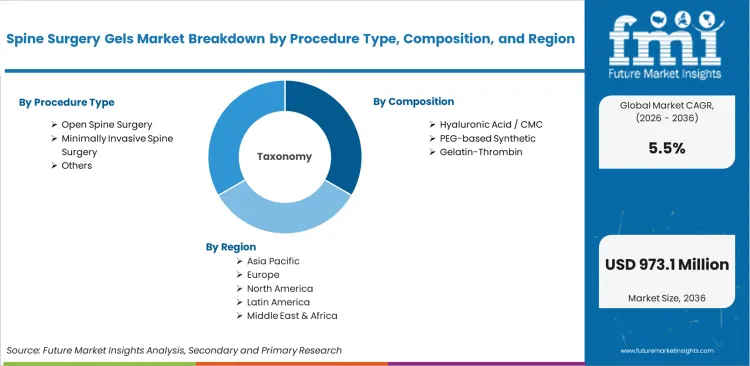
Spine Surgery Gels Market
The Spine Surgery Gels Market is segmented by Product Function (Anti-adhesion barrier gels, Hemostatic gels, Dural / tissue sealant gels, Bone graft carrier gels, Lubricating / access gels), Procedure Type (Open spine surgery, Minimally invasive spine surgery, Revision spine surgery, Deformity correction), Composition (Hyaluronic acid / CMC, PEG-based synthetic, Gelatin-thrombin, Alginate / chitosan, Other polymers), End User (Hospitals, ASCs, Specialty spine centers) and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Spine Surgery Gels Market Size, Market Forecast and Outlook By FMI
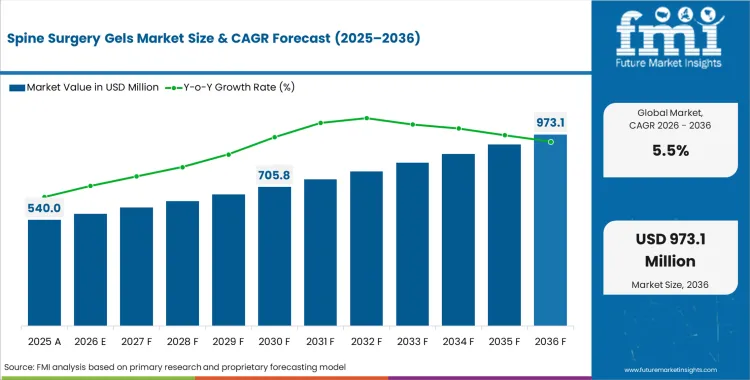
In 2026, the spine surgery gels market was valued at USD 540.0 million. Based on Future Market Insights analysis, demand for spine surgery gels is estimated to grow to USD 920.7 million by 2036. FMI projects a CAGR of 5.5% during the forecast period.
Absolute dollar growth of USD 380.7 million over the decade points to measured expansion rather than a rapid category shift. As per FMI, demand is expected to be supported by steady spine procedure volume and greater use of adjunctive gels to manage adhesion control, sealing, hemostasis, and access support, while product evaluation cycles, hospital formulary discipline, and surgeon preference concentration are expected to keep switching gradual in mature markets. "Patients who present with both severe leg pain and significant back pain are historically more difficult to treat surgically, this study shows that adding Oxiplex at the time of surgery can meaningfully increase the likelihood of achieving greater pain relief and reducing residual neurological symptoms in this challenging patient population." said Kee Kim, MD, study co-author and Chief of Spinal Neurosurgery at UC Davis Health (Sacramento, CA). [1]
Summary of Spine Surgery Gels Market
- Market Definition
- The market includes gel products that are employed during spine procedures to provide support for surgical handling, tissue protection, hemostasis, sealing, prevention of tissue adhesions, graft placement, and access during open and minimally invasive spine procedures. These products are employed in various settings, including hospitals, ambulatory surgery centers, and specialty spine centers, where surgeons increasingly recognize the importance of employing products that provide support for tissue handling and minimize tissue-related complications during procedures.
- Demand Drivers in the Market
- The increase in spinal procedures, including degenerative, revision, and deformity surgery, is driving the demand for gel-based products to facilitate ease of handling.
- The trend towards increased usage of workflow-efficient adjuncts in spine surgery is driving the adoption of gel products for hemostasis, sealing, anti-adhesive support, and grafting.
- Hospitals and spine specialty centers are focusing more on products that can reduce disruption, increase accuracy, and optimize recovery pathways.
- Key Segments Analyzed in the FMI Report
- By Procedure Type: Open Spine Surgery leads at 46.0% share in 2026, followed by minimally invasive spine surgery at 34.0% share.
- By Composition: Hyaluronic Acid / CMC leads at 28.0% share in 2026, followed by PEG-based synthetic at 22.0% share.
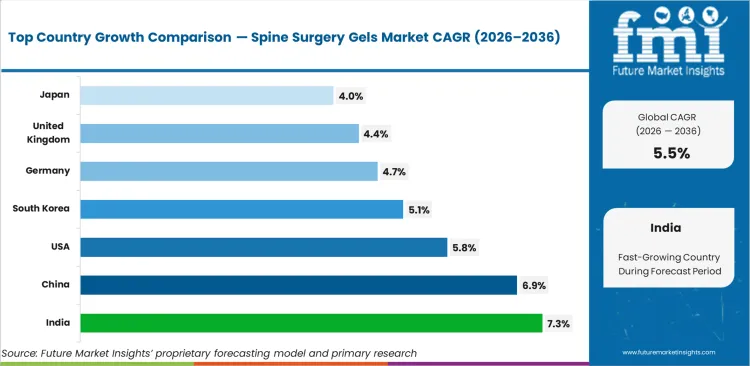
- Geography: India and China show the fastest growth at 7.3% and 6.9% CAGR, supported by rising spinal procedure volumes, improving surgical infrastructure, and growing use of advanced adjunct products in hospital-based spine care.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant at Future Market Insights, opines, “Supplier preference is increasingly being shaped by procedure-specific usability, composition reliability, and the ability to integrate smoothly into demanding spinal workflows, as surgical teams look for gel-based adjuncts that improve control without adding unnecessary complexity to already sensitive operative settings.”
- Strategic Implications and Executive Takeaways
- Build commercial positioning around intraoperative usability, tissue management value, and procedure-specific performance, especially in high-volume hospital spine programs.
- Prioritize open and minimally invasive spine surgery settings where gel-based products can demonstrate clearer workflow and surgical outcome relevance.
- Strengthen growth plans in emerging surgical markets where spine procedure access is expanding and hospitals are upgrading to more advanced operative adjunct portfolios.
- Methodology
- As per FMI, sizing and splits were reconciled using company-level product mapping, procedure-level demand allocation, and composition-based positioning analysis across spine surgery gel categories.
- Segment shares were validated by linking procedure intensity and surgical workflow needs with material preference, product role, and end-user adoption patterns.
- Pricing sensitivity was stress tested under procedure mix and composition mix shifts to confirm revenue response under realistic surgical procurement and utilization scenarios.
Procurement is being shaped by intraoperative usability and outcome-related confidence, with buyers giving weight to handling characteristics, procedural fit, storage simplicity, and compatibility with established surgical routines. Hospitals remain the primary demand center because open and complex spine procedures continue to be concentrated there, while ASC use is rising where minimally invasive spine surgery is expanding and product selection is tied to efficiency and recovery-oriented workflow design.
India (7.3% CAGR, supported by rising spine surgery access and broader hospital expansion) and China (6.9% CAGR, driven by growing procedural volume and specialist center development) are expected to lead growth. The USA (5.8% CAGR) is expected to remain the largest market due to broad specialist use and established adoption of adjunctive surgical materials. Mature markets including South Korea (5.1%), Germany (4.7%), the UK (4.4%), and Japan (4.0%) are expected to contribute more through replacement purchases and selective procedure expansion, constrained by committee review cycles and cautious product standardization.
Spine Surgery Gels Market Definition
The market covers gel-based adjunct products used during spine procedures to support tissue management, hemostasis, sealing, adhesion reduction, graft handling, and instrument or access lubrication. These products are used across open spine surgery, minimally invasive spine surgery, revision procedures, and deformity correction cases, with formulations based on hyaluronic acid or CMC, PEG-based synthetic materials, gelatin-thrombin systems, alginate or chitosan, and other polymer platforms. Buyers include hospitals, ambulatory surgical centers, and specialty spine centers that use these gels to improve procedural control and reduce workflow friction. Demand is shaped by surgeon preference, procedural complexity, and handling performance during surgery.
Spine Surgery Gels Market Inclusions
The report includes global and regional market sizing and a 10-year forecast for 2026 to 2036. Segment-level sizing is provided by product function, procedure type, composition, and end user, with country-level CAGR comparisons across key markets. Coverage also includes assessment of replacement demand, spine procedure mix, and procurement priorities such as handling profile, intraoperative usability, and fit across open and minimally invasive workflows, alongside competitive positioning of major suppliers active in spine surgery gel categories, based on FMI analysis.
Spine Surgery Gels Market Exclusions
The scope excludes non-gel spinal implants, fixation devices, interbody cages, and biologic graft substitutes sold as separate categories. It also omits sutures, mechanical hemostats, barrier films, and surgical instruments that do not deliver gel-based functionality. Procedure fees, hospital service revenue, and rehabilitation-related care are excluded unless directly tied to product value capture. The focus remains on gel-based adjunct products aligned to the listed functions, procedure settings, compositions, end users, and supplier participation.
Spine Surgery Gels Market Research Methodology
- Primary Research: Interviews were conducted with spine surgeons, OR decision-makers, procurement teams, ASC administrators, and supplier commercial teams to validate usage, product preferences, and purchasing criteria.
- Desk Research: Public information was gathered from sources including regulators, spine society references, standards sources, and primary disclosures made by companies to validate product positioning, procedural relevance, and supplier activities.
- Market-Sizing and Forecasting: A hybrid bottom up modelling and top-down approach to forecasting was adopted, utilizing demand for spine procedures, adoption of adjunct products, and utilization of end users to reconcile the results to shares of the segments by product function, type, and composition.
- Data Validation and Update Cycle: The results were validated by internal consistency checks to screen results for variance in total segments and country growth rates.
Segmental Analysis
Spine Surgery Gels Market Analysis by Procedure Type
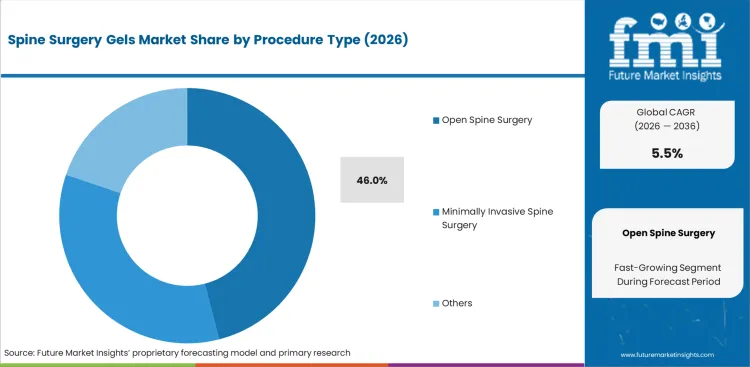
Based on FMI’s report, open spine surgery is estimated to hold 46.0% share in 2026. This lead position is supported by the broader use of gel-based products in more exposed and complex operative settings where bleeding control, tissue separation, sealing support, and adhesion management can play a more visible role. Minimally invasive spine surgery accounts for 34.0% share, while revision spine surgery contributes 12.0% and deformity correction represents 8.0%. Open spine surgery remains the leading segment because procedural exposure is greater, operative complexity is often higher, and the clinical value of adjunctive gels is easier to establish across multiple steps of the surgical workflow.
- Open surgery creates broader need for dural sealing and watertight closure support: Open spine surgery is more often used in decompression-heavy, revision, or complex exposures where incidental dural tears and CSF leak management become an important intraoperative concern. Recent reviews on spinal dural defects and sealants show that sealants and sealing patches are used to reinforce repair and reduce postoperative CSF leakage, making gel- and sealant-based adjuncts more clinically relevant in open workflows where the surgical field is fully exposed and repair options can be applied across multiple steps. [2]
- Greater bleeding burden makes hemostatic gels more relevant in open procedures: Open spine procedures generally involve more tissue exposure, higher intraoperative blood loss, and greater postoperative drainage than less invasive techniques, which increases the practical need for topical hemostatic matrices and flowable gels. Recent comparative studies and reviews report that open lumbar fusion approaches show higher blood loss and drainage than MIS or endoscopic approaches, reinforcing why gel-based hemostatic support is more visible and more routinely useful in open cases. [3]
- Stronger suitability for multi-step operative workflows: Open spine surgery provides a wider procedural setting where gel-based products can be applied across different surgical stages, increasing their practical relevance compared with more limited-access approaches.
Spine Surgery Gels Market Analysis by Composition
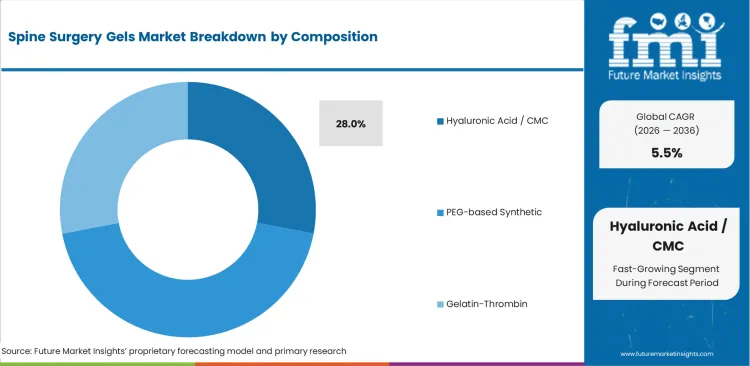
Hyaluronic acid / CMC accounts for 28.0% share in 2026, based on FMI’s report, reflecting its position as the largest composition segment in the market. This leadership is supported by its established role in barrier and tissue-interface applications where smooth handling, biocompatibility, and anti-adhesion utility are valued in spine procedures. PEG-based synthetic holds 22.0% share, gelatin-thrombin contributes 18.0%, while alginate / chitosan and other polymers account for 16.0% each. Hyaluronic acid / CMC leads because it fits well with surgical demand for materials that can support tissue protection and controlled placement without creating unnecessary workflow burden.
- Strong clinical relevance in anti-adhesion use after spine procedures: Recent spinal biomaterials reviews describe epidural fibrosis and postoperative adhesions as persistent concerns after spine surgery, and they specifically note barrier-type hydrogels as a key prevention strategy. HA/CMC is well suited to this role because both materials are widely recognized for forming a temporary physical barrier at the surgical site, which helps separate tissues during healing and limits scar-related attachment. [4]
- Longstanding evidence base and continued relevance versus newer polymer options: HA/CMC benefits from an established evidence base in adhesion prevention and nerve-related scar control, which supports continued surgeon familiarity and product acceptance. Broader adhesion-prevention reviews report that CMC/HA combinations have shown success in both animal and human studies, while experimental work has shown HA-CMC solutions can reduce scar formation and adhesion around repaired nerve tissue. That stronger historical foundation helps explain why HA/CMC remains the leading composition even as newer PEG and synthetic variants expand. [5]
- Established familiarity in adjunctive spinal care: Hyaluronic acid / CMC compositions benefit from broader clinical familiarity in barrier-based surgical applications, which supports stronger acceptance in spine procedures where dependable adjunct performance is preferred.
Spine Surgery Gels Market Drivers, Restraints, and Opportunities
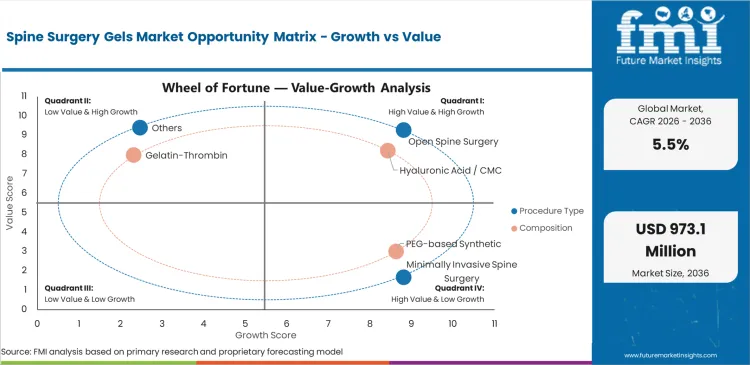
Future Market Insights analysis indicates that this market is developing as spine surgery becomes more adjunct-driven and more focused on products that improve procedural control, tissue protection, and recovery support. Estimated valuation in 2026 is being supported by growing use of gel-based products across open and minimally invasive procedures where surgeons want better management of bleeding, tissue interaction, sealing, and post-operative complication risk. Demand is increasingly being shaped by the need for products that fit naturally into the surgical workflow while offering meaningful functional support in technically sensitive spinal interventions.
At the same time, market expansion is being moderated by product selection complexity, variable surgeon preference, and cost scrutiny in settings where adjunct adoption remains tightly evaluated. Even so, forecast growth remains supported by rising spine procedure volumes, better access to specialized surgical care, and greater familiarity with composition-specific gel functions in operative practice. Based on FMI’s report, the outlook reflects a market where procedure type strongly influences product need, composition affects adoption confidence, and supplier advantage increasingly depends on clinical usability rather than broad portfolio breadth alone.
- Procedure-support Demand: Demand is being supported as spine surgeons increasingly use gels to improve control, protect tissues, and reduce disruption during complex procedures.
- Adoption Variability: Uptake can be slowed where surgeon preference, hospital cost review, and case-by-case product selection create inconsistent usage patterns.
- Advanced Adjunct: Demand is being supported as hospitals and spine centers expand use of specialized operative adjuncts that improve workflow and post-surgical management.
Regional Analysis
Based on the regional analysis, spine surgery gels market is segmented into North America, East Asia, South Asia & Pacific, and Western Europe across 40+ countries. Regional performance is interpreted through case mix across open and minimally invasive spine procedures, surgeon preference for anti-adhesion and hemostatic support materials, and purchasing patterns among hospitals, ASCs, and specialty spine centers, as per FMI. The full report also offers market attractiveness analysis based on regional trends.
<.webp)
| Country | CAGR |
|---|---|
| United States | 5.8% |
| Germany | 4.7% |
| United Kingdom | 4.4% |
| Japan | 4.0% |
| China | 6.9% |
| India | 7.3% |
| South Korea | 5.1% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Spine Surgery Gels Market Analysis
North America remains a procedure-led market, where gel adoption is closely linked to surgeon preference, hospital protocol familiarity, and the complexity of open and revision spine cases. Integra LifeSciences holds visibility through dural and adjunct surgical repair portfolios, while Medtronic and Stryker remain relevant through broad spine surgery relationships and established hospital access. Baxter and Johnson & Johnson (Ethicon) also influence the market where hemostatic and sealant products are evaluated within broader OR purchasing pathways. FMI analysts note that hospitals account for the largest demand share, while ASCs contribute more selectively in lower-complexity settings.
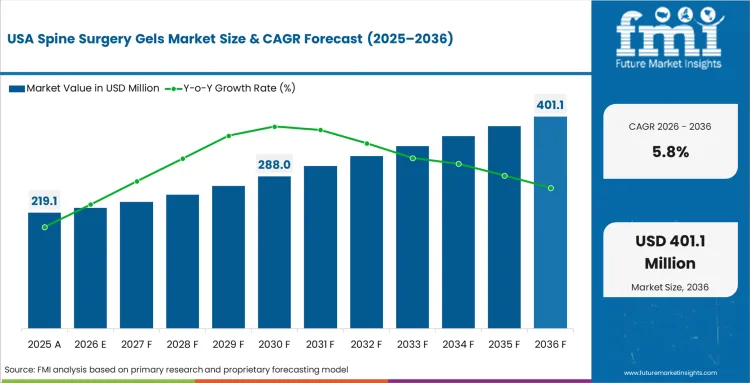
- USA: In the USA, spine surgery gels are expected to advance at 5.8% CAGR through 2036, supported by stable open spine volumes, continued use in minimally invasive procedures, and growing attention to adjunct products that help improve intraoperative handling and post-surgical management.
FMI’s report includes a detailed analysis of the growth in the North American region, along with a country-wise assessment that includes the USA, Canada and Mexico. Readers can also find regional trends, regulations, and market growth based on different segments and countries in the North America region.
East Asia Spine Surgery Gels Market Analysis
East Asia's development is characterized by increasing degenerative spine cases, higher surgical volumes, and stronger use of adjunctive materials in tertiary centers. Product adoption is influenced by surgeons' familiarity with open and minimal access techniques, whereas purchasing decisions are largely influenced by value considerations. Integra LifeSciences, Medtronic, Stryker, and Globus Medical are key companies in this region due to their presence in the hospital ecosystem in the spine business. Other companies in this region include local suppliers and established adjunct companies. FMI's opinion is that the development in this region is increasingly associated with high-volume hospitals.
- China: China leads in this group at a 6.9% CAGR through 2036, and growth is fueled by increasing spine surgery volumes in large urban hospitals and better utilization of adjunctive materials in open and minimally invasive spine procedures. Procedure demand is fueled by degenerative spine conditions and an increasing spine surgery base in large urban hospitals, where there is a greater likelihood that spine surgeons will consider and evaluate gel materials for their ability to improve tissue handling, reduce adhesions, and provide hemostatic support.
- Japan: The Japanese market is growing at a 4.0% CAGR through 2036, and growth in this market is fueled by a mature spine surgery market and a more gradual adoption pattern for adjunctive gel materials. The market is fueled by steady volumes in open and revision spine procedures, rather than rapid increases in spine procedures.
- South Korea: South Korea is expected to expand at 5.1% CAGR through 2036, supported by an active surgical environment and rising use of adjunctive materials in high-volume spine centers. Minimally invasive spine surgery is contributing to demand, though open procedures still account for the largest share of gel usage because they more frequently require tissue protection and bleeding control support.
FMI’s report includes a detailed analysis of the growth in the East Asia region, along with a country-wise assessment that includes China, Japan and South Korea. Readers can also find regional trends, regulations, and market growth based on different segments and countries in the East Asia region.
South Asia & Pacific Spine Surgery Gels Market Analysis
South Asia & Pacific is emerging as a faster-growth opportunity, where rising access to spine surgery, improving hospital infrastructure, and expanding specialist care networks are increasing awareness of adjunctive gels. Product demand is being shaped by a mix of open spine procedures, minimally invasive adoption in private hospitals, and broader use of supportive materials in complex spinal cases. Medtronic, Globus Medical, and Integra LifeSciences remain relevant where premium hospital systems and specialist surgeons evaluate advanced procedure support tools. FMI analysts note that the region’s momentum is being built more through concentrated urban demand than through evenly distributed market maturity.
- India: India has a forecasted CAGR of 7.3% till 2036. This makes it the highest-growing country in this dataset. This has been achieved due to the rising number of spine surgeries in metro hospitals, improved adoption of minimally invasive techniques in private centers, and the willingness to use adjunctive gels in complex open cases.
The report published by FMI provides readers with a detailed analysis of the growth in the South Asia & Pacific region. In addition to this, readers can also find information regarding country-wise analysis, including India, ASEAN Countries, Australia & New Zealand, and Rest of South Asia. Moreover, readers can find information regarding regional trends, regulations, and market growth based on different segments and countries in the South Asia & Pacific region.
Western Europe Spine Surgery Gels Market Analysis
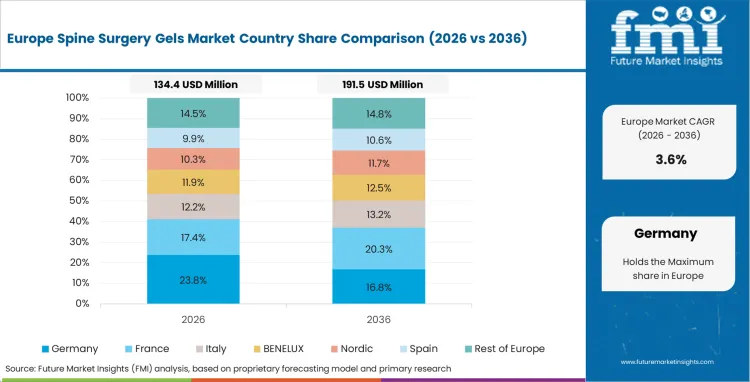
Western Europe continues to be a structured and clinically selective market, with adoption rates being influenced by surgeon familiarity, evidence demands, and hospital procurement discipline. The use of Spine Surgery Gels is more prevalent in environments that favor open surgeries, revision procedures, and complex spinal corrections that require the use of adjunctive materials. Integra LifeSciences, Medtronic, Stryker, and Baxter continue to be significant contributors through established spine and surgical support structures, with product selection being a function of known product handling and cost discipline. FMI opines that the market has potential for steady growth; however, this growth is a function of selective clinical use rather than any form of standardization.
- Germany: The German market is expected to grow at a CAGR of 4.7% until 2036, driven by stable spine surgery volumes and the continued use of gels in open and revision surgeries for tissue management and adhesion reduction.
- United Kingdom: The UK market is expected to grow at a CAGR of 4.4% until 2036, driven by stable volumes and the continued use of gels in open spine surgeries and selected minimally invasive spine surgeries. The use of gels is not widespread and is typically more prominent in those cases in which the surgeons have a clear history of use and the product fits into the workflow of the procedure.
- France: France is expected to rise at 4.8% CAGR through 2036, supported by a steady role for adjunctive gels in open spine surgery, deformity correction, and selected revision cases.
FMI’s analysis of spine surgery gels market in Western Europe consists of country-wise assessment that includes Germany, UK, France, Italy, Spain, BENELUX, Nordic Countries and Rest of Western Europe. Readers can know various regulations and latest trends in the regional market.
Competitive Aligners for Market Players
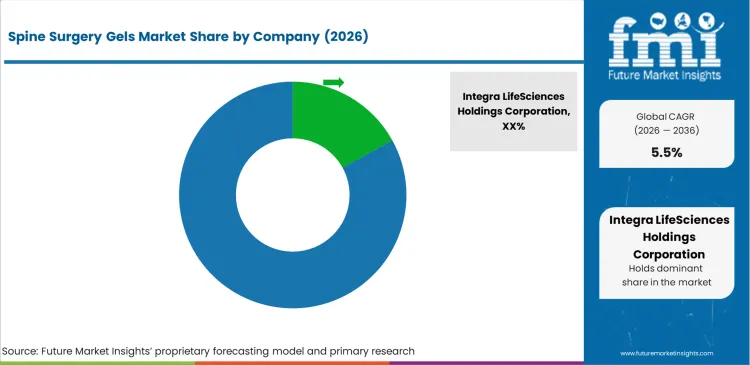
Market structure remains fragmented, yet practical competition is concentrated among a limited set of suppliers that can deliver consistent gel handling in the operative field, maintain sterile manufacturing and shelf-life control, and support hospital contracting with dependable availability. The supplied dataset indicates a clear scale leader at about 11.0% share, while the remaining demand is distributed across diversified surgical and spine-focused manufacturers. The main competitive factor is predictable intraoperative performance tied to surgeon workflow, not short-cycle price movement, because adoption is reinforced through standardised protocols and repeat use in high-volume spine programs. Product function and procedure fit shape rivalry since adhesion barriers and hemostatic gels are used in different steps of care and are evaluated on different clinical expectations, as per FMI.
Companies with structural advantage generally have access to a strong channel in the spine space and also have a broad product offering in surgical consumables to support bundled contracting and access to purchasing committees. Suppliers that have access to multiple formulations to support targeted use cases, such as hyaluronic acid, PEG-based synthetic matrices, and gelatin thrombin, can provide surgeons with their preferred choice while also minimizing the need to access alternate suppliers. However, companies with access to scale and quality systems also have a competitive advantage, as hospitals review suppliers based on their sterility assurance, viscosity, and completeness of kits. Backorder issues also impact suppliers, as hospitals penalize companies for impacting case scheduling. Companies with existing clinical education also have a competitive advantage, as technique plays a critical role in product performance, creating repeat business, according to FMI’s report.
Customer concentration reinforces buyer leverage. Hospitals remain the main purchasing centers and commonly manage supplier dependency through approved alternates, competitive tenders, and value analysis reviews that compare clinical utility against overall consumable cost per case. ASCs enforce tighter price discipline and standardise to fewer SKUs where case mix is predictable, while specialty spine centers place higher weight on surgeon preference and supply continuity. This purchasing behavior limits pricing power for routine use, while measured premiums are retained mainly where a gel reduces operative time, improves field control, or lowers rework risk in complex cases, Future Market Insights analysis.
Recent Developments
The report includes full coverage of key trends from competitive benchmarking. Some of the recent developments covered in the reports:
- In March 2026, Fziomed, Inc., a recognized global leader in postsurgical adhesion prevention with the best-in-class synthetic, absorbable gel technology platform, announced results from a prospective, randomized, double-blinded, multi-center clinical study demonstrating that Oxiplex intraoperative gel improves pain and neurological outcomes for patients with severe leg and back pain when used as an adjuvant during lumbar spine surgery. [1]
- In March 2025, Pramand LLC announced the US commercial launch of the SpineSeal Spine Sealant System following recent premarket approval by the US Food and Drug Administration. SpineSeal is indicated for use as an adjunct to sutured dural repair during spinal surgery to provide watertight closure. [6]
- In July 2025, Medcura, Inc. has formed a Spinal Surgery Advisory Board comprising nine expert spine surgeons to guide the development of its LifeGel™ Absorbable Hemostatic Gel. Led by Chief Medical Officer Dr. Kenneth Renkens, the board will support clinical trials, regulatory strategy, and commercialization, aiming to address unmet surgical needs and improve patient outcomes.
Key Players in Spine Surgery Gels Market
- Integra LifeSciences Holdings Corporation
- Medtronic plc
- Stryker Corporation
- Johnson & Johnson
- Baxter International Inc.
- Anika Therapeutics, Inc.
- Globus Medical, Inc.
- Seikagaku Corporation
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 540.0 Mn (2026) to USD 920.7 Mn (2036), at a CAGR of 5.5% |
| Market Definition | The spine surgery gels market comprises global production and trade of gel-based products used during spine procedures to support adhesion prevention, hemostasis, sealing, graft handling, and surgical access, where demand is shaped by procedure mix, surgeon workflow preferences, and hospital protocol selection for intraoperative use. |
| Product Function Segmentation | Anti-adhesion barrier gels, Hemostatic gels, Dural or tissue sealant gels, Bone graft carrier gels, Lubricating or access gels |
| Procedure Type Coverage | Open spine surgery, Minimally invasive spine surgery, Revision spine surgery, Deformity correction |
| Composition Coverage | Hyaluronic acid or CMC, PEG-based synthetic, Gelatin-thrombin, Alginate or chitosan, Other polymers |
| End User Coverage | Hospitals, ASCs, Specialty spine centers |
| Regions Covered | North America, Latin America, East Asia, South Asia & Pacific, Western Europe, Eastern Europe, and Middle East & Africa. |
| Countries Covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, China, India, Japan, South Korea, Indonesia, Australia and 40+ countries |
| Key Companies Profiled | Integra LifeSciences Holdings Corporation, Medtronic plc, Stryker Corporation, Johnson & Johnson, Baxter International Inc., Anika Therapeutics, Inc., Globus Medical, Inc., Seikagaku Corporation |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top down and bottom up market modeling validated through primary interviews with spine surgeons, hospital procurement teams, and suppliers, supported by procedure volume triangulation and product use mapping, as per FMI. |
Spine Surgery Gels Market Analysis by Segments
Product Function
- Anti-adhesion barrier gels
- Hemostatic gels
- Dural or tissue sealant gels
- Bone graft carrier gels
- Lubricating or access gels
Procedure Type
- Open spine surgery
- Minimally invasive spine surgery
- Revision spine surgery
- Deformity correction
Composition
- Hyaluronic acid or CMC
- PEG-based synthetic
- Gelatin-thrombin
- Alginate or chitosan
- Other polymers
End User
- Hospitals
- ASCs
- Specialty spine centers
Region
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- East Asia
- China
- Japan
- South Korea
- South Asia & Pacific
- India
- ASEAN Countries
- Australia & New Zealand
- Rest of South Asia
- Western Europe
- Germany
- UK
- France
- Italy
- Spain
- BENELUX
- Nordic Countries
- Rest of Western Europe
- Eastern Europe
- Russia
- Hungary
- Poland
- Rest of Eastern Europe
- Middle East & Africa
- Saudi Arabia
- Türkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- [1] Fziomed, Inc. (2026). Fziomed Announces Clinical Results from Level I Study Evaluating Oxiplex Absorbable Gel for Spine Surgery.
- [2] PubMed Central (2023). Clinical management of dural defects: A review.
- [3] PubMed Central (2026). Long-term comparative study of Open-TLIF, MIS-TLIF, and UBE-TLIF in single-level degenerative lumbar spondylolisthesis
- [4] PubMed Central (2024). Polymeric Dural Biomaterials in Spinal Surgery: A Review
- [5] PubMed Central (2022). Barrier materials for prevention of surgical adhesions: systematic review.
- [6] Pramand LLC (2025). Pramand Launches SpineSeal Spine Sealant to Give Surgeons a New Option for Spinal Dural Repair.
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
This Report Addresses
- Market intelligence to enable structured decision making across intraoperative adjunct selection and spine program planning
- Market size estimation and 10-year revenue forecasts from 2026 to 2036 for spine surgery gel demand
- Growth opportunity mapping across product function, procedure type, composition, and end user purchasing pools
- Segment and regional revenue forecasts aligned to the stated taxonomy and covered geographies
- Competition strategy assessment including portfolio positioning, procedure fit, and channel coverage benchmarking
- Product and compliance tracking focused on sterile use expectations, labeling discipline, and procurement documentation requirements
- Regulatory impact analysis covering medical device approval expectations and facility protocol alignment affecting adoption planning
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive and operational use
Frequently Asked Questions
How large is the demand for Spine Surgery Gels in the global market in 2026?
Demand for Spine Surgery Gels in the global market is estimated to be valued at USD 540.0 Mn in 2026, as per FMI.
What will be the market size of Spine Surgery Gels in the global market by 2036?
Market size for Spine Surgery Gels is projected to reach USD 920.7 Mn by 2036.
What is the expected demand growth for Spine Surgery Gels in the global market between 2026 and 2036?
Demand for Spine Surgery Gels in the global market is expected to grow at a CAGR of 5.5% between 2026 and 2036.
Which product function is poised to lead global demand by 2026?
Anti-adhesion barrier gels are expected to lead, capturing 38.0% share in 2026.
Which procedure type is expected to account for the largest share in 2026?
Open spine surgery is expected to hold the highest share at 46.0% in 2026.
How significant is hospital demand in the 2026 end user mix?
Hospitals are projected to account for 74.0% share of end user demand in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Procedure Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Procedure Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Procedure Type , 2026 to 2036
- Open Spine Surgery
- Minimally Invasive Spine Surgery
- Others
- Open Spine Surgery
- Y to o to Y Growth Trend Analysis By Procedure Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Procedure Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Composition
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Composition, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Composition, 2026 to 2036
- Hyaluronic Acid / CMC
- PEG-based Synthetic
- Gelatin-Thrombin
- Hyaluronic Acid / CMC
- Y to o to Y Growth Trend Analysis By Composition, 2021 to 2025
- Absolute $ Opportunity Analysis By Composition, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Procedure Type
- By Composition
- By Country
- Market Attractiveness Analysis
- By Country
- By Procedure Type
- By Composition
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Procedure Type
- By Composition
- By Country
- Market Attractiveness Analysis
- By Country
- By Procedure Type
- By Composition
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Procedure Type
- By Composition
- By Country
- Market Attractiveness Analysis
- By Country
- By Procedure Type
- By Composition
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Procedure Type
- By Composition
- By Country
- Market Attractiveness Analysis
- By Country
- By Procedure Type
- By Composition
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Procedure Type
- By Composition
- By Country
- Market Attractiveness Analysis
- By Country
- By Procedure Type
- By Composition
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Procedure Type
- By Composition
- By Country
- Market Attractiveness Analysis
- By Country
- By Procedure Type
- By Composition
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Procedure Type
- By Composition
- By Country
- Market Attractiveness Analysis
- By Country
- By Procedure Type
- By Composition
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Composition
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Composition
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Composition
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Composition
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Composition
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Composition
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Composition
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Composition
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Composition
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Composition
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Composition
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Composition
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Composition
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Composition
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Composition
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Composition
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Composition
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Composition
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Composition
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Composition
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Composition
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Procedure Type
- By Composition
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Procedure Type
- By Composition
- Competition Analysis
- Competition Deep Dive
- Integra LifeSciences Holdings Corporation
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Medtronic plc
- Stryker Corporation
- Johnson & Johnson
- Baxter International Inc.
- Anika Therapeutics, Inc.
- Integra LifeSciences Holdings Corporation
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Procedure Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Composition, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Procedure Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Composition, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Procedure Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Composition, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Procedure Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Composition, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Procedure Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Composition, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Procedure Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Composition, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Procedure Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Composition, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Procedure Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Composition, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Procedure Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Procedure Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Procedure Type
- Figure 6: Global Market Value Share and BPS Analysis by Composition, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Composition, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Composition
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Procedure Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Procedure Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Procedure Type
- Figure 23: North America Market Value Share and BPS Analysis by Composition, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Composition, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Composition
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Procedure Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Procedure Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Procedure Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Composition, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Composition, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Composition
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Procedure Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Procedure Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Procedure Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Composition, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Composition, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Composition
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Procedure Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Procedure Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Procedure Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Composition, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Composition, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Composition
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Procedure Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Procedure Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Procedure Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Composition, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Composition, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Composition
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Procedure Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Procedure Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Procedure Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Composition, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Composition, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Composition
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Procedure Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Procedure Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Procedure Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Composition, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Composition, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Composition
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

