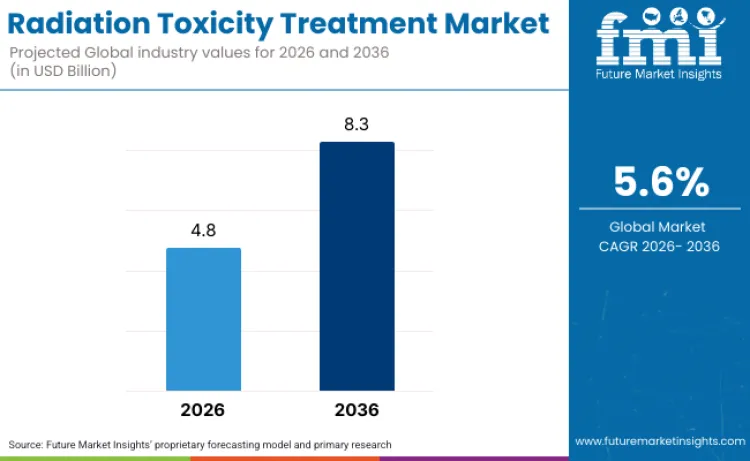
Radiation toxicity treatment industry valuation is expected to reach USD 4.8 billion in 2026, supported by two demand centers that are expanding in parallel: national preparedness for radiological or nuclear incidents and the growing use of medical radiation that requires structured toxicity management pathways. As per FMI’s estimates, revenue is projected to expand at a 5.6% CAGR through 2036, taking the sector to USD 8.3 billion. Government regulation is a direct demand lever because it determines which countermeasures can be procured and stockpiled. In the United States, multiple hematopoietic countermeasures have FDA indications for hematopoietic syndrome of acute radiation syndrome under the FDA Animal Rule, including filgrastim (Neupogen) approved March 2015, pegfilgrastim (Neulasta) approved November 2015, sargramostim (Leukine) approved March 2018, and romiplostim (Nplate) approved January 2021, reinforcing the commercial weight of colony stimulating factor and related hematopoietic recovery products.
Healthcare systems are moving from supportive care to protocolized pharmacologic intervention built around hematopoietic recovery, infection risk control, and multi organ complication management guided by triage logic and biodosimetry where available. Procurement is further supported by stockpile policies, with key cytokines referenced as included in Strategic National Stockpile inventory programs. Outside the USA, safety and preparedness rules institutionalize demand: the EU Basic Safety Standards Directive 2013/59/Euratom strengthens governance for medical exposure and incident prevention in radiology and radiotherapy, while IAEA safety requirements and national regulations such as NRC 10 CFR Part 20 drive radiation protection programs and emergency planning that translate into sustained countermeasure readiness.
| Metric | Value |
|---|---|
| Industry Size (2026) | USD 4.8 Billion |
| Industry Value (2036) | USD 8.3 Billion |
| CAGR (2026-2036) | 5.6% |
Source: FMI analysis based on primary research and proprietary forecasting model
Colony stimulating factor adoption in emergency preparedness is strengthening as regulators and public health agencies formalize their role in treating radiation-induced myelosuppression, and as more products carry labeled use for hematopoietic acute radiation syndrome scenarios. Clinical guidance also supports earlier, protocol-driven use to reduce the duration and consequences of severe neutropenia, which increases demand for rapid-deployment formats such as prefilled injections that fit emergency response workflows.
In provider economics, the most decision-relevant metrics sit in Financial and Operational Performance, including Net Patient Revenue, Operating Margin, Days Cash on Hand, AR Days, and Cost Per Patient Day, because biologic acquisition, storage readiness, and surge delivery can shift working capital and cost structure. Revenue Optimization hinges on Payer Mix, Adjusted Revenue Per Encounter, and Revenue Cycle Efficiency, especially when emergency use and documentation requirements affect charge capture and reimbursement timing. Cost and Profitability is reflected through Supply Chain Cost Ratio and EBITDA, since preparedness procurement and cold-chain management must be balanced against utilization uncertainty and quality assurance obligations.
Global marketplace is systematically segmented to address varied requirements of the radiation emergency medicine industry, categorizing sector by product type, indication, radiation type, and end-user. This structural organization enables stakeholders to identify specific therapeutic opportunities, such as increasing demand for potassium iodide in nuclear emergency preparations or preference for prussian blue in internal contamination scenarios. Through analyzing these segments, manufacturers can customize production strategies to meet distinct requirements of government stockpiling programs versus hospital emergency departments. Segmentation also reveals transition from basic supportive care to sophisticated pharmacological countermeasures. As emergency preparedness frameworks advance, detailed segmentation becomes essential for understanding therapeutic pathways and regulatory compliance, ensuring product development aligns with evolving radiation emergency standards.
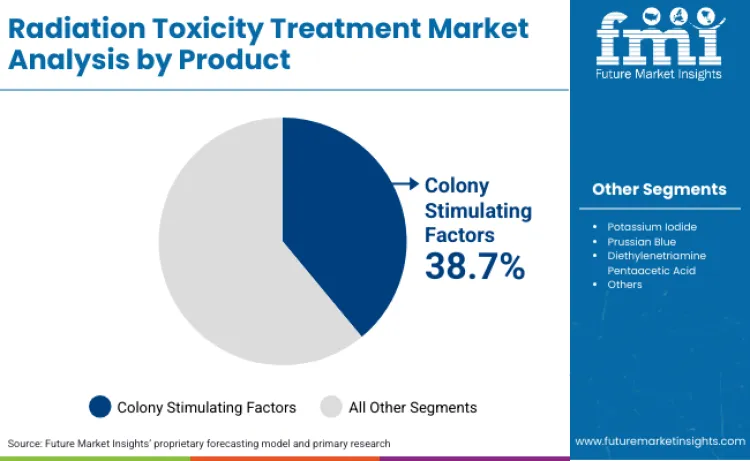
Colony stimulating factors hold 38.7% of product type share in 2026 because they are among the most clinically established options for accelerating hematopoietic recovery in radiation-related scenarios, making them a default choice in preparedness planning and emergency use protocols. Their position is reinforced by familiarity in neutropenia management, predictable response patterns, and ongoing formulation advances that improve dosing practicality and treatment continuity in high-pressure care settings.
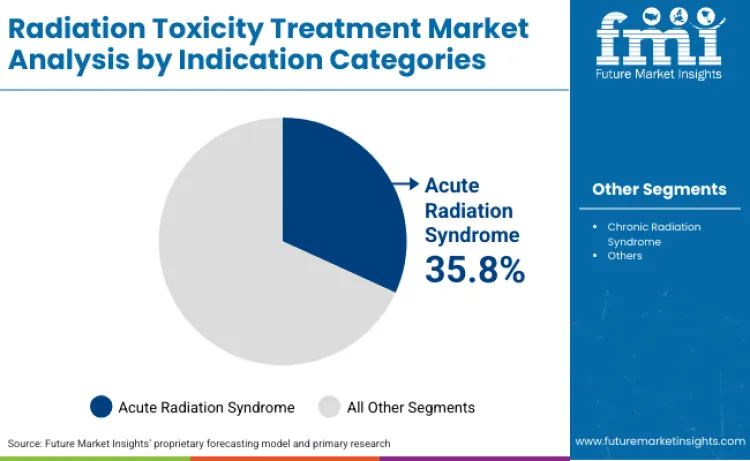
Acute radiation syndrome represents 35.8% of indication share in 2026 because it concentrates the most urgent therapeutic demand, where treatment decisions are driven by severity, speed, and the need to stabilize multi-system complications. Health systems prioritize indications that require immediate intervention and standardized protocols, which pushes spending toward therapies and regimens designed to support survival and reduce downstream complications across hematopoietic and other affected systems.
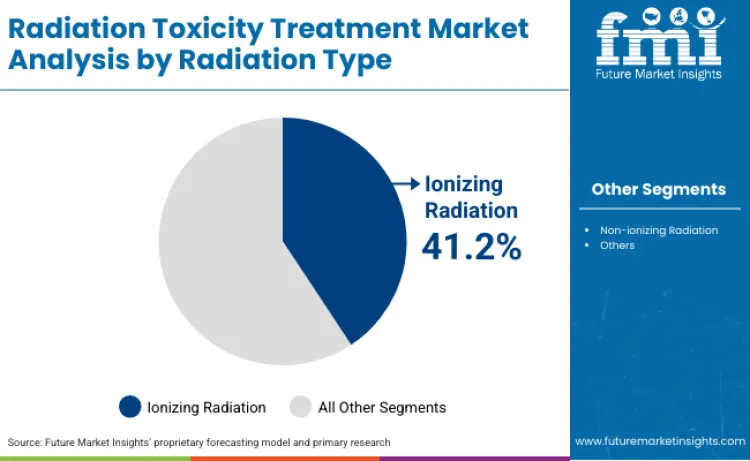
Ionizing radiation accounts for 41.2% of radiation type share in 2026 because it is central to the exposure profiles most often addressed in medical, industrial, and nuclear emergency planning, where countermeasure readiness is structured around cellular damage risk and monitoring requirements. Its dominance reflects how response programs prioritize scenarios with defined mechanisms of injury and established clinical management approaches, creating a clearer pathway from risk assessment to procurement and protocol adoption.
Expansion of civilian nuclear capacity is likely to raise baseline demand for radiation countermeasures because new and upgraded facilities typically bring tighter preparedness expectations, larger planning populations, and more frequent procurement cycles for stockpiles. As reactor deployments increase, utilities, public health agencies, and designated treatment centers tend to expand inventory depth for agents that support hematopoietic recovery, infection risk reduction, and supportive care pathways across different exposure severities. The shift toward newer reactor designs can also broaden scenario planning, pushing buyers to formalize protocols that cover mixed exposure profiles and to prioritize products with clear labeling, stable shelf life, and scalable supply. For suppliers, this translates into a market environment shaped by multi-year framework contracts, replenishment schedules driven by expiry management, and growing emphasis on validated performance in occupational and community preparedness use cases.
Modern preparedness programs are moving from static stockpiles to coordinated deployment models, which changes what healthcare systems purchase and how they evaluate products. Real-time coordination tools, regional referral pathways, and standardized triage algorithms increase the value of therapies that can be administered quickly, stored and transported reliably, and integrated into stepwise treatment protocols across multiple facilities. As response models become networked, procurement also shifts toward interoperable inventory tracking, pre-positioned caches, and distribution plans that can surge to designated centers based on exposure assessment. This raises demand for suppliers that can support training, clear administration guidance, and logistics readiness alongside the product, because operational usability becomes a differentiator in emergency tenders and preparedness contracting.
Regulatory evolution is reshaping the countermeasure pipeline by rewarding sponsors that can prove efficacy through accepted non-clinical pathways while maintaining high manufacturing and quality thresholds. In this domain, development often relies on robust animal efficacy packages paired with human safety and pharmacology data, which can compress timelines but increases the burden on study design, translational relevance, and post-approval commitments. Emergency use mechanisms can accelerate access during crises, but they also raise expectations for lot-to-lot consistency, stability programs, and traceable supply, since products may be deployed under intense scrutiny. As regulators align standards across regions, manufacturers gain clearer routes to broader commercialization, yet they must invest more in quality systems, validated processes, and shelf-life assurance to meet procurement requirements tied to national preparedness and public health readiness.
Global landscape for radiation emergency medicine is characterized by diverse preparedness frameworks, influenced by nuclear energy policies and emergency management systems. Established markets prioritize comprehensive countermeasure stockpiles and advanced treatment protocols, whereas developing economies focus on basic radiation emergency capabilities and accessible therapeutic options. United States is emerging as preparedness leader due to extensive biodefense programs and advanced pharmaceutical development infrastructure. Conversely, European markets emphasize regulatory coordination and cross-border emergency response integration models. As per FMI's projection, government initiatives in developed nations supporting nuclear security ensure sustained demand for radiation countermeasures across all emergency preparedness categories.
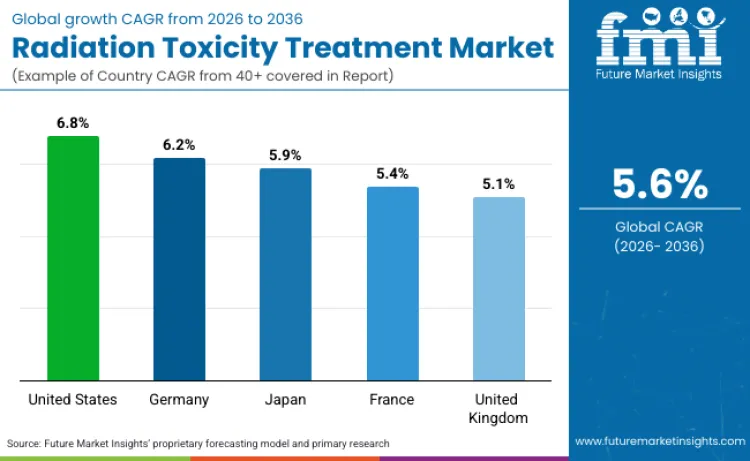
| Country | CAGR (2026 to 2036) |
| United States | 6.8% |
| Germany | 6.2% |
| Japan | 5.9% |
| France | 5.4% |
| United Kingdom | 5.1% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Development of radiation countermeasures in the United States is positioned to grow at 6.8% CAGR through 2036 because the category is directly tied to federal preparedness procurement and product development pipelines for radiological and nuclear incidents. The Strategic National Stockpile is managed by HHS ASPR and is designed to maintain and deliver critical medical assets for emergencies, which creates recurring demand for stockpiled radiation and trauma therapeutics and the logistics to deploy them. FDA has approved specific medical countermeasures for acute radiation syndrome indications, which supports structured purchasing, protocol adoption, and lifecycle replenishment rather than one time buying. BARDA’s CBRN portfolio investment also sustains development and manufacturing readiness for medical countermeasures, strengthening the supply base that feeds federal and state preparedness channels.
Demand for radiation emergency therapeutics in Germany is projected to advance at 6.2% CAGR because preparedness is implemented through formal radiation protection arrangements that include population level thyroid blocking and structured distribution responsibilities. Germany’s Federal Office for Radiation Protection explains iodine blockade and notes that federal states are responsible for storage and distribution of high dose iodine tablets, which supports continuous planning, replacement cycles, and readiness related procurement. BFS has also reported replacement and replenishment of iodine tablet stocks procured federally and distributed by state governments, indicating an ongoing readiness supply chain that sustains demand for medical countermeasure inventory management and validated products.
Radiation toxicity treatment demand in Japan is set to expand at 5.9% CAGR because national and local preparedness systems were strengthened after Fukushima and continue to emphasize readiness for rapid protective actions and medical response coordination. Japan’s emergency preparedness materials and international documentation describe planning that includes stable iodine use and decision structures for nuclear emergencies, which supports recurring demand for stocked countermeasures and distribution capability. Prefectural guidance also shows advance distribution planning for stable iodine near nuclear facilities, indicating locally executed readiness programs that sustain procurement and clinical protocol integration. Japan’s medical community has also issued professional guidance on iodine prophylaxis for nuclear accidents, reinforcing structured clinical expectations that support demand for validated countermeasure products and training.
Sales of radiation countermeasures in France are set to grow at 5.4% CAGR because preparedness is anchored in formal public protection actions, including preventive stable iodine distribution and national medical guidance for radiological events. France’s public service communications describe an iodine tablet distribution campaign for people living near nuclear power plants, implemented via pharmacies, which indicates an established, recurring distribution model that drives ongoing inventory and readiness demand. France’s nuclear safety regulator publications include a national guide on medical response in nuclear or radiological emergencies, supporting standardized protocols and consistent requirements for emergency treatment readiness across healthcare settings. Regulator reporting also references stable iodine distribution expectations around emergency planning perimeters, reinforcing a standing preparedness mechanism that sustains countermeasure planning and procurement.
Revenue from radiation countermeasures in the United Kingdom is positioned to grow at 5.1% CAGR because preparedness is governed through a defined regulatory framework that requires planning and public information arrangements for radiation emergencies. The Health and Safety Executive sets out the purpose of REPPIR 2019 as establishing a framework of preparedness measures for radiation emergencies, which drives structured planning and recurring readiness requirements that translate into demand for protocols, training, and countermeasure availability. UK government guidance for the public on radiation emergencies explicitly references urgent protective actions including taking stable iodine if advised, reinforcing that stable iodine planning remains an active part of preparedness, which supports ongoing stock management and distribution planning.
Since 2023, competition has become more execution-driven than broad-based, because most near-term demand is tied to deployable, labeled medical countermeasures for acute radiation syndrome and their fit with national preparedness protocols. A key differentiator is operational usability in emergencies, for example, the FDA highlighted approval of an auto-injector presentation for pegfilgrastim (Udenyca) for H-ARS, which matters because simpler administration can improve real-world deployment under time pressure.
Another differentiator is readiness integration: Partner Therapeutics has stated that LEUKINE is held for use by the ASPR Strategic National Stockpile for H-ARS, reinforcing the importance of government-linked positioning, supply assurance, and protocol alignment. As a result, companies are prioritizing rapid-acting delivery formats, clear dosing guidance, and interoperability with emergency management workflows, while pipeline efforts increasingly focus on improved tolerability and combination approaches for varied exposure scenarios. FMI is of the opinion that competitive advantage will accrue to players that can pair credible clinical/animal-rule evidence packages with deployment-ready formats and dependable stockpile-oriented manufacturing.
| Items | Values |
|---|---|
| Quantitative Units | USD Billion |
| Product Segments | Colony Stimulating Factors; Potassium Iodide; Prussian Blue; Diethylenetriamine Pentaacetic Acid; Others |
| Indication Categories | Acute Radiation Syndrome; Chronic Radiation Syndrome; Others |
| Radiation Type Categories | Ionizing Radiation; Non-ionizing Radiation; Others |
| End-User Categories | Hospitals; Research & Academic Institutes; Others |
| Regions Covered | North America, Europe, Asia-Pacific, Latin America, Middle East & Africa |
| Key Countries | United States, Germany, Japan, France, United Kingdom |
| Key Companies Profiled | Amgen; Partner Therapeutics, Inc.; Novartis AG; Mylan NV; Coherus BioSciences Inc; Jubilant Life Sciences; Siegfried Holdings; Heyl Chemisch-pharmazeutische Fabrik GmbH & Co. KG; Recipharm AB; Mission Pharmacal Company |
| Additional Attributes | Dollar sales measured for radiation toxicity treatments used in emergency medicine and countermeasure applications, specified by therapeutic mechanism (hematopoietic stimulation, chelation, radioprotection), product approval status, emergency use authorization, application focus (acute exposure, contamination, prophylaxis), procurement model (government stockpile vs. hospital), and compliance alignment with evolving biodefense standards and emergency preparedness regulations. |
How big is the global radiation toxicity treatment market?
The global radiation toxicity treatment market is valued at USD 4.8 billion in 2026.
What is the growth outlook over the next 10 years?
The market is projected to grow at a 5.6% CAGR from 2026 to 2036, reaching USD 8.3 billion.
Which therapeutic areas or applications drive demand?
Demand is driven primarily by acute radiation syndrome management, hematopoietic recovery therapies, biodefense preparedness, and nuclear emergency medicine.
How does market behavior differ by region?
North America leads demand through government stockpiling and biodefense programs, while Europe and Asia-Pacific emphasize nuclear safety compliance and coordinated emergency preparedness.
What are the key risks, regulatory, or development constraints?
Key constraints include limited clinical trial pathways, high development costs, reliance on government procurement cycles, and stringent quality and shelf-life requirements.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.