Malaria Treatment Market
The Malaria Treatment Market is segmented by Drug Type (ACTs, Quinine, Antifolates, Atovaquone, Others); Route of Administration (Oral, Injectable); Distribution Channel (Public Sales, Private Sales); Product Type (Generic, Branded); Patient Group (Children, Adults, Infants), and Region. Forecast for 2026 to 2036.
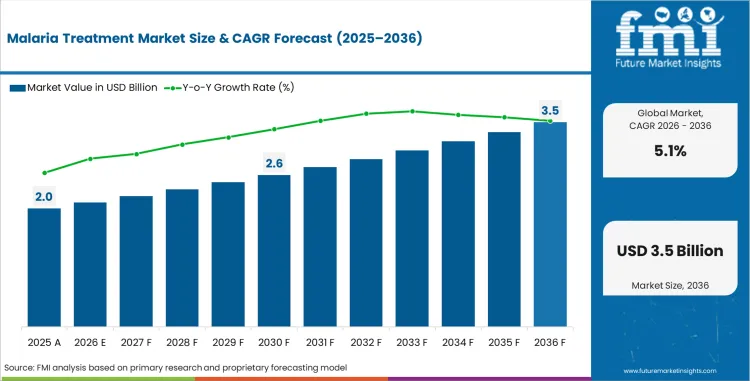
Malaria Treatment Market Size, Market Forecast and Outlook By FMI
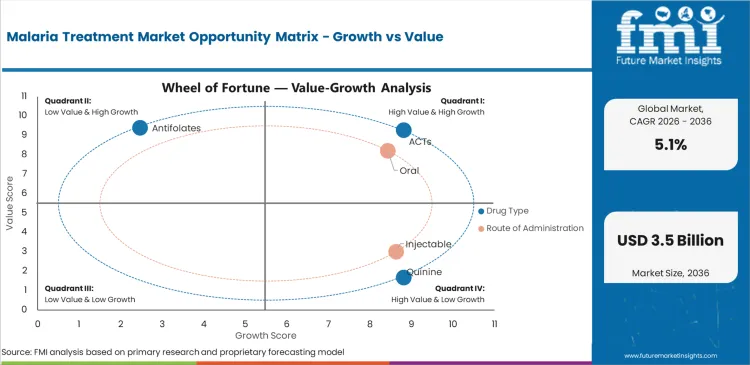
The Malaria Treatment Market is expected to move from USD 1.9 billion in 2025 to USD 2 billion in 2026. Demand is forecast to reach USD 3.3 billion by 2036 at a 5.1% CAGR from 2026 to 2036. ACTs are projected to account for 45.6% of drug type demand in 2026. Oral formulations are estimated to represent 70.7% of route demand in 2026.
Summary of the Malaria Treatment Market
- Demand and Growth Drivers
- Confirmed diagnosis supports appropriate antimalarial use across community and hospital care. Severe malaria treatment keeps injectable therapies important in referral hospitals.
- Pediatric dosing needs are creating a clearer role for age-appropriate formulations. Donor-backed and government programs keep medicine availability central to disease control.
- Product and Segment View
- ACTs are preferred for first-line treatment where falciparum malaria remains the main concern. Oral formulations suit outpatient care because they are easier to dispense and complete.
- Public sales dominate access in endemic countries through health ministries and donor programs. Generic products hold value where tender authorities need affordable and compliant medicines.
- Geography and Competitive Outlook
- India is projected to lead growth among the selected countries as district-level malaria programs support treatment demand. Brazil is expected to follow with steady demand across Amazon basin states and riverine health networks.
- The USA, UK, Germany, France, Japan and China are expected to show lower growth through imported cases and travel medicine use. Novartis, Sanofi, Cipla, GSK and Ipca compete through branded and generic antimalarial portfolios.
- Analyst Opinion
- Sabyasachi Ghosh, Associate Vice President at FMI says, “I view malaria treatment as a public health access market before I view it as a classic pharmaceutical growth market. Product use comes from confirmed fever care, severe malaria protocols, and child-focused dosing gaps. Cost pressure comes from national tenders and donor-backed purchasing.”
- Malaria Treatment Market Value Analysis
- The malaria treatment market is developing as a public health access market tied to confirmed case management.
- Adoption is being supported by continued use of ACTs in first-line malaria treatment programs.
- The market is benefiting from oral formulations that suit community and outpatient care.
- Demand is further supported by pediatric dosing needs and reliable public medicine supply.
Malaria Treatment Market Definition
The market covers medicines used to treat uncomplicated and severe malaria in human patients. Scope includes ACTs, quinine-based drugs, antifolates, atovaquone-based drugs, injectable artesunate, and supportive antimalarial regimens. It excludes mosquito control products, diagnostics sold without therapy, travel insurance services, and vaccines used only for prevention.
Malaria Treatment Market Inclusions
Included products cover ACTs, quinine, antifolates, atovaquone, oral tablets, injectable medicines, generic supply, branded supply, public sales, private sales, and treatment use across all major regions.
Malaria Treatment Market Exclusions
Excluded areas include bed nets, repellents, mosquito lamps, diagnostic kits sold without treatment, prevention-only vaccines, vector control chemicals, and non-malarial fever therapies.
Malaria Treatment Market Research Methodology
- Primary Research: FMI analysts reviewed input from public health tender authorities and hospital pharmacists serving endemic countries. Interviews focused on dosage choice and stock continuity across high burden regions.
- Desk Research: FMI reviewed WHO treatment guidance and regulator approvals. The addressable pool was defined around medicines used after malaria diagnosis or severe malaria admission.
- Market Sizing and Forecasting: The model combined 2026 baseline values and ACT share with country level treatment intensity. Forecasts were tested against disease burden and funding stability.
- Data Validation: Forecasts were checked against WHO burden data and official product approvals. Company portfolios were reviewed to confirm current participation in malaria treatment supply.
Why is the Malaria Treatment Market Growing?
- Confirmed case treatment keeps ACT use as malaria diagnosis moves closer to community care points.
- Severe malaria admissions protect injectable demand as hospitals need fast parasite clearance in high-risk patients.
- Infant dosing gaps create new demand for weight-specific medicines after recent pediatric approvals.
- Public program purchasing sustains volume since endemic countries need reliable stock before seasonal peaks.
Case burden places treatment demand inside health systems and public care pathways. The infectious disease diagnostics base supports medicine targeting through confirmed testing. Suppliers benefit when ACT packs match national guidelines and rural dispensing routines. Any stock gap can move fever care back toward poor-quality medicines. Reliable supply becomes a direct advantage for approved manufacturers.
Access funding turns disease burden into medicine volume across public systems. Treatment suppliers must meet price targets and quality checks together. Donor-backed purchasing reduces brand flexibility for public tender authorities. It rewards firms able to supply large orders with stable documentation. The forecast gains most when funding is predictable across high-burden African countries.
Market Segmentation Analysis
- ACTs are projected to account for 45.6% of drug type demand in 2026 as first-line malaria care depends on combination therapy.
- Oral formulations are estimated to represent 70.7% of route demand in 2026 since most uncomplicated malaria cases are treated outside hospitals.
- Public sales are expected to capture 70.0% of distribution demand in 2026 as national malaria programs manage large-volume medicine access.
- Generic products are projected to hold 62.0% of product type demand in 2026 as public tenders need affordable and quality-approved supply.
- Children are expected to account for 52.8% of patient group demand in 2026 as pediatric malaria care drives public treatment planning.
The Malaria Treatment Market is segmented by drug type, route of administration, distribution channel, product type, patient group, and region. Drug type includes ACTs, quinine, antifolates, atovaquone, and other antimalarial medicines. Route of administration includes oral and injectable therapies. Distribution channel covers public sales and private sales. Product type includes generic and branded medicines. Patient group includes children, adults, and infants.
Insights into the ACTs Drug Type Segment
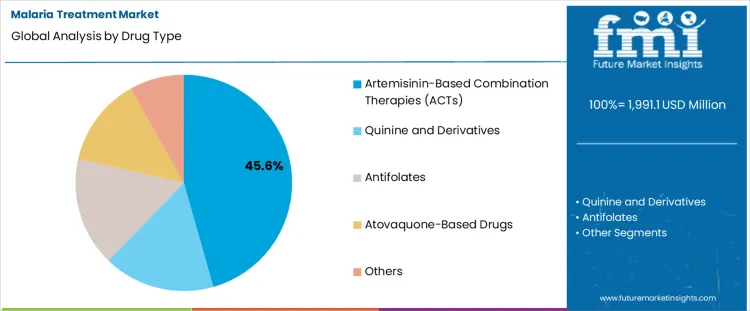
- ACTs are projected to account for 45.6% of drug type demand in 2026 as combination therapy supports first-line treatment across falciparum malaria cases.
- Quinine and atovaquone-based medicines serve narrower clinical needs where resistance risk or patient condition requires another option.
Insights into the Oral Route of Administration Segment
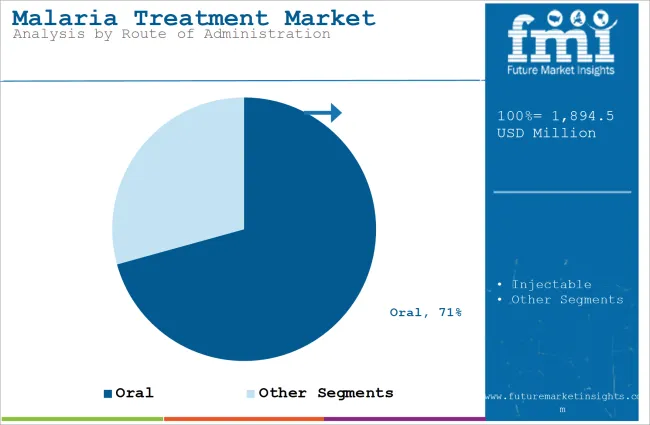
- Oral formulations are estimated to represent 70.7% of route demand in 2026 as tablets and dispersible products fit outpatient treatment workflows.
- Injectable therapies are used mainly for severe malaria, where hospitals need fast treatment for high-risk patients.
Insights into the Public Sales Distribution Channel Segment
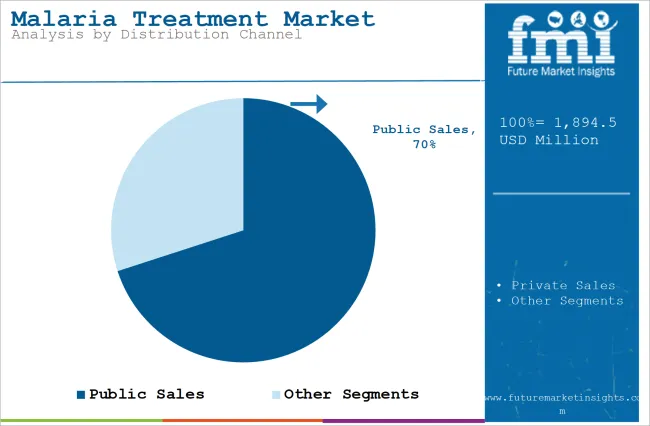
- Public sales are expected to capture 70.0% of distribution demand in 2026 as ministries and donor funded programs manage treatment access.
- Private sales support travel medicine, urban clinics and self-paid treatment in countries with smaller or imported malaria case volumes.
Insights into the Generic Product Type Segment
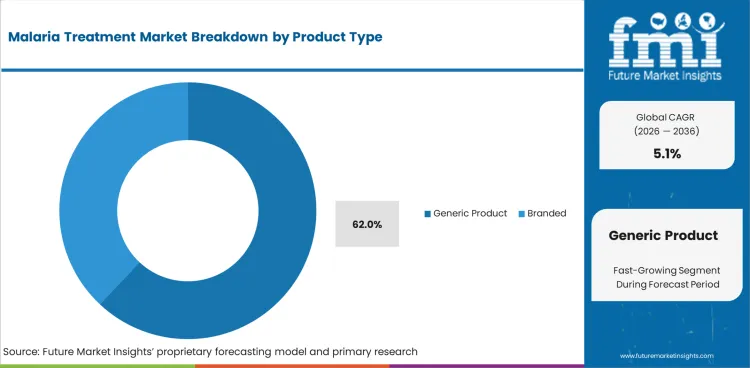
- Generic products are projected to hold 62.0% of product type demand in 2026 as public tender suthprities place price and approved quality together.
- Branded medicines retain demand in regulated hospital and travel clinics where prescriber confidence is important.
Insights into the Children Patient Group Segment
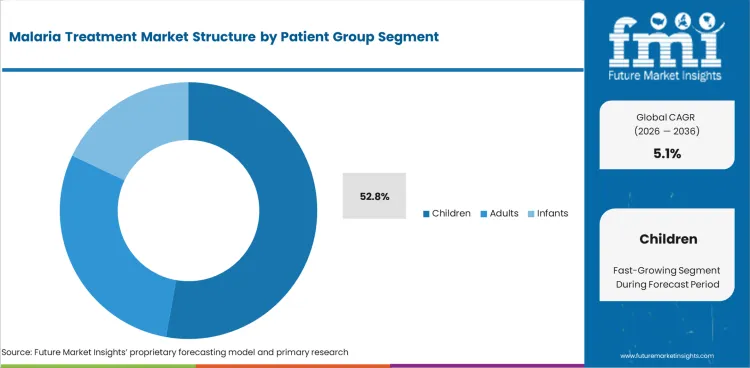
- Children are expected to account for 52.8% of patient group demand in 2026 as pediatric malaria care drives treatment planning in endemic areas.
- Adult demand is concentrated in travel medicine and imported case care, especially across the US, UK, Germany, France, Japan and China.
Malaria Treatment Market Drivers, Restraints, and Opportunities
- Guideline-based ACT use protects baseline demand across endemic countries with high falciparum malaria burden.
- Infant dosing approvals open a focused opportunity in children previously treated with older child formulations.
- Resistance surveillance can shift volumes toward newer regimens once partner drug failure increases in a country.
- Funding gaps are the main restraint as public channels depend on budgets and donor grants.
ACT Guideline Pull
Treatment demand follows clinical protocol across malaria-endemic countries. WHO guidance keeps ACTs central for uncomplicated falciparum malaria. Approved combination suppliers a predictable role in national formularies. The anti-infective vaccines rollout continue treatment need since breakthrough cases require medicine. Tender authorities look for blister packs and clear dosing tables that pass tender checks. Suppliers improve access when product match the local regimen or shelf-life needs. Regimen alignment and documentation decide access before sales messaging begins.
Infant Dosing Gap
Pediatric malaria care is moving from improvised dosing toward weight-specific products. Swissmedic authorized Riamet Baby in July 2025 for infants weighing 2 kg to 5 kg. This approval closes a treatment gap for very young patients who were often dosed with older child tablets. The decision supports smaller dose packs and caregiver-friendly administration. It creates a new product tier priced for public access. Suppliers with pediatric formulation skill gain a clearer path into child health tenders.
Analysis of Malaria Treatment Market by Key Countries
.webp)
| Country | CAGR |
|---|---|
| India | 4.8% |
| Brazil | 4.3% |
| United States | 3.8% |
| France | 3.6% |
| United Kingdom | 3.5% |
| Germany | 3.4% |
| China | 3.2% |
| Japan | 2.9% |
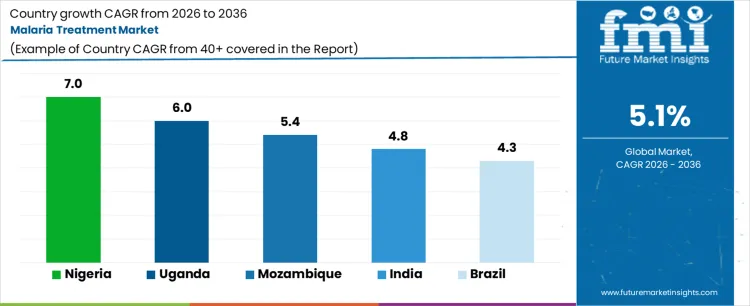
Source: FMI analysis based on primary research and proprietary forecasting model.
Malaria Treatment Market CAGR Analysis by Country
- India is projected to record a 4.8% CAGR through 2036 as treatment demand stays concentrated in endemic districts.
- Brazil is expected to expand at 4.3% CAGR through 2036 since Amazon basin states need reliable antimalarial access.
- The United States is forecast to grow at 3.8% CAGR through 2036 as imported malaria cases support hospital and travel medicine demand.
- France is expected to advance at 3.6% CAGR through 2036 given imported case care and overseas territory treatment needs.
- The United Kingdom is projected to rise at 3.5% CAGR through 2036 as travel-linked malaria cases sustain specialist treatment use.
- Germany is forecast at 3.4% CAGR through 2036 with demand tied to travel medicine and hospital-managed imported cases.
- China is expected to post 3.2% CAGR through 2036 as imported malaria surveillance supports limited treatment demand.
- Japan is projected to grow at 2.9% CAGR through 2036 as demand remains focused on imported cases and travel clinics.
Demand Outlook for Malaria Treatment Market in India
India is projected to record a 4.8% CAGR through 2036 as malaria treatment demand remains concentrated in specific districts. Demand is high in forest and tribal regions where public health workers manage confirmed cases. Odisha, Chhattisgarh, and Mizoram keep treatment needs visible despite reduced national incidence. The National Center for Vector Borne Diseases Control guides treatment standards across state programs. Suppliers mainly compete on affordable generics and reliable public channel supply. Better district surveillance would make antimalarial stock planning more precise.
- Odisha stays important due to tribal districts with recurring malaria clusters during monsoon periods.
- Mizoram needs focused treatment supply as northeastern transmission pockets require steady program coverage.
- Chhattisgarh supports district-level demand through forest-area surveillance and public treatment access.
Sales Analysis of Malaria Treatment Market in Brazil
The Brazilian Unified Health System supports standardized treatment in malaria-prone areas. Amazonas and Pará account for the clearest need through public health networks and river-based care routes. Brazil is expected to expand at 4.3% CAGR through 2036 as malaria treatment demand stays centered on the Amazon basin. Demand is concentrated across endemic municipalities. Suppliers need registration readiness and supply models suited to remote locations. Improved access along forest and mining corridors would increase confirmed treatment use.
- Amazonas State is the core geography for public malaria treatment planning.
- Pará State needs antimalarial access across forest and riverine municipalities.
- The Brazilian Unified Health System supports standardized care across endemic areas.
Demand Analysis of Malaria Treatment Market in the United States
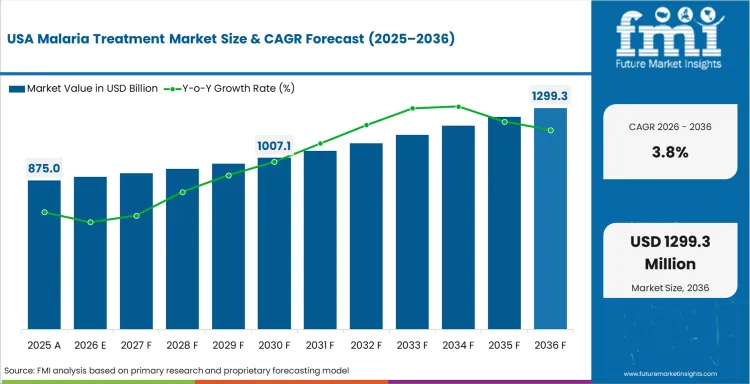
Demand in United States comes from hospitals, travel clinics and military-linked health systems. The United States is forecast to grow at 3.8% CAGR through 2036 as malaria cases are mainly imported. Severe malaria cases create need for injectable therapy in regulated hospital channels. CDC guidance supports case recognition and treatment selection across clinical care. Suppliers compete through regulatory approval and product documentation. Any increase in overseas travel to endemic regions would raise treatment use in specialist care.
- CDC guidance supports malaria diagnosis and treatment decisions across US clinical practice.
- Large hospitals in New York and Houston manage imported fever cases from global travel.
- Military health facilities need antimalarial readiness for personnel returning from endemic regions.
Opportunity Analysis of Malaria Treatment Market in France
Paris-area hospitals manage many tropical disease referrals tied to travel and migration. French Guiana adds a direct endemic treatment need within French public health coverage. France is expected to advance at 3.6% CAGR through 2036 as imported case care and overseas territory treatment needs support demand. Demand favors regulated products with clear prescribing guidance. Suppliers need hospital access and strong documentation for specialist care. Better surveillance across overseas territories would improve stock planning for public treatment channels.
- French Guiana creates a direct malaria care need within the French health system.
- Paris hospitals support imported malaria care through tropical disease units.
- Santé publique France helps track malaria risk through surveillance and public health guidance.
Future Outlook for Malaria Treatment Market in the United Kingdom
The United Kingdom is projected to rise at 3.5% CAGR through 2036 as travel-linked malaria cases sustain specialist treatment use. London and other large cities see cases tied to travel from endemic regions. Hospital care and travel medicine clinics decide most product use. NHS guidance and specialist infectious disease units support treatment decisions. Suppliers need regulated access and clear dosage information for clinical users. Growth depends on travel patterns and awareness among returning travelers with fever.
- London hospitals manage imported malaria cases through infectious disease and tropical medicine services.
- UKHSA surveillance supports public health tracking of travel-linked malaria.
- NHS travel health guidance helps direct patients toward prevention and prompt treatment.
In-depth Analysis of Malaria Treatment Market in Germany
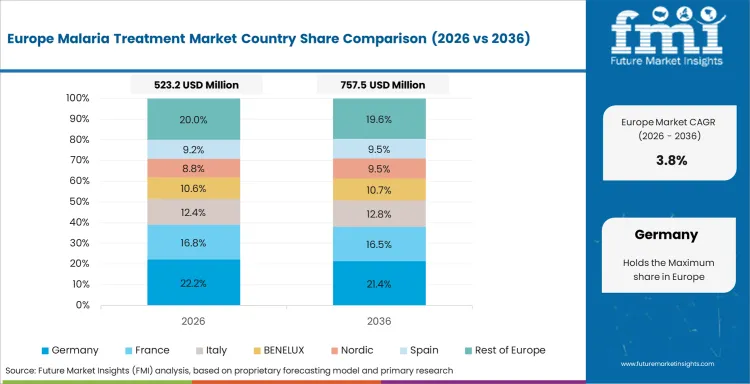
Germany is forecast at 3.4% CAGR through 2036 with malaria treatment demand tied to travel medicine and hospital-managed imported cases. Large cities with international travel links account for most clinical need. Hospitals and travel medicine specialists guide product use rather than broad retail channels. Demand favors approved medicines with clear safety documentation. Suppliers depend on hospital listings and physician familiarity. A rise in travel to endemic countries would increase treatment need across specialist clinics.
- Berlin and Frankfurt support imported case management through large hospital networks.
- Robert Koch Institute surveillance helps track malaria cases reported in Germany.
- Travel medicine clinics guide prevention advice and post-travel fever referrals.
Demand Outlook for Malaria Treatment Market in China
Current use in China is mainly tied to imported cases among travelers and overseas workers. China is expected to post 3.2% CAGR through 2036 as imported malaria surveillance supports limited treatment demand. Provincial surveillance helps identify cases before wider spread can occur. Hospitals in major travel hubs manage most treatment needs. Suppliers need registration strength and access to regulated hospital channels. Demand can rise when overseas labor and travel to endemic regions increase.
- Yunnan stays important due to border surveillance and imported case monitoring.
- Shanghai hospitals support treatment for travelers returning from endemic countries.
- China CDC guidance supports case detection and malaria elimination safeguards.
Sales Analysis of Malaria Treatment Market in Japan
Japan is projected to grow at 2.9% CAGR through 2036 as demand remains focused on imported cases and travel clinics. Demand is concentrated in travel medicine and hospital infectious disease care. Tokyo and Osaka account for much of the clinical access base. Prescribers need approved medicines with clear dosing and safety guidance. Suppliers are evaluated on specialist availability and regulated clinical access. Higher outbound travel to endemic countries would raise low-volume treatment demand.
- Tokyo hospitals manage imported malaria through infectious disease and tropical medicine units.
- Osaka travel medicine clinics support prevention advice and post-travel referral.
- Japan’s public health surveillance helps track imported infectious disease risks.
Competitive Landscape and Strategic Positioning
- Market concentration is moderate as branded ACT leaders compete with generic suppliers in public tenders.
- Large firms hold advantage through WHO-aligned dossiers and multi-country registration coverage.
- Regional players compete when they can meet price targets and maintain steady ACT supply.
- Entry barriers include clinical evidence, quality assurance, tropical stability, and public tender access.
Competition centers on supply trust more than promotional reach. Novartis strengthened its position when Coartem Baby received Swissmedic approval in July 2025. The approval is important because infants had a dosing gap in many endemic countries. A supplier solving a defined dosing problem can defend value even in price-controlled channels. The vaccines rollout may lower some severe cases over time. Treatment firms need resilient public access as breakthrough infections will require therapy.
Regulatory milestones can change tender eligibility before they change shipment volume. WHO prequalified Coartem Baby in April 2026 for newborns and young infants. This status supports donor-funded and public sector buying by UN-linked channels. Companies with strong regulatory functions can move faster after a new formulation is cleared. Generic suppliers answer through scale and cost control. The quinoline value chain is relevant where chemistry and antimalarial compounds overlap.
Key Companies in the Malaria Treatment Market
The competitive structure mixes branded formulation leaders with generic suppliers and severe care specialists.
- Global Leaders: Novartis, Sanofi, and GSK have broad malaria care visibility through branded therapies and prevention portfolios. Novartis has the clearest treatment position through Coartem and infant-specific Coartem Baby.
- Generic Suppliers: Cipla, Ipca Laboratories, and Strides Pharma compete through ACTs and lower-cost antimalarial supply across high-burden regions.
- Specialist Access Players: Pfizer and Amivas support selected antimalarial portfolios or hospital access pathways.
Competitive Benchmarking: Malaria Treatment Market
| Company | ACT Portfolio Depth | Pediatric Formulation Reach | Tender Supply Readiness | Geographic Footprint |
|---|---|---|---|---|
| Novartis | High | High | Strong | Global |
| Sanofi | High | Medium | Strong | Global |
| Cipla | High | Medium | Strong | Africa and Asia |
| GSK | Medium | High | Strong | Global |
| Pfizer | Medium | Low | Moderate | Global |
| Ipca Laboratories | High | Medium | Strong | Asia and Africa |
| Strides Pharma | Medium | Low | Moderate | Asia and Africa |
| Amivas | Low | Low | Moderate | United States and regulated markets |
Source: Future Market Insights competitive analysis, 2026. Ratings reflect relative positioning based on ACT portfolio depth, pediatric formulation reach, and tender supply readiness.
Key Developments in Malaria Treatment Market
- In October 2025, Novartis and Medicines for Malaria Venture launched Coartem Baby in Ghana. Ghana became the first malaria-endemic country to launch the infant-specific malaria treatment.
- In July 2025, Ipca Laboratories had its Primaquine 15 mg tablets included in the WHO list of prequalified medicines for malaria treatment. The product supports radical cure for Plasmodium vivax and Plasmodium ovale malaria.
Key Players in the Malaria Treatment Market
Global Leaders
- Novartis
- Sanofi
- GSK
Generic Suppliers
- Cipla
- Ipca Laboratories
- Strides Pharma
Specialist Access Players
- Pfizer
- Amivas
Report Scope and Coverage
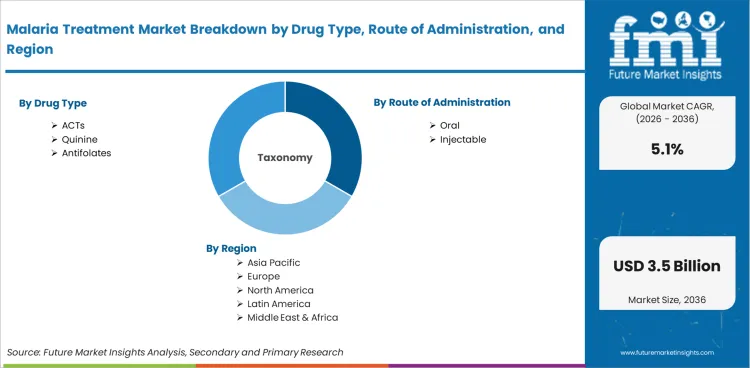
Malaria Treatment Market Breakdown By Drug Type, Route, Distribution Channel, Product Type, Patient Group, and Region
| Parameter | Details |
|---|---|
| Quantitative Units | USD 2 billion in 2026 to USD 3.3 billion by 2036, at a CAGR of 5.1% |
| Market Definition | Medicines used to treat uncomplicated and severe malaria in human patients across public and private care channels. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | UK, US, Japan, China, India, Germany, France, Brazil, and 30+ countries |
| Key Companies Profiled | Novartis, Sanofi, Cipla, GSK, Pfizer, Ipca Laboratories, Strides Pharma |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down methodology starting with verified treatment access signals and country burden patterns |
Malaria Treatment Market by Segments
Malaria Treatment Market Segmented by Drug Type:
- ACTs
- Quinine
- Antifolates
- Atovaquone
- Others
Malaria Treatment Market Segmented by Route of Administration:
- Oral
- Injectable
Malaria Treatment Market Segmented by Distribution Channel:
- Public Sales
- Private Sales
Malaria Treatment Market Segmented by Product Type:
- Generic
- Branded
Malaria Treatment Market Segmented by Patient Group:
- Children
- Adults
- Infants
Malaria Treatment Market by Region:
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan and Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia and New Zealand
- Rest of South Asia and Pacific
- Middle East and Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East and Africa
Research Sources and Bibliography
- Novartis. (2025, July 8). Novartis receives approval for first malaria medicine for newborn babies and young infants.
- Ipca Laboratories Limited. (2025). WHO Public Assessment Report: Primaquine 15 mg tablets. World Health Organization.
- World Health Organization. (2025, December 4). World malaria report 2025. World Health Organization.
- The Global Fund. (2025). Results report 2025. The Global Fund to Fight AIDS, Tuberculosis and Malaria.
- UNICEF. (2025, November 23). Gavi and UNICEF announce equitable pricing deal for malaria vaccine to protect 7 million more children by end of decade. UNICEF.
- Swissmedic. (2025, July). Swissmedic grants authorisation for a paediatric formulation of an antimalarial medicine. Swiss Agency for Therapeutic Products.
- Medicines for Malaria Venture. (2026, April 24). WHO prequalification of Coartem Baby marks breakthrough in treating malaria. MMV.
The bibliography is provided for reader reference and uses primary non-commercial government, regulatory, academic, or official trade body publications where possible.
This Report Answers
- What is the estimated value of malaria treatment demand in 2026 and by 2036?
- What CAGR is expected for malaria treatment between 2026 and 2036?
- Which drug type is projected to account for the leading share in 2026?
- Which route of administration is expected to lead malaria treatment use in 2026?
- How do public sales channels affect medicine access across endemic countries?
- Which countries are forecast to record faster malaria treatment growth through 2036?
- How do infant formulations change supplier positioning in malaria care?
- Which companies compete across branded ACTs and generic antimalarial supply?
- What products are included and excluded from the market definition?
- How does FMI validate malaria treatment forecasts using burden and channel evidence?
Frequently Asked Questions
What is the global market demand for malaria treatment in 2026?
In 2026, the global market is expected to be worth USD 2 billion with public programs guiding most volume.
What is the forecast value by 2036?
The market is forecast to reach USD 3.3 billion by 2036 as confirmed treatment access improves in endemic countries.
What CAGR is projected from 2026 to 2036?
Demand is expected to advance at 5.1% CAGR through 2036 based on treatment access and program continuity.
Which drug type is expected to lead in 2026?
ACTs are projected to account for 45.6% share in 2026 as combination therapy stays central to malaria care.
Which route of administration is expected to lead?
Oral formulations are estimated to represent 70.7% share in 2026 as outpatient and community care use tablets.
Which country is projected to grow fastest in the malaria treatment market?
The USA is projected to record the fastest growth at 5.7% CAGR through 2036 due to travel medicine and imported case care.
What does the market include?
The market includes ACTs, quinine, antifolates, atovaquone, injectable therapies and supportive antimalarial regimens for human treatment.
How was the forecast built?
FMI combined treatment value anchors, burden patterns, route shares and company portfolio checks to validate the forecast.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Drug Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Drug Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Drug Type , 2026 to 2036
- ACTs
- Quinine
- Antifolates
- ACTs
- Y to o to Y Growth Trend Analysis By Drug Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Drug Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Route of Administration
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Route of Administration, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Route of Administration, 2026 to 2036
- Oral
- Injectable
- Oral
- Y to o to Y Growth Trend Analysis By Route of Administration, 2021 to 2025
- Absolute $ Opportunity Analysis By Route of Administration, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Public Sales
- Private Sales
- Public Sales
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type, 2026 to 2036
- Generic Product
- Branded
- Generic Product
- Y to o to Y Growth Trend Analysis By Product Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Patient Group
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Patient Group, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Patient Group, 2026 to 2036
- Children
- Adults
- Infants
- Children
- Y to o to Y Growth Trend Analysis By Patient Group, 2021 to 2025
- Absolute $ Opportunity Analysis By Patient Group, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- By Country
- Market Attractiveness Analysis
- By Country
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Drug Type
- By Route of Administration
- By Distribution Channel
- By Product Type
- By Patient Group
- Competition Analysis
- Competition Deep Dive
- Novartis
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Sanofi
- GSK
- Cipla
- Ipca Laboratories
- Strides Pharma
- Novartis
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Drug Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Patient Group, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Drug Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Patient Group, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Drug Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Patient Group, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Drug Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Patient Group, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Drug Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Patient Group, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Drug Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Patient Group, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Drug Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Patient Group, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Drug Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Patient Group, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Drug Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Drug Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Drug Type
- Figure 6: Global Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Route of Administration
- Figure 9: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Distribution Channel
- Figure 12: Global Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Product Type
- Figure 15: Global Market Value Share and BPS Analysis by Patient Group, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Patient Group, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Patient Group
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Drug Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Drug Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Drug Type
- Figure 32: North America Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Route of Administration
- Figure 35: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Distribution Channel
- Figure 38: North America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Product Type
- Figure 41: North America Market Value Share and BPS Analysis by Patient Group, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Patient Group, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Patient Group
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Drug Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Drug Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Drug Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Route of Administration
- Figure 51: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 54: Latin America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Product Type
- Figure 57: Latin America Market Value Share and BPS Analysis by Patient Group, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Patient Group, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Patient Group
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Drug Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Drug Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Drug Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Route of Administration
- Figure 67: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 70: Western Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Product Type
- Figure 73: Western Europe Market Value Share and BPS Analysis by Patient Group, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Patient Group, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Patient Group
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Drug Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Drug Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Drug Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Route of Administration
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Patient Group, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Patient Group, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Patient Group
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Drug Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Drug Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Drug Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Route of Administration
- Figure 99: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 102: East Asia Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Product Type
- Figure 105: East Asia Market Value Share and BPS Analysis by Patient Group, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Patient Group, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Patient Group
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Drug Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Drug Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Drug Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Route of Administration
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Patient Group, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Patient Group, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Patient Group
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Drug Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Drug Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Drug Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Route of Administration
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Patient Group, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Patient Group, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Patient Group
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

