The influenza vaccine patch market size was valued at USD 19.5 million in 2025. The microneedle flu vaccine market is poised to surpass USD 24.0 million in 2026 at a CAGR of 23.00% during the forecast period. Demand outlook propels the valuation to USD 190.2 million through 2036 as public health procurement models switch from cold-chain dependent liquid vials to thermostable solid-dose formats, establishing the positive trend for influenza vaccine patch forecast 2036.
Immunization programs are fundamentally re-evaluating their delivery infrastructure to eliminate the logistical friction of refrigerated transport and sharps disposal. Buyers exploring the influenza vaccine patch market are transitioning from purchasing doses to acquiring complete delivery modalities, forcing microneedle drug delivery systems out of experimental pilot phases and into commercial supply agreements. National health agencies that delay integrating these solid-state formats forfeit the ability to rapidly distribute a pandemic influenza microneedle patch outside of clinical settings. The actual driver of this transition is not patient preference for pain-free administration, but the operational imperative to decouple vaccination rates from the availability of trained healthcare personnel.
Microneedle Patch Influenza Vaccine Delivery Market Definition
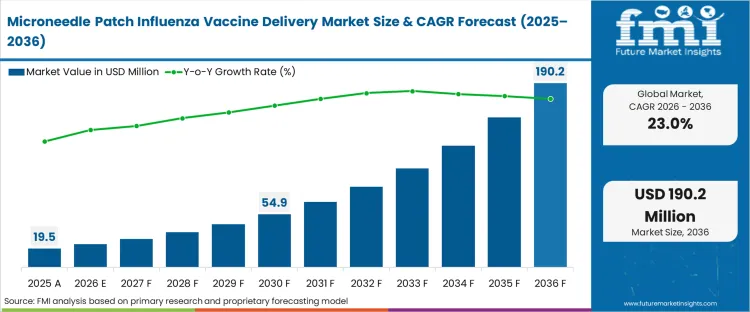
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 24.0 million |
| Industry Value (2036) | USD 190.2 million |
| CAGR (2026-2036) | 23.00% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Before this needle-free influenza vaccine patch can displace conventional syringes at scale, national regulatory authorities must establish standardized monographs for evaluating combined vaccine-device immunogenicity. Once this regulatory gateway is defined, biopharma developers can seamlessly transition legacy liquid formulations to patch substrates without re-running phase three efficacy trials from scratch.
Australia is expected to advance at 25.1%, followed closely by the United States at 24.6% and the United Kingdom at 22.9%. Germany and Canada track at 22.4% and 22.1% respectively, while the China influenza vaccine patch opportunity is anticipated to expand at 21.8%, and India garners 21.2%. The divergence across these jurisdictions stems directly from their baseline integration of pharmacy-led and self-administered immunization frameworks, rather than mere population size.
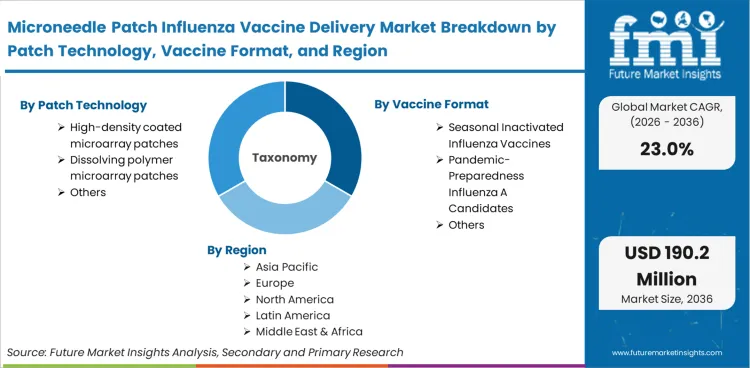
The microneedle patch influenza vaccine delivery market encompasses the commercialization and procurement of solid-state transdermal delivery devices specifically formulated with influenza antigens. This market is analytically distinct from general wearable injectors and legacy transdermal drug patches because it explicitly tracks the convergence of vaccine stabilization chemistry with micro-scale epidermal disruption technology, capturing the value of the delivery device, the stabilization matrix, and the specific application to seasonal or pandemic influenza formats.
Scope inclusions encompass an influenza vaccine microarray patch, including high-density coated patches, dissolving polymer matrices, and silk-based architectures pre-loaded with inactivated, live-attenuated, or mRNA influenza antigens. The market captures revenue generated through early-stage clinical procurement, public health stockpiling contracts, and direct-to-pharmacy seasonal distributions. Additionally, microarray patch licensing milestones and influenza vaccine patch licensing opportunities tied directly to commercialization are fully included in the valuation.
Standalone liquid influenza vaccines delivered via conventional needle and syringe are explicitly excluded, as they do not possess the thermostable or self-administration characteristics that define this market's boundary. Hollow microneedle devices that require a separate liquid reservoir and plunger mechanism are excluded if they do not feature pre-loaded, integrated influenza formulations. General transdermal patches for continuous small-molecule drug delivery are also excluded to isolate the specific dynamics of epidermal vaccine immunology.
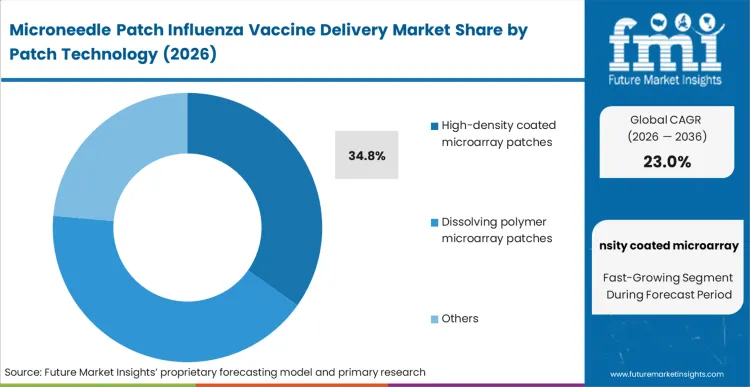
The legacy approach of liquid injection inherently relies on muscle tissue, which is immunologically sparse compared to the skin. The HD-MAP influenza vaccine dominates this dimension, securing a 34.8% share in 2026, because it successfully targets the antigen-presenting Langerhans cells in the epidermis, enabling profound dose sparing.
According to FMI's estimates, this influenza patch dose-sparing technology is the critical economic lever that makes the format viable for seasonal campaigns where antigen yield is frequently a manufacturing bottleneck. These seasonal influenza vaccines formulators leverage coating techniques that dry the active ingredient instantly, bypassing the degradation risks associated with thermal or chemical integration methods. Developers who commit to a dissolving microneedle influenza vaccine matrix instead must clear significantly higher regulatory hurdles regarding the clearance of the polymer from the human body.
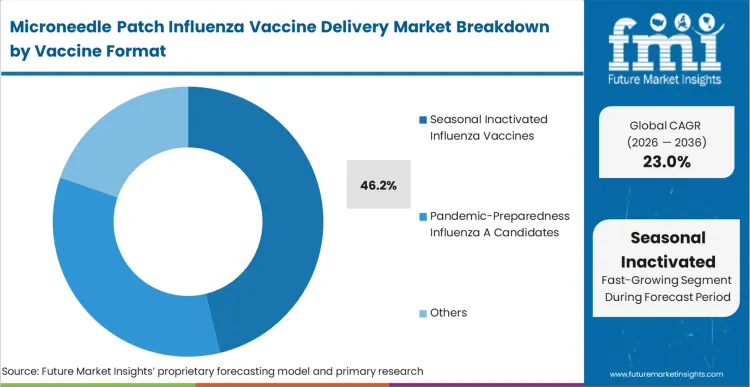
Seasonal inactivated influenza vaccines hold 46.2% of the market in 2026, primarily because this format provides the massive, predictable volume required to justify the capital expenditure of building custom patch manufacturing lines. The established safety profile of inactivated antigens reduces the combination-product regulatory risk, allowing developers to focus purely on the delivery mechanism rather than defending a novel immunology pathway. Biopharma sponsors utilize these established vaccines profile to advance clinical trials at an accelerated pace, securing non-dilutive government funding based on clear feasibility. However, as manufacturing yields scale, the integration within the mRNA vaccine patch market will drastically alter the value proposition. Formulators who fail to prove their patch technology is compatible with next-generation mRNA lipid nanoparticles will be excluded from future pandemic preparedness contracts entirely.
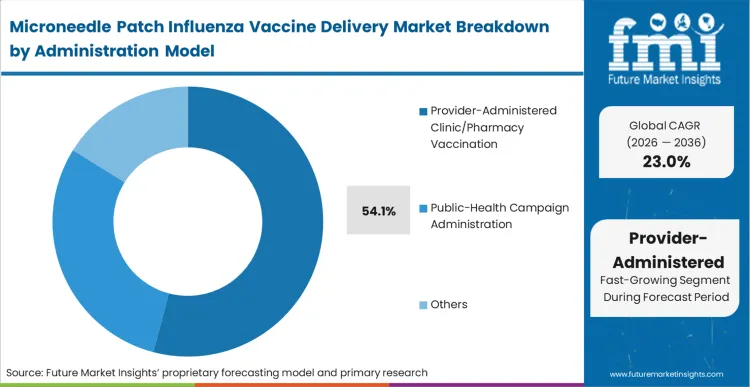
Provider-administered clinic and pharmacy vaccination holds 54.1% of the market in 2026 as it represents the mandatory first step in commercializing any novel delivery device. Regulatory agencies globally require extensive post-market surveillance in controlled settings before authorising full direct-to-patient deployment.
In FMI's view, this institutional oversight phase is not a long-term limitation, but a necessary bridge to establish real-world immunogenicity equivalence. Retail pharmacy chains deploy these drug delivery technology formats to increase their throughput during peak seasonal demand, effectively treating more patients per hour than is possible with traditional sharps. Health systems that delay integrating a self-administered flu vaccine patch pathway will rapidly lose their commercial distribution footprint once competitors roll out verified mail-to-home models.
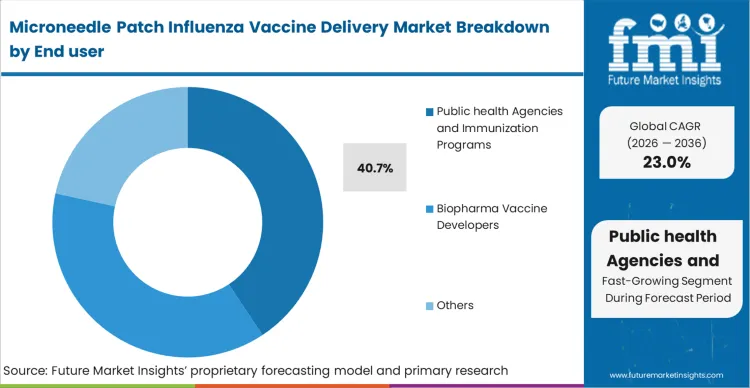
A public health immunization microneedle patch deployment accounts for 40.7% of the market in 2026, functioning as the primary commercial catalyst because agency purchasing power dictates the survival of early-stage needle-free injection system innovators. The massive upfront capital required to build continuous microarray production lines cannot be justified by fragmented retail demand; it requires the volume guarantees that only national stockpiles can provide.
FMI analysts opine that these agencies are actively moving forward-purchasing agreements specifically to derisk the industrialization phase for preferred device partners. Biopharma developers operate in parallel, licensing the technology to protect their legacy antigen portfolios from obsolescence. Manufacturers that cannot secure public-sector validation find themselves unable to raise the capital necessary to reach commercial manufacturing scale.
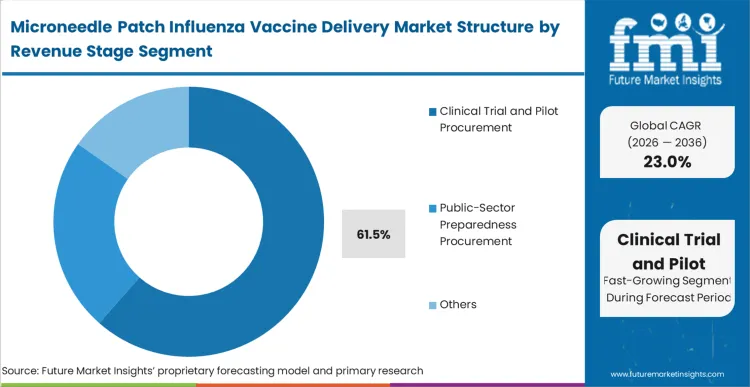
Clinical trial and pilot procurement dominates this dimension with 61.5% share in 2026, an accurate reflection of a market operating right at the commercial inflection point. The vast majority of immediate capital flow consists of milestone payments, phase II/III material supply contracts, and non-dilutive defense grants rather than routine seasonal sales. This needle-free injectors revenue will invert violently over the next 48 months as late-stage candidates transition into approved commercial products. Public-sector preparedness procurement acts as the immediate secondary tier, absorbing pilot capacity as it comes online. Vendors who mismanage their clinical material supply chain jeopardize not only their trial timelines but their ability to demonstrate industrial readiness to major government buyers.
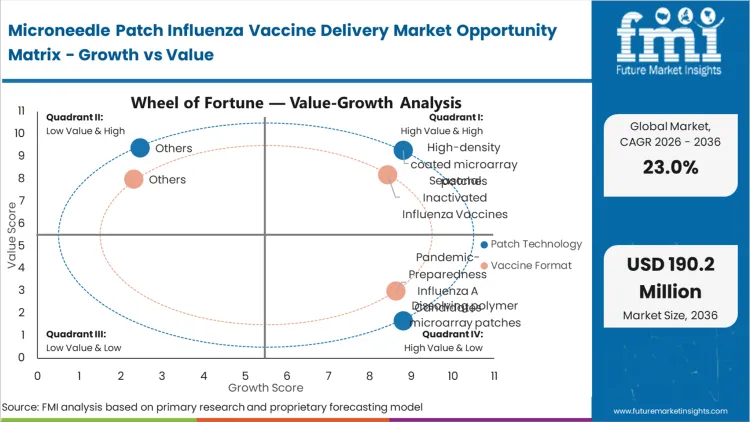
The necessity to deploy countermeasures outside of established clinical infrastructure compels national defense and public health agencies to aggressively fund thermostable delivery solutions. The vulnerability of centralized cold-chain networks during crisis scenarios forces logistics directors to seek alternatives that can sit in ambient warehouses for years and be distributed via conventional mail. This pressure moves needle-free vaccine injectors from an optional consumer convenience to a national security imperative. Agencies that fail to secure advance purchase agreements for solid-state formats will remain dangerously tethered to the availability of refrigerated transport and certified nursing staff.
The primary structural friction dragging adoption is the total absence of a unified regulatory monograph for evaluating solid-dose epidermal vaccine-device combinations, which represents one of the steepest influenza vaccine patch commercialization barriers. Unlike conventional syringes which are universally characterized, every microarray architecture currently requires a bespoke regulatory pathway, dramatically increasing the cost and timeline for phase III validation. While the FDA and EMA are issuing ad-hoc guidance for specific clinical trial sponsors, this fragmented approach prevents smaller formulation developers from entering the space.
The regional assessment divides the microneedle patch influenza vaccine delivery market into North America, Europe, and the Asia Pacific region, covering more than 40 countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| Australia | 25.1% |
| United States | 24.6% |
| United Kingdom | 22.9% |
| Germany | 22.4% |
| Canada | 22.1% |
| China | 21.8% |
| India | 21.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
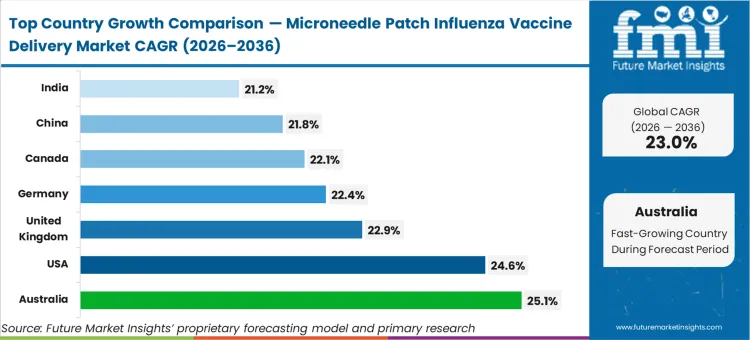
Australia's mandate to aggressively integrate premium immunization formats stems from its highly proactive national influenza program, which actively rewards innovation in administration technology. As per FMI's projection, this dynamic provides a secure landing zone for high-density microarray platforms to achieve immediate commercial viability before expanding globally. The region's vast geographic disparities further amplify the utility of vaccine packaging formats that decouple distribution from refrigerated transport.
FMI's report includes secondary Asian markets such as Japan and South Korea. These advanced healthcare economies demonstrate a distinct preference for self-administration paradigms, driven by severe demographic aging and healthcare workforce limitations.

The intense concentration of Biomedical Advanced Research and Development Authority (BARDA) funding directly shapes the commercial timeline for North American patch developers. This government-backed underwriting effectively eliminates the early-stage capitalization risks that typically kill novel vaccine shippers hardware. North American biopharma leaders utilize this public-private framework to advance clinical trials at a pace unmatched in other geographies.
FMI's report includes secondary North American and Caribbean markets. These territories function primarily as technology importers, heavily reliant on the regulatory pathways established by the FDA and Health Canada to authorize domestic distribution.
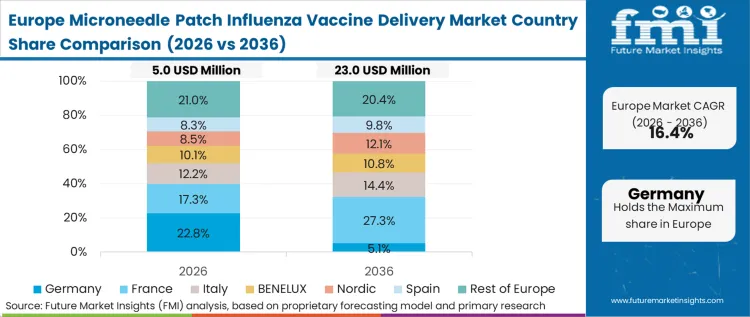
European adoption is heavily influenced by the continent's stringent extended producer responsibility regulations regarding medical waste and sharps disposal. Procurement frameworks in the EU actively penalize the utilization of legacy isothermal boxes and single-use plastics inherent in syringe distribution. Based on FMI's assessment, this environmental compliance pressure forces national health systems to assign a definitive financial premium to dissolving microneedle platforms.
FMI's report includes broad coverage of the European Union member states. A unified regulatory posture from the European Medicines Agency regarding combination products serves as the critical gate unlocking simultaneous multi-country commercialization.
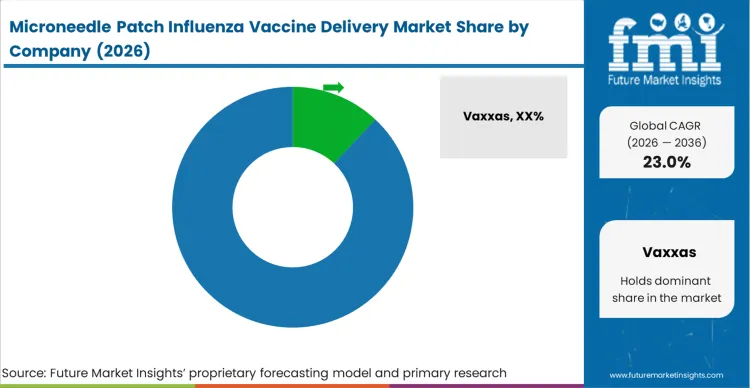
The competitive structure of this market is intensely concentrated, defined entirely by the ownership of proprietary microarray engineering patents rather than raw vaccine stabilizers production capacity. Leading entities dominate early-stage negotiations because they have successfully navigated the transition from academic cleanrooms to scalable, continuous roll-to-roll manufacturing. Procurement directors at global biopharma firms utilize demonstrated manufacturing scalability, not simply phase I immunogenicity data, as the sole variable separating qualified device partners from unviable academic projects.
Incumbents holding functional, high-density coated or dissolving polymer architectures possess an advantage protected by extreme technological moats. Replicating an anti-infective vaccines patch requires challengers to simultaneously master epidermal micro-molding, ambient-temperature desiccation chemistry, and automated packaging alignment. A challenger entering the space must build a completely novel coating mechanism to circumvent existing IP, a process that requires years of specialized capital expenditure.
Large public health buyers actively resist lock-in by funding multiple competing patch architectures simultaneously, ensuring no single device manufacturer monopolizes future pandemic supply chains. The tension between biopharma entities demanding exclusive device rights for their influenza antigens and device manufacturers seeking agnostic, multi-partner platforms will dictate commercial behaviour through 2036. The market is projected to consolidate heavily around three or four foundational device architectures, as the sheer cost of late-stage clinical trials forces smaller players into acquisition or licensing models.
| Metric | Value |
|---|---|
| Quantitative Units | USD 24.0 million to USD 190.2 million, at a CAGR of 23.00% |
| Market Definition | The commercialization and procurement of solid-state transdermal delivery devices specifically formulated with influenza antigens, capturing the value of the delivery device, stabilization matrix, and application. |
| Patch technology Segmentation | High-density coated microarray patches, Dissolving polymer microarray patches, Silk-based sustained-release microneedle patches, Hollow / liquid-compatible microneedle patches |
| Vaccine format Segmentation | Seasonal inactivated influenza vaccines, Pandemic-preparedness influenza A candidates, mRNA / broadly protective influenza vaccines, Adjuvanted influenza formulations |
| Administration model Segmentation | Provider-administered clinic/pharmacy vaccination, Public-health campaign administration, Workplace / institutional immunization, Self-administration / mail-to-home models |
| End user Segmentation | Public health agencies and immunization programs, Biopharma vaccine developers, Hospitals and retail pharmacy chains, Preparedness and stockpile agencies |
| Regions Covered | North America, Europe, Asia Pacific |
| Countries Covered | United States, Australia, United Kingdom, Germany, Canada, China, India, and 40 plus countries |
| Key Companies Profiled | Vaxxas, Vaxess Technologies, Micron Biomedical, LTS Lohmann Therapie-Systeme AG, BioNet, Zipcode Bio, PopVax |
| Forecast Period | 2026 to 2036 |
| Approach | Primary interviews targeted procurement directors and clinical development leads. Baseline valuation anchors to verifiable pilot procurement and trial material contracts. Forecasts were cross-validated against published national immunization schedules and scale-up timelines. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
What is the current size of the microneedle patch influenza vaccine delivery market?
The market reaches USD 24.0 million in 2026. This initial valuation represents milestone payments, clinical material supply, and very early-stage pilot procurements, signalling an industry positioned exactly at its commercial evolution.
How large could the market become by 2036?
The valuation expands to USD 190.2 million by 2036. This scale reflects the wholesale change of major national stockpiles away from cold-chain dependent liquid vials toward integrated solid-state mrna synthesis materials distribution paradigms.
Give me the CAGR, key players, and barriers in the influenza vaccine patch market?
The market registers a 23.00% CAGR during the forecast period. Vaxxas, Vaxess Technologies, and Micron Biomedical lead the clinical landscape. The primary barrier is the absence of a unified regulatory monograph for evaluating solid-dose epidermal vaccine-device combinations, forcing bespoke validation pathways.
Which companies are developing flu vaccine patches?
Leading developers include Vaxxas, Vaxess Technologies, Micron Biomedical, LTS Lohmann Therapie-Systeme AG, BioNet, Zipcode Bio, and PopVax. These firms are actively advancing clinical stage programs and establishing continuous manufacturing partnerships.
How do HD-MAP and dissolving microneedle patch platforms differ?
High-density microarray patches (HD-MAP) feature solid structures coated with rapidly evaporating antigen layers, avoiding chemical crosslinking. Dissolving patches integrate the antigen directly into a polymer matrix that safely clears into the body, completely eliminating post-administration medical waste.
Compare HD-MAP and dissolving microneedle flu vaccine platforms?
HD-MAP excels in preserving complex protein structures because the antigen is coated onto the surface, making it ideal for established inactivated strains. Dissolving platforms offer superior waste management profiles but present higher regulatory hurdles regarding the clearance of the proprietary polymer matrix from the patient's system.
Why are microneedle patches being developed for influenza vaccines?
The need to rapidly deploy pandemic countermeasures outside of established clinical infrastructure forces adoption. Stockpile directors mandate thermostable formats because traditional cold-chain networks fail immediately under the logistical stress of population-wide crisis distribution.
What are the main commercialization barriers in this market?
The total absence of a unified regulatory monograph for evaluating solid-dose epidermal vaccine-device combinations throttles the market. Without standardized guidelines, every new patch architecture requires a completely bespoke and highly expensive validation pathway.
Which countries are likely to adopt influenza vaccine patches first?
Australia outpaces the United States (25.1% vs 24.6%) because its national immunization program actively integrates and subsidizes premium delivery modalities. The robust domestic ecosystem of academic microneedle engineering seamlessly translates into accelerated public health procurement.
Are any flu vaccine patches approved for commercial use yet?
While significant phase I and II clinical trial milestones have been achieved by major developers, regulatory bodies have not yet granted full commercial approval for an integrated influenza microarray patch. The market currently operates on pilot procurement and material supply contracts.
Can flu vaccine patches be self-administered at home?
Yes, but regulatory agencies require extensive post-market surveillance in controlled pharmacy or clinic settings first. Once this institutional oversight phase validates immunogenicity and safety equivalence, true mail-to-home models will be authorized for the public.
What role does thermostability play in influenza vaccine patch adoption?
Thermostability completely decouples vaccination distribution from refrigerated transport. This allows public health agencies to pre-position pandemic stockpiles in ambient warehouses and ship seasonal doses via conventional mail, radically lowering the logistical baseline of national health budgets.
microneedle flu vaccine vs intramuscular flu shot: how do they compare?
Microneedle delivery targets the immune-rich Langerhans cells in the epidermis, enabling powerful dose-sparing advantages over intramuscular shots, which rely on immunologically sparse muscle tissue. Furthermore, patches eliminate sharps disposal costs and cold-chain dependency entirely.
Show me the latest outlook for the microneedle patch influenza vaccine delivery market?
The outlook is defined by a rapid transition from clinical exploration to massive public health procurement. Driven by defense mandates and the need to overcome pharmacy workforce bottlenecks, the sector will evolve heavily around proprietary IP licensing and exclusive biopharma device partnerships through 2036.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.