Magnesium Stearate Market
The Magnesium Stearate Market is segmented by Form (Powder, Granular, Flakes), End Use (Pharmaceuticals, Dietary Supplements, Personal Care, Plastics and Polymers, Others), Application (Lubricant, Emulsifier, Binder, Others), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Magnesium Stearate Market Size, Market Forecast and Outlook By FMI
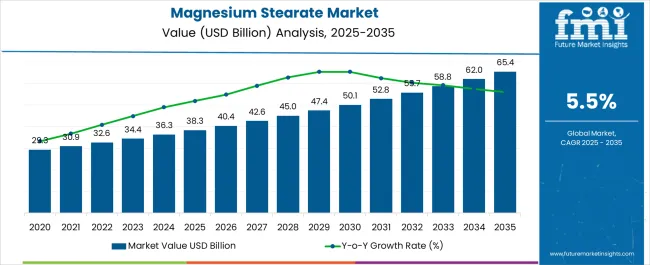
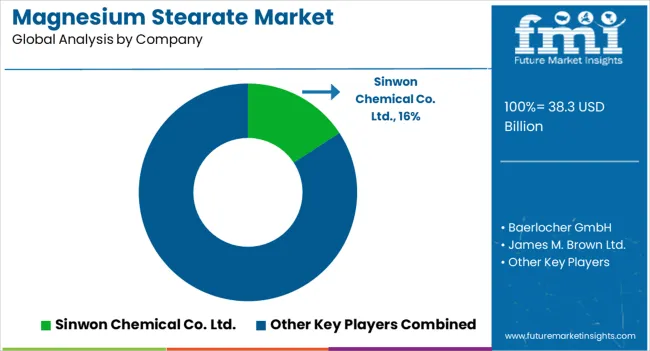
In 2025, the magnesium stearate market surpassed a value of USD 38.3 billion. Based on Future Market Insights’ analysis, demand is estimated to rise to USD 40.4 billion in 2026 and USD 69.2 billion by 2036. FMI projects the market to expand at a CAGR of 5.5% between 2026 and 2036, reflecting accelerating pharmaceutical production, rising dietary supplement consumption, and sustained reliance on high-purity excipients across regulated global manufacturing environments.
Pharmaceutical formulators are standardizing excipient selection to navigate rigorous regulatory reviews. Shifting toward high-purity metallic soaps reduces batch failure rates during tableting processes. As per FMI's projection, formulators actively revise ingredient matrices to replace highly volatile thickeners with cost-effective compounds.
Summary of Magnesium Stearate
- Magnesium Stearate Market Definition:
- The industry covers the industrial production and commercial distribution of essential chemical lubricants used in solid oral dosage manufacturing.
- Demand Drivers in the Market:
- Expanding solid-dose pharmaceutical output requires reliable equipment lubrication.
- Surging dietary supplement consumption relies heavily on efficient capsule filling.
- Polymer processing operations utilize metallic soaps as effective mold-release agents.
- Key Segments Analyzed in the FMI Report:
- Form: Powder configurations dominate due to superior surface area coverage.
- End Use: Pharmaceuticals capture a 21.2% share driven by strict formulation guidelines.
- Geography: Asian volume manufacturing contrasts with highly regulated Western environments.
- Analyst Opinion at FMI:
- Nandini Roy Choudhury, Principal Consultant for Food & Beverage at Future Market Insights, opines, “As nutraceutical brands pivot toward plant-based positioning, magnesium stearate sourcing is under sharper scrutiny. My team’s findings show that consumers are increasingly aware of excipient origins, compelling brands to prioritize non-GMO and allergen-free vegetable sources. In this environment, excipient selection becomes part of brand storytelling, not just formulation science, reshaping procurement strategies across supplement value chains.”
- Strategic Implications/Executive Takeaways:
- Secure long-term supply agreements to buffer against oleochemical feedstock fluctuations.
- Invest in low-endotoxin manufacturing lines to target premium biopharma accounts.
- Expand regional distribution networks to support localized tablet production hubs.
- Methodology:
- Validated through first-hand corporate production and capacity data.
- Zero reliance on speculative third-party market research reports.
- Based on verifiable industrial benchmarks and verified sources.
“Excipients, such as magnesium stearate, silicon dioxide, stearic acid, talc, etc., are just a few of the various excipients consumers are looking to replace,” said Prestipino. “Some of the many reasons for these requests are potential health concerns, widely covered lawsuits and sustainability awareness.” [11]
Magnesium stearate industry growth is strongest in Asia, where China and India, at 7.4% and 6.9% CAGR, benefit from rapid expansion in generic drug production and rising contract manufacturing capacity. Germany, growing at 6.3%, is supported by high value biopharma output and strong healthcare spending. The United Kingdom and United States continue to generate stable demand, backed by established solid dosage manufacturing bases and strict excipient quality standards. Brazil posts moderate growth as domestic pharmaceutical manufacturing improves. Across mature Western markets, expansion remains driven more by compliance requirements and supplier consolidation than by sharp volume increases.
Magnesium Stearate Market Key Takeaways
| Metric | Value |
|---|---|
| Industry Size (2026) | USD 40.4 Billion |
| Industry Value (2036) | USD 69.2 Billion |
| CAGR (2026 to 2036) | 5.5% |
Magnesium Stearate Market Definition
Magnesium stearate operates as a functional chemical compound comprising magnesium salts of fatty acids. Formulators utilize the fine powder extensively as an essential lubricating agent during tablet compression and capsule filling. The ingredient prevents pharmaceutical materials from adhering to manufacturing equipment surfaces.
Magnesium Stearate Market Inclusions
Coverage encompasses a comprehensive analysis of industrial consumption across pharmaceutical, nutraceutical, and personal care formulations. Analysts quantify specific metallic stearates utilized explicitly as tableting lubricants or cosmetic emulsifiers. Trade values and capacities from primary chemical processors are fully integrated into the baseline models.
Magnesium Stearate Market Exclusions
The scope omits separate functional additives such as butyl stearate unless integrated into a hybrid slip-agent compound. Downstream finished goods including bottled dietary supplements or blister-packed tablets are entirely excluded. Focus remains strictly on business-to-business excipient procurement.
Magnesium Stearate Market Research Methodology
- Primary Research: Analysts conducted targeted interviews with global chemical synthesis firms, excipient distributors, and pharmaceutical procurement heads.
- Desk Research: Import trade flow proxies provided baseline consumption metrics for chemical lubricants.
- Market-Sizing and Forecasting: Analysts deployed a bottom-up conversion model tracking processed tablet output against standard recipe inclusion rates.
- Data Validation and Update Cycle: Supply-side capacity disclosures were cross-referenced with regional chemical trade flows to verify final estimates.
Segmental Analysis
Magnesium Stearate Market Analysis by Form
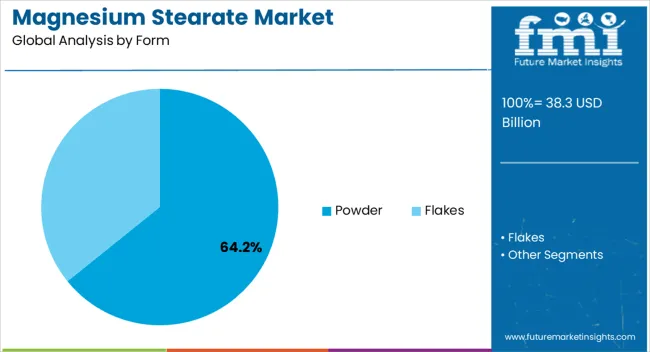
Powder forms hold a 64.2% share in 2026, supported by their strong fit with core tableting requirements. Fine particles provide effective coating of active pharmaceutical ingredients, helping reduce friction during high speed compression. FMI estimates indicate that consistent particle distribution supports smoother machine performance across large generic drug production lines. As a result, procurement teams prioritize validated texture modifying excipients to limit unplanned downtime and protect throughput.
- Procurement trigger: Buyers prefer ultra-fine powder grades to guarantee maximum blending homogeneity. Purchasing managers lock in volume contracts to ensure continuous supply for high-speed manufacturing lines.
- Technology shift: Advanced milling technologies produce tighter particle size distributions. Formulators achieve consistent dissolution profiles, ensuring final medications meet strict bioavailability standards.
- Quality signal: Low-moisture specifications act as a baseline requirement for moisture-sensitive active ingredients. Suppliers delivering verified dry powder formats secure lucrative contracts from multinational pharmaceutical processors.
Magnesium Stearate Market Analysis by End Use
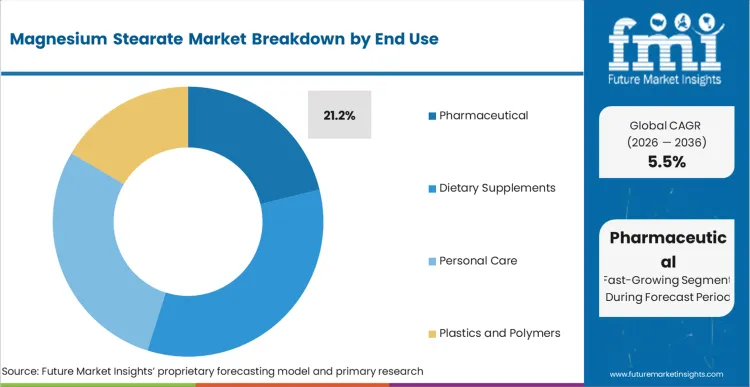
Pharmaceutical applications represent 21.2% of consumption in 2026, reflecting the essential lubrication role magnesium stearate plays in solid dose production. Formulators rely on it to support clean tablet release from metal dies during compression. FMI analysts opine that continued commercialization of new therapies is sustaining baseline demand. The FDA’s approval of 50 novel drugs in 2024 also signals ongoing expansion in pharmaceutical manufacturing activity. [3]
- Buyer priority: Drug manufacturers require certified excipients backed by exhaustive regulatory documentation. Sourcing compliant materials prevents disastrous product recalls and protects institutional brand equity.
- Formulation shift: Complex drug delivery systems demand excipients with minimal active ingredient interaction. Product development scientists select highly purified grades to maintain delicate chemical stability.
- Supply-side move: Chemical conglomerates execute strategic acquisitions to consolidate excipient portfolios. Developing specialized lubricant grades captures premium pricing from advanced biopharmaceutical clients.
Magnesium Stearate Market Analysis by Application
As a lubricating agent, the compound provides critical friction reduction across solid dose matrices. Performance under high-compression conditions makes it indispensable for contract manufacturing organizations. Integration with sorbitan esters enables the creation of highly tailored delivery systems for poorly soluble drugs. BASF targeted clean-label demand by launching the Emulgade Verde natural-based emulsifier line. [4]
- Performance trigger: Tableting facilities require stabilizers that withstand high mechanical stress. The excipient maintains formulation integrity, preventing capping or lamination in final retail products.
- Cross-industry adoption: Personal care formulators integrate metallic soaps to replace synthetic emulsifiers. Biodegradable rheology modifiers meet rising consumer expectations for sustainable cosmetics.
- Synergy mechanism: Combining specific lubricants alters the flow properties of complex powder blends. Precise ratios allow scientists to dial in exact compression characteristics, differentiating premium brands.
Magnesium Stearate Market Drivers, Restraints, and Opportunities
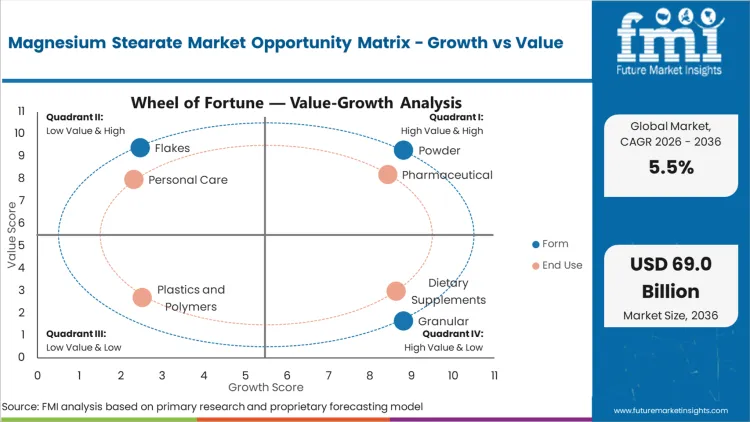
Rising retail pharmaceutical expenditure provides a massive fundamental driver for excipient consumption globally. Aging populations and expanding access to generic medications directly increase total tablet production volumes. Manufacturing facilities operating at maximum capacity consume vast quantities of chemical lubricants to maintain throughput. As per FMI's projection, intense machinery utilization requires consistent, high-grade lubrication inputs. The FDA approved 143 NDAs and BLAs under the PDUFA program in FY 2025, reflecting an enormous baseline for commercial medication output. [5]
Oleochemical supply chain volatility presents a severe structural restraint for excipient manufacturers. Fluctuations in stearic acid feedstocks create unpredictable margin compression for chemical synthesis. Chemical processors attempt to mitigate this exposure through long-term raw material contracts. Sourcing managers continuously monitor international trade flows to anticipate pricing shifts. The United States retains a 52% net import reliance for magnesium compounds, highlighting structural dependency on foreign raw materials. [6]
- Nutraceutical Expansion: Dietary supplement consumption creates parallel demand for efficient capsule-filling lubricants. Consumer shift toward preventative health routines generates volume requirements across high-speed packaging lines.
- Cosmetics Penetration: Personal care brands increasingly substitute synthetic thickeners with naturally derived metallic soaps. Cosmetic chemists utilize these compounds to stabilize emulsions and improve texture profiles.
- Polymer Processing: Industrial plastic manufacturers require stable internal lubricants for efficient extrusion. Expanding regional polymer production capacity drives concurrent procurement of high-performance metallic soaps.
Regional Analysis
Based on the regional analysis, the magnesium stearate market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania and Middle East & Africa across 40+ countries. The full report also offers market attractiveness analysis based on regional trends.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 7.4% |
| India | 6.9% |
| Germany | 6.3% |
| United Kingdom | 5.2% |
| United States | 4.7% |
| Brazil | 4.1% |
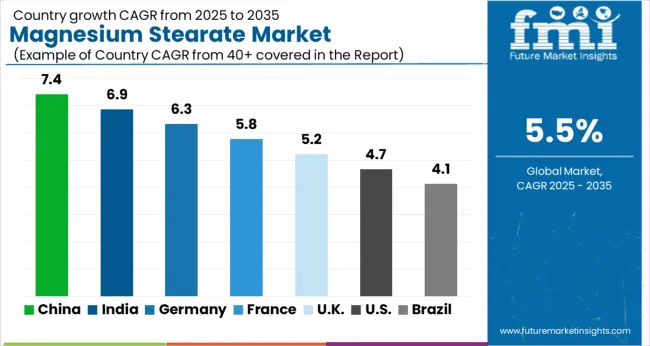
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Magnesium Stearate Market Analysis
North America exhibits mature demand driven by premium pharmaceutical manufacturing and an enormous nutraceutical sector. Leading processing facilities maintain strict auditing standards for all excipient inputs. Rigorous regulatory environments force buyers to prioritize supplier reliability over raw commodity pricing. According to FMI's estimates, this massive infrastructure commitment requires substantial, localized lubricant supply chains to function efficiently.
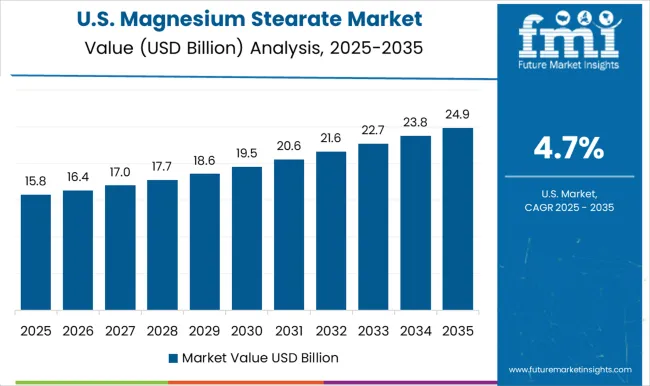
- United States: Demand is expected to rise at 4.7% CAGR through 2036, supporting a high-value pharmaceutical sector before formulation cycles translate into commercial manufacturing volumes. Formulators rely heavily on steady excipient sourcing to support massive retail healthcare networks.
FMI’s report includes a detailed assessment of North American supply dynamics. Canada and Mexico present complementary avenues as domestic processing sectors modernize. Procurement specialists monitor United States regulatory shifts to align export-oriented standards with strict magnesium oxide requirements.
Europe Magnesium Stearate Market Analysis
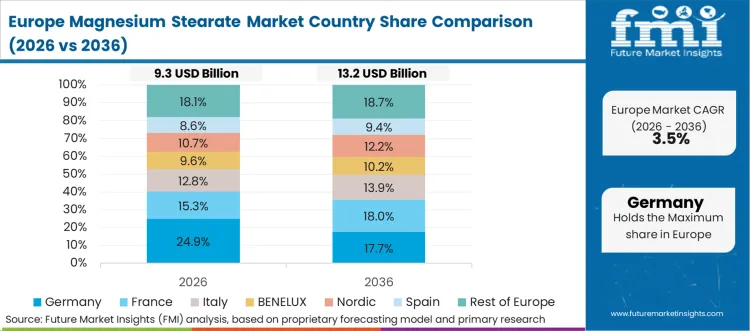
Europe operates under stringent quality mandates, with manufacturing concentrated around advanced biopharmaceuticals. Formulators actively seek highly purified grades boasting exceptionally low endotoxin limits. Regulatory compliance dictates purchasing decisions across entire continental blocks. Chemical suppliers must maintain flawless documentation to secure contracts within this territory.
- Germany: Consumption is forecast to increase at 6.3% CAGR during 2026 to 2036, with growth catalyzed by immense retail healthcare infrastructure that pulls forward continuous tableting operations. Premium medication output is supported by per capita retail pharmaceutical spending reaching EUR 743. [7]
- United Kingdom: Sales are projected to advance at 5.2% CAGR from 2026 to 2036, raising addressable pipeline constraints for vendors with robust local distribution networks. Domestic pharmaceutical hubs consume steady volumes to support National Health Service prescription fulfillment.
FMI’s report includes extensive coverage of European regulatory frameworks and ingredient sourcing impacts. France, Italy, and Spain act as vital processing centers for dietary supplements and cosmetics. Sourcing managers continuously seek sustainable sorbitan stearate alternatives to satisfy eco-conscious consumer demographics.
Asia Pacific Magnesium Stearate Market Analysis
Asia Pacific dominates global volume consumption through massive generic pharmaceutical and contract manufacturing expansion. Massive populations drive unprecedented demand for basic solid oral dosage medications. Regional processing plants operate immense high-speed tableting lines requiring constant bulk lubrication. FMI analysts opine that intense domestic competition keeps regional excipient pricing highly competitive.
- China: Growth is estimated at 7.4% CAGR through 2036, driven by colossal industrial throughput that increases the value of supply chain reliability for procurement teams. Unprecedented manufacturing scale requires massive upstream raw material availability, prompting PMC Biogenix to expand regional capacity by 50%. [8]
- India: Expansion indicates a 6.9% CAGR from 2026 to 2036 as thriving pharmaceutical export sectors prompt procurement teams to accelerate vendor qualification processes. Expanding solid-dose manufacturing capacity increases total lubricant consumption substantially.
FMI’s report includes strategic mapping of Asian chemical production capacities. Japan, South Korea, and Indonesia offer expanding avenues for high-grade polyglyceryl-6 stearate applications. Elevating regional manufacturing standards directly increases demand for premium imported excipients.
Latin America Magnesium Stearate Market Analysis
Latin America represents a steady growth frontier as domestic healthcare access improves across massive populations. Local pharmaceutical manufacturers scale production to reduce reliance on expensive imported medications. This localization trend requires reliable regional excipient distribution networks. Chemical suppliers establishing local warehousing gain commercial advantages.
- Brazil: Trajectory points to 4.1% CAGR through 2036, underpinned by improving healthcare infrastructure that steadily raises adoption intensity among domestic drug manufacturers. Expanding regional capacity is supported by an estimated per capita health spend of USD 1,700. [9]
FMI’s report includes tactical analysis of Latin American formulation trends. Mexico, Argentina, and Chile remain integral to regional volume projections. Supply chain executives monitor evolving local regulations regarding magnesium carbonate usage in pharmaceutical applications.
Competitive Aligners for Market Players
Strategic acquisitions fundamentally alter competitive positioning among top-tier excipient suppliers. Massive chemical conglomerates absorb specialized entities to construct comprehensive, globally integrated formulation portfolios. Roquette completed its acquisition of IFF Pharma Solutions, strengthening global supply capabilities across the excipient landscape. [10]
Manufacturing localization emerges as a critical strategy to capture regional market share. Establishing production or packaging facilities near major pharmaceutical hubs drastically reduces logistical friction for downstream buyers. As per FMI's projection, proximity guarantees rapid delivery schedules required for uninterrupted commercial drug production.
Product innovation targets highly specific regulatory and formulation challenges to secure premium pricing. Developing ultra-pure grades addresses critical concerns surrounding active ingredient degradation. Upgrading purification processes allows chemical manufacturers to differentiate commodity lubricants into high-value specialty ingredients.
Recent Developments:
The report includes full coverage of key trends from competitive benchmarking. Some of the recent developments covered in the reports:
- In October 2024, Clariant launched eight new pharmaceutical ingredient products at CPHI Milan 2024. [2]
- In October 2025, ACG announced a USD 200 million phased investment for its first United States capsule manufacturing facility. [1]
Key Players in Magnesium Stearate
- Roquette
- Clariant
- BASF
- Sudeep Pharma
- Valtris Specialty Chemicals
- Baerlocher
- PMC Biogenix
- Faci Spa
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 40.4 billion (2026) to USD 69.2 billion (2036), at a CAGR of 5.5% |
| Market Definition | Magnesium stearate is a functional chemical compound comprising magnesium salts of fatty acids, utilized extensively as an essential lubricating agent during tablet compression and capsule filling. |
| Form Segmentation | Powder, Granular, Flakes |
| End Use Segmentation | Pharmaceuticals, Dietary Supplements, Personal Care, Plastics and Polymers, Others |
| Application Coverage | Lubricant, Emulsifier, Binder, Others |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | United States, United Kingdom, China, India, Germany, Brazil, and 40+ countries |
| Key Companies Profiled | Roquette, Clariant, BASF, Sudeep Pharma, Valtris Specialty Chemicals, Baerlocher |
| Forecast Period | 2026 to 2036 |
| Approach | Bottom-up formulation analysis validated through primary extraction capacity interviews and international trade flow proxies. |
Magnesium Stearate Market Analysis by Segments
Form:
- Powder
- Granular
- Flakes
End Use:
- Pharmaceuticals
- Dietary Supplements
- Personal Care
- Plastics and Polymers
- Others
Application:
- Lubricant
- Emulsifier
- Binder
- Others
Region:
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Bibliography
- [1] ACG. (2025). ACG to invest $200 million in USA capsule manufacturing facility.
- [2] Clariant. (2024). Clariant introduces new excipients at CPHI Milan.
- [3] USA Food and Drug Administration. (2024). New Drug Therapy Approvals 2024.
- [4] BASF. (2024). BASF introduces Emulgade Verde.
- [5] USA Food and Drug Administration. (2025). FY2025 PDUFA Real Time totals.
- [6] USA Geological Survey. (2025). Magnesium Compounds Mineral Commodity Summaries 2025.
- [7] OECD. (2025). Germany Country Health Profile 2025.
- [8] Business Wire. (2024). PMC Biogenix announces expansion in South Korea.
- [9] OECD. (2025). Institutionalizing health accounts in Brazil.
- [10] Roquette Frères. (2025). Roquette completes acquisition of IFF Pharma Solutions.
- [11] NIE Magazine. (2024). Excipient evolution: The active ingredients may be the star of the show, but excipients are the workhorses behind the scenes.
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
This Report Addresses
- Market intelligence to enable structured strategic decision making across global chemical excipient economies.
- Market size estimation and 10 year revenue forecasts from 2026 to 2036, supported by validated capacity benchmarks.
- Growth opportunity mapping across powder and granular formats with an emphasis on pharmaceutical transitions.
- Segment and regional revenue forecasts covering dietary supplement, personal care, and polymer applications.
- Competition strategy assessment including corporate acquisitions and supply chain integration models.
- Product reformulation tracking including clean-label and biopharmaceutical active ingredient requirements.
- Regulatory impact analysis covering FDA compliance standards and international quality testing mandates.
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive use.
Frequently Asked Questions
How large is the demand for Magnesium Stearate in the global market in 2026?
Demand for Magnesium Stearate in the global market is estimated to be valued at USD 40.4 billion in 2026.
What will be the market size of Magnesium Stearate in the global market by 2036?
Market size for Magnesium Stearate is projected to reach USD 69.2 billion by 2036.
What is the expected demand growth for Magnesium Stearate in the global market between 2026 and 2036?
Demand for Magnesium Stearate in the global market is expected to grow at a CAGR of 5.5% between 2026 and 2036.
Which Form is poised to lead global sales by 2026?
Powder formats dominate global sales due to superior surface area coverage required for efficient high-speed tableting.
How significant is the role of Pharmaceuticals End Use in driving Magnesium Stearate adoption in 2026?
Pharmaceuticals capture a 21.2% share, driven by indispensable lubrication requirements across solid-dose drug manufacturing facilities.
What is driving demand in China?
Colossal industrial throughput and rapid contract manufacturing expansion drive massive adoption of chemical lubricants.
What compliance standards or regulations are referenced for China?
Strict vendor qualification processes and local industrial processing guidelines dictate excipient procurement specifications.
What is the China growth outlook in this report?
China is projected to grow at a CAGR of 7.4% during 2026 to 2036.
Why is Europe described as a priority region in this report?
Rigorous biopharmaceutical quality mandates compel drug manufacturers to source ultra-pure, low-endotoxin metallic soaps.
What type of demand dominates in Europe?
Demand revolves entirely around highly traceable, premium-grade lubricants for advanced medical formulations.
What is United States growth outlook in this report?
The United States is projected to expand at a CAGR of 4.7% during 2026 to 2036.
Does the report cover United Kingdom in its regional analysis?
Yes, the United Kingdom is included within the Europe regional scope.
What are the sources referred to for analyzing United Kingdom?
OECD health expenditure datasets and official trade flow proxies serve as primary quantitative anchors.
What is the main demand theme linked to United Kingdom in its region coverage?
Procurement relies heavily on compliant excipients to support robust National Health Service prescription fulfillment.
Does the report cover India in its regional analysis?
Yes, India is included within the Asia Pacific regional scope.
What is the main India related demand theme in its region coverage?
Thriving pharmaceutical export sectors prompt procurement teams to accelerate vendor qualification for bulk lubricants.
Which product formats or configurations are strategically important for North America supply chains?
Ultra-fine powder configurations remain vital to survive extended transcontinental shipping and satisfy homogeneous blending requirements.
What is Magnesium Stearate and what is it mainly used for?
It is a functional chemical compound comprising magnesium salts of fatty acids, primarily used as a tableting lubricant.
What does Magnesium Stearate mean in this report?
It refers to the global supply, trade, and industrial application of the essential slip agent in manufacturing.
What is included in the scope of this Magnesium Stearate report?
Scope covers consumption by form and end use across industrial pharmaceutical, dietary supplement, and personal care sectors.
What is excluded from the scope of this report?
Downstream finished retail products like bottled dietary supplements and blister-packed tablets are excluded from core volume metrics.
What does market forecast mean on this page?
The forecast represents a model-based projection mapping industrial tablet output against standard excipient inclusion rates.
How does FMI build and validate the Magnesium Stearate forecast?
Analysts deploy a bottom-up model validated through primary chemical synthesis capacity interviews and international trade flow proxies.
What does zero reliance on speculative third party market research mean here?
Primary interviews and verifiable public customs datasets are utilised to ensure baseline accuracy without secondary guessing.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Form
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Form , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Form , 2026 to 2036
- Powder
- Granular
- Flakes
- Powder
- Y to o to Y Growth Trend Analysis By Form , 2021 to 2025
- Absolute $ Opportunity Analysis By Form , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Pharmaceutical
- Dietary Supplements
- Personal Care
- Plastics and Polymers
- Pharmaceutical
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Form
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Form
- By End Use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Form
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Form
- By End Use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Form
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Form
- By End Use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Form
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Form
- By End Use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Form
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Form
- By End Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Form
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Form
- By End Use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Form
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Form
- By End Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By End Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By End Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By End Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By End Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By End Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By End Use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By End Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By End Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By End Use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By End Use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By End Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By End Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By End Use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By End Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By End Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By End Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By End Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By End Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By End Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By End Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By End Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Form
- By End Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Form
- By End Use
- Competition Analysis
- Competition Deep Dive
- Roquette
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Clariant
- BASF
- Sudeep Pharma
- Valtris Specialty Chemicals
- Baerlocher
- Roquette
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Form , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Form , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Form , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Form , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Form , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Form , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Form , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Form , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Form , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Form , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Form
- Figure 6: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by End Use
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Form , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Form , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Form
- Figure 23: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by End Use
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Form , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Form , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Form
- Figure 30: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by End Use
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Form , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Form , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Form
- Figure 37: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by End Use
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Form , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Form , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Form
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Form , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Form , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Form
- Figure 51: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by End Use
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Form , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Form , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Form
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Form , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Form , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Form
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

