical decision uncertainty and personalization requirements shape adoption?
Risk stratification testing addresses situations where standard clinical indicators fail to clearly guide treatment intensity or surveillance strategy. Demand increases when clinicians must distinguish patients who benefit from aggressive intervention from those suitable for conservative management. Oncology applications use genomic scores to assess recurrence risk and chemotherapy benefit, reducing overtreatment exposure. Cardiovascular and inherited disease settings apply stratification to identify patients requiring early intervention. Test value depends on validated correlation between genomic markers and long-term outcomes. Integration into guidelines and care pathways accelerates adoption. Turnaround time and interpretability influence clinician confidence. Adoption reflects reliance on genomics to resolve decision ambiguity in high-stakes, preference-sensitive treatment scenarios.
How do evidence thresholds, reimbursement alignment, and data integration limits influence market scalability?
Genomic risk tests require robust clinical validation linking scores to actionable outcomes. Demand sensitivity rises where evidence remains population-specific or limited to retrospective data. Reimbursement decisions depend on demonstrated cost avoidance or outcome improvement, delaying coverage expansion. Data integration into electronic health records adds workflow complexity. Interpretation requires clinician education and genetic counseling support. Assay cost affects uptake in early-stage or screening contexts. Regulatory oversight varies by test classification and region. Rapid evolution of genomic knowledge necessitates frequent assay updates. Scalability remains constrained by proof requirements, payer acceptance, and infrastructure readiness for genomics-driven decision support.
How Is Demand for Genomic Risk Stratification Testing Evolving Globally?
Demand for genomic risk stratification testing is expanding globally as precision medicine reshapes clinical decision-making across oncology, cardiology, and inherited disorders. Clinicians increasingly rely on genomic scores to guide therapy intensity, surveillance frequency, and preventive interventions. Reimbursement alignment and guideline inclusion support routine adoption. Laboratory automation and bioinformatics capacity enable higher testing throughput. Growth rates in India at 12.6%, China at 12.4%, Brazil at 12.0%, USA at 10.8%, and Germany at 10.7% indicate sustained expansion driven by protocol-mandated risk assessment, therapy stratification requirements, and health system emphasis on outcome optimization rather than exploratory genetic testing.
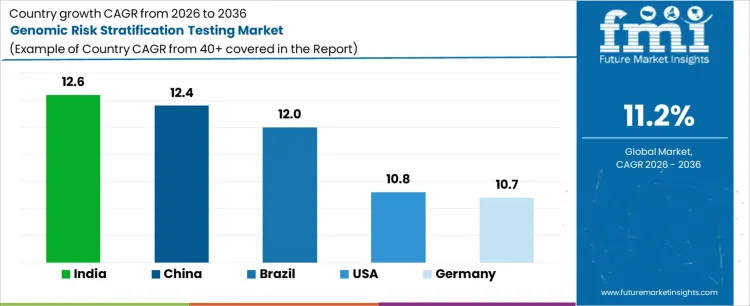
| Country | CAGR (%) |
|---|---|
| India | 12.6% |
| China | 12.4% |
| Brazil | 12.0% |
| USA | 10.8% |
| Germany | 10.7% |
What Is Driving Rapid Expansion of Genomic Risk Stratification Demand in India?
Genomic risk stratification testing demand in India is growing at a CAGR of 12.6%, reflecting rapid integration into oncology and cardiology care pathways. Private hospitals expand in-house genomic panels to reduce turnaround times. Rising use of targeted therapies increases dependence on validated risk scores. Medical insurers increasingly recognize genomic testing for treatment planning. Domestic laboratory networks improve affordability and geographic reach. Growth reflects therapy-linked diagnostic necessity and infrastructure scaling rather than population-wide preventive screening adoption.
- Expansion of in-house genomic testing at private hospitals
- Therapy-driven reliance on validated risk scores
- Gradual insurance recognition for clinical genomics
- Improved affordability through domestic lab networks
Why Is China Experiencing Strong Uptake of Genomic Risk Stratification Testing?
Genomic risk stratification demand in China is expanding at a CAGR of 12.4%, supported by standardized precision medicine initiatives. Large tertiary hospitals integrate genomic scoring into routine care protocols. Public reimbursement inclusion for selected tests improves access. Centralized sequencing facilities enable high-throughput analysis. Alignment with domestic drug development reinforces testing relevance. Growth reflects protocolized adoption at scale rather than discretionary genetic exploration.
- Precision medicine initiatives embedding genomic scoring
- Reimbursement support for selected risk tests
- Centralized high-throughput sequencing infrastructure
- Alignment with domestic therapeutic pipelines
How Is Brazil Sustaining Growth in Genomic Risk Stratification Testing?
Genomic risk stratification testing demand in Brazil is growing at a CAGR of 12.0%, influenced by expansion of comprehensive care models. Oncology and cardiology centers adopt genomic scores to optimize therapy selection. Increased access to advanced treatments elevates need for risk-based decision tools. Laboratory partnerships extend testing beyond metropolitan hubs. Legal pathways occasionally broaden access to advanced diagnostics. Growth reflects institutional adoption and therapy access expansion rather than broad preventive testing programs.
- Comprehensive care models adopting genomic scoring
- Advanced therapy access increasing risk-based decisions
- Laboratory partnerships extending regional reach
- Legal mechanisms supporting diagnostic access
What Factors Are Driving Genomic Risk Stratification Growth in United States?
Genomic risk stratification testing demand in the United States is expanding at a CAGR of 10.8%, driven by guideline-endorsed precision care. Broad payer coverage supports use in oncology and cardiovascular risk assessment. High penetration of next-generation sequencing enables multi-gene scoring. Clinical evidence continues to refine risk algorithms. Integrated electronic records support test utilization. Growth reflects standardized adoption and evidence accumulation rather than increased testing frequency per patient.
- Guideline endorsement across major indications
- Broad payer coverage for genomic risk tests
- High availability of NGS platforms
- Integration with electronic clinical workflows
Why Is Germany Experiencing Sustained Expansion in Genomic Risk Stratification Testing?
Genomic risk stratification demand in Germany is growing at a CAGR of 10.7%, shaped by evidence-based medicine frameworks. University hospitals and certified centers apply genomic scoring to guide therapy intensity. Reimbursement pathways support validated tests with clinical utility. Central laboratory networks ensure analytical consistency. Cost-effectiveness assessments influence adoption scope. Growth remains sustained, reflecting structured integration and quality governance rather than rapid decentralization of genomic services.
- Evidence-based application in certified centers
- Reimbursement for clinically validated tests
- Central laboratory quality assurance networks
- Cost-effectiveness guiding adoption boundaries
What is the competitive landscape of demand for the genomic risk stratification testing market globally?
Demand for genomic risk stratification testing arises from precision treatment planning, population screening expansion, and payer emphasis on outcome-linked decision support. Clinical teams evaluate predictive validity, gene panel relevance, analytical sensitivity, reproducibility across laboratories, and correlation with therapeutic response. Laboratories consider sample requirements, turnaround reliability, automation compatibility, data interpretation clarity, and secure reporting within clinical workflows. Procurement behavior reflects long-term reagent agreements, platform standardization, regulatory clearance status, and vendor support for validation studies. Trend in the genomic risk stratification testing market shows movement toward multigene signatures guiding treatment intensity and surveillance frequency.
Illumina holds leading positioning through sequencing platforms enabling high-throughput risk modeling and standardized pipelines across research and clinical settings. Roche supports demand via clinically validated assays and integration with diagnostic ecosystems used in oncology and hereditary disease programs. Thermo Fisher Scientific competes by offering scalable instruments, flexible content options, and informatics suited for decentralized laboratory networks. Qiagen maintains relevance with sample-to-insight solutions emphasizing workflow simplicity and consistent performance across varied specimen types. Competitive differentiation centers on clinical evidence depth, data interoperability, regulatory reach, and trusted collaboration with healthcare systems.
Key Players in the Genomic Risk Stratification Testing Market
- Illumina
- Roche
- Thermo Fisher Scientific
- Qiagen
- Agilent Technologies
- Others
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Test Kit Type | NGS-based tests; Microarray tests; PCR panels; Others |
| Application | Oncology risk profiling; Cardiovascular risk; Inherited disease risk |
| End User | Diagnostic laboratories; Hospital; Direct-to-provider services |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, China, Brazil, USA, Germany, and 40+ countries |
| Key Companies Profiled | Illumina; Roche; Thermo Fisher Scientific; Qiagen; Agilent Technologies; Others |
| Additional Attributes | Dollar sales by test kit type, application, end user, and sales channel; analytical depth and variant detection accuracy across NGS, microarray, and PCR platforms; clinical utility in oncology, cardiovascular, and inherited disease risk stratification; turnaround time and data interpretation workflows in laboratory and hospital settings; regulatory oversight and data privacy considerations; procurement dynamics shaped by centralized diagnostics, hospital adoption, and provider-integrated testing services. |
Genomic Risk Stratification Testing Market by Segment
Test Kit Type:
- NGS-based tests
- Microarray tests
- PCR panels
- Others
Application:
- Oncology risk profiling
- Cardiovascular risk
- Inherited disease risk
End User:
- Diagnostic laboratories
- Hospital
- Direct-to-provider services
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- European Society of Cardiology. (2024). Cardiovascular disease prevention in clinical practice: Incorporating genetic and genomic risk assessment. European Heart Journal.
- European Society for Medical Oncology. (2024). ESMO recommendations on the clinical use of genomic assays for risk stratification and treatment decision-making in oncology. Annals of Oncology.
- Organisation for Economic Co-operation and Development. (2024). Genomic medicine and risk stratification in value-based healthcare systems. OECD Publishing.
- U.S. Food and Drug Administration, Center for Devices and Radiological Health. (2024). Clinical validation and regulatory considerations for genomic tests used in risk stratification. U.S. Food and Drug Administration.
- World Health Organization. (2024). Genomics and public health: Applications of genetic risk stratification. World Health Organization.
Frequently Asked Questions
How big is the genomic risk stratification testing market in 2026?
The global genomic risk stratification testing market is estimated to be valued at USD 4.2 billion in 2026.
What will be the size of genomic risk stratification testing market in 2036?
The market size for the genomic risk stratification testing market is projected to reach USD 12.1 billion by 2036.
How much will be the genomic risk stratification testing market growth between 2026 and 2036?
The genomic risk stratification testing market is expected to grow at a 11.2% CAGR between 2026 and 2036.
What are the key product types in the genomic risk stratification testing market?
The key product types in genomic risk stratification testing market are ngs-based tests, microarray tests, pcr panels and others.
Which application segment to contribute significant share in the genomic risk stratification testing market in 2026?
In terms of application, oncology risk profiling segment to command 46.0% share in the genomic risk stratification testing market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Test Kit Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Test Kit Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Test Kit Type , 2026 to 2036
- NGS-based tests
- Microarray tests
- PCR panels
- Others
- NGS-based tests
- Y to o to Y Growth Trend Analysis By Test Kit Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Test Kit Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Oncology risk profiling
- Cardiovascular risk
- Inherited disease risk
- Oncology risk profiling
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Diagnostic laboratories
- Hospital
- Direct-to-provider services
- Diagnostic laboratories
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Test Kit Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Kit Type
- By Application
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Test Kit Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Kit Type
- By Application
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Test Kit Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Kit Type
- By Application
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Test Kit Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Kit Type
- By Application
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Test Kit Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Kit Type
- By Application
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Test Kit Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Kit Type
- By Application
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Test Kit Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Kit Type
- By Application
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Kit Type
- By Application
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Kit Type
- By Application
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Kit Type
- By Application
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Kit Type
- By Application
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Kit Type
- By Application
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Kit Type
- By Application
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Kit Type
- By Application
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Kit Type
- By Application
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Kit Type
- By Application
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Kit Type
- By Application
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Kit Type
- By Application
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Kit Type
- By Application
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Kit Type
- By Application
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Kit Type
- By Application
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Kit Type
- By Application
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Kit Type
- By Application
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Kit Type
- By Application
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Kit Type
- By Application
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Kit Type
- By Application
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Kit Type
- By Application
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Kit Type
- By Application
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Test Kit Type
- By Application
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Test Kit Type
- By Application
- By End User
- Competition Analysis
- Competition Deep Dive
- Illumina
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Roche
- Thermo Fisher Scientific
- Qiagen
- Agilent Technologies
- Others
- Illumina
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Test Kit Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Test Kit Type , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Test Kit Type , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Test Kit Type , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Test Kit Type , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Test Kit Type , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Test Kit Type , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Test Kit Type , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Test Kit Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Test Kit Type , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Test Kit Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Test Kit Type , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Test Kit Type , 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Test Kit Type
- Figure 26: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Application
- Figure 29: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by End User
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Test Kit Type , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Test Kit Type , 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Test Kit Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Application
- Figure 39: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by End User
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Test Kit Type , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Test Kit Type , 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Test Kit Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Application
- Figure 49: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by End User
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Test Kit Type , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Test Kit Type , 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Test Kit Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Application
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End User
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Test Kit Type , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Test Kit Type , 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Test Kit Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Application
- Figure 69: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by End User
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Test Kit Type , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Test Kit Type , 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Test Kit Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Test Kit Type , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Test Kit Type , 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Test Kit Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

