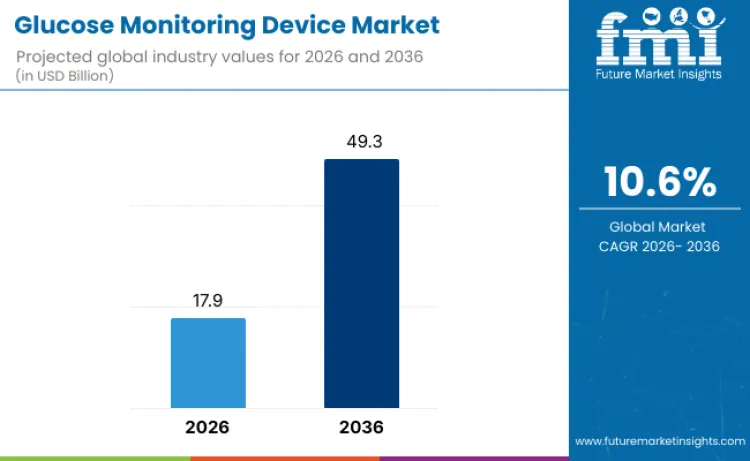
Glucose monitoring device industry valuation is set to reach USD 17.9 billion in 2026, driven by escalating diabetes prevalence worldwide and growing emphasis on continuous blood glucose management. As per FMI's projection, revenue from the sales of glucose monitoring devices is anticipated to expand at a robust CAGR of 10.6% through 2036, pushing total sector worth beyond USD 49.3 billion by the conclusion of this assessment period. Rising geriatric population demographics and advancing sensor technology integration act as primary catalysts for this financial trajectory.
Medical device manufacturers are channeling substantial investment into innovative monitoring technologies, aiming to validate accuracy performance through real-time glucose tracking protocols without compromising patient comfort standards. Healthcare providers are shifting focus from traditional fingerstick testing methods to continuous glucose monitoring systems, redefining diabetes management practices in clinical and home care environments. Such behavioral changes encourage medical technology companies to introduce comprehensive glucose monitoring portfolios utilizing advanced biosensor technologies.
FMI is of the opinion that growing awareness of diabetes complications and preventive care requirements are significantly influencing industry expansion. Endocrinologists emphasize evidence-based monitoring approaches, utilizing specific sensor configurations and data analytics to create validated glycemic control experiences. Research investigating the clinical benefits of continuous glucose monitoring is creating demand for standardized measurement protocols, instilling confidence among healthcare professionals regarding future treatment specifications.
| Metric | Value |
|---|---|
| Industry Size (2026) | USD 17.9 Billion |
| Industry Value (2036) | USD 49.3 Billion |
| CAGR (2026-2036) | 10.6% |
Source: Future Market Insights’ proprietary forecasting model and primary research
Shift toward patient-centric diabetes management is serving as a significant catalyst for broader acceptance of portable blood glucose testing systems. Recent breakthroughs in glucometer technology have complemented self-care protocols by preserving measurement accuracy and user convenience characteristics effectively. This empowerment approach appeals to diabetic patients seeking evidence-based glucose control protection. Research success of smartphone-connected glucometers in home care settings has expanded application possibilities among diverse patient lifestyle requirements. Education programs highlighting the role of self-monitoring in glycemic awareness are driving diabetes educator professional recommendations. Healthcare companies are capitalizing on these trends by introducing specialized self-testing variants for insulin-dependent diabetes management. FMI is of the opinion that integrating cloud-based data storage into comprehensive diabetes care strategies is becoming standard practice, ensuring optimal glucose tracking without compromising data accessibility.
Global landscape is intricately segmented to address diverse requirements of the diabetes care industry, categorizing sector by product type, indication, end-user setting, and technology platform. Structural division allows stakeholders to identify specific monitoring targets, such as rising demand for continuous glucose monitoring in hospital settings or preference for self-monitoring devices in home care applications. By analyzing these segments, manufacturers can tailor device development to meet distinct needs of Type 1 diabetes patients versus Type 2 diabetes management requirements. Segmentation also highlights shift from generic monitoring approaches to targeted indication-specific interventions. As industry matures, granular segmentation becomes essential for understanding patient pathways and clinical specifications, ensuring device development aligns with evolving diabetes care guidelines.
Self-monitoring devices account for 58.2% of product share in 2026, driven by extensive patient adoption supporting convenience and affordability benefits. Leadership position is underpinned by proven usability characteristics from diabetic patients recognizing ease-of-use claims. Segment benefits from continuous design improvements, such as smaller blood sample requirements enhancing patient comfort specifications. Research focused on glucometer accuracy is renewing interest among healthcare providers. Critical role in managing daily glucose tracking further solidifies standing, as these devices enable effective diabetes control under various lifestyle conditions. As diabetes guidelines prioritize patient empowerment strategies, demand for user-friendly monitoring concentrates is expected to remain robust.
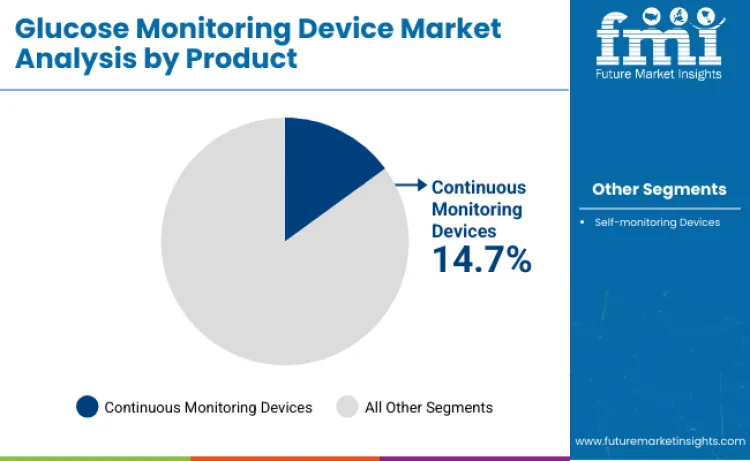
Continuous monitoring devices command 14.7% growth rate of sector in 2026, reflecting healthcare preference for real-time glucose data delivery. Growth acceleration is linked to superior clinical insights compared to periodic testing alternatives. Healthcare professionals rely on specialized sensor technologies to ensure effective trend analysis and glycemic pattern recognition. These systems are essential for managing insulin pump therapy and intensive diabetes management, driving priority status among endocrinology brands. Relentless pursuit of glycemic optimization ensures products designed for high-risk diabetes conditions remain innovation choice. High accuracy acceptance necessitates advanced sensor capacity, bolstering sustained revenue streams for continuous monitoring suppliers.
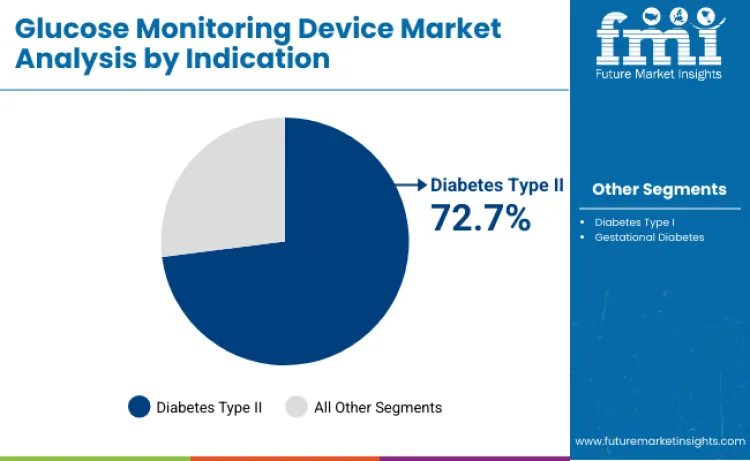
Diabetes Type II applications capture 72.7% of market share in 2026, owing to widespread disease prevalence requiring glucose monitoring intervention. Dominance stems from clinical guidelines recommending regular glucose testing alongside lifestyle modification methods for optimal diabetes management. Patients prefer these devices for their disease control benefits and complication prevention optimization. Usage in Type 2 diabetes management drives continuous volume growth among primary care providers. Compatibility with various treatment protocols allows for integrated solutions across different diabetes severity categories. Manufacturers focusing on Type 2-specific monitoring algorithms and lifestyle management formulations for these high-prevalence applications secure significant loyalty among primary care practices.
Diabetes care transition toward intelligent monitoring systems is set to reshape sales landscapes by demanding AI-enhanced glucose tracking formulations. As per FMI's estimates, machine learning trends evolve, manufacturers access technologies with enhanced pattern recognition for complex glycemic conditions. Shift toward predictive analytics creates demand for multi-algorithm formulations addressing multiple diabetes management requirements simultaneously. Manufacturers failing to position devices as AI-compatible risk losing visibility among tech-savvy healthcare providers. Availability of automated insulin adjustment options for closed-loop systems drives complementary consumption patterns. Rise of digital diabetes management specifications stimulates innovation, with companies differentiating offerings through algorithmic prediction integration.
Healthcare digitization adoption is revolutionizing glucose monitoring device utilization by enabling comprehensive remote patient management protocols. Integration with telehealth platforms allows real-time monitoring of glucose patterns, triggering automated clinical recommendations based on individual patient conditions. Medical consultations are driving specialized remote monitoring applications, with clinicians prescribing specific device formulations through telemedicine platforms. Digital health channels are expanding rapidly, offering remote support and competitive monitoring services compared to traditional in-clinic methods. FMI is of the opinion that mobile health applications featuring glucose tracking and virtual consultation create sustained engagement with diabetes management programs.
Evolving diabetes care frameworks are creating both opportunities and challenges for glucose monitoring device producers. Stricter reimbursement standards demand higher clinical evidence investments while improving patient confidence in device efficacy. Insurance coverage validation approvals from healthcare payers enable companies to market specific monitoring benefits, differentiating products in competitive markets. International harmonization of diabetes care standards facilitates global market expansion for established brands. Clinical outcome requirements are increasing, forcing manufacturers to invest in real-world evidence studies validating device effectiveness claims.
Global landscape for diabetes management technology is characterized by diverse healthcare frameworks, influenced by reimbursement systems and clinical practice standards. Established markets prioritize evidence-based specifications and regulatory-approved devices, whereas emerging economies focus on accessible solutions and basic glucose monitoring protection. Asia-Pacific is emerging as rapid growth hub due to increasing diabetes prevalence and expanding healthcare technology awareness. Conversely, North American sectors pivot towards advanced monitoring systems and personalized diabetes protocols. Government initiatives in developing nations supporting diabetes prevention ensure sustained demand for proven monitoring solutions across all patient categories.
| Country | CAGR (2026 to 2036) |
|---|---|
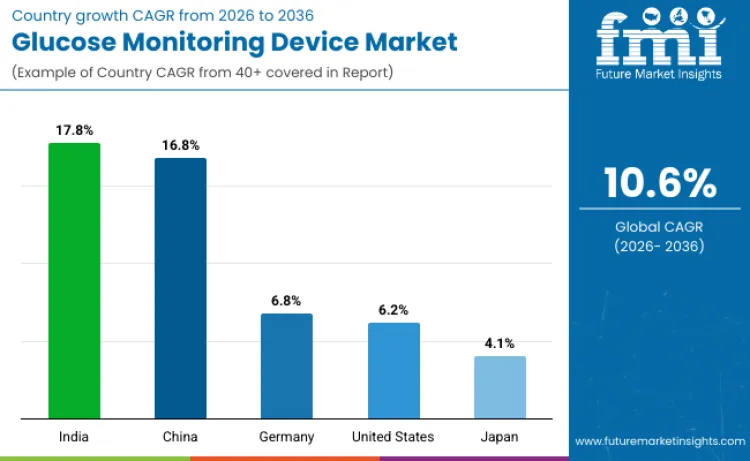
| United States | 6.2% |
| China | 16.8% |
| Japan | 4.1% |
| Germany | 6.8% |
| India | 17.8% |
Source: Future Market Insights analysis, supported by a proprietary forecasting model and primary research
Sales of diabetes monitoring products in United States are set to grow at 6.2% CAGR through 2036. Expansion is underpinned by established healthcare infrastructure and increasing continuous glucose monitoring integration requirements among diabetes care providers. Growth is fueled by Medicare coverage reforms emphasizing diabetes prevention and monitoring optimization management. Rising diabetes awareness investment drives specialized device adoption per patient among evidence-focused healthcare systems. As per FMI's projection, presence of major medical device companies establishing advanced sensor capabilities streamlines innovation cycles and reduces regulatory approval costs. Emphasis on traditional diabetes care integration with modern monitoring technologies continues to be primary driver, maintaining United States position as critical innovation engine for diabetes management solutions.
Demand for diabetes technology solutions in China is projected to rise at 16.8% CAGR during assessment period. Initiatives emphasizing healthcare modernization and diabetes awareness foster favorable environment for advanced monitoring platforms. Increasing investment in medical device manufacturing creates massive distribution channels for glucose monitoring products. Rising diabetic population requiring effective blood glucose management options prompts domestic brands to develop cost-effective formulations. Shift is supported by government policies encouraging medical device innovation, accelerating uptake of Chinese monitoring brands across healthcare facilities and fueling sector expansion through improved affordability.
Glucose monitoring device industry in Japan is poised to expand at 4.1% CAGR. Growth is largely attributed to increasing awareness of diabetes complications among aging populations. Government focus on preventive healthcare initiatives addressing chronic disease challenges encourages patients to adopt monitoring measures. FMI is of the opinion that increasing partnerships between local healthcare providers and international device suppliers ensure availability of high-quality monitoring systems. Economic stability combined with growing healthcare technology investment attracts significant resources to advanced diabetes care products. Strong regulatory framework ensuring device safety and clinical validation further drives patient confidence in monitoring protocols.
Sales of diabetes monitoring systems in Germany are set to grow at 6.8% CAGR. Expansion is underpinned by comprehensive healthcare system recognizing diabetes prevention benefits. Growth is fueled by increasing healthcare costs driving patients toward self-monitoring solutions. Widespread adoption of digital health standards and automated glucose tracking creates steady demand for validated diabetes management. High standards of clinical evidence and regulatory oversight prompt brands to invest in evidence-based formulations. Emphasis on patient empowerment and technology innovation continues to be primary driver, maintaining Germany position as quality leader in diabetes monitoring applications.
Revenue from diabetes monitoring products in India is set to grow at 17.8% CAGR. Expansion is underpinned by rapidly growing diabetic population recognition of monitoring benefits. Growth is fueled by increasing awareness of diabetes complications prevention needs. Emerging adoption of affordable monitoring technologies and healthcare accessibility drives revenue generation in diabetes care segments. Presence of established healthcare institutions ensures growing quality product demand and clinical validation. As per FMI's estimates, emphasis on preventive healthcare and technology adoption continues to be primary driver, maintaining India position as high-growth market for accessible diabetes monitoring systems.
Market participants are increasingly focusing on accuracy validation to differentiate their diabetes monitoring device formulations. Leading players like Abbott Laboratories and Dexcom Inc. are directing investment toward research partnerships with clinical institutions for device efficacy studies. Significant trend involves developing specialized sensor platforms to customize glucose monitoring based on individual patient glycemic profiles. Companies actively engage in strategic partnerships with healthcare providers to integrate monitoring devices into diabetes care protocols.
Innovation emerges as competitive frontier, with providers exploring advanced sensor systems for enhanced monitoring reliability. Consolidation activities remain moderate as specialty medical device companies acquire sensor technology suppliers to control quality and costs against established healthcare technology giants. Abbott Laboratories holds a significant share, leveraging its global diabetes care network and continuous monitoring investments.
| Items | Values |
|---|---|
| Quantitative Units | USD Billion |
| Product Segments | Continuous Monitoring Devices; Self-monitoring Devices |
| Indication Categories | Diabetes Type I; Diabetes Type II; Gestational Diabetes |
| End-User Categories | Hospitals; Clinics; Home Care Settings |
| Technology Categories | Invasive; Non-invasive; Minimally Invasive |
| Regional Coverage | North America, Latin America, East Asia, South Asia & Pacific, Western Europe, Eastern Europe, Middle East & Africa |
| Key Countries | United States, China, Japan, Germany, India |
| Key Companies Profiled | Abbott Laboratories; Dexcom Inc.; F. Hoffmann-La Roche Ltd; Medtronic; LifeScan IP Holdings LLC; Ascensia Diabetes Care Holdings AG; ARKRAY Inc.; ACON Laboratories Inc.; NIPRO; Nova Biomedical |
| Additional Attributes | Dollar sales measured for glucose monitoring devices used in diabetes management and blood glucose tracking applications, specified by device type (continuous monitoring, self-monitoring), sensor technology, measurement method preference, end-use focus (hospitals, home care, clinics), indication target (Type 1, Type 2, gestational diabetes), and compliance alignment with evolving clinical standards and diabetes care regulations. |
How big is the glucose monitoring device market in 2026?
The glucose monitoring device industry is valued at USD 17.9 billion in 2026.
What will be the size of the glucose monitoring device market in 2036?
The glucose monitoring device market is projected to reach USD 49.3 billion by 2036.
How much will the glucose monitoring device market grow between 2026 and 2036?
The market is expected to expand at a CAGR of 10.6% during the 2026 to 2036 forecast period.
Which product segment leads the glucose monitoring device market?
Self-monitoring devices lead the product category, accounting for 58.2% share in 2026.
Which indication segment holds the dominant share in the glucose monitoring device market?
Diabetes Type II dominates the indication landscape with a 72.7% share in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.