High Performance Liquid Chromatography-Tandem Mass Spectrometry System Market
This report furnishes a complete analysis of the high performance liquid chromatography-tandem mass spectrometry system market, spanning market size, projected revenue, competitive terrain, demand outlook, key growth pillars, market impediments, application trends, supply chain developments, and strategic growth recommendations.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
High Performance Liquid Chromatography-Tandem Mass Spectrometry System Market size, market forecast and outlook by FMI
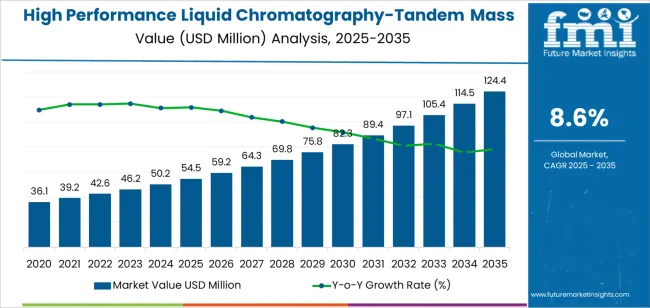
The high performance liquid chromatography-tandem mass spectrometry system market was valued at USD 54.50 billion in 2025, projected to reach USD 59.19 billion in 2026, and is forecast to expand to USD 135.06 billion by 2036 at a 8.6% CAGR. Expanding regulatory testing mandates across pharmaceutical quality control, food safety monitoring, and clinical diagnostics are accelerating LC-MS/MS system deployment. Drug development laboratories require high-throughput tandem mass spectrometry platforms for bioanalytical method validation and pharmacokinetic studies. Food safety authorities are tightening maximum residue limit enforcement for pesticides and veterinary drug residues, creating non-discretionary instrument procurement demand across government and contract testing laboratories.
Summary of High Performance Liquid Chromatography-Tandem Mass Spectrometry System Market
- Market Snapshot
- The high performance liquid chromatography-tandem mass spectrometry system market is valued at USD 54.50 billion in 2025 and is projected to reach USD 135.06 billion by 2036.
- The industry is expected to grow at a 8.6% CAGR from 2026 to 2036, creating an incremental opportunity of USD 75.87 billion.
- The market functions as a regulated analytical instrumentation category where detection sensitivity, throughput automation, and regulatory method compliance define competitive positioning.
- Demand and Growth Drivers
- Demand is accelerating as pharmaceutical regulatory agencies expand bioanalytical testing requirements for drug approval submissions, mandating LC-MS/MS method validation across clinical and preclinical studies.
- Food safety testing mandate enforcement across pesticide residue, mycotoxin, and veterinary drug monitoring programs is creating non-discretionary instrument procurement demand at government and contract testing laboratories.
- Growth is further supported by clinical diagnostic laboratory expansion deploying LC-MS/MS platforms for therapeutic drug monitoring, toxicology screening, and endocrine disorder testing panels.
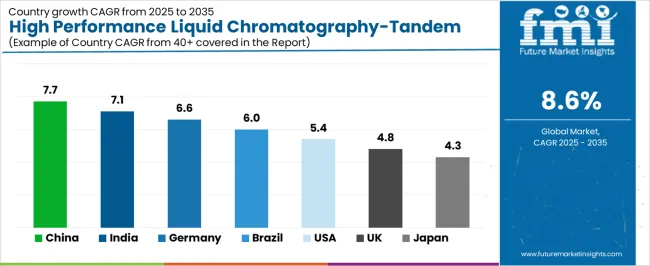
- Among key countries, China leads at 7.7% CAGR, India at 7.1%, Germany at 6.6%, Brazil at 6.0%, USA at 5.4%, UK at 4.8%.
- Product and Segment View
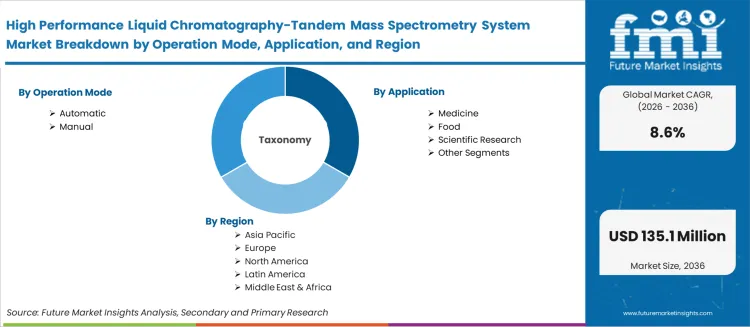
- The market includes automatic and manual LC-MS/MS system platforms deployed across medicine, food safety, scientific research, and other analytical testing applications.
- Automatic leads by Operation Mode with 71.9% share in 2026.
- Medicine leads by Application with 25.2% share in 2026.
- Scope includes Operation Mode, Application segmentation with regional coverage across seven global regions.
- Geography and Competitive Outlook
- China and India are the fastest-growing markets, while North America and Europe remain mature demand bases.
- Competition is shaped by product quality, technical specifications, and supply chain reliability, with key players including AB Sciex Pte. Ltd., Waters, EAGU, AELAB, Agilent, Drawell, Beijing BGI-GBI Biotech Co., Ltd., Hunan Kailaipu Biotechnology Co., Ltd., Puju Medical, Wuhan BGI Biomedical Engineering Co., Ltd., and Anhui Wanyi Science and Technology Co., Ltd..
High Performance Liquid Chromatography-Tandem Mass Spectrometry System Market key takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 59.19 billion |
| Industry Value (2036) | USD 135.06 billion |
| CAGR (2026 to 2036) | 8.6% |
Source: Future Market Insights, 2026
Based on FMI's report, the high performance liquid chromatography-tandem mass spectrometry system sector is transitioning from research-only instrumentation to routine analytical workflow deployment across regulated testing environments. Automatic operation mode systems account for the dominant share as high-throughput laboratories require unattended sample processing capability to manage escalating testing volumes. Manual systems maintain a defined role in method development and specialized research applications where operator intervention is required during analytical runs. Medicine leads the application segment as pharmaceutical bioanalytical testing, therapeutic drug monitoring, and clinical toxicology generate the highest per-laboratory instrument utilization rates. Food safety testing and scientific research applications contribute expanding demand as regulatory testing mandates intensify.
Country-level growth trajectories reflect pharmaceutical industry density and food safety regulation enforcement intensity. China leads at 7.7% CAGR, driven by pharmaceutical manufacturing quality control laboratory expansion and food safety testing mandate enforcement. India follows at 7.1%, where contract research organization growth and government laboratory modernization sustain instrument procurement. Germany at 6.6% benefits from pharmaceutical R&D laboratory density and clinical reference laboratory capacity. Brazil at 6.0% and USA at 5.4% contribute demand through regulatory testing infrastructure investment and bioanalytical services expansion.
High Performance Liquid Chromatography-Tandem Mass Spectrometry System Market definition
High Performance Liquid Chromatography-Tandem Mass Spectrometry System encompasses integrated analytical instrument platforms combining liquid chromatography separation with tandem mass spectrometry detection for quantitative and qualitative analysis of complex sample matrices. The market covers automatic and manual operation mode systems deployed across pharmaceutical bioanalysis, food safety testing, clinical diagnostics, environmental monitoring, and scientific research applications. These systems deliver picogram-level detection sensitivity required for regulatory compliance testing and trace analyte quantification.
High Performance Liquid Chromatography-Tandem Mass Spectrometry System Market inclusions
Market scope includes global and regional market sizes, a 2026 to 2036 forecast period, segment breakdowns by Operation Mode, Application, and regional analysis across North America, Latin America, Europe, East Asia, South Asia, Oceania, and Middle East and Africa. Pricing trend analysis and competitive positioning assessments are incorporated.
High Performance Liquid Chromatography-Tandem Mass Spectrometry System Market exclusions
The scope excludes standalone HPLC systems without tandem mass spectrometry detectors, single-quadrupole LC-MS platforms, and GC-MS/MS instruments. Consumable columns, reagents, and sample preparation kits are excluded as separate product categories.
High Performance Liquid Chromatography-Tandem Mass Spectrometry System Market research methodology
- Primary Research: Analysts engaged with pharmaceutical bioanalytical laboratory directors, food safety testing coordinators, and clinical laboratory instrument procurement managers to map system selection criteria and throughput capacity requirements.
- Desk Research: Data collection aggregated pharmaceutical regulatory guidance documents, food safety maximum residue limit databases, clinical laboratory accreditation standards, and instrument vendor product registration filings from FDA, EMA, and NMPA.
- Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of product-level revenues, applying region-specific adoption curves to project future demand velocity.
- Data Validation and Update Cycle: Projections are tested against publicly reported expenditure guidance from leading industry participants and cross-referenced with regulatory filings.
Why is the High Performance Liquid Chromatography-Tandem Mass Spectrometry System Market Growing?
Market expansion rests on three fundamental shifts driving adoption across the pharmaceutical, food safety, and scientific research sectors. First, analytical precision demand creates compelling operational advantages through mass spectrometry systems that provide immediate compound identification and quantitative analysis without compromising testing accuracy, enabling laboratories to meet stringent regulatory standards while maintaining research productivity and reducing analytical errors. Second, laboratory automation modernization accelerates as pharmaceutical companies worldwide seek advanced analytical systems that complement traditional testing processes, enabling precise method optimization and quality control that align with industry standards and regulatory requirements.
Third, pharmaceutical research enhancement drives adoption from drug development laboratories and clinical testing facilities requiring effective analytical solutions that maximize testing throughput while maintaining operational accuracy during sample preparation and analysis operations. Growth faces headwinds from equipment cost challenges that vary across analytical suppliers regarding the sourcing of mass spectrometry components and detector systems, which may limit adoption in cost-sensitive environments. Technical limitations also persist regarding maintenance complexity and operator training requirements that may reduce effectiveness in resource-constrained laboratory scenarios, which affect system performance and operational requirements.
Opportunity Pathways - High Performance Liquid Chromatography-Tandem Mass Spectrometry System Market
The high performance liquid chromatography-tandem mass spectrometry system market represents a specialized yet critical analytical opportunity driven by expanding global pharmaceutical research, laboratory automation modernization, and the need for superior detection precision in diverse testing applications. As laboratories worldwide seek to achieve advanced analytical effectiveness, reduce testing errors, and integrate automated systems with laboratory platforms, mass spectrometry systems are evolving from basic analytical instruments to sophisticated testing solutions ensuring measurement accuracy and regulatory compliance leadership.
The market's growth trajectory from USD 54.5 million in 2025 to USD 125.2 million by 2035 at an 8.6% CAGR reflects fundamental shifts in pharmaceutical industry testing requirements and analytical precision optimization. Geographic expansion opportunities are particularly pronounced in China and India markets, while the dominance of automatic operation systems and medicine applications provides clear strategic focus areas.
Pathway A - Automatic Operation Leadership & Advanced System Optimization
Strengthening the dominant automatic operation segment through enhanced automation formulations, superior analytical precision, and integrated laboratory systems. This pathway focuses on optimizing system configuration, improving testing throughput, extending operational effectiveness to advanced detection rates, and developing specialized configurations for diverse applications. Market leadership consolidation through advanced automation engineering and laboratory integration enables premium positioning while defending competitive advantages against manual operation alternatives. Expected revenue pool: USD 8.0-11.0 million
Pathway B - China Market Expansion & Laboratory Localization
Rapid pharmaceutical research and laboratory infrastructure growth across China creates substantial expansion opportunities through local manufacturing capabilities and technology transfer partnerships. Growing drug development activity and government analytical standards initiatives drive steady demand for advanced mass spectrometry systems. Localization strategies reduce import costs, enable faster technical support, and position companies advantageously for procurement programs while accessing growing domestic markets. Expected revenue pool: USD 7.0-9.5 million
Pathway C - Medicine Market Dominance & Pharmaceutical Integration
Expansion within the dominant medicine segment through specialized system designs addressing pharmaceutical testing standards and high-volume analytical requirements. This pathway encompasses automated sample processing, regulatory compliance integration, and compatibility with diverse pharmaceutical research processes. Premium positioning reflects superior detection precision and comprehensive quality compliance supporting modern drug development. Expected revenue pool: USD 6.0-8.5 million
Pathway D - Food Application Diversification
Strategic expansion into food applications requires enhanced detection capabilities and specialized system configurations addressing food safety operational requirements. This pathway addresses contaminant analysis, residue testing, and quality assurance applications with advanced separation engineering for demanding analytical standards. Premium pricing reflects specialized detection requirements and extended sensitivity standards. Expected revenue pool: USD 5.0-7.0 million
Pathway E - Scientific Research & Other Application Innovation
Development of specialized system configurations for scientific research applications and other segments, addressing specific analytical requirements and research testing demands. This pathway encompasses method development, environmental analysis, and cost-effective alternatives for emerging research segments. Technology differentiation through proprietary configurations enables diversified revenue streams while reducing dependency on single application platforms. Expected revenue pool: USD 4.5-6.5 million
Pathway F - Manual Operation Development & Cost-Effective Positioning
Expansion of manual operation segment through accessible system designs, operator-friendly interfaces, and budget-conscious laboratory requirements. This pathway encompasses academic research applications, small-scale testing facilities, and specialized applications requiring hands-on analytical control. Market development through simplified operation engineering enables differentiated positioning while accessing cost-sensitive markets requiring reliable analytical solutions. Expected revenue pool: USD 3.5-5.0 million
Pathway G - Regulatory Compliance & Method Validation Development
Development of regulatory-compliant system configurations addressing pharmaceutical validation and food safety requirements across medicine and scientific research applications. This pathway encompasses method validation protocols, regulatory documentation, and comprehensive compliance systems. Premium positioning reflects regulatory expertise and validation capabilities while enabling access to regulated testing programs and compliance-driven laboratory partnerships. Expected revenue pool: USD 3.0-4.5 million
What factors drive the dominance of the automatic operation mode and medicine application segments in the high performance liquid chromatography tandem mass spectrometry system market?
Primary Classification: The market segments by operation mode into Manual and Automatic categories, representing the evolution from basic operator-controlled systems to specialized automated solutions for comprehensive analytical optimization.
Secondary Classification: Application segmentation divides the market into Medicine, Food, Scientific Research, and Other Segments sectors, reflecting distinct requirements for analytical precision, testing capacity, and detection standards.
Regional Classification: Geographic distribution covers China, India, Germany, Brazil, United States, United Kingdom, Japan, and other regions, with developed markets leading adoption while emerging economies show accelerating growth patterns driven by laboratory expansion programs.
The segmentation structure reveals technology progression from standard manual-based systems toward specialized automated configurations with enhanced precision and testing capabilities, while application diversity spans from pharmaceutical analysis to specialized food safety and research applications requiring precise analytical solutions.
By Operation Mode, the Automatic Segment Accounts for Dominant Market Share
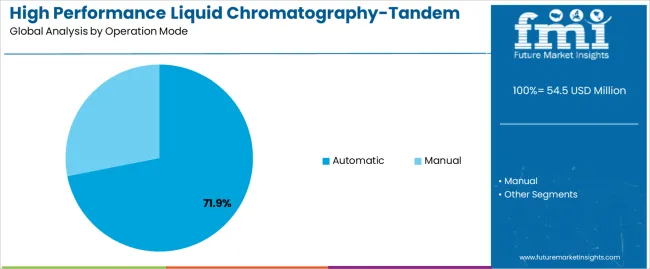
Market Position: Automatic operation systems command the leading position in the market with approximately 71.9% market share through advanced automation properties, including superior testing throughput, precision analytical capability, and operation optimization that enable laboratories to achieve optimal analytical performance across diverse pharmaceutical and research environments.
Value Drivers: The segment benefits from laboratory preference for reliable automated systems that provide consistent testing performance, reduced operator error, and workflow optimization without requiring significant process modifications. Advanced automation features enable integrated sample processing, analytical consistency, and integration with existing laboratory equipment, where system performance and testing reliability represent critical operational requirements.
Competitive Advantages: Automatic operation systems differentiate through proven operational stability, consistent analytical characteristics, and integration with laboratory information management systems that enhance testing effectiveness while maintaining optimal precision suitable for diverse pharmaceutical applications.
Key market characteristics:
- Advanced automation configurations with optimized workflow integration and precision control capabilities
- Extended operational effectiveness, enabling advanced detection with consistent analytical quality
- Laboratory compatibility, including sample management systems, data processing, and method integration for pharmaceutical testing operations
Manual Operation Shows Strong Market Presence
Manual operation systems maintain cost-effective positioning in the market with approximately 28.1% market share due to their accessible operation properties and hands-on analytical advantages. These systems appeal to laboratories requiring operator-controlled instruments with adequate performance for research and educational applications. Market adoption is driven by academic laboratory expansion, focusing reliable analytical solutions and operational flexibility through operator-controlled system configurations while maintaining competitive pricing structures.
By Application, the Medicine Segment Accounts for the Largest Market Share
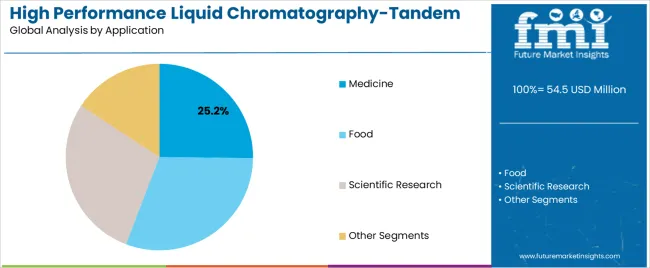
Market Context: Medicine applications dominate the market with approximately 25.2% market share due to widespread adoption of precision analytical testing and increasing focus on pharmaceutical development, drug monitoring, and quality assurance applications that minimize testing failures while maintaining pharmaceutical industry standards.
Appeal Factors: Pharmaceutical manufacturers prioritize analytical reliability, detection consistency, and integration with existing drug development infrastructure that enables coordinated testing application across multiple research programs. The segment benefits from substantial pharmaceutical industry investment and regulatory programs that emphasize the acquisition of mass spectrometry systems for quality control and compound identification applications.
Growth Drivers: Drug development programs incorporate mass spectrometry systems as standard analytical tools for pharmaceutical operations, while clinical testing growth increases demand for precision analytical capabilities that comply with regulatory standards and minimize measurement uncertainty.
Market Challenges: Varying pharmaceutical standards and drug formulation differences may limit analytical standardization across different testing facilities or operational scenarios.
Application dynamics include:
- Strong growth in pharmaceutical research and clinical diagnostic facilities requiring precision analytical capabilities
- Increasing adoption in drug metabolism studies and therapeutic monitoring applications for pharmaceutical development
- Rising integration with automated sample preparation systems for testing optimization and quality assurance
Food Applications Demonstrate Strong Growth
Food applications capture approximately 5.7% market share through specialized analytical requirements in food safety testing, contaminant analysis, and quality control applications. These facilities demand robust analytical systems capable of detecting trace compounds while providing exceptional sensitivity and regulatory compliance capabilities.
Scientific Research and Other Segments Show Diversified Demand
Scientific research applications account for market share, while other segments include environmental testing, forensic analysis, and specialized research applications requiring mass spectrometry capabilities for analytical precision and method development.
What are the Drivers, Restraints, and Key Trends of the High Performance Liquid Chromatography-Tandem Mass Spectrometry System Market?
Growth Accelerators: Pharmaceutical research expansion drives primary adoption as mass spectrometry systems provide superior analytical detection capabilities that enable research laboratories to meet stringent regulatory standards without excessive equipment costs, supporting drug development and pharmaceutical missions that require precise compound identification applications. Laboratory automation infrastructure demand accelerates market expansion as pharmaceutical companies seek effective automated systems that minimize operator intervention while maintaining analytical effectiveness during sample testing and method validation scenarios. Pharmaceutical industry spending increases worldwide, creating steady demand for precision analytical systems that complement traditional laboratory processes and provide testing differentiation in competitive research markets.
Growth Inhibitors: Equipment cost challenges vary across analytical suppliers regarding the sourcing of mass spectrometry detectors and separation components, which may limit operational flexibility and market penetration in regions with budget constraints or cost-sensitive laboratory operations. Technical performance limitations persist regarding maintenance requirements and operator training needs that may reduce effectiveness in resource-limited facilities, small-scale laboratories, or academic research conditions, affecting system reliability and operational requirements. Market fragmentation across multiple pharmaceutical standards and analytical methods creates compatibility concerns between different system suppliers and existing laboratory infrastructure.
Market Evolution Patterns: Adoption accelerates in pharmaceutical research and clinical testing sectors where analytical precision justifies system costs, with geographic concentration in developed markets transitioning toward mainstream adoption in emerging economies driven by laboratory capacity expansion and regulatory awareness. Technology development focuses on enhanced automation capabilities, improved detection sensitivity, and compatibility with laboratory information systems that optimize analytical throughput and testing effectiveness. The market could face disruption if alternative analytical technologies or portable testing innovations significantly limit the deployment of mass spectrometry-based systems in laboratory applications, though the technology's unique combination of precision detection, compound identification, and quantitative analysis continues to make it preferred in pharmaceutical applications.
Analysis of the High Performance Liquid Chromatography-Tandem Mass Spectrometry System Market by Key Country
The market demonstrates varied regional dynamics with Growth Leaders including China (7.7% CAGR) and India (7.1% CAGR) driving expansion through pharmaceutical capacity additions and laboratory infrastructure programs. Steady Performers encompass Germany (6.6% CAGR), Brazil (6.0% CAGR), and United States (5.4% CAGR), benefiting from established pharmaceutical industries and advanced analytical technology adoption. Mature Markets feature United Kingdom (4.8% CAGR) and Japan (4.3% CAGR), where specialized pharmaceutical applications and precision testing integration support consistent growth patterns.
.webp)
| Country | CAGR (2026-2036) |
|---|---|
| China | 7.7% |
| India | 7.1% |
| Germany | 6.6% |
| Brazil | 6.0% |
| United States | 5.4% |
| United Kingdom | 4.8% |
| Japan | 4.3% |
Regional synthesis reveals China and India markets leading adoption through pharmaceutical manufacturing expansion and laboratory infrastructure development, while European countries maintain steady expansion supported by pharmaceutical technology advancement and regulatory standardization requirements. North American markets show moderate growth driven by drug development applications and analytical system integration trends.
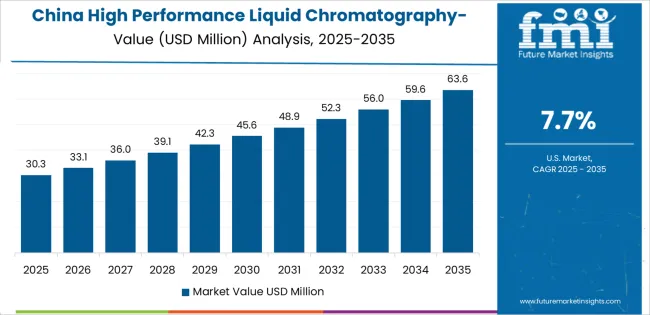
Why does China demonstrate rapid market expansion?
The Chinese market emphasizes advanced analytical features, including precision detection control and integration with comprehensive pharmaceutical platforms that manage testing quality, method optimization, and regulatory applications through unified laboratory systems. The country demonstrates strong growth at 7.7% CAGR, driven by pharmaceutical manufacturing expansion, laboratory infrastructure initiatives, and emerging drug development programs that support mass spectrometry integration. Chinese pharmaceutical laboratories prioritize operational effectiveness with analytical systems delivering consistent testing performance through advanced automation capabilities and method adaptation features.
Technology deployment channels include major pharmaceutical manufacturers, specialized analytical equipment suppliers, and research institute procurement programs that support professional applications for complex drug analysis and compound identification applications. Laboratory platform integration capabilities with established pharmaceutical systems expand market appeal across diverse operational requirements seeking precision detection and regulatory compliance benefits. The expanding pharmaceutical sector and accelerating drug development market create steady demand, while innovative applications in traditional medicine analysis and biosimilar testing open new growth avenues.
Performance Metrics:
- Pharmaceutical research facilities in Beijing, Shanghai, and Guangzhou leading mass spectrometry adoption for drug development and quality control operations
- Contract research organizations maintaining significant market share for analytical testing integration applications
- Academic research institutions supporting substantial system acquisitions across university laboratory facilities
- Laboratory platform compatibility with major pharmaceutical systems driving procurement selection criteria
How does Germany maintain analytical technology leadership?
Germany's advanced pharmaceutical market demonstrates sophisticated mass spectrometry deployment with documented operational effectiveness in drug development applications and research facilities through integration with existing analytical systems and laboratory infrastructure. The country leverages engineering expertise in precision instrumentation and pharmaceutical systems integration to maintain strong growth at 6.6% CAGR. Industrial centers, including Frankfurt, Munich, and Hamburg, showcase premium installations where mass spectrometry systems integrate with comprehensive laboratory platforms and quality management systems to optimize pharmaceutical testing and analytical effectiveness.
German pharmaceutical companies prioritize system reliability and EU compliance in analytical development, creating demand for precision systems with advanced features, including method validation and automated data processing. The market benefits from established pharmaceutical infrastructure and a willingness to invest in analytical technologies that provide long-term research benefits and compliance with international regulatory and quality standards.
Market Intelligence Brief:
- Engineering focuses on EU regulatory standards and precision quality compliance, driving pharmaceutical segment growth
- Pharmaceutical partnerships providing faster method development cycles
- Technology collaboration between German analytical equipment manufacturers and international pharmaceutical companies
- Regulatory training programs expanding mass spectrometry system integration in pharmaceutical testing scenarios
Why does the United States emphasize pharmaceutical innovation?
The USA mass spectrometry market demonstrates sophisticated deployment across pharmaceutical applications with documented effectiveness in drug development and clinical testing facilities through integration with comprehensive laboratory management systems and regulatory infrastructure. The country leverages advanced research capabilities in analytical innovation and pharmaceutical technologies to maintain moderate growth at 5.4% CAGR. Industrial centers, including Boston, San Francisco, and Research Triangle, showcase premium installations where mass spectrometry systems integrate with comprehensive pharmaceutical platforms and drug discovery networks to optimize analytical performance and testing effectiveness.
American pharmaceutical companies prioritize testing precision and regulatory compliance in analytical development, creating demand for advanced systems with sophisticated features, including high-resolution detection and automated sample processing. The market benefits from established pharmaceutical infrastructure and willingness to invest in analytical technologies that provide long-term research benefits and compliance with FDA and international standards.
Market Intelligence Brief:
- Pharmaceutical innovation focuses on regulatory standards and precision quality compliance, driving drug development segment growth
- Pharmaceutical partnerships providing accelerated analytical method cycles
- Technology collaboration between American analytical manufacturers and global pharmaceutical companies
- FDA validation programs expanding mass spectrometry system integration in pharmaceutical testing scenarios
How does the United Kingdom drive pharmaceutical research integration?
The U.K. mass spectrometry market demonstrates advanced pharmaceutical deployment with documented operational effectiveness in drug development applications and research facilities through integration with existing regulatory systems and laboratory infrastructure. The country leverages pharmaceutical expertise in analytical testing and research systems integration to maintain steady growth at 4.8% CAGR. Industrial centers, including Oxford, Cambridge, and London, showcase premium installations where mass spectrometry systems integrate with comprehensive pharmaceutical platforms and research management systems to optimize regulatory compliance and analytical effectiveness.
British pharmaceutical companies prioritize system validation and regulatory compliance in analytical development, creating demand for certified systems with advanced features, including method qualification and regulatory documentation. The market benefits from established pharmaceutical infrastructure and commitment to invest in analytical technologies that provide long-term research benefits and compliance with UK and EU regulatory standards. Drug development applications, clinical research systems, and pharmaceutical quality programs drive diversified demand across multiple application segments.
Strategic Market Indicators:
- Regulatory compliance focuses on UK pharmaceutical standards and precision quality requirements, driving research segment growth
- Pharmaceutical partnerships providing faster analytical validation cycles
- Technology collaboration between British analytical manufacturers and international pharmaceutical companies
- Regulatory certification programs expanding mass spectrometry system integration in pharmaceutical testing scenarios
Why is India emerging as a high-growth market?
India's mass spectrometry market demonstrates rapid expansion deployment with documented operational effectiveness in pharmaceutical applications and research facilities through integration with emerging laboratory systems and drug development infrastructure. The country leverages growing pharmaceutical capabilities in analytical testing and research systems integration to achieve high growth at 7.1% CAGR. Industrial centers, including Mumbai, Hyderabad, and Bangalore, showcase expanding installations where mass spectrometry systems integrate with comprehensive pharmaceutical platforms and testing networks to optimize market penetration and analytical effectiveness.
Indian pharmaceutical companies prioritize analytical precision and quality standards in system development, creating demand for sophisticated analytical instruments with advanced features, including automated operation and regulatory compliance. The market benefits from expanding pharmaceutical infrastructure and willingness to invest in international-standard analytical technologies that provide testing differentiation and compliance with global quality standards.
Market Intelligence Brief:
- Pharmaceutical manufacturing focuses on international quality standards and analytical precision, driving pharmaceutical market segment growth
- Contract research partnerships providing accelerated market entry cycles
- Technology collaboration between Indian manufacturers and international analytical companies
- Quality certification programs expanding mass spectrometry system integration in pharmaceutical testing scenarios
How does Japan emphasize analytical precision excellence?
Japan's mass spectrometry market demonstrates precision deployment with documented operational effectiveness in pharmaceutical applications and research facilities through integration with advanced laboratory systems and quality control infrastructure. The country leverages engineering excellence in precision instrumentation and analytical systems integration to maintain steady growth at 4.3% CAGR. Industrial centers, including Tokyo, Osaka, and Yokohama, showcase premium installations where mass spectrometry systems integrate with comprehensive quality platforms and pharmaceutical testing systems to optimize analytical excellence and detection effectiveness.
Japanese pharmaceutical companies prioritize system precision and analytical excellence in instrument development, creating demand for high-precision systems with advanced features, including ultra-high sensitivity detection and precision control systems. The market benefits from established pharmaceutical infrastructure and commitment to invest in highest-quality analytical technologies that provide superior testing performance and compliance with stringent Japanese quality standards.
Strategic Market Indicators:
- Engineering focuses on precision quality standards and analytical excellence, driving pharmaceutical segment growth
- Pharmaceutical partnerships providing enhanced method development cycles
- Technology collaboration between Japanese analytical specialists and international pharmaceutical companies
- Precision manufacturing programs expanding mass spectrometry system integration in pharmaceutical testing scenarios
What is the market split by country in Europe?
The high performance liquid chromatography-tandem mass spectrometry system market in Europe is projected to grow substantially over the forecast period, with Germany expected to maintain its leadership position with a significant market share supported by its advanced pharmaceutical infrastructure and major research centers in Frankfurt and Munich. United Kingdom follows with strong market presence, driven by comprehensive pharmaceutical research programs and clinical testing initiatives. France holds substantial market share through specialized drug development activities, analytical testing applications, and pharmaceutical manufacturing. Italy commands notable market presence through pharmaceutical research and quality control projects. Spain accounts for growing market share aided by pharmaceutical manufacturing expansion and analytical testing adoption. The Netherlands maintains steady share driven by specialty pharmaceutical applications and research laboratory demand. The Rest of Europe region is anticipated to show steady adoption, reflecting consistent growth in Nordic countries, pharmaceutical research expansion in Central European markets, and laboratory infrastructure upgrades across Eastern European research facilities.
Why does automatic operation dominate analytical testing demand in Japan?
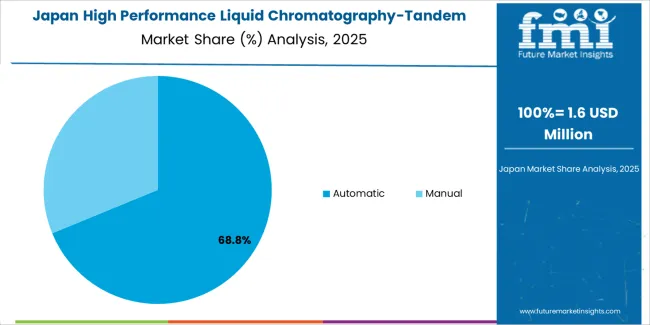
In Japan, the market prioritizes automatic operation systems, which capture the dominant share of pharmaceutical and research installations due to their advanced features, including precision workflow optimization and seamless integration with existing laboratory infrastructure. Japanese pharmaceutical laboratories emphasize reliability, precision, and long-term operational excellence, creating demand for automatic systems that provide consistent analytical capabilities and superior testing performance based on regulatory requirements and quality standards. Manual operation maintains secondary positions primarily in academic research applications and educational laboratory installations where operator-controlled functionality meets analytical requirements without compromising testing efficiency.
Market Characteristics:
- Premium focus on automatic operation systems with advanced automation capabilities and precision analytical properties
- Integration requirements with existing pharmaceutical platforms and laboratory information systems
- Focus on operational reliability and long-term performance in pharmaceutical applications
Why do analytical equipment manufacturers lead laboratory services in South Korea?
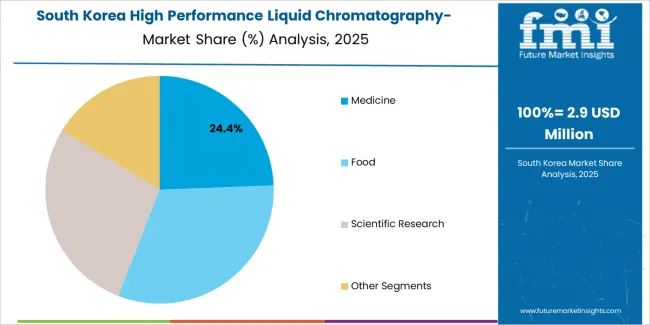
In South Korea, the market structure favors international analytical manufacturers, including AB Sciex, Waters, and Agilent, which maintain dominant positions through comprehensive product portfolios and established pharmaceutical industry networks supporting both drug development and clinical testing installations. These providers offer integrated solutions combining advanced mass spectrometry systems with professional technical services and ongoing analytical support that appeal to Korean pharmaceutical companies seeking reliable precision testing systems. Local laboratory equipment distributors and technical service providers capture moderate market share by providing localized service capabilities and competitive pricing for standard pharmaceutical testing installations, while domestic manufacturers focus on specialized applications and cost-effective solutions tailored to Korean pharmaceutical market characteristics.
Channel Insights:
- International analytical brands maintaining premium market positioning through advanced testing offerings
- Local pharmaceutical service networks expanding to support growing demand for professional technical and analytical support
- Method development capabilities becoming a key differentiator for pharmaceutical and research applications
What factors define the competitive landscape of the high performance liquid chromatography tandem mass spectrometry system market?
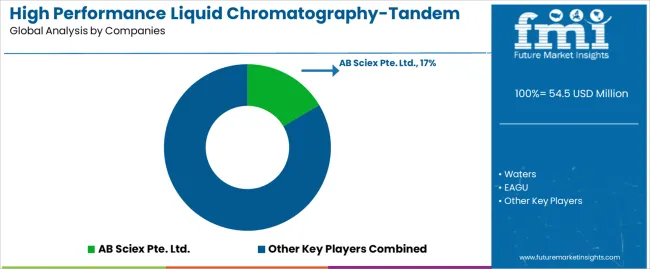
The market operates with moderate concentration, featuring approximately 10-12 meaningful participants, where leading companies control roughly 55-60% of the global market share through established pharmaceutical relationships and comprehensive analytical system portfolios. Competition emphasizes advanced detection capabilities, automation integration, and pharmaceutical laboratory compatibility rather than price-based rivalry. The market structure reflects specialized analytical instrumentation requirements where technology expertise and regulatory compliance create significant barriers to entry.
Market Leaders encompass AB Sciex Pte. Ltd., Waters, and Agilent, which maintain competitive advantages through extensive mass spectrometry expertise, global pharmaceutical laboratory networks, and comprehensive method development capabilities that create customer loyalty and support premium pricing. These companies leverage decades of analytical technology experience and ongoing innovation investments to develop advanced systems with precision detection control and automation features. Technology Innovators include EAGU, AELAB, and regional specialists, which compete through specialized system configuration focus and innovative automation capabilities that appeal to laboratories seeking advanced analytical solutions and testing differentiation.
These companies differentiate through rapid method development cycles and specialized pharmaceutical application focus. Regional Specialists feature analytical equipment manufacturers focusing on specific geographic markets and specialized applications, including automated laboratory systems and integrated pharmaceutical solutions. Market dynamics favor participants that combine reliable detection performance with advanced automation capabilities, including precision analytical control and comprehensive method optimization features. Competitive pressure intensifies as traditional laboratory equipment suppliers expand into pharmaceutical testing systems, while specialized analytical companies challenge established players through innovative detection solutions and automated platforms targeting drug development and clinical testing segments.
Key Players in the High Performance Liquid Chromatography-Tandem Mass Spectrometry System Market
- AB Sciex Pte. Ltd.
- Waters
- EAGU
- AELAB
- Agilent
- Drawell
- Beijing BGI-GBI Biotech Co., Ltd.
- Hunan Kailaipu Biotechnology Co., Ltd.
- Puju Medical
- Wuhan BGI Biomedical Engineering Co., Ltd.
- Anhui Wanyi Science and Technology Co., Ltd.
Scope of the report
| Metric | Value |
|---|---|
| Quantitative Units | USD 59.19 billion to USD 135.06 billion, at a CAGR of 8.6% |
| Market Definition | High Performance Liquid Chromatography-Tandem Mass Spectrometry System encompasses integrated analytical instrument platforms combining liquid chromatography separation with tandem mass spectrometry d... |
| Operation Mode Segmentation | Automatic, Manual |
| Application Segmentation | Medicine, Food, Scientific Research, Other Segments |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | China, India, Germany, Brazil, USA, UK, Japan, and 40 plus countries |
| Key Companies Profiled | AB Sciex Pte. Ltd., Waters, EAGU, AELAB, Agilent, Drawell, Beijing BGI-GBI Biotech Co., Ltd., Hunan Kailaipu Biotechnology Co., Ltd., Puju Medical, Wuhan BGI Biomedical Engineering Co., Ltd., and Anhui Wanyi Science and Technology Co., Ltd. |
| Forecast Period | 2026 to 2036 |
| Approach | Forecasting models apply a bottom-up methodology starting with global installed base metrics and projecting the conversion rate to advanced product adoption. |
High Performance Liquid Chromatography-Tandem Mass Spectrometry System Market by Segments
Operation Mode:
- Manual
- Automatic
Application:
- Medicine
- Food
- Scientific Research
- Other Segments
Country:
- China
- India
- Germany
- Brazil
- United States
- United Kingdom
- Japan
Bibliography
- 1. USA Food and Drug Administration. (2024). Bioanalytical Method Validation Guidance for Industry. FDA.
- 2. European Medicines Agency. (2024). Guideline on Bioanalytical Method Validation. EMA.
- 3. Codex Alimentarius Commission. (2024). Maximum Residue Limits for Pesticides in Food. FAO/WHO.
- 4. International Organization for Standardization. (2024). ISO 17025: General Requirements for Competence of Testing Laboratories. ISO.
- 5. The World Bank. (2024). World Development Indicators: Health and Food Safety. World Bank Group.
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
Frequently Asked Questions
How large is the demand for High Performance Liquid Chromatography-Tandem Mass Spectrometry System in the global market in 2026?
Demand for high performance liquid chromatography-tandem mass spectrometry system in the global market is estimated to be valued at USD 59.19 billion in 2026.
What will be the market size of High Performance Liquid Chromatography-Tandem Mass Spectrometry System in the global market by 2036?
Market size for high performance liquid chromatography-tandem mass spectrometry system is projected to reach USD 135.06 billion by 2036.
What is the expected demand growth for High Performance Liquid Chromatography-Tandem Mass Spectrometry System between 2026 and 2036?
Demand for high performance liquid chromatography-tandem mass spectrometry system is expected to grow at a CAGR of 8.6% between 2026 and 2036.
Which Operation Mode is poised to lead global sales by 2026?
Automatic accounts for 71.9% share in 2026 as it addresses the primary demand requirements across global markets.
How significant is the role of Medicine in driving high performance liquid chromatography-tandem mass spectrometry system adoption in 2026?
Medicine represents 25.2% of segment share as end-user demand consolidates around established application pathways.
What is driving demand in China?
China is projected to grow at a CAGR of 7.7% during 2026 to 2036, driven by expanding infrastructure investment and industrial capacity growth.
What does market forecast mean on this page?
The market forecast represents a model-based projection built on defined industry and supply chain assumptions for strategic planning purposes.
How does FMI build and validate the high performance liquid chromatography-tandem mass spectrometry system forecast?
Forecasting models apply a bottom-up methodology starting with global installed base metrics and cross-validate projections against quarterly industry revenue volumes.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Operation Mode
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Operation Mode , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Operation Mode , 2026 to 2036
- Automatic
- Manual
- Automatic
- Y to o to Y Growth Trend Analysis By Operation Mode , 2021 to 2025
- Absolute $ Opportunity Analysis By Operation Mode , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Medicine
- Food
- Scientific Research
- Other Segments
- Medicine
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Operation Mode
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Operation Mode
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Operation Mode
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Operation Mode
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Operation Mode
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Operation Mode
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Operation Mode
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Operation Mode
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Operation Mode
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Operation Mode
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Operation Mode
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Operation Mode
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Operation Mode
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Operation Mode
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Operation Mode
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Operation Mode
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Operation Mode
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Operation Mode
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Operation Mode
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Operation Mode
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Operation Mode
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Operation Mode
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Operation Mode
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Operation Mode
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Operation Mode
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Operation Mode
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Operation Mode
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Operation Mode
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Operation Mode
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Operation Mode
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Operation Mode
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Operation Mode
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Operation Mode
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Operation Mode
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Operation Mode
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Operation Mode
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Operation Mode
- By Application
- Competition Analysis
- Competition Deep Dive
- AB Sciex Pte. Ltd.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Waters
- EAGU
- AELAB
- Agilent
- Drawell
- Beijing BGI-GBI Biotech Co., Ltd.
- Hunan Kailaipu Biotechnology Co., Ltd.
- Puju Medical
- Wuhan BGI Biomedical Engineering Co., Ltd.
- Anhui Wanyi Science and Technology Co., Ltd.
- AB Sciex Pte. Ltd.
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Operation Mode , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Operation Mode , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Operation Mode , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Operation Mode , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Operation Mode , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Operation Mode , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Operation Mode , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Operation Mode , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Operation Mode , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Operation Mode , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Operation Mode
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Operation Mode , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Operation Mode , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Operation Mode
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Operation Mode , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Operation Mode , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Operation Mode
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Operation Mode , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Operation Mode , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Operation Mode
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Operation Mode , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Operation Mode , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Operation Mode
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Operation Mode , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Operation Mode , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Operation Mode
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Operation Mode , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Operation Mode , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Operation Mode
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Operation Mode , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Operation Mode , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Operation Mode
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

