Radiation-Free Fetal Heart Rate Monitor Market
The Radiation-Free Fetal Heart Rate Monitor Market is segmented by Product Type (Doppler Fetal Heart Rate Monitors, Photoelectric Sensor Fetal Heart Rate Monitors, Others), End User (Clinics, Home Use, Others), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Radiation-Free Fetal Heart Rate Monitor Market Forecast and Outlook By FMI
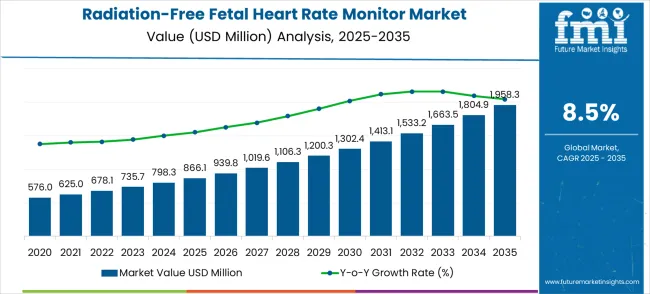
In 2025, the radiation-free fetal heart rate monitor market was valued at USD 866.1 million. Based on Future Market Insights’ analysis, demand for radiation-free fetal heart rate monitors is estimated to grow to USD 939.7 million in 2026 and USD 2,124.8 million by 2036. FMI projects a CAGR of 8.5% during the forecast period.
An absolute dollar growth of USD 1,185.1 million between 2026 and 2036 reflects a structural shift from standalone Doppler devices used during scheduled clinic visits toward connected monitoring ecosystems that enable continuous, data‑driven prenatal care. FMI projects that this growth depends on the successful integration of fetal monitors with maternal health platforms, the expansion of home‑use prescriptions for high‑risk pregnancies, and the replacement of legacy ultrasound systems with radiation‑free alternatives in emerging market maternity centers.
Summary of the Radiation-Free Fetal Heart Rate Monitor Market
- Market Definition
- The market comprises non‑invasive medical devices using Doppler ultrasound or photoelectric sensors to detect fetal heart rate without ionizing radiation, deployed across hospital, clinic, and home settings for prenatal surveillance.
- Demand Drivers
- WHO and national guidelines now explicitly recommend Doppler ultrasound as the preferred method for routine fetal heart rate auscultation, displacing Pinard stethoscopes and radiation‑based alternatives. [2]
- Rising prevalence of high‑risk pregnancy conditions (gestational diabetes, hypertensive disorders) requiring more frequent monitoring than standard care schedules permit.
- Expansion of remote patient monitoring programs in developed health systems, enabling clinic‑grade fetal assessment in home environments.
- Key Segments Analyzed
- Product Type: Doppler monitors lead, accounting for 60.0% share in 2025 due to clinical gold‑standard status and real‑time audio feedback valued by both clinicians and parents.
- End‑User: Clinics dominate at 65.0% share, reflecting protocol‑driven monitoring at scheduled antenatal visits and the capital equipment budgets of hospital obstetrics departments.
- Geography: China leads growth through infrastructure standardization; Germany leads European value through DiGA reimbursement integration for home‑use devices.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant for Healthcare & Life Sciences at Future Market Insights, observes: ‘In this reassessed forecast for the radiation‑free fetal heart rate monitor market, we see a category moving beyond the binary of "safe versus unsafe" toward a paradigm of "actionable versus episodic." The February 2025 update to the WHO antenatal care recommendations explicitly endorses Doppler for fetal heart rate detection and calls for integration with maternal health records, a directive that compels manufacturers to prioritize data interoperability as a core product feature. The market winners through 2036 will be those whose devices generate clinically structured data streams, not just audible heartbeats.’
- Strategic Implications/Executive Takeaways
- Pursue cloud‑based data platform partnerships with electronic medical record vendors to ensure fetal monitoring data flows directly into prenatal charts, reducing manual transcription errors.
- Develop home‑use SKUs with prescription‑only feature sets (trend analysis, automated alert thresholds) to capture DiGA reimbursement in Germany and similar digital health pathways in France and Japan.
- Invest in local clinical validation studies in China and India to support NMPA and CDSCO registration timelines, which are increasingly determinant of market access speed.
- Methodology
- Built on primary interviews with fetal monitor manufacturers and obstetrics procurement executives.
- Benchmarked against birth statistics, maternal care facility counts, and medical device registration filings.
- Validated using shipment audit data and internal peer review as per FMI modelling standards.
In May 2025, Philips introduced the next generation of its Avalon CL Fetal Monitor series, incorporating artificial intelligence‑based signal processing that reduces artifact interference by an estimated 40% compared to previous models, while adding seamless data transmission to the Philips IntelliSpace Perinatal platform. [1] This launch signals the convergence of hardware reliability with software‑driven workflow efficiency, a key requirement for hospital systems seeking to standardize prenatal monitoring across multiple sites.
China (11.5% CAGR) leads global expansion, supported by the National Health Commission’s 2024 guidelines for maternal near‑miss surveillance, which recommend continuous electronic fetal monitoring in all secondary and tertiary maternity centers. According to FMI, India (10.6% CAGR) follows, driven by the expansion of the government’s Janani Suraksha Yojana cash transfer scheme, which now includes incentives for facilities demonstrating adoption of radiation‑free monitoring technologies. Germany (9.8% CAGR) grows on the back of digital health applications (DiGA) reimbursement pathways that now include certain home‑use fetal monitors prescribed for high‑risk pregnancies.
Mature markets such as the United States (8.1%), the United Kingdom (7.2%), and Japan (6.4%) generate comparatively slower expansion, where demand is shaped primarily by replacement cycles in established hospital infrastructure and conversion from intermittent Doppler monitoring to continuous wearable solutions, with device saturation in tertiary centers acting as a structural constraint on unit growth.
Radiation-Free Fetal Heart Rate Monitor Market Definition
The radiation-free fetal heart rate monitor market covers the global production and supply of non‑invasive medical devices that detect and record fetal cardiac activity without emitting ionizing radiation. These devices primarily employ Doppler ultrasound or photoelectric plethysmography to capture fetal heart rate, providing audible and visual feedback for prenatal assessment. They are used in hospital obstetrics departments, ambulatory clinics, and home‑care settings for routine antenatal surveillance, high‑risk pregnancy management, and expectant parent reassurance.
Market Inclusions
The report includes global and regional market size estimates in value terms, with a forecast period from 2026 to 2036. It provides segmentation by Product Type, End‑User, and Region. The analysis incorporates manufacturing technology trends, sensor miniaturization advances, wireless connectivity integration, and regulatory clearance pathways for home‑use devices. Regional demand dynamics, country‑level growth rates, and competitive positioning are also evaluated as per FMI methodology.
Market Exclusions
The scope excludes fetal monitors that rely on ionizing radiation (X‑ray, CT), invasive fetal scalp electrodes requiring ruptured membranes, and non‑medical novelty devices lacking clinical validation for heart rate accuracy. It also excludes revenues from telemedicine consultation services not bundled with proprietary hardware and focuses strictly on the medical device manufacturing layer. Custom research‑grade fetal monitoring systems produced in sub‑commercial volumes are not considered due to negligible market contribution.
Research Methodology
- Primary Research: Interviews were conducted with fetal monitor manufacturers, obstetrics department procurement managers, maternal‑fetal medicine specialists, and digital health platform integrators.
- Desk Research: Public company filings, medical device registration databases (FDA 510(k), CE‑MDR, NMPA), maternal health epidemiology studies, and healthcare technology assessment reports were reviewed.
- Market‑Sizing and Forecasting: A hybrid top‑down and bottom‑up model was constructed, reconstructing demand from birth rates, hospital delivery volumes, per‑facility device penetration ratios, and home‑use prescription trends across key geographies.
- Data Validation and Update Cycle: Outputs were subjected to variance testing against production shipment data, trade flow benchmarking under relevant HS codes, and structured internal review prior to publication.
Radiation-Free Fetal Heart Rate Monitor Market Drivers, Restraints, and Opportunities
According to FMI, the market has historically existed as a clinically driven, hospital‑concentrated category tied to scheduled antenatal visits. Its baseline scale is anchored in the universal protocol of fetal heart rate auscultation at each prenatal contact, a standard that generates predictable replacement cycles for hospital‑based Doppler units.
FMI analysts observe a clear bifurcation between entry‑level Doppler devices optimized for audible heart rate confirmation and advanced monitors that incorporate maternal heart rate differentiation, contraction timing, and cloud‑based trend visualization. Standard units face margin compression where procurement is centralized by group purchasing organizations, while growth is concentrated in SKUs that justify higher pricing through validated algorithm performance, seamless EMR integration, and documented reduction in unscheduled clinic visits for high‑risk pregnancies.
- WHO antenatal care recommendation update: The World Health Organization’s February 2025 revision to its antenatal care guidelines explicitly recommends Doppler ultrasound for fetal heart rate detection over Pinard stethoscopes and adds a new directive to “integrate fetal monitoring data into the continuum of maternal health records.” This guidance influences procurement specifications in UN‑funded maternal health programs and national tenders across low‑ and middle‑income countries. [2]
- FDA guidance on wireless medical devices: The USA Food and Drug Administration’s final guidance on wireless medical device interoperability, published in September 2024, clarifies expectations for cybersecurity, data formatting, and real‑time transmission reliability. Manufacturers incorporating Bluetooth or cellular connectivity in home‑use fetal monitors must now demonstrate compliance with these standards before 510(k) clearance, raising development costs but reducing market entry for uncertified competitors. [3]
- Maternal mortality review committee recommendations: Rising number of USA states mandating maternal mortality review committees has increased scrutiny of prenatal surveillance adequacy. Committee reports frequently cite delayed detection of fetal distress as a contributing factor in preventable stillbirths, driving hospital systems to adopt continuous or near‑continuous monitoring protocols that favor rental or subscription models for advanced fetal monitors rather than one‑time capital purchase. [4]
Segmental Analysis
Radiation-Free Fetal Heart Rate Monitor Market Analysis by Product Type
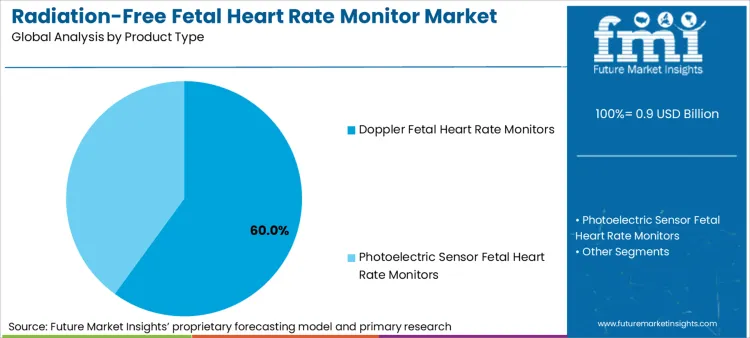
Based on FMI's radiation-free fetal heart rate monitor market report, demand for Doppler fetal heart rate monitors remains the leading product stream, capturing 60.0% of the market share. Rising preference for Doppler monitors is sustained by it’s inclusion in every major clinical guideline for fetal assessment, its ability to provide real‑time auditory confirmation that reassures both clinicians and parents, and its compatibility with existing clinical workflows without requiring retraining.
- Regulatory alignment: The UK National Institute for Health and Care Excellence updated its intrapartum care guideline in January 2025, reinforcing Doppler ultrasound as the preferred method for intermittent auscultation during labor and recommending against routine use of cardiotocography for low‑risk pregnancies. This specification preserves Doppler’s central role while limiting unwarranted substitution by more expensive continuous monitoring systems. [5]
- Product engineering: Huntleigh Healthcare launched the FD3+ Pocket Doppler in April 2025, featuring a rechargeable battery system that supports 12 hours of continuous use and an integrated heart rate display that eliminates the need for separate counting timers. The device is specifically designed for community midwifery programs in sub‑Saharan Africa and South Asia, where grid power availability is inconsistent. [6]
- Technology convergence: FMI's analysis shows that manufacturers are increasingly combining Doppler with maternal vital signs monitoring in single devices. Dräger’s 2024 introduction of the Infinity M540 fetal/maternal monitor allows simultaneous display of fetal heart rate, maternal ECG, and uterine activity on a single screen, reducing equipment footprint in labor and delivery suites. [7]
Radiation-Free Fetal Heart Rate Monitor Market Analysis by End‑User
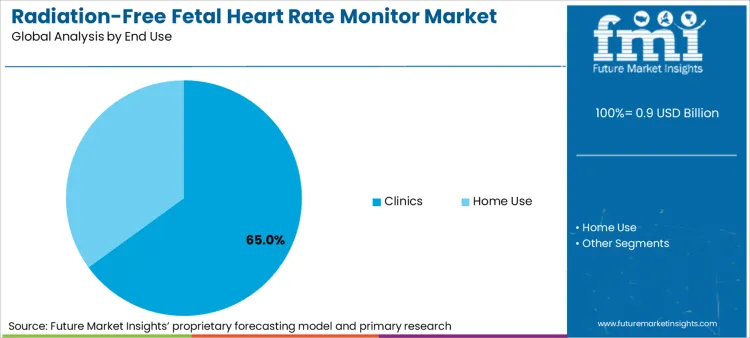
Clinics generate maximum demand at 65.0% for radiation-free fetal heart rate monitors because the vast majority of prenatal contacts occur in ambulatory settings where brief, intermittent monitoring suffices for routine risk stratification. FMI attributes the dominance of clinics to capital budgeting cycles that favor durable, multi‑patient devices over single‑patient disposables, and by infection control policies that discourage device sharing between hospital and home settings.
- Channel expansion: Beurer expanded its distribution agreement with Boots UK in March 2025 to include the Beurer BY90 Doppler fetal monitor in 400 retail pharmacy locations, marking the first large‑scale availability of a clinical‑grade fetal monitor outside traditional medical equipment channels. The devices are positioned as “for home use under midwife guidance” rather than as consumer electronics, preserving clinical credibility while expanding accessibility. [8]
- Procurement consolidation: Inclination towards group purchasing organization consolidation in the USA hospital market is compressing margins for standalone Doppler units but creating volume guarantees for manufacturers that can supply integrated monitoring platforms. The Vizient 2025 maternal health contract award favored suppliers offering fetal monitors that natively interface with Epic and Cerner electronic health records, a specification that excluded several incumbent Doppler‑only vendors. [9]
- Home‑use prescription acceleration: The National Health Service England published a service specification for remote fetal monitoring in high‑risk pregnancies in August 2024, authorizing integrated care boards to commission home‑use fetal Doppler services with cloud‑based data review by community midwives. The specification requires devices to transmit structured data directly into the BadgerNet maternity information system, creating a technical barrier that favors established health IT integrators over consumer‑electronics entrants. [10]
Regional Analysis
The radiation-free fetal heart rate monitor market is segmented geographically across North America, Europe, East Asia, South Asia & Pacific, Latin America, and Middle East & Africa. Growth dynamics differ based on maternal care infrastructure density, digital health reimbursement policies, and the prevalence of high‑risk pregnancy surveillance programs. The full report also provides market attractiveness analysis across regions.
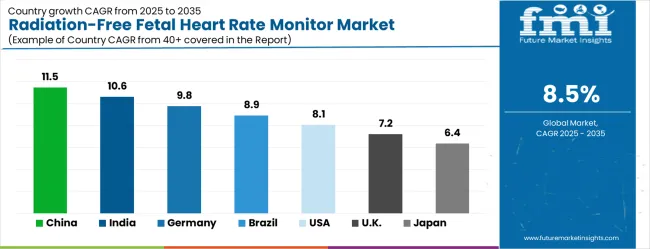
| Country | CAGR (2026-2036) |
|---|---|
| China | 11.5% |
| India | 10.6% |
| Germany | 9.8% |
| Brazil | 8.9% |
| USA | 8.1% |
| UK | 7.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Market Analysis
North America functions as a technology‑validation and reimbursement‑benchmark region for the global fetal monitor market. According to FMI, hospital purchasing decisions are heavily influenced by compatibility with dominant electronic health record platforms (Epic, Cerner) and by inclusion in value‑based care contracts that tie obstetric reimbursement to patient outcome metrics.
- United States: Demand for radiation-free fetal heart rate monitors in the United States is forecasted to advance at 8.1% CAGR through 2036. Growth is driven not by birth rate increases-which remain near historic lows-but by the substitution of intermittent Doppler monitoring with near‑continuous wearable systems for the 20-25% of pregnancies classified as high‑risk. The Centers for Medicare & Medicaid Services’ 2024 introduction of a new remote physiologic monitoring reimbursement code specific to pregnancy has accelerated health system investment in home‑use fetal monitor programs. [11]
FMI's analysis shows that unlike emerging markets, where growth is volume‑led through facility expansion, USA growth is value‑led through technology upgrade cycles and the conversion of capital purchase to device‑as‑a‑service subscription models that include continuous software updates and clinical decision support algorithms.
FMI’s analysis of radiation-free fetal heart rate monitor market in North America consists of country‑wise assessment that includes the United States and Canada. Readers can find CMS reimbursement code tracking, EHR integration case studies, and group purchasing organization contract award analysis.
Europe Market Analysis
Europe acts as the regulatory‑stringent, digital‑health‑integrated frontier for fetal monitoring adoption. As per FMI's analysis, the region’s fragmented reimbursement landscape, with statutory health insurance in Germany, NHS in the UK, and mixed public‑private systems elsewhere, creates varied market access pathways that manufacturers must navigate product‑by‑product.
- Germany: Demand for radiation-free fetal heart rate monitors in Germany is projected to rise at 9.8% CAGR through 2036. Growth is influenced by the Federal Institute for Drugs and Medical Devices (BfArM) DiGA fast‑track reimbursement pathway, which as of 2025 includes two home‑use fetal monitors meeting the criteria for provisional 12‑month reimbursement. Inclusion requires demonstration of positive care effects through comparative studies. FMI opines that this creates a premium tier in the German market: DiGA‑listed devices commanding ASPs exceeding €500, while non‑listed consumer Doppler devices compete below €150 in purely discretionary retail channels. [12]
FMI’s analysis of radiation-free fetal heart rate monitor market in Europe consists of country‑wise assessment that includes Germany, United Kingdom, France, Italy, and Spain. Readers can find DiGA application trend analysis, MDR transitional compliance tracking, and NHS England remote monitoring service specification analysis.
Asia-Pacific Market Analysis
Asia‑Pacific remains the volume expansion engine and manufacturing center of gravity for the global fetal monitor market. The region benefits from rising institutional birth rates, government‑led maternal health infrastructure investments, and dense medical device supply chains capable of producing cost‑optimized devices for price‑sensitive procurement tenders.
- China: Radiation-free fetal heart rate monitor sales in China are projected to rise at 11.5% CAGR through 2036. Growth is supported by the National Health Commission’s 2024 “Action Plan for Further Improvement of Maternal Safety,” which mandates that all secondary and tertiary hospitals maintain continuous electronic fetal monitoring capability in labor and delivery suites. The plan also includes procurement subsidies for county‑level maternal health centers to upgrade from Pinard stethoscopes to Doppler devices. [13]
China’s strength lies in its ability to rapidly scale domestic manufacturing of NMPA‑registered devices through companies such as CONTEC and Banglijian, which compete aggressively on price in public hospital tenders while Philips and Dräger capture the premium academic medical center segment. According to FMI, the market is increasingly bifurcated between domestically produced devices meeting baseline specifications and internationally branded devices offering advanced analytics and EMR integration.
FMI’s analysis of radiation-free fetal heart rate monitor market in Asia‑Pacific consists of country‑wise assessment that includes China, India, Japan, and South Korea. Readers can find NMPA registration tracking, public tender award analysis, and domestic manufacturing scale‑up trends.
- India: Demand for radiation-free fetal heart rate monitors in India is projected to rise at 10.6% CAGR through 2036. The Ministry of Health and Family Welfare’s LaQshya program, which certifies labor rooms and maternity operating theaters meeting quality standards including availability of functional Doppler fetal monitors, support growth. As of 2025, over 1,200 district hospitals and medical colleges have received LaQshya certification, each requiring multiple monitoring devices. [14]
Unlike China’s emphasis on continuous monitoring in tertiary centers, India’s growth is weighted toward basic Doppler devices for high‑volume public facilities and emerging demand for home‑use monitors in urban private practice. The government’s “Pradhan Mantri Surakshit Matritva Abhiyan” fixed‑day antenatal care program creates concentrated demand for portable devices that can be transported to outreach camps in rural areas.
FMI’s analysis of radiation-free fetal heart rate monitor market in Asia‑Pacific consists of country‑wise assessment that includes China, India, Japan, and South Korea. Readers can find LaQshya certification trends, public procurement pipeline analysis, and domestic manufacturing incentive scheme tracking.
Competitive Aligners for Market Players
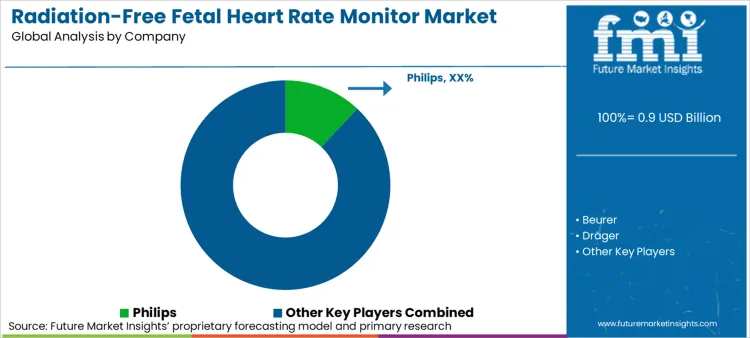
The radiation-free fetal heart rate monitor market is moderately consolidated at the global premium tier and highly fragmented at the domestic volume tier. FMI's analysis shows that the top five global players-Philips, Dräger, Medtronic, Nihon Kohden, and Huntleigh Healthcare-collectively control approximately 45-50% of global value revenue, particularly in hospital‑grade Doppler and integrated monitoring systems.
The primary competitive variable is no longer raw device sensitivity but software ecosystem integration, regulatory registration velocity in emerging markets, and the ability to supply total cost‑of‑ownership packages including training, maintenance, and clinical decision support algorithms.
Large players benefit from dedicated regulatory teams managing multi‑country medical device registrations and from global service networks that support multi‑year equipment warranties. Domestic manufacturers in China and India compete on price and local tender responsiveness but face margin compression in commodity Doppler segments and limited access to the premium connected‑device market requiring advanced software development.
Buyer behavior in this market reflects two distinct procurement modes. Hospital systems and group purchasing organizations exhibit extended evaluation cycles requiring device validation studies, interoperability testing, and service level agreements. Public sector tenders in emerging markets emphasize lowest‑cost technically compliant bids, favoring domestic manufacturers with localized production and lower overhead structures.
Recent Developments
- In May 2025, Philips introduced AI‑enhanced Avalon CL Fetal Monitor series with 40% artifact reduction and seamless IntelliSpace Perinatal platform integration. [1]
- Huntleigh Healthcare launched the FD3+ Pocket Doppler in April 2025. This device comes with a 12‑hour rechargeable battery for community midwifery programs in off‑grid settings. [6]
- In 2024, Dräger introduced Infinity M540 fetal/maternal monitor combining fetal heart rate, maternal ECG, and uterine activity display on single screen. [7]
- In March 2025, Beurer expanded UK distribution to 400 Boots pharmacy locations for BY90 Doppler fetal monitor under midwife‑guidance positioning. [8]
Key Players in the Radiation-Free Fetal Heart Rate Monitor Market
- Philips
- Beurer
- Dräger
- Medtronic
- Nihon Kohden
- Huntleigh Healthcare
- Babyfun
- CONTEC
- Banglijian
Scope of Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 939.7 million (2026) to USD 2,124.8 million (2036), at a CAGR of 8.5% |
| Market Definition | The radiation-free fetal heart rate monitor market comprises the global production and supply of non‑invasive medical devices using Doppler ultrasound or photoelectric sensors to detect fetal heart rate without ionizing radiation, for use in hospital obstetrics, clinics, and home‑care settings. |
| Product Type Segmentation | Doppler Fetal Heart Rate Monitors, Photoelectric Sensor Fetal Heart Rate Monitors, Others |
| End‑User Segmentation | Clinics, Home Use, Others |
| Region Segmentation | North America, Europe, East Asia, South Asia & Pacific, Latin America, Middle East & Africa |
| Countries Covered | United States, Canada, Germany, United Kingdom, France, Italy, Spain, China, India, Japan, South Korea, Brazil and 40+ countries |
| Key Companies Profiled | Philips, Beurer, Dräger, Medtronic, Nihon Kohden, Huntleigh Healthcare, Babyfun, CONTEC, Banglijian |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top‑down and bottom‑up market modeling validated through primary interviews with fetal monitor manufacturers, obstetrics procurement managers, and digital health platform integrators, supported by trade benchmarking and medical device registration tracking |
Radiation-Free Fetal Heart Rate Monitor Market Analysis by Segments
Product Type
- Doppler Fetal Heart Rate Monitors
- Photoelectric Sensor Fetal Heart Rate Monitors
- Others
End‑User
- Clinics
- Home Use
- Others
Region
- North America (USA, Canada, Mexico)
- Latin America (Brazil, Chile, Rest of Latin America)
- Western Europe (Germany, UK, Italy, Spain, France, Nordic, BENELUX, Rest of Western Europe)
- Eastern Europe (Russia, Poland, Hungary, Balkan & Baltic, Rest of Eastern Europe)
- East Asia (China, Japan, South Korea)
- South Asia and Pacific (India, ASEAN, Australia & New Zealand, Rest of South Asia and Pacific)
- Middle East & Africa (Kingdom of Saudi Arabia, Other GCC Countries, Turkiye, South Africa, Other African Union, Rest of Middle East & Africa)
Bibliography
- [1] Philips. (2025, May 14). Philips introduces next‑generation Avalon CL Fetal Monitor with AI‑based signal processing. Philips Press Release.
- [2] World Health Organization. (2025, February 20). WHO recommendations on antenatal care for a positive pregnancy experience: 2025 update. Geneva: WHO.
- [3] USA Food and Drug Administration. (2024, September 27). Wireless Medical Devices: Interoperability Considerations - Guidance for Industry and Food and Drug Administration Staff. Silver Spring, MD: FDA.
- [4] Centers for Disease Control and Prevention. (2024). Enhancing Reviews and Surveillance to Eliminate Maternal Mortality (ERASE MM) Program: Annual Report 2024. Atlanta: CDC.
- [5] National Institute for Health and Care Excellence. (2025, January 15). Intrapartum care for healthy women and babies (NG235) - Update. London: NICE.
- [6] Huntleigh Healthcare. (2025, April 3). Huntleigh launches FD3+ Pocket Doppler for community midwifery programs. Huntleigh Corporate News.
- [7] Drägerwerk AG & Co. KGaA. (2024, October 22). Dräger introduces Infinity M540 fetal/maternal monitor for integrated labor suite monitoring. Dräger Medical Press Release.
- [8] Beurer GmbH. (2025, March 11). Beurer expands UK distribution of BY90 Doppler fetal monitor to 400 Boots pharmacy locations. Beurer Corporate News.
- [9] Vizient Inc. (2025, February 5). Vizient announces 2025 maternal health technology contract awards. Vizient Press Release.
- [10] NHS England. (2024, August 29). Service specification: Remote fetal monitoring in high‑risk pregnancy (E08/S/a). London: NHS England.
- [11] Centers for Medicare & Medicaid Services. (2024, November 1). Medicare Program; CY 2025 Payment Policies under the Physician Fee Schedule and Other Changes to Medicare Part B (CMS‑1807‑F). Federal Register, 89(212), 87654-87658.
- [12] Bundesinstitut für Arzneimittel und Medizinprodukte. (2025, January 29). DiGA Directory Update: Inclusion of fetal monitoring devices. BfArM Notice 2025‑003. Bonn: BfArM.
- [13] National Health Commission of the People's Republic of China. (2024, June 12). Action Plan for Further Improvement of Maternal Safety (2024-2027). Beijing: NHC.
- [14] Ministry of Health and Family Welfare, Government of India. (2025, January 10). LaQshya Program: Progress Report and Certified Facilities List as of December 2024. New Delhi: MoHFW.
- [15] Israel Ministry of Health. (2024, December 5). Digital health strategy 2025-2030: Remote monitoring in pregnancy. Jerusalem: MoH Israel.
This Report Addresses
- Market intelligence to enable structured strategic decision‑making across fetal monitor manufacturers, maternal health program administrators, and digital health platform integrators.
- Market size estimation and 10‑year revenue forecasts from 2026 to 2036, including validated CAGR and absolute dollar opportunity sizing.
- Growth opportunity mapping across Product Type, End‑User, and regional demand pockets, with emphasis on conversion of episodic clinic monitoring to continuous connected surveillance for high‑risk pregnancies.
- Segment and regional forecasting covering Doppler and photoelectric sensor technologies, alongside end‑use demand trajectories across hospital obstetrics, ambulatory clinics, and home‑care settings.
- Competition strategy assessment, including EMR integration capability, regulatory registration velocity, and clinical evidence generation shaping supplier retention in hospital system contracts.
- Product and compliance tracking focused on wireless interoperability standards, AI‑based artifact reduction validation, and real‑world evidence generation for DiGA reimbursement dossiers.
- Regulatory impact analysis covering WHO antenatal guideline revisions, FDA wireless device guidance, and national maternal health program specifications influencing procurement and market access.
- Report delivery in PDF, Excel, and presentation‑ready formats to support executive planning, R&D portfolio alignment, and market access strategy development.
Frequently Asked Questions
How large is the demand for radiation-free fetal heart rate monitors in the global market in 2026?
Demand for radiation-free fetal heart rate monitors is estimated to be valued at USD 939.7 million in 2026.
What will be the market size of radiation-free fetal heart rate monitors in the global market by 2036?
Market size for radiation-free fetal heart rate monitors is projected to reach USD 2,124.8 million by 2036.
What is the expected demand growth for radiation-free fetal heart rate monitors between 2026 and 2036?
Demand for radiation-free fetal heart rate monitors is expected to grow at an 8.5% CAGR between 2026 and 2036.
Which product type is poised to lead global sales?
Doppler fetal heart rate monitors are expected to remain the leading product type, anchored by clinical gold standard status and real time audio feedback, holding 60.0% share in 2025.
Which end user segment dominates the radiation-free fetal heart rate monitor market?
Clinics are the dominant end user segment at 65.0% share in 2025, reflecting protocol driven monitoring at scheduled antenatal visits and capital budgets of hospital obstetrics departments.
What is driving demand in China?
The National Health Commission’s 2024 maternal safety action plan mandating continuous electronic fetal monitoring in all secondary and tertiary hospitals, plus procurement subsidies for county level centers, is driving demand in China.
What is China's growth outlook in this report?
China is projected to expand at an 11.5% CAGR during 2026 to 2036.
What is driving demand in India?
The Ministry of Health’s LaQshya certification program requiring functional Doppler monitors in labor rooms and the Pradhan Mantri Surakshit Matritva Abhiyan outreach camps are driving demand in India.
What is India's growth outlook in this report?
India is projected to expand at a 10.6% CAGR during 2026 to 2036.
Why is Germany described as a strategic digital health market in this report?
Germany's DiGA fast track reimbursement pathway for home use fetal monitors prescribed for high risk pregnancies creates a premium tier with ASPs exceeding €500, compared to <€150 for non listed consumer devices.
What is Germany's growth outlook in this report?
Germany is projected to expand at a 9.8% CAGR during 2026 to 2036.
Does the report cover the United Kingdom in its regional analysis?
Yes, the United Kingdom is included within Europe under the regional coverage framework.
What is the United Kingdom's growth outlook in this report?
The United Kingdom is projected to expand at a 7.2% CAGR during 2026 to 2036.
Does the report cover the United States in its regional analysis?
Yes, the United States is included within North America and is covered in the country wise assessment.
What is the United States growth outlook in this report?
The United States is projected to expand at an 8.1% CAGR during 2026 to 2036.
Does the report cover Japan in its regional analysis?
Yes, Japan is included within East Asia under the regional scope of analysis.
What is Japan's growth outlook in this report?
Japan is projected to expand at a 6.4% CAGR during 2026 to 2036.
Does the report cover Brazil in its regional analysis?
Yes, Brazil is included within Latin America and is covered in the country wise assessment.
What is Brazil's growth outlook in this report?
Brazil is projected to expand at an 8.9% CAGR during 2026 to 2036.
What is a radiation-free fetal heart rate monitor and what is it mainly used for?
A radiation-free fetal heart rate monitor is a non invasive medical device using Doppler ultrasound or photoelectric sensors to detect and record fetal cardiac activity without ionizing radiation, used for routine prenatal surveillance, high risk pregnancy management, and expectant parent reassurance.
What does the radiation-free fetal heart rate monitor market mean in this report?
It refers to the global manufacturing and supply of radiation-free fetal heart rate monitors by product type, end user, and region, measured in value terms at the medical device manufacturing layer.
What is included in the scope of this radiation-free fetal heart rate monitor market report?
The report’s scope includes market sizing and forecasting for 2026 to 2036, segmentation by product type, end user, and region, along with competitive assessment, regulatory impact analysis, and country wise outlook.
What is excluded from the scope of this report?
The radiation-free fetal heart monitor market report excludes fetal monitors using ionizing radiation (X ray, CT), invasive fetal scalp electrodes requiring ruptured membranes, non medical novelty devices lacking clinical validation, and telemedicine consultation services not bundled with proprietary hardware.
How are regulatory developments shaping the radiation-free fetal heart rate monitor market?
Regulatory developments including WHO antenatal guideline revisions, FDA wireless interoperability standards, NMPA maternal safety mandates, and German DiGA reimbursement integration are shifting competitive advantage from hardware sensitivity to data interoperability, clinical evidence generation, and regulatory registration velocity.
What is the competitive landscape of the radiation-free fetal heart rate monitor market?
The market is moderately consolidated at the global premium tier (Philips, Dräger, Medtronic, Nihon Kohden, Huntleigh Healthcare controlling 45–50% of value revenue) and highly fragmented at the domestic volume tier. Competition is shifting from raw device sensitivity to software ecosystem integration, regulatory registration speed, and total cost of ownership packages including training and clinical decision support.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Doppler Fetal Heart Rate Monitors
- Photoelectric Sensor Fetal Heart Rate Monitors
- Doppler Fetal Heart Rate Monitors
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Clinics
- Home Use
- Clinics
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End Use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End Use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End Use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End Use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End Use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By End Use
- Competition Analysis
- Competition Deep Dive
- Philips
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Beurer
- Dräger
- Medtronic
- Nihon Kohden
- Huntleigh Healthcare
- Philips
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by End Use
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Product Type
- Figure 23: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by End Use
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Product Type
- Figure 30: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by End Use
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by End Use
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Product Type
- Figure 51: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by End Use
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

