The nerve capping devices market is expected to grow from USD 326.2 million in 2026 to USD 1,059.3 million by 2036, reflecting a compound annual growth rate (CAGR) of 12.5%. Nerve capping devices are medical devices used to protect and seal nerve endings, particularly in surgical procedures where nerve damage can occur. These devices are designed to improve patient outcomes by reducing complications related to nerve injury, such as chronic pain and sensory dysfunction. The increasing demand for nerve capping devices is driven by advancements in surgical techniques, the growing prevalence of neurological conditions, and the rising number of surgeries requiring nerve protection.
The market's growth is also supported by the increasing adoption of minimally invasive surgeries, which require precise nerve protection during procedures. With the advancement of biomaterials and technology, nerve capping devices are becoming more effective, further expanding their application in both neurosurgery and orthopedic procedures. Additionally, the growing focus on improving patient recovery and minimizing postoperative complications is contributing to the increasing demand for these devices in the global healthcare market.
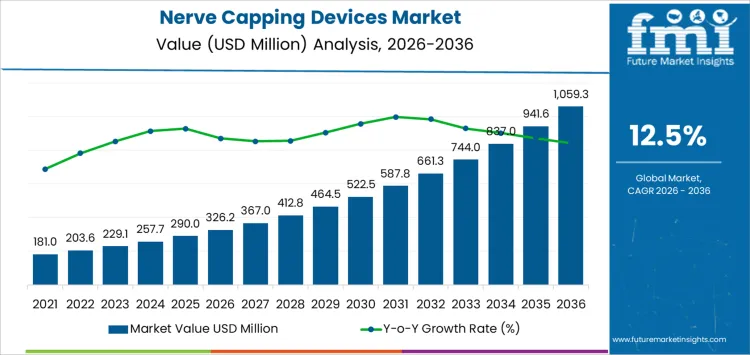
The 5-year growth block analysis for the nerve capping devices market indicates steady and consistent growth over the forecast period, with significant acceleration in the later years. Starting at USD 326.2 million in 2026, the market grows to USD 367 million in 2027 and USD 412.9 million in 2028, reflecting gradual adoption as surgical centers begin to recognize the benefits of nerve capping devices in improving surgical outcomes.
From 2028 to 2030, the market sees more substantial growth, reaching USD 464.5 million in 2029 and USD 522.7 million in 2030. This phase marks an acceleration as nerve capping devices gain wider acceptance in clinical practice, driven by improved clinical evidence and more surgeons incorporating these devices into their procedures. Between 2030 and 2035, the market continues to expand rapidly, reaching USD 588.5 million in 2031 and projected to increase further as the technology matures and more healthcare providers adopt these devices for nerve protection during complex surgeries.
By 2035, the market is projected to reach USD 1,059.3 million, reflecting significant growth due to the increasing application of nerve capping devices in multiple surgical fields. This growth is driven by technological advancements, higher adoption rates, and a greater focus on reducing postoperative complications, contributing to a promising market outlook for the coming decade.
| Metric | Value |
|---|---|
| Industry Sales Value (2026) | USD 326.2 million |
| Industry Forecast Value (2036) | USD 1,059.3 million |
| Industry Forecast CAGR (2026-2036) | 12.5% |
The global demand for nerve capping devices is driven by the need to improve outcomes in peripheral nerve injury repair and to reduce the incidence of neuroma formation and chronic pain. Peripheral nerve trauma can result from trauma, surgical procedures, or amputations, and standard repair techniques often face challenges in preventing aberrant nerve growth at injury sites. Nerve capping devices are designed to cover or contain the proximal nerve end, providing a physical barrier that guides regenerating axons and reduces the likelihood of painful neuroma development. Growing awareness among surgeons of the benefits of targeted strategies to prevent chronic neuropathic pain, as well as increasing volumes of surgical procedures involving nerve repair, stimulate adoption of these devices. The expansion of reconstructive and trauma surgery services, especially where limb salvage and functional restoration are priorities, also supports market growth. Improvements in biomaterials and device design, such as biocompatible, resorbable caps that integrate with native tissue while providing effective guidance, encourage clinician confidence and broader clinical use.
Future demand for nerve capping devices is expected to rise as evidence of clinical benefit, including reduced pain and improved functional outcomes, continues to accumulate and as surgical practices evolve toward tissue preserving and pain minimising techniques. Innovations that improve ease of use, customisation for varied nerve sizes, and integration with adjunctive therapies such as growth factors or cellular guidance cues will expand clinical applications. Expansion of healthcare infrastructure in emerging economies will increase access to advanced surgical solutions, supporting device uptake beyond high income regions. Regulatory frameworks that recognise the therapeutic value of devices designed to prevent long term complications may facilitate more widespread adoption and reimbursement coverage, reducing cost barriers for providers. Training and education for surgeons in best practices for nerve repair and the role of capping devices will further reinforce utilisation. As populations age and incidents of traumatic injury or surgical nerve exposure remain significant, the demand for nerve capping devices as part of comprehensive nerve repair strategies is likely to grow steadily across global healthcare settings.
The nerve capping devices market is driven by two key segments: material types and indications. Among the materials, bioabsorbable polymers dominate, accounting for 44% of the market share, followed by biologic/collagen-based and silicone/non-absorbable materials. Bioabsorbable polymers are favored for their ability to safely degrade in the body, reducing the need for surgical removal. The market is also shaped by indications such as amputation neuroma prevention, which accounts for 46% of the market, and peripheral nerve repair adjunct, pain management/neuroma treatment, and other indications. The demand for nerve capping devices varies across different medical conditions, with a strong focus on improving patient outcomes in nerve repair and pain management. Regional factors, especially in North America and Europe, play a significant role in the growth of these segments as healthcare facilities continue to adopt advanced nerve capping solutions to improve post-surgical recovery and minimize complications.
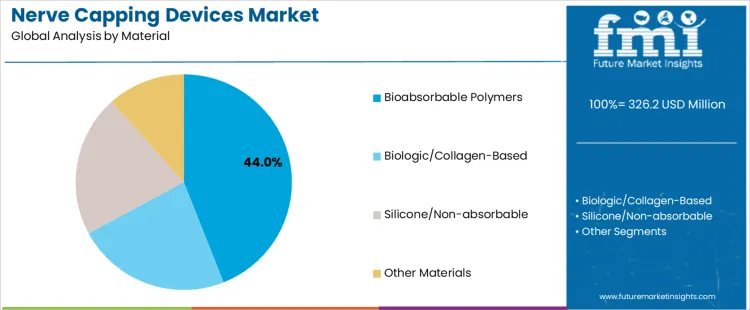
Bioabsorbable polymers are the leading material type in the nerve capping devices market, comprising 44% of the market share. These polymers are preferred for their unique property of safely dissolving within the body, eliminating the need for removal surgeries after nerve capping procedures. The use of bioabsorbable polymers is particularly beneficial in nerve repair and amputation neuroma prevention, where their biocompatibility and degradation properties promote natural healing. As the demand for minimally invasive and effective surgical solutions increases, bioabsorbable polymers are becoming the material of choice for surgeons seeking long-term, patient-friendly options. Their application in nerve repair procedures is driven by the need for materials that not only provide support but also encourage tissue regeneration and reduce the risk of complications. The ongoing development of more advanced bioabsorbable polymers with improved mechanical properties and faster degradation rates is expected to further fuel growth in this segment. Additionally, these materials align with current trends in the healthcare industry, which prioritizes patient safety, faster recovery times, and the reduction of surgical interventions. As a result, bioabsorbable polymers are projected to maintain their dominant position in the market.
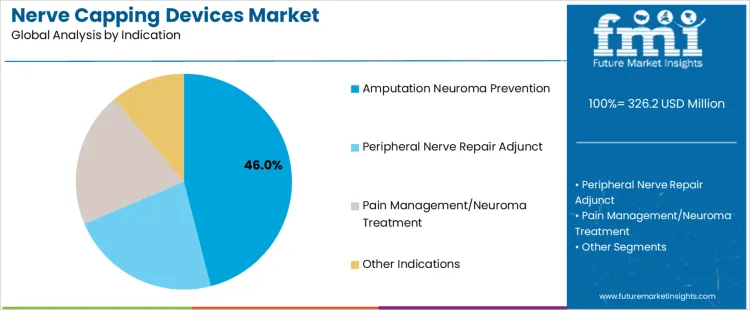
Amputation neuroma prevention is the largest indication within the nerve capping devices market, accounting for 46% of the market share. This segment’s growth is primarily driven by the increasing prevalence of amputations, which often result in painful neuromas benign growths of nerve tissue that form at the site of a nerve cut. Preventing these neuromas is critical in improving the quality of life for amputees, as untreated neuromas can lead to chronic pain and complications. Nerve capping devices play a crucial role in reducing the formation of neuromas by providing a protective barrier and promoting the natural healing of nerve endings. As surgical techniques continue to advance and more healthcare providers adopt nerve capping solutions, the demand for amputation neuroma prevention devices is increasing. In particular, the growing focus on post-operative care and pain management in amputee patients has driven the adoption of these devices in clinical settings. The effectiveness of nerve capping devices in preventing neuroma formation, combined with their minimal invasiveness, positions this indication as a key growth driver in the market. As awareness of neuroma prevention grows and the importance of post-amputation recovery is emphasized, this segment is expected to continue its upward trajectory.
The global nerve capping devices market is expanding as surgical management of peripheral nerve injuries and neuromas becomes more sophisticated. Nerve capping devices are used to protect severed nerve ends, reduce pain, and guide healthy tissue regeneration after trauma or surgery. Growth reflects rising incidence of nerve damage from accidents, orthopedic and oncologic procedures, and expanding surgical adoption of nerve preservation techniques. Interest in devices that improve functional recovery and reduce complications is increasing among surgeons and health systems. Use spans hospitals, ambulatory surgical centers and specialty practices focused on reconstructive and hand surgery worldwide.
Rising rates of peripheral nerve injuries and surgical procedures motivate demand for solutions that support nerve repair and regeneration. Clinicians value nerve capping devices that help manage neuromas and reduce pain, improving postoperative quality of life for patients. Advances in biomaterials and device design that enhance biocompatibility and integration with host tissue support clinical confidence. Growth of reconstructive, trauma and microsurgery procedures increases procedural volume. Awareness among surgeons about long term functional outcomes drives adoption. Expansion of healthcare infrastructure and investment in advanced surgical tools and implants contribute to broader deployment of nerve capping technology.
One restraint is the specialized nature of these devices, which can command higher prices and require justification within surgical budgets. Hospitals and surgical centers with tight capital constraints may prioritize other technologies or traditional repair techniques. Some surgeons remain cautious until long term clinical evidence clearly demonstrates comparative benefit over standard care in specific injury types. Regulatory approval processes for new nerve repair devices vary by region and can delay commercial availability. Variation in training and familiarity with advanced nerve management tools may limit adoption in practices with fewer microsurgical cases or limited access to specialist education.
A key trend is innovation in advanced biomaterials that support nerve protection while minimizing foreign body response and promoting regeneration. Manufacturers are focusing on bioresorbable polymers and hybrid designs that provide structural support and degrade as healing progresses. Integration of nerve capping devices with regenerative medicine approaches and growth factor delivery is gaining research interest. Surgeons are adopting tools that combine ease of use with enhanced clinical performance in complex injuries. Investment in clinical studies, real world outcome tracking and surgeon training programs is increasing. Multi disciplinary collaboration between device developers and surgical specialists helps refine design, education and market acceptance.
The nerve capping devices market is expected to grow significantly across key regions, driven by rising incidences of nerve injuries, the demand for minimally invasive surgeries, and ongoing technological advancements in medical devices. Countries like China, the United States, and the United Kingdom are projected to see considerable growth due to the increasing adoption of innovative healthcare solutions and the aging population that demands effective nerve injury treatments. The demand for nerve capping devices is also fueled by advancements in surgical precision, which allow for better patient outcomes and shorter recovery times. In particular, China is anticipated to lead the growth with a projected CAGR of 14%, while other countries such as the United States, United Kingdom, and Germany follow with steady growth. As the healthcare systems in these regions continue to modernize, the market for nerve capping devices is expected to experience consistent expansion. By focusing on market trends and growth forecasts for each country, businesses can tailor strategies to meet the unique needs of each region, optimizing the adoption of nerve capping devices.
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 14% |
| United States | 12% |
| United Kingdom | 12% |
| Germany | 11.9% |
| Japan | 10.5% |
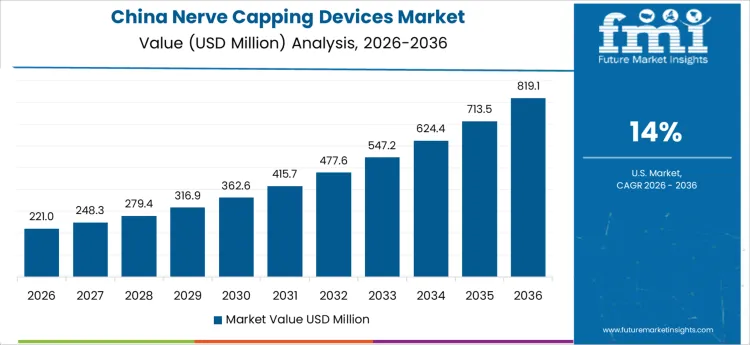
The nerve capping devices market in China is projected to grow at the highest rate, with a CAGR of 14%. This strong growth is primarily driven by China’s expanding healthcare infrastructure, coupled with the increasing demand for treatments related to nerve injuries, particularly those affecting the elderly. The country’s government has prioritized healthcare modernization and innovation, creating a favorable environment for the adoption of advanced medical technologies. With a large and growing population, the need for effective and accessible nerve injury treatments is more pressing than ever. China’s healthcare system continues to improve, ensuring greater access to medical innovations like nerve capping devices. The rise in chronic conditions, along with the increasing adoption of minimally invasive surgeries, contributes to the significant demand for these devices. Furthermore, China’s rapid advancements in healthcare technology are expected to sustain the market’s growth over the forecast period. As the nation continues to focus on modernizing its healthcare facilities and improving patient outcomes, the nerve capping devices market will likely continue to experience robust growth.
The nerve capping devices market in the United States is expected to grow at a solid pace, with a projected CAGR of 12%. The U.S. has long been a leader in medical technology innovation, and the demand for nerve capping devices is fueled by the country’s large healthcare infrastructure and increasing rates of nerve injuries. With a rapidly aging population, the need for treatments to address age-related nerve conditions is expanding. In addition, the growing focus on minimally invasive procedures, which offer quicker recovery times and less trauma to patients, is driving the adoption of nerve capping devices. The U.S. market benefits from a highly receptive healthcare system that is eager to integrate innovative medical devices that enhance patient care and surgical precision. Furthermore, the strong regulatory environment and advanced research institutions support the introduction of new technologies into the U.S. healthcare system. As the market continues to expand, the adoption of nerve capping devices is expected to increase in both clinical and surgical settings, making the U.S. one of the largest markets globally.
The nerve capping devices market in the United Kingdom is projected to grow at a CAGR of 12%. The UK’s healthcare system, known for its advanced technology and emphasis on patient care, provides a solid foundation for the adoption of innovative medical solutions like nerve capping devices. The increasing prevalence of nerve injuries, particularly among the elderly, is contributing to the rising demand for advanced treatment options. The National Health Service (NHS) plays a significant role in the integration of these devices into the healthcare system, ensuring their widespread adoption across the country. With a growing focus on improving surgical outcomes and patient recovery times, healthcare providers in the UK are increasingly turning to minimally invasive techniques, such as nerve capping procedures, to meet patient needs. Furthermore, the UK’s robust regulatory framework for medical devices ensures that the market remains conducive to the introduction of advanced technologies. As a result, the nerve capping devices market in the UK is expected to continue its upward trajectory in the coming years.
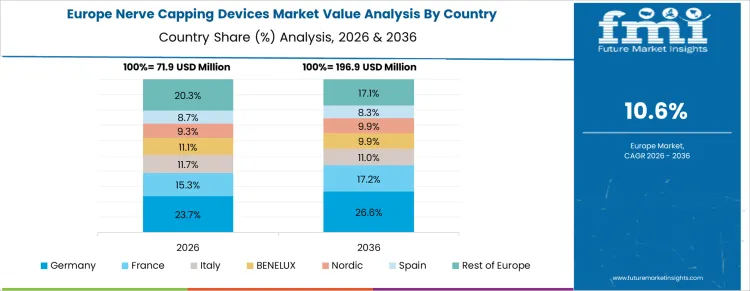
The nerve capping devices market in Germany is expected to grow at a moderate pace, with a projected CAGR of 11.9%. Germany’s advanced healthcare infrastructure, strong emphasis on medical research, and focus on improving surgical techniques support the growth of the nerve capping devices market. With an aging population and a rising number of cases involving nerve-related injuries, there is an increasing demand for effective treatments. Germany’s healthcare system is highly efficient and well-equipped to integrate new technologies, providing an ideal environment for the adoption of nerve capping devices. The country's strong regulatory environment further supports the market's growth by ensuring that only the most innovative and effective solutions are brought to market. Additionally, Germany’s emphasis on high-quality medical care ensures that devices like nerve capping technologies are increasingly used in clinical settings. As the demand for minimally invasive surgical options grows, Germany is expected to remain a key player in the European nerve capping devices market.
The nerve capping devices market in Japan is expected to grow at a CAGR of 10.5%. Japan’s aging population is a major factor driving the demand for advanced medical technologies, including nerve capping devices. The country’s healthcare system is one of the most sophisticated in the world, and there is a strong focus on improving patient care through innovative, minimally invasive procedures. With a growing number of nerve injuries related to both aging and lifestyle, the need for more effective treatments is on the rise. Japan’s healthcare providers are increasingly adopting new technologies that promise better outcomes and faster recovery times for patients. The government’s support for healthcare innovation and the country’s focus on improving medical device accessibility further boost the market's growth potential. Although the growth rate is slightly slower compared to some other countries, Japan’s highly developed healthcare infrastructure and focus on advanced treatments will continue to drive the demand for nerve capping devices. As the market matures, adoption of these devices is expected to increase, supported by ongoing technological advancements in medical procedures.
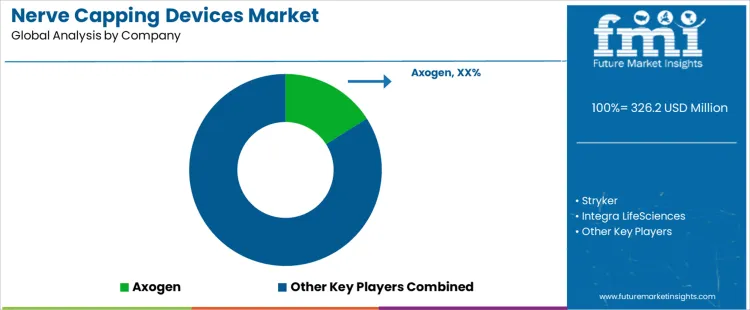
The nerve capping devices market is growing, driven by the increasing demand for effective solutions to manage nerve injuries and improve recovery outcomes. Axogen leads the market with its advanced nerve capping devices, providing cutting-edge technologies designed to protect and repair damaged nerves. Their focus on regenerative medicine and nerve repair positions them as a key player in the market. Stryker, Integra LifeSciences, and Johnson & Johnson (Ethicon) are major competitors, offering their own nerve repair and protection solutions. Stryker specializes in innovative surgical devices, while Integra LifeSciences provides a range of neuroregenerative products. Johnson & Johnson (Ethicon), through its strong portfolio, enhances treatment options for nerve injuries with its advanced products in the nerve capping space.
BD, B. Braun, Medtronic, Smith+Nephew, DePuy Synthes (J&J), and Coloplast contribute further competition with their extensive medical device portfolios. BD and B. Braun offer devices designed for precision nerve repair, focusing on enhancing surgical outcomes. Medtronic and Smith+Nephew bring advanced technologies that improve nerve repair through better surgical techniques and device integration. DePuy Synthes (J&J) and Coloplast provide specialized products that cater to nerve repair and management, positioning them as significant competitors in the market. These companies compete by focusing on product innovation, surgical precision, and the ability to offer effective solutions for nerve repair and protection.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD Million |
| Material | Bioabsorbable Polymers, Biologic/Collagen-Based, Silicone/Non-absorbable, Other Materials |
| Indication | Amputation Neuroma Prevention, Peripheral Nerve Repair Adjunct, Pain Management/Neuroma Treatment, Other Indications |
| Setting | Hospitals, Ambulatory Surgery Centers, Specialty Clinics |
| Surgical Specialty | Orthopedics/Trauma, Plastic/Reconstructive, Vascular & Amputation Care, Other Specialties |
| Companies | Axogen, Stryker, Integra LifeSciences, Johnson & Johnson (Ethicon), BD, B. Braun, Medtronic, Smith+Nephew, DePuy Synthes (J&J), Coloplast |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, South Asia and Pacific, East Asia, Middle East & Africa |
| Countries Covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, Netherlands, China, India, Japan, South Korea, ANZ, GCC Countries, South Africa |
| Additional Attributes | Dollar by sales by material, indication, setting, surgical specialty, and region. Includes market trends in nerve capping devices, focusing on bioabsorbable polymers, biologic/collagen-based materials, and silicone/non-absorbable options. Highlights the role of nerve capping devices in amputation neuroma prevention, peripheral nerve repair, and pain management. Focus on cost-effectiveness, regulatory compliance, market share, and competitive positioning of key companies in nerve capping solutions. |
How big is the nerve capping devices market in 2026?
The global nerve capping devices market is estimated to be valued at USD 326.2 million in 2026.
What will be the size of nerve capping devices market in 2036?
The market size for the nerve capping devices market is projected to reach USD 1,059.3 million by 2036.
How much will be the nerve capping devices market growth between 2026 and 2036?
The nerve capping devices market is expected to grow at a 12.5% CAGR between 2026 and 2036.
What are the key product types in the nerve capping devices market?
The key product types in nerve capping devices market are bioabsorbable polymers, biologic/collagen-based, silicone/non-absorbable and other materials.
Which indication segment to contribute significant share in the nerve capping devices market in 2026?
In terms of indication, amputation neuroma prevention segment to command 46.0% share in the nerve capping devices market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.