Mucopolysaccharidosis Treatment Market Analysis - Size, Share & Forecast 2026 to 2036
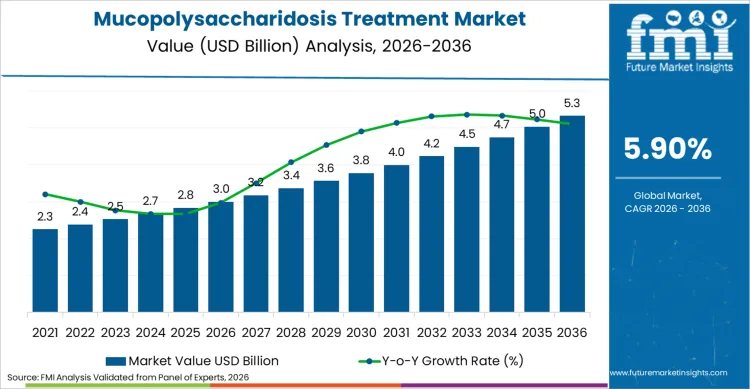
Mucopolysaccharidosis treatment industry valuation is set to reach USD 3.0 billion in 2026, driven by advancements in biotechnology and genetic diagnostics. Revenue generation is projected to expand at a steady CAGR of 5.9% through 2036, pushing total sector worth beyond USD 5.3 billion by conclusion of this assessment period. Increasing prevalence of rare genetic disorders acts as primary catalyst for this financial trajectory. Pharmaceutical entities are channeling significant capital into developing novel enzyme replacement therapies, aiming to address unmet medical needs in lysosomal storage diseases.
Clinicians are shifting focus from palliative care to disease-modifying interventions, redefining prescription habits in major healthcare economies. Such behavioral changes encourage biotech firms to introduce comprehensive patient support programs alongside therapeutic products.
Rising awareness regarding neonatal screening programs is significantly influencing industry expansion. Healthcare professionals emphasize early intervention, utilizing precise diagnostic tools to initiate treatment before irreversible damage occurs. Clinical trials investigating gene editing techniques are creating needs for specialized delivery vectors, instilling confidence among investors regarding future product pipelines.
Growing understanding of pathophysiology continues to fuel demand for targeted therapies, creating sustained opportunities for orphan drug developers. Government health initiatives targeting rare disease management are facilitating faster regulatory approvals for breakthrough medications. Researchers are combining enzyme replacement therapy with blood-brain barrier crossing technologies to treat neurological symptoms. Companies are witnessing robust growth potential in developing nations, where improving diagnostic infrastructure aligns perfectly with cost-effective treatment adoption.
Innovations in protein engineering are transforming how enzymes are delivered to affected tissues. Development of substrate reduction therapies is gaining traction, offering oral alternatives to infusions. Focus is shifting toward comprehensive chaperone-based therapeutics capable of stabilizing misfolded enzymes. Advancements in autoimmune disease therapeutics enable researchers to mitigate immunogenic responses to exogenous proteins, enhancing therapeutic efficacy while significantly reducing adverse events.
| Metric | Value |
|---|---|
| Industry Size (2026) | USD 3.0 Billion |
| Industry Value (2036) | USD 5.3 Billion |
| CAGR (2026-2036) | 5.9% |
Source: Future Market Insights’ proprietary forecasting model and primary research
Shift toward orphan drug development is serving as significant catalyst for broader acceptance of specialized treatments. Recent breakthroughs in fabry disease research have complemented treatment protocols by establishing regulatory pathways for lysosomal storage disorders. This holistic approach appeals to payers managing high-cost claims. Clinical success of sandhoff disease therapeutics in preclinical models has expanded research horizons. Education programs highlighting benefits of early diagnosis are driving screening proactivity. Pharmaceutical firms are capitalizing on these trends by introducing gaucher and pompe diseases enzyme replacement therapy ERT insights into MPS pipelines. Integrating phenylketonuria therapeutics strategies regarding metabolic management is becoming considered, ensuring diverse metabolic needs are met effectively.
Global landscape is intricately segmented to address diverse requirements of lysosomal storage disease management, categorizing sector by therapy type, MPS type, and end-user settings. Structural division allows stakeholders to identify specific growth pockets, such as rising demand for stem cell transplants in severe phenotypes or preference for enzyme infusions in attenuated forms. By analyzing these segments, manufacturers can tailor production lines to meet distinct needs of large referral centers versus local clinics. Segmentation also highlights shift from symptomatic management to curative approaches like gene therapy. As industry matures, granular segmentation becomes essential for understanding flow of capital and innovation, ensuring product development aligns perfectly with evolving clinical protocols of genetic medicine community.
Enzyme Replacement Therapy accounts for 56.4% of share in 2026, driven by its established efficacy in clearing glycosaminoglycans from somatic tissues. Leadership position is underpinned by clinical guidelines recommending ERT as standard of care for MPS I, II, IV, and VI. Segment benefits from continuous formulation improvements, such as pediatric diabetes therapeutic delivery modifications applicable to chronic infusions. Research focused on extending half-life is renewing interest among patients seeking reduced dosing frequency. Critical role in managing gastrointestinal rare diseases treatment symptoms further solidifies standing, as these therapies alleviate visceral organ enlargement. As healthcare systems prioritize proven outcomes, demand for high-quality enzymes is expected to remain robust.
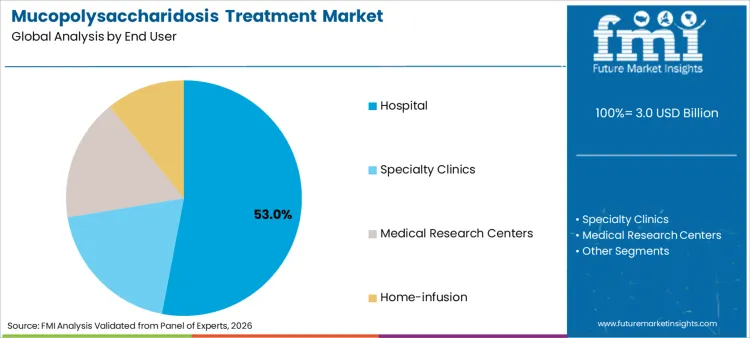
Hospitals command 53.0% share of sector in 2026, reflecting global reliance on specialized centers for complex genetic treatments. Dominance is inextricably linked to need for multidisciplinary care teams including geneticists, cardiologists, and neurologists. Hospitals rely on specialized human identification testing to confirm diagnoses before initiating therapy. These settings are essential for managing infusion reactions, driving priority status in healthcare budgets. Relentless pursuit of patient safety ensures products designed specifically for controlled hospital environments remain top priority. High patient volume necessitates dynamic drug supply, bolstering sustained revenue streams for pharmaceutical manufacturers.
Gene therapy adoption is set to reshape sales landscapes by offering potential one-time cures. As rare inflammatory disease treatment platforms evolve, patients access viral vectors capable of restoring enzyme production endogenously. Shift toward curative medicine creates demand for long-term follow-up monitoring devices. Manufacturers failing to invest in genomic medicine risk losing visibility among forward-thinking clinicians. Availability of thrombocytopenia treatment protocols to manage conditioning regimens drives complimentary consumption. Rise of inflammatory diseases research stimulates innovation, with companies differentiating offerings by bundling gene therapies with immune modulation support.
Global landscape for MPS treatment is characterized by diverse growth patterns, influenced by reimbursement policies and genetic screening infrastructure. Established markets prioritize innovative therapies and newborn screening, whereas emerging economies rely on international aid and compassionate use programs. North America is emerging as rapid growth hub due to favorable orphan drug legislation and high diagnosis rates. Conversely, Asian sectors pivot towards increasing access to basic ERT. Government initiatives in developing nations supporting rare disease funding ensure sustained demand for essential medications across all geographies.
| Country | CAGR (2026 to 2036) |
|---|---|
| United States | 7.4% |
| Germany | 7.1% |
| Japan | 6.3% |
| UK | 5.4% |
| China | 4.1% |
Source: Future Market Insights’ proprietary forecasting model and primary research
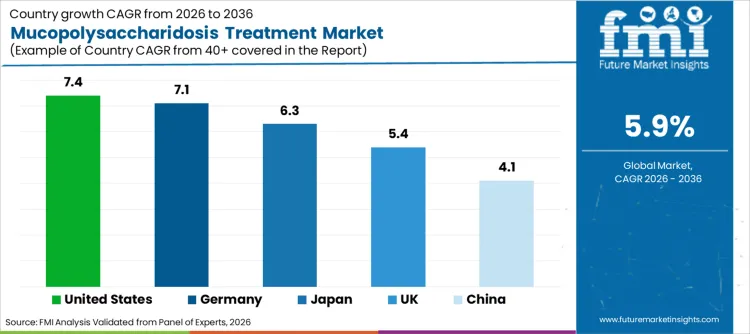
Demand for MPS therapies in United States is set to grow at 7.4% CAGR through 2036. Expansion is underpinned by robust insurance coverage for high-cost orphan drugs. Growth is fueled by widespread implementation of recommended uniform screening panels for newborns. Rapid adoption of next-generation biologics drives revenue generation per patient. Presence of major biotechnology players headquartered within country streamlines supply chains. Emphasis on personalized medicine continues to be primary driver, maintaining United States position as critical revenue generator.
Sales of treatment options in Germany are projected to rise at 7.1% CAGR during assessment period. Initiatives emphasizing equitable access to healthcare foster favorable environment for domestic and international vendors. Increasing investment in university hospital research centers creates massive supply channel for clinical trial participation. Rising patient registry numbers requiring longitudinal care prompts local administrators to seek sustainable funding models. Shift is supported by government policies encouraging rare disease research, accelerating uptake of novel therapies across country and fueling sector expansion.
MPS treatment industry in Japan is poised to expand at 6.3% CAGR. Surge is largely attributed to strong government support for intractable disease sufferers. Government focus on streamlining approval processes for orphan drugs encourages facilities to opt for latest global therapies. Increasing partnerships between Japanese pharma companies and global innovators necessitate availability of high-quality biologics. High healthcare standards combined with comprehensive reimbursement systems attract significant investment. Strict adherence to clinical guidelines further drives adoption of evidence-based treatments.
Revenue from MPS drugs in UK is set to grow at 5.4% CAGR. Expansion is underpinned by NHS specialized commissioning for lysosomal storage disorders. Growth is fueled by strong patient advocacy groups driving awareness and access. Moderate adoption of highly specialized treatments drives revenue generation in expert centers. Presence of leading genetic research institutions ensures rapid uptake of innovation. Emphasis on holistic patient care continues to be primary driver, maintaining UK position as steady revenue generator.
Sales of rare disease drugs in China are set to grow at 4.1% CAGR. Expansion is underpinned by recent regulatory reforms accelerating orphan drug review. Growth is fueled by vast population base and improving diagnostic capabilities in tier-1 cities. Gradual inclusion of rare disease drugs in national reimbursement lists drives revenue generation. Presence of emerging domestic biotech firms aiming to develop affordable biosimilars streamlines market access. Emphasis on reducing out-of-pocket costs for families continues to be primary driver, establishing China as emerging revenue generator.
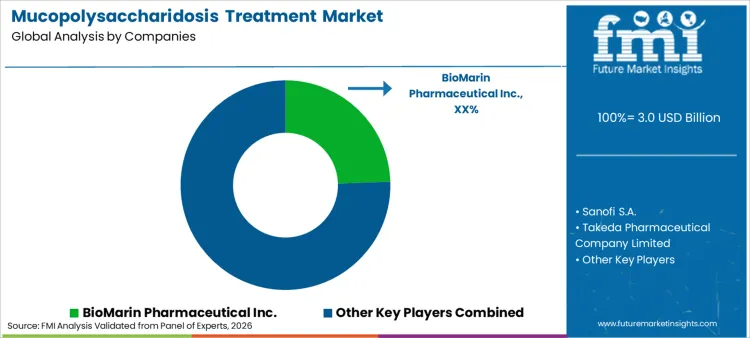
Market participants are increasingly focusing on pipeline diversification to capture value beyond established enzymes. Leading players like BioMarin Pharmaceutical Inc. and Sanofi S.A. are directing investment toward gene therapy vectors. Significant trend involves developing therapies that cross blood-brain barrier to address neurocognitive decline. Companies actively engage in strategic collaborations with patient organizations to improve diagnosis rates. Sustainability emerges as competitive frontier, with providers exploring efficient biomanufacturing processes. Consolidation activities remain high as large pharma conglomerates acquire specialized rare disease firms to broaden their portfolios against generic competition. BioMarin holds a significant 24.5% share, leveraging its diverse portfolio of approved MPS therapies.
| Items | Values |
|---|---|
| Quantitative Units | USD Billion |
| Therapy Type | Enzyme Replacement Therapy, Stem cell therapy (Bone marrow transplantation; Umbilical cord blood transplantation) |
| Type of MPS | MPS I, MPS II, MPS IV A, MPS VI, MPS VII |
| End User | Hospital, Specialty Clinics, Medical Research Centers, Home-infusion |
| Regions | North America, Europe, East Asia, South Asia, Latin America, MEA |
How large is the demand for Mucopolysaccharidosis Treatment in the global market in 2026?
Demand for Mucopolysaccharidosis Treatment in global market is estimated to be valued at USD 3.0 billion in 2026.
What will be the market size of Mucopolysaccharidosis Treatment in the global market by 2036?
Market size for Mucopolysaccharidosis Treatment is projected to reach USD 5.3 billion by 2036.
What is the expected demand growth for Mucopolysaccharidosis Treatment in the global market between 2026 and 2036?
Demand for Mucopolysaccharidosis Treatment in global market is expected to grow at CAGR of 5.9% between 2026 and 2036.
Which Therapy Type is poised to lead global sales by 2026?
Enzyme Replacement Therapy is expected to be dominant therapy type, capturing approximately 56.4% of global market share in 2026 due to clinical efficacy.
How significant is the role of Hospitals in driving Mucopolysaccharidosis Treatment adoption in 2026?
Hospitals represent critical end user, projected to hold substantial 53.0% share of total market in 2026 as facilities manage complex infusions.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.