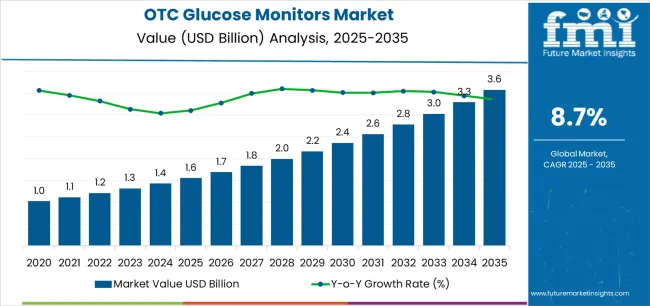
The otc glucose monitors market was valued at USD 1.55 billion in 2025, projected to reach USD 1.68 billion in 2026, and is forecast to expand to USD 3.88 billion by 2036 at a 8.7% CAGR. Based on FMI's analysis, FMI analysts observe that the OTC glucose monitor market is undergoing a structural expansion beyond its traditional diabetic patient base into the general wellness and metabolic health consumer segment. The FDA clearance of over-the-counter continuous glucose monitors, beginning with Abbott's Lingo and Dexcom's Stelo in 2024, has removed the prescription barrier that previously limited CGM access to diagnosed diabetics.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 1.68 Billion |
| Industry Value (2036) | USD 3.88 Billion |
| CAGR (2026-2036) | 8.7% |
Source: Future Market Insights, 2026
The core transition is the shift from episodic fingerstick testing to continuous sensor-based monitoring, which now accounts for 55.0% of OTC glucose monitor sales by value. CGM sensors generate 288 data points per day compared to the 2 to 4 measurements typical of fingerstick testing, enabling users and their healthcare providers to identify glycemic patterns that episodic testing cannot detect. The sensor replacement cycle of 10 to 14 days creates a recurring consumable revenue stream that is 4x to 6x the annual consumable spend of traditional meter users. As per FMI, country-level growth trajectories reflect distinct market structures. China leads at 11.7% CAGR, India registers at 10.9% CAGR, Germany registers at 10.0% CAGR, USA registers at 8.3% CAGR, UK registers at 7.4% CAGR, Japan registers at 6.5% CAGR.
OTC glucose monitors are over-the-counter blood glucose and interstitial glucose monitoring devices that consumers can purchase without a prescription for self-management of diabetes or general metabolic health tracking. The category includes traditional fingerstick blood glucose meters, continuous glucose monitors (CGMs) with adhesive sensor patches, and smartphone-connected biosensors. CGMs use subcutaneous sensors to measure interstitial glucose levels every 1 to 5 minutes, transmitting data to mobile applications that display trends, alerts, and historical patterns.
Market scope covers OTC glucose monitoring devices and associated consumables (test strips, sensors, lancets) sold globally through retail pharmacies, online channels, and direct-to-consumer platforms. The report includes regional and country-level market sizes, forecast values from 2026 to 2036, segmentation by product, indication, technology, and distribution channel, along with competitive landscape and regulatory pathway analysis.
The scope excludes prescription-only continuous glucose monitors that require physician ordering, hospital-grade point-of-care glucose analyzers, laboratory hemoglobin A1c testing equipment, and insulin delivery devices such as pumps and pens. Non-invasive glucose monitoring prototypes that have not received regulatory clearance for OTC sale are also omitted.
The OTC glucose monitors market grows by enabling diabetes patients to achieve superior glycemic control and lifestyle flexibility while reducing dependence on invasive fingerstick testing. Diabetes patients face mounting pressure to maintain tight glucose management preventing complications, with continuous glucose monitoring typically providing 10-15 additional glucose readings per hour compared to traditional testing methods, making real-time monitoring essential for optimal diabetes management outcomes. The patient empowerment movement's need for accessible self-monitoring tools creates demand for OTC glucose monitor solutions that can deliver actionable insights, enable proactive interventions, and ensure consistent tracking without prescription requirements across diverse care settings.
Government initiatives promoting diabetes awareness and preventive healthcare drive adoption in Type I diabetes, Type II diabetes, and gestational diabetes applications, where early detection and continuous management have direct impact on long-term health outcomes and healthcare cost reduction. The global shift toward digital health integration and telemedicine accelerates OTC glucose monitor demand as patients seek connected devices enabling remote monitoring and virtual care coordination. Affordability barriers for sensor-based systems and limited insurance coverage for OTC purchases may limit adoption rates among uninsured populations and regions with constrained healthcare budgets.
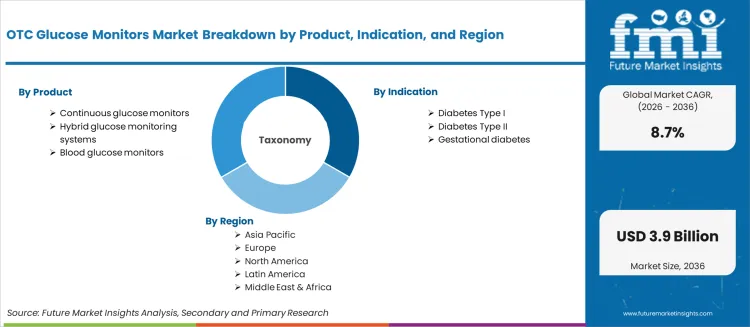
The market is segmented by product, indication, technology, distribution channel, and region. By product, the market is divided into continuous glucose monitors, hybrid glucose monitoring systems, and blood glucose monitors. Based on indication, the market is categorized into diabetes Type I, diabetes Type II, and gestational diabetes. By technology, the market includes sensor-based glucose monitoring, strip-based glucose monitoring, and hybrid technology. Based on distribution channel, the market encompasses drug stores, online pharmacies, retail pharmacies, specialty stores, and hypermarkets & supermarkets. Regionally, the market is divided into Asia Pacific, Europe, North America, Latin America, and Middle East & Africa.
The continuous glucose monitors segment represents the dominant force in the OTC glucose monitors market, capturing approximately 55.0% of total market share in 2026. This advanced category encompasses devices featuring subcutaneous sensors with real-time glucose tracking capabilities, delivering comprehensive trend analysis with predictive alerts for hypoglycemia and hyperglycemia prevention. The Continuous glucose monitors segment's market leadership stems from its exceptional convenience eliminating fingerstick testing, superior data density providing 288+ readings daily, and compatibility with smartphone applications enabling remote monitoring and data sharing with healthcare providers.
The Hybrid glucose monitoring systems segment maintains a substantial 25.0% market share, serving patients requiring combination approaches through integrated CGM-pump systems for automated insulin delivery in Type I diabetes management and individuals seeking flexible monitoring with periodic calibration verification. The Blood glucose monitors segment accounts for 20.0% market share, featuring traditional fingerstick meters for episodic testing and budget-conscious users requiring basic glucose measurements without continuous tracking capabilities.
Key advantages driving the Continuous glucose monitors segment include:
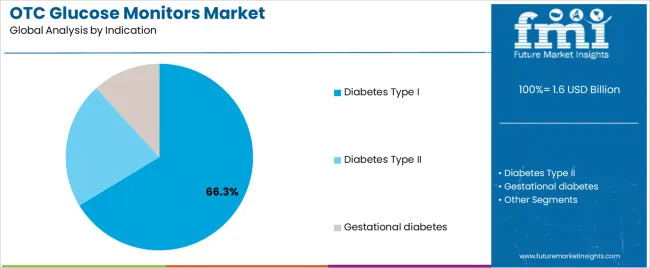
Diabetes Type I applications dominate the OTC glucose monitors market with approximately 66.3% market share in 2026, reflecting the critical importance of intensive glucose monitoring in insulin-dependent patients requiring multiple daily insulin adjustments. The Diabetes Type I segment's market leadership is reinforced by clinical guidelines recommending continuous monitoring for all Type I patients, higher technology adoption rates among younger patient populations, and insurance coverage supporting CGM access for autoimmune diabetes management.
The Diabetes Type II segment represents 21.9% market share through applications including insulin-requiring Type II diabetes, newly diagnosed patients establishing glycemic patterns, and individuals optimizing oral medication regimens requiring periodic glucose assessment. The Gestational diabetes segment accounts for 11.8% market share, featuring temporary monitoring during pregnancy for women developing glucose intolerance requiring dietary intervention and fetal protection.
Key market dynamics supporting indication preferences include:
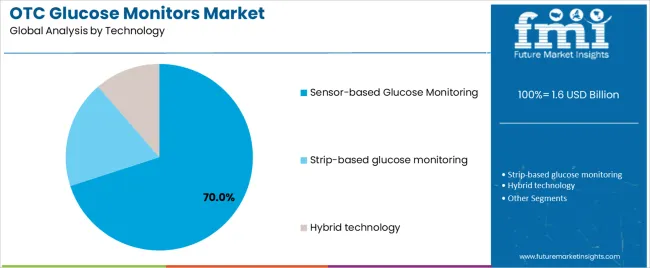
Sensor-based glucose monitoring dominates the OTC glucose monitors market with approximately 70.0% market share in 2026, reflecting the technological shift toward continuous electrochemical sensors providing interstitial glucose measurements. The Sensor-based segment's market leadership is reinforced by accuracy improvements achieving MARD (Mean Absolute Relative Difference) values below 10%, extended sensor wear times reaching 10-14 days, and factory calibration eliminating fingerstick calibration requirements.
The Strip-based glucose monitoring segment represents 20.0% market share through traditional blood glucose meters utilizing enzymatic test strips for capillary blood sampling. The Hybrid technology segment accounts for 10.0% market share, featuring systems combining continuous sensing with periodic strip-based calibration verification.
Key market dynamics supporting technology preferences include:
The market is driven by three concrete demand factors tied to diabetes prevalence and technology advancement. First, global diabetes epidemic creates increasing monitoring device requirements, with IDF projecting 783 million adults with diabetes by 2045 requiring daily glucose assessment, and OTC availability expanding access beyond prescription-dependent healthcare systems enabling self-directed management for 60-70% of patients previously underserved by traditional medical channels. Second, continuous glucose monitoring clinical evidence accelerates adoption, with landmark studies demonstrating HbA1c reductions of 0.5-1.0% and 40% decrease in hypoglycemic events driving patient demand and physician recommendations despite prescription requirements creating market spillover into OTC channels. Third, digital health ecosystem integration enhances value propositions, with smartphone connectivity, cloud-based analytics, and telehealth platform compatibility creating comprehensive diabetes management solutions worth 30-50% price premiums over standalone monitoring devices.
Market restraints include high upfront and ongoing costs affecting accessibility, particularly for continuous glucose monitors where device costs of USD 1,000-3,000 annually plus sensor replacement expenses of USD 200-400 monthly create affordability barriers for uninsured patients and those in developing markets where out-of-pocket healthcare spending constraints limit adoption. Reimbursement limitations for OTC devices pose adoption challenges, as insurance coverage typically requires prescriptions and medical necessity documentation, creating administrative burdens and reducing OTC channel attractiveness compared to prescription pathways offering partial or complete coverage. Accuracy concerns in OTC devices without medical supervision create safety considerations, as patients self-interpreting glucose data without healthcare provider guidance may make inappropriate treatment decisions, particularly during sensor warm-up periods or when physiological conditions affect measurement accuracy.
Key trends indicate accelerated adoption of factory-calibrated CGM systems, with elimination of fingerstick calibration requirements improving user experience and reducing compliance barriers previously limiting continuous monitoring acceptance. Technology advancement trends toward extended sensor wear times reaching 14-180 days reduce replacement frequency and system costs, with implantable sensors offering 90-180 day longevity transforming OTC value propositions through reduced ongoing expenses. The market thesis could face disruption if non-invasive glucose monitoring technologies achieve clinical accuracy standards enabling reliable measurement without skin penetration, eliminating sensor replacement costs and skin irritation concerns, or if regulatory authorities impose prescription requirements on current OTC devices due to safety concerns about unsupervised diabetes management reducing market addressability.
.webp)
| Country | CAGR (2026-2036) |
|---|---|
| China | 11.7% |
| India | 10.9% |
| Germany | 10.0% |
| USA | 8.3% |
| UK | 7.4% |
| Japan | 6.5% |
Source: FMI analysis based on primary research and proprietary forecasting model
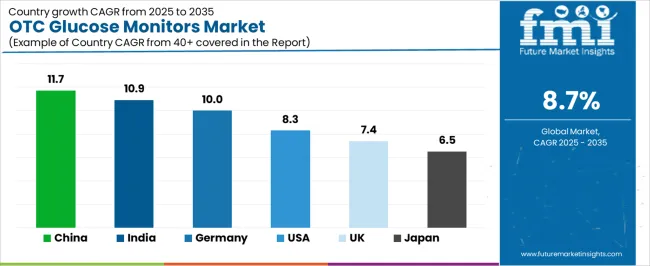
The OTC glucose monitors market is gaining momentum worldwide, with China taking the lead due to massive diabetes population growth and digital health infrastructure development programs. Close behind, India benefits from rising diabetes prevalence and increasing healthcare awareness, positioning itself as a strategic growth hub in the Asia-Pacific region. Germany shows strong advancement, where universal healthcare coverage and diabetes management excellence strengthen its role in European continuous monitoring adoption. The USA demonstrates robust growth through technology innovation leadership and direct-to-consumer marketing expansion, signaling continued investment in accessible diabetes management solutions. The UK stands out for its NHS diabetes prevention programs and patient empowerment initiatives, while Japan continues to record steady progress driven by aging population health management and quality-focused medical device preferences.
The report covers an in-depth analysis of 40+ countries, with top-performing countries are highlighted below.
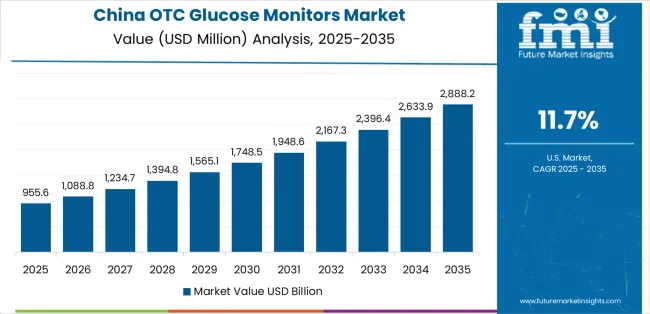
China demonstrates the strongest growth potential in the OTC Glucose Monitors Market with a CAGR of 11.7% through 2036. The country's leadership position stems from comprehensive diabetes epidemic response programs, intensive digital health infrastructure development, and aggressive chronic disease management targets driving adoption of self-monitoring technologies. Growth is concentrated in major urban centers, including Beijing, Shanghai, Guangzhou, and Shenzhen, where rising middle-class populations, increasing health awareness, and established e-commerce pharmacy infrastructure are facilitating OTC glucose monitor accessibility for Type I diabetes patients, newly diagnosed Type II patients, and health-conscious consumers seeking preventive monitoring. Distribution channels through online pharmacies, retail pharmacy chains, and specialty diabetes care stores expand deployment across metropolitan areas and tier-2 cities with growing diabetes populations. The country's Healthy China 2030 initiative provides policy support for chronic disease prevention, including diabetes screening programs and digital health technology integration.
Key market factors:
In major metropolitan areas, tier-1 cities, and urban centers, the adoption of OTC glucose monitoring systems is accelerating across newly diagnosed diabetics, gestational diabetes patients, and health-conscious individuals, driven by rising diabetes awareness and pharmacy chain expansion. The market demonstrates strong growth momentum with a CAGR of 10.9% through 2036, linked to comprehensive diabetes epidemic emergence and increasing focus on preventive healthcare solutions. Indian consumers are implementing self-monitoring programs and digital health tracking systems to improve glycemic control while managing healthcare costs across private healthcare utilization and out-of-pocket medication management. The country's expanding pharmacy infrastructure creates sustained demand for accessible monitoring devices, while growing health insurance penetration drives adoption of preventive health technologies.
Advanced healthcare system in Germany demonstrates sophisticated implementation of OTC glucose monitoring programs, with documented case studies showing integration with statutory health insurance coverage achieving 70-80% CGM adoption among Type I diabetes patients through prescription pathways complemented by OTC purchases for backup devices. The country's diabetes care infrastructure in major population centers, including Berlin, Munich, Hamburg, and Frankfurt, showcases integration of continuous monitoring technologies with existing diabetes management systems, leveraging expertise in medical device regulation and patient education. German patients emphasize technology reliability and data accuracy, creating demand for clinically validated monitoring systems that support intensive diabetes management protocols and integrate with electronic health records. The market maintains strong growth through focus on digital health integration and preventive care, with a CAGR of 10.0% through 2036.
Key development areas:
The USA market leads in glucose monitoring technology innovation based on robust R&D investment, expedited FDA approval pathways, and direct-to-consumer marketing enabling OTC accessibility. The country shows solid potential with a CAGR of 8.3% through 2036, driven by diabetes prevalence affecting 37+ million Americans and increasing emphasis on patient-directed care across major population centers, including California, Texas, Florida, and New York. American consumers are adopting OTC glucose monitors for diabetes management independence, real-time dietary feedback, and wellness tracking applications, particularly among Type I diabetes patients requiring backup devices and Type II diabetics seeking lifestyle optimization tools beyond prescribed monitoring requirements. Distribution channels through retail pharmacies, online health stores, and direct manufacturer sales expand coverage across diverse consumer segments.
Leading market segments:
The UK's OTC glucose monitors market demonstrates mature implementation focused on complementing NHS provision with consumer-directed purchases, with documented integration in Type I diabetes management achieving comprehensive CGM coverage through prescription pathways supplemented by OTC sensor purchases during supply interruptions. The country maintains steady growth momentum with a CAGR of 7.4% through 2036, driven by NHS diabetes prevention programs and patient empowerment initiatives supporting self-monitoring adoption. Major population regions, including London, Southeast England, and Midlands, showcase advanced glucose monitoring applications where OTC purchases integrate with existing prescription CGM programs and diabetes specialty pharmacy services aligned with NICE guidelines and NHS treatment protocols.
Key market characteristics:
OTC glucose monitors market in Japan demonstrates sophisticated implementation focused on accurate measurement technology and integration with comprehensive diabetes management protocols, with documented preference for clinically validated devices achieving superior measurement accuracy and reliability. The country maintains steady growth momentum with a CAGR of 6.5% through 2036, driven by aging population health management priorities and emphasis on preventive healthcare supporting chronic disease monitoring. Major population centers, including Tokyo, Osaka, and Nagoya, showcase advanced deployment of glucose monitoring technologies with comprehensive quality verification that integrates seamlessly with existing diabetes care delivery systems and patient support programs meeting Japanese healthcare standards.
Key market characteristics:
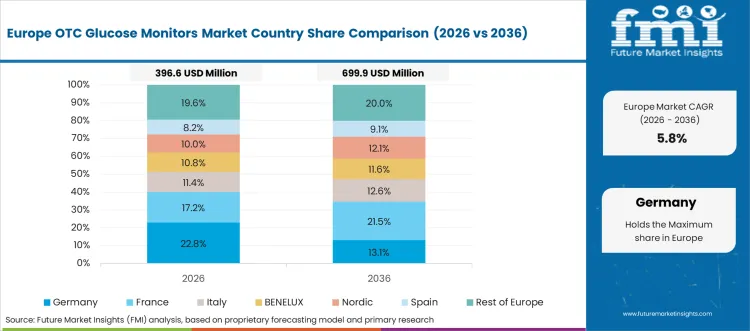
The OTC glucose monitors market in Europe is projected to grow from USD 410.8 million in 2026 to USD 942.5 million by 2036, registering a CAGR of 8.6% over the forecast period. Germany is expected to maintain its leadership position with a 30.5% market share in 2026, declining slightly to 30.2% by 2036, supported by its comprehensive diabetes care infrastructure and major population centers, including Berlin, Munich, and Hamburg metropolitan regions.
The United Kingdom follows with a 19.8% share in 2026, projected to reach 19.5% by 2036, driven by comprehensive NHS diabetes programs and patient empowerment initiatives implementing self-monitoring standards. France holds a 16.3% share in 2026, expected to rise to 16.5% by 2036 through ongoing diabetes management optimization and digital health integration. Italy commands a 12.8% share in both 2026 and 2036, backed by universal healthcare coverage and diabetes specialty care networks. Spain accounts for 9.5% in 2026, rising to 9.7% by 2036 on diabetes awareness campaigns and pharmacy access expansion. The Netherlands maintains 5.6% in 2026, reaching 5.8% by 2036 on preventive healthcare leadership and digital health adoption. The Rest of Europe region is anticipated to hold 5.5% in 2026, expanding to 5.8% by 2036, attributed to increasing OTC glucose monitor adoption in Nordic countries and emerging Central & Eastern European diabetes management programs.
The South Korean OTC glucose monitors market is characterized by growing international medical device manufacturer presence, with companies maintaining significant positions through comprehensive technical capabilities and innovation leadership for diabetes management applications. The market demonstrates increasing emphasis on digital health connectivity and smartphone integration, as Korean consumers increasingly demand advanced glucose monitoring devices that integrate with domestic health app ecosystems and sophisticated telemedicine platforms deployed across major healthcare networks. Regional medical device distributors are gaining market share through strategic partnerships with international manufacturers, offering specialized services including Korean regulatory compliance support and customized patient support programs for diabetes care applications. The competitive landscape shows increasing collaboration between multinational device companies and Korean digital health specialists, creating hybrid service models that combine international sensor technology expertise with local healthcare system integration capabilities and patient education networks.
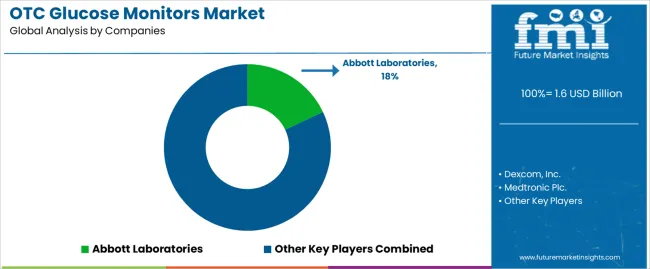
The OTC glucose monitors market features approximately 15-20 meaningful players with moderate concentration, where the top three companies control roughly 50-55% of global market share through established technology platforms and comprehensive distribution networks. Competition centers on sensor accuracy, user experience design, and digital ecosystem integration rather than price competition alone. Abbott Laboratories leads with approximately 18% market share through its FreeStyle Libre continuous glucose monitoring system portfolio.
Market leaders include Abbott Laboratories, Dexcom, Inc., and Medtronic Plc., which maintain competitive advantages through proprietary sensor technologies achieving superior accuracy and longevity, extensive clinical validation supporting medical professional recommendations and patient confidence, and comprehensive digital health platforms providing data analytics and remote monitoring capabilities across multiple patient populations, creating reliability and ecosystem advantages with diabetes patients and healthcare providers. These companies leverage research and development capabilities in miniaturized sensor engineering and ongoing innovation relationships to defend market positions while expanding into non-invasive technologies and predictive analytics algorithms.
Challengers encompass Roche Diabetes Care and Senseonics Holdings, Inc., which compete through specialized product offerings and strong regional presence in key diabetes markets. Product specialists focus on specific technology approaches or patient segments, offering differentiated capabilities in implantable sensors, hybrid monitoring systems, and specialized applications for gestational diabetes or pediatric populations.
Regional players and emerging digital health-integrated providers create competitive pressure through localized distribution advantages and innovative business models, particularly in high-growth markets including China and India, where smartphone-first designs and affordable sensor pricing provide advantages in cost-sensitive populations. Market dynamics favor companies that combine proven sensor accuracy with comprehensive patient support offerings including mobile app connectivity, remote monitoring capabilities, predictive alert algorithms, and healthcare provider data sharing that address complete diabetes management requirements across Type I intensive management, Type II lifestyle optimization, and gestational diabetes temporary monitoring throughout multi-year patient relationships.
OTC glucose monitors represent patient-directed monitoring systems that enable diabetes patients to achieve 10-15 additional glucose readings per hour compared to fingerstick methods, delivering superior glycemic insights and treatment optimization with proven HbA1c reductions of 0.5-1.0% in demanding diabetes management applications. With the market projected to grow from USD 1,552.9 million in 2026 to USD 3,576.3 million by 2036 at an 8.7% CAGR, these accessible monitoring systems offer compelling advantages - treatment independence, lifestyle flexibility, and complication prevention - making them essential for Diabetes Type I applications (66.3% market share), Type II diabetes management, and patient populations seeking alternatives to prescription-dependent monitoring that limits accessibility and creates healthcare system bottlenecks. Scaling market adoption and regulatory framework development requires coordinated action across diabetes policy, device safety standards, technology manufacturers, healthcare providers, and digital health investment capital.
| Metric | Value |
|---|---|
| Quantitative Units | USD 1.68 Billion to USD 3.88 Billion, at a CAGR of 8.7% |
| Market Definition | OTC glucose monitors are over-the-counter blood glucose and interstitial glucose monitoring devices that consumers can purchase without a prescription for self-management of diabetes or general metabolic health tracking. The category includes traditional fingerstick blood glucose meters, continuous glucose monitors (CGMs) with adhesive sensor patches, and smartphone-connected biosensors. |
| Product Segmentation | Continuous glucose monitors, Hybrid glucose monitoring systems, Blood glucose monitors |
| Indication Segmentation | Diabetes Type I, Diabetes Type II, Gestational diabetes |
| Technology Segmentation | Sensor-based glucose monitoring, Strip-based glucose monitoring, Hybrid technology |
| Distribution Channel Segmentation | Retail pharmacies, Drug stores, Online pharmacies, Specialty stores, Hypermarkets & supermarkets |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | China, India, Germany, USA, UK, Japan, and 40 plus countries |
| Key Companies Profiled | Abbott Laboratories, Dexcom, Inc., Medtronic Plc., Roche Diabetes Care, Senseonics Holdings, Inc. |
| Forecast Period | 2026 to 2036 |
| Approach | Forecasting models apply a bottom-up methodology starting with global installed base metrics and projecting conversion rates based on regulatory and technology adoption curves. |
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
How large is the demand for OTC Glucose Monitors in the global market in 2026?
Demand for otc glucose monitors in the global market is estimated to be valued at USD 1.68 billion in 2026.
What will be the market size of OTC Glucose Monitors in the global market by 2036?
Market size for otc glucose monitors is projected to reach USD 3.88 billion by 2036.
What is the expected demand growth for OTC Glucose Monitors in the global market between 2026 and 2036?
Demand for otc glucose monitors is expected to grow at a CAGR of 8.7% between 2026 and 2036.
Which Product is poised to lead global sales by 2026?
Continuous glucose monitors accounts for 55.0% share in 2026, driven by established adoption patterns and buyer preference across primary end-use applications.
What is driving demand in China?
China leads with a 11.7% CAGR through 2036, driven by expanding capacity and increasing regulatory alignment with international standards.
What is the India growth outlook in this report?
India is projected to grow at a CAGR of 10.9% during 2026 to 2036.
What is OTC Glucose Monitors and what is it mainly used for?
OTC glucose monitors are over-the-counter blood glucose and interstitial glucose monitoring devices that consumers can purchase without a prescription for self-management of diabetes or general metabolic health tracking. The category includes traditional fingerstick blood glucose meters, continuous glucose monitors (CGMs) with adhesive sensor patches, and smartphone-connected biosensors.
How does FMI build and validate the OTC Glucose Monitors forecast?
Future Market Insights analysis applies a bottom-up methodology starting with global installed base metrics and cross-validates projections against reported industry expenditure and regulatory compliance data.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.