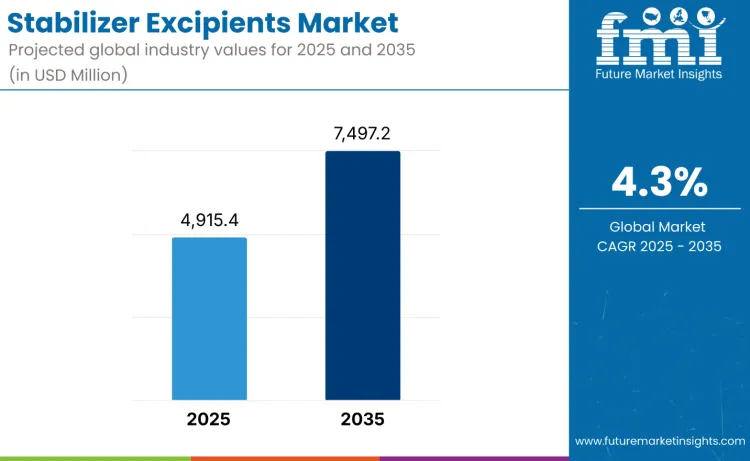
The stabilizer excipients market is set for steady expansion, reaching USD 7,497.2 million by 2035 from USD 4,915.4 million in 2025 at a CAGR of 4.3%. Growth is supported by rising biologics development, increasing formulation complexity across injectable, oral, and inhalation products, and stronger focus on shelf-life extension in pharmaceutical manufacturing. Drug developers face increasing pressure to ensure long-term stability as products move through global supply chains. While adoption continues to rise, progress remains moderated by strict raw material purity expectations, lengthy excipient qualification processes, and regulatory scrutiny surrounding the introduction of novel stabilizing agents.
Market momentum reflects deeper shifts in formulation science, where stabilizer excipients play a central role in preserving molecular integrity and therapeutic performance throughout a product’s lifecycle. Biopharmaceutical manufacturers increasingly rely on stabilizers to control degradation, aggregation, and chemical instability, particularly in protein- and peptide-based therapies. These excipients demonstrate significant improvements in stability during lyophilization, aseptic processing, and temperature stress, making them essential in advanced drug delivery systems. Their role has expanded as biologics, vaccines, and specialty injectables demand greater formulation robustness under increasingly variable storage and transport conditions.
Innovation continues across carbohydrate protectants, amino acid systems, polymeric stabilizers, and sugar alcohol platforms designed to minimize oxidative, thermal, and moisture-related degradation. High-purity grades and co-processed formats improve compatibility with monoclonal antibodies, gene therapies, nucleic acid products, and sustained-release formulations. Government-backed manufacturing expansion, increased bioprocess investment, and stronger academic-industry collaboration further support adoption. Growing cold-chain infrastructure and advanced fill-finish capabilities reinforce demand for stabilizer systems that maintain molecular integrity during large-scale global distribution.
Between 2025 and 2030, the stabilizer excipients market is expected to expand from USD 4,915.4 million to USD 6,070.6 million, adding USD 1,155.2 million and accounting for 44.7% of total decade growth. Expansion is driven by rising biologics manufacturing volumes, increasing formulation complexity, and wider use of high-purity stabilizers in protein therapies, vaccines, and nucleic acid products. Pharmaceutical companies are strengthening positions through investment in excipient-drug interaction modeling, advanced purification processes, and strategic supply partnerships to ensure GMP-grade material continuity and risk mitigation.
From 2030 to 2035, the market is projected to grow from USD 6,070.6 million to USD 7,497.2 million, contributing 55.3% of overall expansion. Demand will be shaped by next-generation biologics, multifunctional stabilizers, sustainability priorities, and deeper collaboration between excipient developers and formulation teams.
| Metric | Value |
|---|---|
| Market Value (2025) | USD 4,915.4 million |
| Market Forecast Value (2035) | USD 7,497.2 million |
| Forecast CAGR (2025 to 2035) | 4.3% |
The stabilizer excipients market expands as pharmaceutical and biopharmaceutical manufacturers seek to preserve drug potency, molecular structure, and long-term stability across increasingly complex formulations. Rising development of monoclonal antibodies, recombinant proteins, mRNA vaccines, and high-concentration injectables has increased reliance on sugars, polyols, amino acids, polymers, and co-processed stabilizers that maintain consistent therapeutic performance through manufacturing, storage, and distribution. These excipients reduce degradation risk, improve formulation success rates, and support regulatory acceptance across biologic, vaccine, and nucleic acid platforms.
Adoption is further driven by global immunization programs, temperature-sensitive therapies, and extended-shelf-life requirements across lyophilized, liquid, and sustained-release formats. Growth in cell and gene therapies and sterile injectable manufacturing strengthens demand for high-purity stabilizers. Government investment supports utilization, though high costs, lengthy qualification, and strict regulatory review can constrain uptake.
The market is segmented by product type, application, and end user. By product type, the market is divided into polymers, sugars and polyols, proteins and amino acids, surfactants, and other stabilizing excipients. Based on application, the market is categorized into biologics and vaccine formulations, small-molecule formulations, nutraceuticals and food supplements, cell and gene therapy formulations, and diagnostic and lyophilized reagent applications. End users include pharmaceutical manufacturers, biopharmaceutical companies, CDMOs/CROs, and academic and research institutes. Regionally, the market is divided into Asia Pacific, Europe, North America, Latin America, and Middle East & Africa.
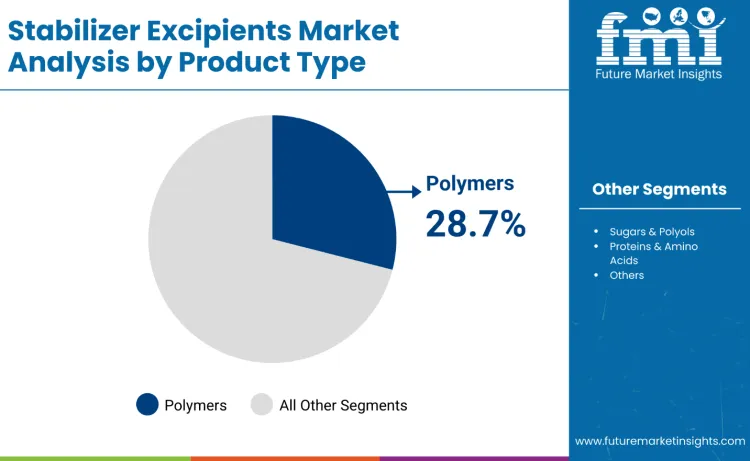
Polymers represent the leading category in the stabilizer excipients market, accounting for 28.7% share in 2025. Pharmaceutical-grade polymers such as PVP, HPMC, and PVA are widely used to enhance physical and chemical stability, control viscosity, and prevent aggregation in sensitive formulations. Their application spans sterile injectables, controlled-release oral systems, lyophilized biologics, and high-concentration parenteral products.
Leadership is driven by functional versatility, compatibility with biologics and small molecules, and reliable performance under freezing, sterilization, and filling conditions. Polymers support dispersion uniformity, moisture resistance, and thermal protection, reinforcing their role in regulated formulations. Established safety profiles, broad regulatory acceptance, and growing demand for advanced biologics continue to strengthen polymer adoption across global formulation pipelines.
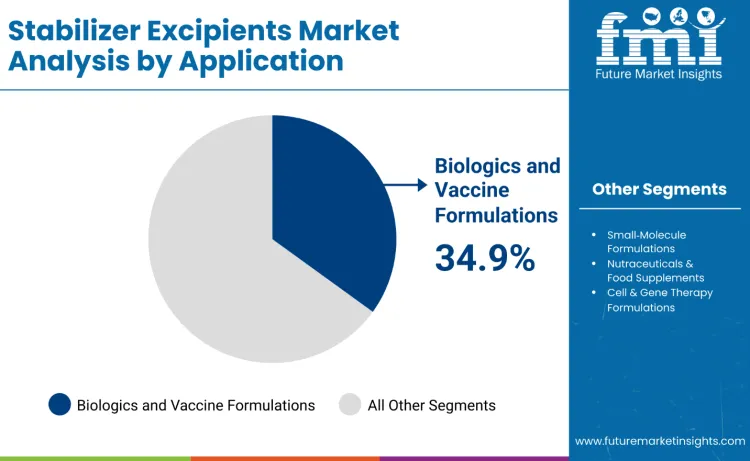
Biologics and vaccine formulations lead the stabilizer excipients market in 2025 with a 34.9% share, supported by expanding monoclonal antibody pipelines, recombinant proteins, RNA-based vaccines, and global immunization programs. Stabilizer excipients play a critical role in preserving molecular structure, potency, and long-term stability across cold-chain and lyophilized formats. Sugars, polyols, amino acids, and polymers protect against denaturation, aggregation, and oxidative stress during processing and distribution.
Growth is reinforced by preventive healthcare initiatives, pandemic preparedness, and demand for thermostable vaccines. Regulatory focus on stability and quality consistency drives extensive formulation screening and adoption of pharmacopeial-compliant excipients compatible with scalable commercial manufacturing.
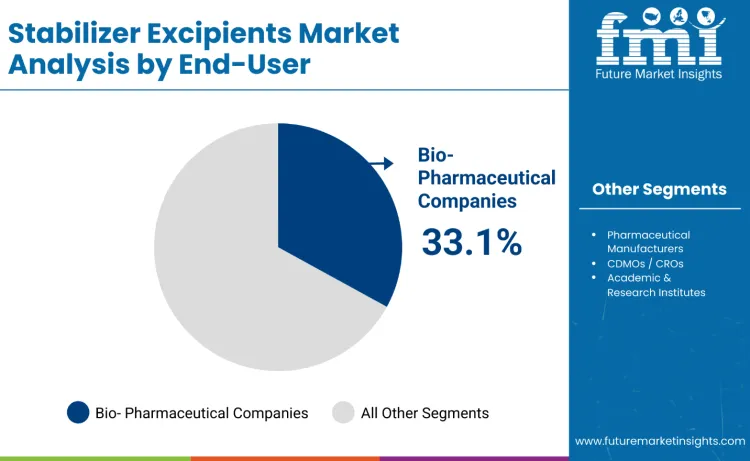
Biopharmaceutical companies represent about 33.1% of stabilizer excipient demand in 2025, reflecting sustained investment in biologics, sterile injectables, and advanced therapy manufacturing. These companies incorporate stabilizers from early formulation through commercial production, prioritizing excipients that enhance molecular stability, extend shelf life, and meet strict GMP and regulatory standards.
Growth in antibody therapies, nucleic acid drugs, and cell-based treatments increases reliance on high-purity stabilizer systems. In-house formulation and analytical teams conduct extensive compatibility and stability testing, ensuring performance under freeze-thaw stress, temperature variation, and long-term storage. Strong internal capabilities, expanding biologics pipelines, and global bioproduction scale reinforce consistent stabilizer excipient adoption.
The stabilizer excipients market is driven by rapid expansion of biologics, vaccines, and advanced injectable therapies. Sustained double-digit growth in monoclonal antibodies, mRNA platforms, and recombinant proteins increases demand for excipients that preserve molecular stability during manufacturing, freezing, lyophilization, and distribution. Biopharmaceutical companies and CDMOs are strengthening formulation capabilities, adopting high-purity sugars, amino acids, and polymer-based stabilizers to prevent aggregation and degradation. Expanding cold-chain infrastructure, fill-finish capacity, and global immunization programs further elevate demand for excipients that maintain potency and shelf-life across commercial and clinical supply chains.
Market restraints include stringent regulatory scrutiny, as stabilizer excipients require extensive compatibility data, toxicology validation, and pharmacopeial compliance, slowing adoption of novel excipient classes. High-purity grades designed for biologics command premium pricing, creating cost pressures for emerging manufacturers and contract producers. Technical challenges related to excipient-API interaction studies, scale-up validation, and long-term stability testing extend development timelines. These complexities require advanced analytical expertise and risk-mitigation strategies to avoid formulation failure, batch variability, or unexpected degradation in high-value biologics and sterile injectable products.
Key trends include rising adoption of co-processed and multifunctional stabilizer systems that improve formulation robustness and manufacturing efficiency in biologics and advanced therapies. Development of thermo-stable and ready-to-use formulations, including room-temperature biologics and next-generation vaccines, is driving innovation toward excipients that reduce cold-chain dependency. Increasing use of digital formulation tools, predictive degradation modeling, and Quality-by-Design approaches is refining excipient selection. Collaboration between excipient suppliers, biopharma companies, and academic institutions is accelerating screening platforms that enhance formulation optimization, regulatory readiness, and time-to-market performance.
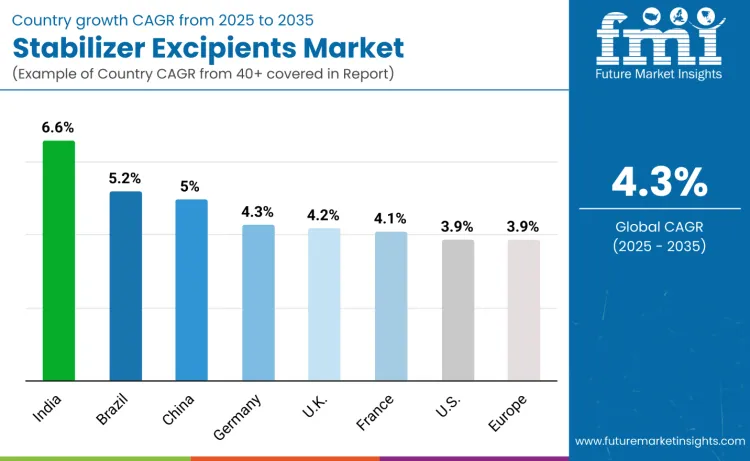
| Country | CAGR (2025 to 2035) |
|---|---|
| USA | 3.9% |
| Brazil | 5.2% |
| China | 5.0% |
| India | 6.6% |
| Europe | 3.9% |
| Germany | 4.3% |
| France | 4.1% |
| UK | 4.2% |
The stabilizer excipients market is expanding globally, led by India with 6.6% growth, supported by rapid biologics manufacturing expansion and vaccine production initiatives. Brazil follows at 5.2%, driven by immunization programs and biosimilar investments, while China records 5.0% growth through large-scale biomanufacturing and high-purity excipient localization. Germany advances at 4.3% on strong biologics innovation and EU GMP compliance. The UK grows at 4.2% and France at 4.1% through biopharma R&D and cell-therapy investments. The United States remains stable at 3.9%, while Europe overall also grows at 3.9%, supported by biosimilar commercialization and stringent quality standards.
The report covers an in-depth analysis of 40+ countries top-performing countries are highlighted below.
China shows one of the strongest growth trajectories in the stabilizer excipients landscape, expanding at a CAGR of 5.0% through 2035. Momentum is driven by large-scale biologics and vaccine manufacturing expansion, national self-reliance programs, and targeted investment in high-purity excipient localization.
Growth clusters around Shanghai, Suzhou, Beijing, Shenzhen, and Guangzhou, where vaccine developers, biosimilar producers, and CDMOs increase consumption of sugars, polymers, amino acids, and surfactants to meet stability requirements for monoclonal antibodies and mRNA platforms. Policy support under Healthy China 2030 and pharmaceutical industrial upgrade initiatives accelerates adoption of GMP-compliant stabilizers across sterile injectable and lyophilized manufacturing environments.
India demonstrates strong stabilizer excipient adoption with a CAGR of 6.6% through 2035, supported by rapid biologics capacity expansion and vaccine manufacturing growth. Biopharma clusters in Hyderabad, Bengaluru, Pune, and Ahmedabad integrate high-purity sugars, polymers, and amino acids into antibody, peptide, and mRNA formulation pipelines. Make-in-India initiatives, BIRAC funding programs, and national biotechnology strategies strengthen formulation science capabilities and GMP-grade raw-material adoption.
Rising biosimilar exports and expanding aseptic fill-finish and lyophilization infrastructure further reinforce demand. Strategic collaboration between global excipient suppliers and Indian CDMOs improves quality alignment, supply security, and regulatory readiness for US FDA, EMA, and WHO-aligned production.
Germany sustains steady stabilizer excipient growth at a CAGR of 4.3% through 2035, driven by a quality-centric biopharmaceutical ecosystem. Biomanufacturers and research institutions across Munich, Berlin, Heidelberg, and Frankfurt integrate high-purity sugars, polymers, and multifunctional stabilizers into monoclonal antibody and recombinant protein formulations.
Strong emphasis on GMP compliance, precision chemistry, and advanced analytical testing supports excipient adoption in lyophilized, cold-chain, and high-concentration biologic formats. Collaboration between universities, research institutes, and excipient producers strengthens formulation science training and excipient characterization. Strategic partnerships ensure EU pharmacopeia compliance, secured supply, and robust documentation across clinical and commercial biologics manufacturing.
Brazil leads Latin America in stabilizer excipient adoption, growing at a CAGR of 5.2% through 2035. Expansion is driven by increasing vaccine production, biosimilar development, and government-backed pharmaceutical self-sufficiency initiatives. Biomanufacturing hubs in São Paulo, Rio de Janeiro, and Brasília integrate sugars, polyols, amino acids, and stabilizing polymers into monoclonal antibody and recombinant protein formulations.
Public immunization programs and rising sterile injectable capacity strengthen stability requirements across lyophilized and liquid biologics. Partnerships with international excipient suppliers, expansion of fill-finish facilities, and growing cold-chain infrastructure further support adoption, ensuring potency preservation and regulatory compliance across domestic and regional biologics pipelines.
The United States maintains stable stabilizer excipient growth at a CAGR of 3.9% through 2035, supported by advanced biologics pipelines and extensive commercial manufacturing networks. Biopharma clusters in Massachusetts, California, North Carolina, and New Jersey deploy high-purity sugars, polymers, amino acids, and surfactants to support formulation stability, lyophilization efficiency, and cold-chain durability.
Strong regulatory expectations drive standardized excipient qualification and analytical validation. Established supply networks ensure consistent access to GMP-grade stabilizers with audit-ready documentation. Collaboration between academia, biotech innovators, CDMOs, and excipient suppliers accelerates formulation optimization, reinforcing adoption across monoclonal antibodies, mRNA vaccines, and injectable biologics.
The United Kingdom records steady stabilizer excipient growth with a CAGR of 4.2% through 2035, supported by biologics innovation and expanding CDMO activity. Biotechnology clusters in London, Cambridge, Oxford, and Manchester integrate stabilizers into monoclonal antibody, RNA, and advanced therapy formulations to enhance stability and shelf life.
National life-sciences strategy investments and expansion of cell- and gene-therapy manufacturing reinforce demand for high-purity excipients. Academic-industry collaboration strengthens formulation training and excipient characterization. Partnerships with global excipient suppliers ensure GMP alignment, technical support, and supply reliability, enabling competitive positioning in European and global biologics manufacturing networks.
The stabilizer excipients market has a consolidated structure, led by a small group of global suppliers that bring strong formulation expertise and dependable manufacturing scale. Competition is shaped more by excipient purity, regulatory readiness, and supply reliability than by pricing alone. Companies with broad portfolios and multi-site GMP production are preferred partners for pharmaceutical and biopharmaceutical manufacturers, as they can support products from early formulation through commercial launch. BASF stands out through its long-established polymer and solubilization platforms, supported by deep technical knowledge and close collaboration with formulation teams across injectable and oral biologics.
Leading suppliers such as BASF, Roquette, and Ashland continue to strengthen their positions through wide-ranging stabilization solutions, global distribution networks, and consistent regulatory performance. Challenger players including Merck MilliporeSigma and Thermo Fisher Scientific compete by offering integrated bioprocessing and formulation support, while specialists like Evonik, Croda, DFE Pharma, and Avantor focus on high-performance chemistries and ready-to-use GMP formats. Regional manufacturers add competitive pressure with high-purity alternatives, particularly in emerging biopharma hubs.
| Items | Values |
|---|---|
| Quantitative Units | USD 4,915.4 million |
| Product Type | Polymers (PVP, HPMC, PVA), Sugars & Polyols (sucrose, trehalose, mannitol, sorbitol), Proteins & Amino Acids (gelatin, albumin, glycine), Surfactants (polysorbates, poloxamers), Others (organic acids, PEGs, salts) |
| Application | Biologics & Vaccine Formulations, Small‑Molecule Formulations, Nutraceuticals & Food Supplements, Cell & Gene Therapy Formulations, Diagnostic & Lyophilized Reagents |
| End User | Pharmaceutical Manufacturers, Biopharmaceutical Companies, CDMOs / CROs, Academic & Research Institutes |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Country Covered | China, India, Germany, Brazil, USA, UK, Japan, and 40+ countries |
| Key Companies Profiled | BASF (Kollidon/Kolliphor), Ashland (Benecel/Aquarius), Roquette (Mannitol/Sorbitol/Trehalose), Merck MilliporeSigma, Thermo Fisher Scientific, Evonik (EUDRAGIT & additives), Croda (Span/Tween & excipients), DFE Pharma, Avantor, Others |
| Additional Attributes | Dollar sales by stabilizer class and formulation, regional adoption linked to biologics scale, competitive landscape of excipient suppliers and CDMOs, regulatory benchmarks, integration with bioprocessing and fill-finish workflows, and innovations in co-processed, bio-derived, high-purity stabilizers for biologics and mRNA platforms. |
How big is the stabilizer excipients market in 2025?
The global stabilizer excipients market is valued at USD 4,915.4 million in 2025.
What will be the size of the stabilizer excipients market in 2035?
The stabilizer excipients market is projected to reach USD 7,497.2 million by 2035.
How much will the stabilizer excipients market grow between 2025 and 2035?
The stabilizer excipients market is expected to grow at a CAGR of 4.3% between 2025 and 2035.
Which product type leads the stabilizer excipients market in 2025?
Polymers lead the stabilizer excipients market with an approximate 28.7% share in 2025.
Who are the major players in the stabilizer excipients market?
Key players include BASF, Roquette, Ashland, Merck MilliporeSigma, and Thermo Fisher Scientific.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.