Sterilization Monitoring Supplies Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
Sterilization Monitoring Supplies Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Sterilization Monitoring Supplies Market Forecast and Outlook 2026 to 2036
The sterilization monitoring supplies market is projected to reach USD 1,990.6 million in 2026 and expand to USD 4,141.0 million by 2036, at a CAGR of 7.6%. Value growth reflects structural dependence on consumable monitoring tools required for every sterilization cycle across healthcare, pharmaceutical, and device manufacturing environments. Spending is recurring rather than discretionary, anchored in routine use of chemical indicators, biological indicators, and test packs that verify cycle performance under regulatory oversight. Demand intensity tracks procedural volume, audit frequency, and sterilizer utilization rather than capital equipment expansion, supporting predictable revenue formation.
Market expansion is shaped by compliance discipline, infection control accountability, and increasing decentralization of sterile processing activities. Utilization rises where accreditation frameworks mandate documented evidence for each sterilization load, particularly within hospitals and contract sterilization facilities operating high cycle counts. Product selection emphasizes modality specificity, traceability, and integration with electronic quality records, reinforcing supplier lock-in and repeat procurement patterns. Cost pressure, modality diversity, and workflow adherence moderate scalability, keeping growth aligned with operational sterilization activity and regulatory enforcement consistency rather than episodic capacity build-out.
Quick Stats for Sterilization Monitoring Supplies Market
- Sterilization Monitoring Supplies Market Value (2026): USD 1,990.6 million
- Sterilization Monitoring Supplies Market Forecast Value (2036): USD 4,141.0 million
- Sterilization Monitoring Supplies Market Forecast CAGR 2026 to 2036: 7.6%
- Leading Product Category by Demand Share: Chemical indicators
- Fastest-Growing Countries: India, Brazil, China, USA, Germany
- Top Players in Global Demand: STERIS, Getinge, 3M, Mesa Labs, Fortive
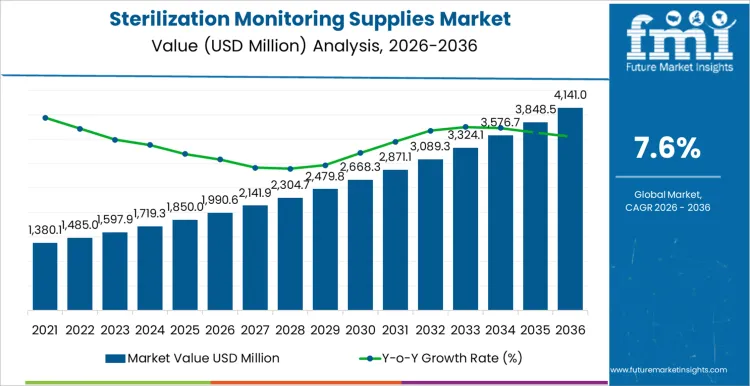
Sterilization Monitoring Supplies Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 1,990.6 million |
| Market Forecast Value (2036) | USD 4,141.0 million |
| Forecast CAGR 2026 to 2036 | 7.6% |
Why is the Demand for Sterilization Monitoring Supplies Growing?
Demand for sterilization monitoring supplies is increasing as healthcare facilities, pharmaceutical manufacturers, and life science laboratories emphasize validated sterilization processes to protect patients, products, and regulatory compliance. Monitoring supplies such as biological indicators, chemical indicators, integrators, and Bowie-Dick tests provide objective evidence that sterilization parameters, temperature, pressure, time, and sterilant concentration, have been met in autoclaves, dry heat ovens, ethylene oxide chambers, and vaporized hydrogen peroxide systems. Infection prevention teams and quality managers specify monitoring supplies that align with sterilizer types and load configurations because effective monitoring reduces the risk of undetected sterilization failure that could lead to contamination, recalls, or compromised instruments. Procurement specialists evaluate product accreditation, lot traceability, reporting formats, and compatibility with electronic documentation systems to ensure reliable integration with quality systems and audit readiness.
Growth in regulatory expectations and accreditation requirements reinforces adoption of advanced monitoring supplies that support documented performance and risk mitigation. Healthcare institutions are expanding sterilization capacity due to increased procedural volumes, ambulatory surgical centers, and outpatient care settings, which elevates reliance on accurate, repeatable monitoring results. Pharmaceutical and medical device manufacturers also integrate monitoring into manufacturing execution systems to maintain batch release records and support compliance with current good manufacturing practice frameworks. Manufacturers of monitoring supplies continue to refine indicator sensitivity, provide visual and electronic readouts, and streamline protocols to reduce time to result, which improves operational efficiency. These operational, compliance, and quality assurance drivers are contributing to sustained demand growth in the sterilization monitoring supplies market.
How Is the Sterilization Monitoring Supplies Industry Segmented by Product Category, Application Area, and End User?
Demand for sterilization monitoring supplies is shaped by regulatory compliance requirements, infection control protocols, and audit readiness across healthcare and manufacturing settings. Facilities prioritize reliable verification of sterilization efficacy across diverse modalities. Adoption reflects increasing procedural volumes, decentralization of sterilization activities, and emphasis on traceable documentation. Selection focuses on compatibility with sterilization methods, ease of interpretation, and integration with quality systems. Segmentation explains how monitoring tools, sterilization processes, and organizational responsibilities influence purchasing and usage patterns.
Which Product Category Accounts for the Largest Share of Sterilization Monitoring Supplies?
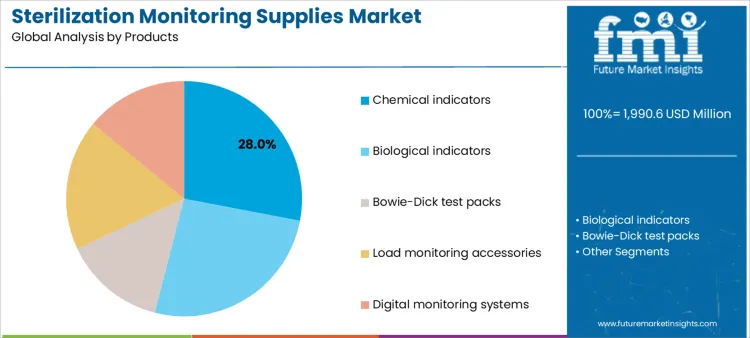
Chemical indicators account for 28.0%, reflecting their routine use for immediate visual confirmation of exposure parameters. These indicators support daily cycle checks across multiple sterilization methods. Biological indicators at 26.0% provide definitive microbial inactivation verification and are central to periodic validation protocols. Load monitoring accessories at 18.0% assist in placement consistency and documentation accuracy. Bowie-Dick test packs at 14.0% assess steam penetration and air removal in pre-vacuum systems. Digital monitoring systems at 14.0% enable electronic tracking and data retention. Product segmentation shows balanced reliance on rapid checks and confirmatory validation tools.
Key Points:
- Chemical indicators support routine cycle verification.
- Biological indicators underpin validation assurance.
- Digital systems strengthen traceability and records.
How Do Sterilization Applications Influence Monitoring Supply Utilization?
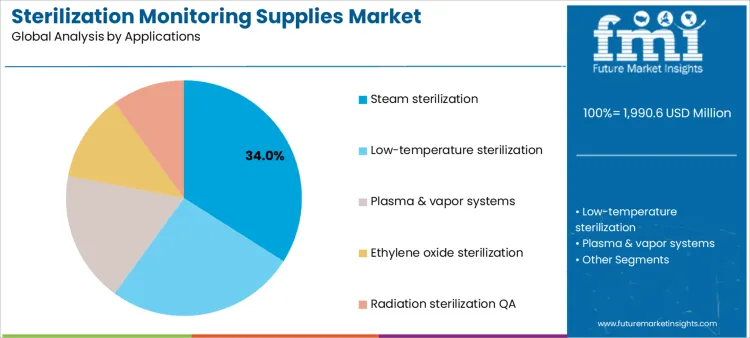
Steam sterilization represents 34.0%, driven by widespread use in hospitals and industrial settings requiring frequent monitoring. Low-temperature sterilization at 26.0% supports heat-sensitive devices needing alternative verification tools. Plasma and vapor systems at 18.0% apply specialized indicators matched to specific process chemistries. Ethylene oxide sterilization at 12.0% requires extended monitoring due to complex cycles and safety controls. Radiation sterilization quality assurance at 10.0% focuses on batch-level verification and documentation. Application-based segmentation highlights higher utilization where process variability and regulatory scrutiny demand consistent monitoring.
Key Points:
- Steam processes drive the highest monitoring frequency.
- Low-temperature systems require modality-specific indicators.
- Complex methods emphasize documentation and assurance.
Which End User Groups Drive Demand for Sterilization Monitoring Supplies?
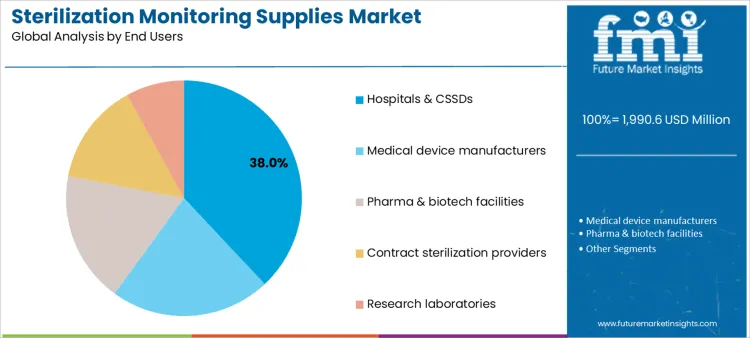
Hospitals and CSSDs account for 38.0%, reflecting continuous sterilization activity and infection prevention mandates. Medical device manufacturers at 22.0% integrate monitoring into production and release workflows. Pharma and biotech facilities at 18.0% apply supplies within aseptic manufacturing and validation programs. Contract sterilization providers at 14.0% manage monitoring across diverse client loads and processes. Research laboratories at 8.0% use monitoring tools for controlled studies and pilot operations. End-user segmentation shows concentration where accountability, throughput, and compliance requirements are highest.
Key Points:
- Hospitals require continuous, routine monitoring.
- Manufacturers embed monitoring into quality systems.
- Service providers manage multi-client verification needs.
What are the Key Dynamics in the Sterilization Monitoring Supplies Market?
Demand for sterilization monitoring supplies reflects need to verify effectiveness of sterilization processes across healthcare, pharmaceutical, laboratory, and device manufacturing environments. Adoption concentrates in hospitals, central sterile departments, contract sterilization facilities, and regulated production sites. Global scope aligns with mandatory process validation and routine compliance monitoring. Usage centers on chemical indicators, biological indicators, and integrators that confirm exposure to defined sterilization parameters during each cycle.
How do compliance verification requirements and risk management practices shape adoption?
Sterilization processes require objective evidence that critical parameters were achieved for every load. Demand increases where patient safety and product sterility depend on documented process control. Chemical indicators provide immediate visual confirmation of temperature, time, or gas exposure, supporting routine load release decisions. Biological indicators deliver higher assurance by challenging cycles with resistant microorganisms, supporting periodic validation and audit readiness. Healthcare facilities rely on monitoring supplies to standardize workflows across varied sterilization modalities. Device manufacturers integrate monitoring into quality systems to meet regulatory expectations. Adoption reflects operational reliance on layered verification to minimize sterilization failure risk and support traceable compliance.
How do modality diversity, workflow integration, and cost pressure influence market scalability?
Monitoring supplies must align precisely with sterilization modality, cycle parameters, and load configuration. Demand sensitivity rises where incorrect indicator selection compromises result relevance. Interpretation variability increases training requirements and error risk. Biological indicator incubation time delays release decisions in time-sensitive operations. Consumable cost accumulates with high cycle volumes, affecting budget planning in large facilities. Inventory management complexity grows with multiple sterilization technologies in use. Regulatory documentation and lot traceability add administrative burden. Scalability remains constrained by modality-specific requirements, recurring consumable cost, and dependence on disciplined workflow execution across decentralized sterilization environments.
How Is Demand for Sterilization Monitoring Supplies Evolving Globally?
Demand for sterilization monitoring supplies is expanding globally as healthcare providers and device manufacturers strengthen assurance of sterile processing outcomes. Biological indicators, chemical indicators, and integrators support routine validation across steam, low-temperature, and alternative sterilization modalities. Regulatory audits and accreditation standards increase monitoring frequency and documentation rigor. Expansion of surgical volumes and centralized sterile services departments influences consumption patterns. Growth rates in India at 10.1%, Brazil at 9.6%, China at 9.2%, the USA at 6.0%, and Germany at 5.9% indicate sustained expansion driven by compliance requirements, process verification needs, and infection control priorities rather than episodic sterilization capacity additions.
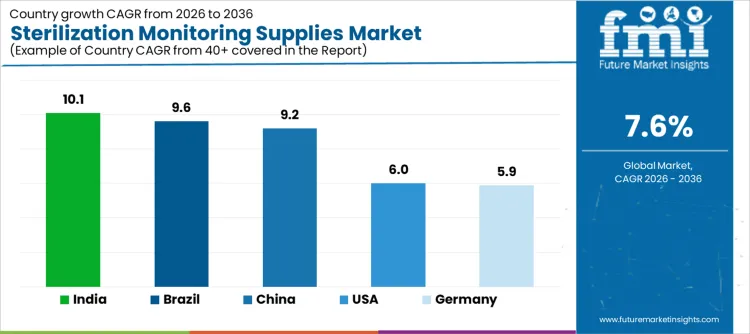
| Country | CAGR (%) |
|---|---|
| India | 10.1% |
| Brazil | 9.6% |
| China | 9.2% |
| USA | 6.0% |
| Germany | 5.9% |
What Is Driving Expansion of Sterilization Monitoring Supply Demand in India?
Demand for sterilization monitoring supplies in India is growing at a CAGR of 10.1%, supported by expansion of hospital infrastructure and surgical throughput. Central sterile services departments increase routine use of indicators to meet accreditation standards. Growth of private hospital chains standardizes sterilization documentation practices. Medical device manufacturing facilities adopt monitoring supplies to satisfy export compliance audits. Training initiatives improve adherence to monitoring protocols. Expansion reflects institutionalization of quality assurance and infection control rather than replacement of sterilization technologies.
- Expansion of centralized sterile services departments
- Accreditation-driven monitoring protocol adoption
- Growth of export-oriented device manufacturing
- Increased staff training on sterilization validation
Why Is Brazil Experiencing Strong Uptake of Sterilization Monitoring Supplies?
Sterilization monitoring supply demand in Brazil is expanding at a CAGR of 9.6%, driven by healthcare system modernization and infection prevention focus. Public hospitals increase indicator usage to support audit readiness. Surgical centers standardize cycle verification to reduce postoperative infection risk. Regulatory oversight encourages consistent biological and chemical monitoring. Regional distributors improve supply availability across states. Growth reflects system-wide quality reinforcement and compliance alignment rather than expansion of sterilization capacity alone.
- Public hospital audit and compliance requirements
- Standardization of surgical sterilization cycles
- Regulatory oversight of infection prevention
- Improved regional distribution coverage
How Is China Sustaining Growth in Sterilization Monitoring Supply Utilization?
Sterilization monitoring supply demand in China is growing at a CAGR of 9.2%, aligned with scale of hospital networks and regulatory enforcement. High patient volumes increase sterilization cycle frequency, elevating indicator consumption. Central hospitals implement standardized monitoring protocols across departments. Medical device manufacturers apply monitoring supplies for process validation. Domestic suppliers provide cost-effective products at scale. Growth reflects systematized compliance and volume-driven utilization rather than discretionary monitoring adoption.
- High sterilization cycle volumes in large hospitals
- Standardized monitoring protocols across departments
- Process validation in device manufacturing
- Scaled domestic supply availability
What Factors Are Supporting Sterilization Monitoring Supply Demand in United States?
Sterilization monitoring supply demand in the United States is expanding at a CAGR of 6.0%, supported by mature infection control frameworks and accreditation requirements. Hospitals maintain routine biological and chemical monitoring across sterilization modalities. Ambulatory surgical centers increase cycle verification volumes. Strong regulatory scrutiny sustains consistent consumption patterns. Replacement and compliance-driven demand dominates purchasing behavior. Growth reflects adherence to established standards and steady procedural volumes rather than expansion of healthcare facilities.
- Accreditation-mandated routine monitoring practices
- Growth of ambulatory surgical center volumes
- Strong regulatory and audit scrutiny
- Stable replacement-driven supply demand
Why Is Germany Showing Measured Growth in Sterilization Monitoring Supplies?
Sterilization monitoring supply demand in Germany is growing at a CAGR of 5.9%, shaped by disciplined healthcare governance and quality assurance standards. Hospitals apply validated monitoring protocols within certified sterile processing units. Documentation accuracy and traceability guide supply selection. Statutory oversight ensures compliance without excessive frequency escalation. Procurement frameworks emphasize reliability and standard conformity. Growth remains measured, reflecting structured utilization and compliance stability rather than rapid procedural expansion.
- Certified sterile processing unit standards
- Emphasis on documentation and traceability
- Statutory oversight of monitoring practices
- Disciplined procurement and utilization controls
What is the competitive landscape for the sterilization monitoring supplies market globally?
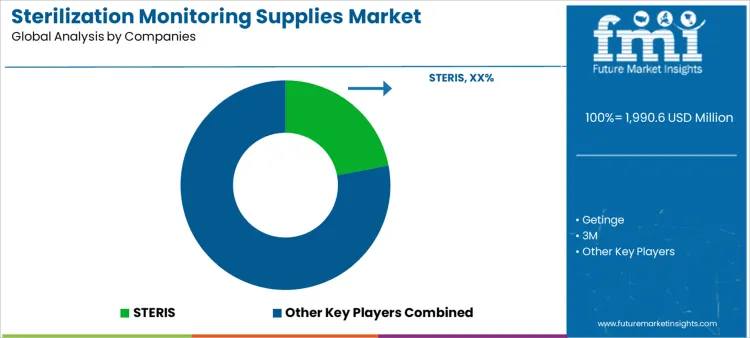
Demand for sterilization monitoring supplies is shaped by infection control standards, regulatory compliance requirements, and rising procedure volumes. Sterile processing departments evaluate indicator accuracy, cycle-specific performance, readability, shelf stability, and compatibility with steam, low-temperature, and gas sterilization methods. Buyer assessment includes documentation traceability, ease of interpretation, training burden, and alignment with audit protocols. Procurement behavior reflects recurring consumable purchasing, validation policy updates, and preference for standardized indicators across facilities. Trend in the sterilization monitoring supplies market shows increased reliance on multi-parameter indicators and digital traceability supporting quality assurance programs.
STERIS leads competitive positioning through comprehensive monitoring portfolios embedded within hospital sterilization workflows and compliance frameworks. Getinge competes by supplying indicators aligned with its sterilization equipment and service ecosystem. 3M supports demand with chemical and biological indicators recognized for consistency and broad modality coverage. Mesa Labs maintains relevance through biological indicators and calibration solutions supporting high-assurance monitoring needs. Fortive participates via specialized monitoring and compliance tools serving regulated healthcare environments. Competitive differentiation centers on indicator reliability, documentation support, modality coverage breadth, and supplier capability to support recurring compliance audits.
Key Players in the Sterilization Monitoring Supplies Market
- STERIS
- Getinge
- 3M
- Mesa Labs
- Fortive
- Others
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Products | Chemical indicators; Biological indicators; Bowie-Dick test packs; Load monitoring accessories; Digital monitoring systems |
| Applications | Steam sterilization; Low-temperature sterilization; Plasma & vapor systems; Ethylene oxide sterilization; Radiation sterilization QA |
| End Users | Hospitals & CSSDs; Medical device manufacturers; Pharma & biotech facilities; Contract sterilization providers; Research laboratories |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, Brazil, China, USA, Germany, and 40+ countries |
| Key Companies Profiled | STERIS; Getinge; 3M; Mesa Labs; Fortive; Others |
| Additional Attributes | Dollar sales by product type, application, and end user; performance reliability of chemical and biological indicators across steam, low-temperature, and alternative sterilization modalities; compliance with international sterilization standards and audit requirements; role of digital monitoring systems in traceability and documentation; adoption patterns across hospital CSSDs, device manufacturing, and contract sterilization facilities; procurement dynamics influenced by regulatory scrutiny, infection control protocols, and recurring consumables demand. |
Sterilization Monitoring Supplies Market by Segment
Products:
- Chemical indicators
- Biological indicators
- Bowie-Dick test packs
- Load monitoring accessories
- Digital monitoring systems
Applications:
- Steam sterilization
- Low-temperature sterilization
- Plasma & vapor systems
- Ethylene oxide sterilization
- Radiation sterilization QA
End Users:
- Hospitals & CSSDs
- Medical device manufacturers
- Pharma & biotech facilities
- Contract sterilization providers
- Research laboratories
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- International Organization for Standardization. (2024). ISO 17665-1: Sterilization of health care products – Moist heat – Requirements for the development, validation, and routine control of a sterilization process. ISO.
- Association for the Advancement of Medical Instrumentation. (2024). AAMI ST79: Comprehensive guide to steam sterilization and sterility assurance in health care facilities. AAMI.
- Centers for Disease Control and Prevention. (2023). Guideline for disinfection and sterilization in healthcare facilities. U.S. Department of Health and Human Services.
- United States Food and Drug Administration. (2023). Reprocessing medical devices in health care settings: Validation methods and labeling. U.S. Department of Health and Human Services.
- European Commission. (2023). Regulation (EU) 2017/745 on medical devices: Annex I and Annex II requirements related to sterilization and process monitoring. Official Journal of the European Union.
- World Health Organization. (2023). Decontamination and reprocessing of medical devices for health-care facilities. World Health Organization.
Frequently Asked Questions
How big is the sterilization monitoring supplies market in 2026?
The global sterilization monitoring supplies market is estimated to be valued at USD 1,990.6 million in 2026.
What will be the size of sterilization monitoring supplies market in 2036?
The market size for the sterilization monitoring supplies market is projected to reach USD 4,141.0 million by 2036.
How much will be the sterilization monitoring supplies market growth between 2026 and 2036?
The sterilization monitoring supplies market is expected to grow at a 7.6% CAGR between 2026 and 2036.
What are the key product types in the sterilization monitoring supplies market?
The key product types in sterilization monitoring supplies market are chemical indicators, biological indicators, bowie-dick test packs, load monitoring accessories and digital monitoring systems.
Which applications segment to contribute significant share in the sterilization monitoring supplies market in 2026?
In terms of applications, steam sterilization segment to command 34.0% share in the sterilization monitoring supplies market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Products
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Products , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Products , 2026 to 2036
- Chemical indicators
- Biological indicators
- Bowie-Dick test packs
- Load monitoring accessories
- Digital monitoring systems
- Chemical indicators
- Y to o to Y Growth Trend Analysis By Products , 2021 to 2025
- Absolute $ Opportunity Analysis By Products , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Applications
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Applications, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Applications, 2026 to 2036
- Steam sterilization
- Low-temperature sterilization
- Plasma & vapor systems
- Ethylene oxide sterilization
- Radiation sterilization QA
- Steam sterilization
- Y to o to Y Growth Trend Analysis By Applications, 2021 to 2025
- Absolute $ Opportunity Analysis By Applications, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Users
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Users, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Users, 2026 to 2036
- Hospitals & CSSDs
- Medical device manufacturers
- Pharma & biotech facilities
- Contract sterilization providers
- Research laboratories
- Hospitals & CSSDs
- Y to o to Y Growth Trend Analysis By End Users, 2021 to 2025
- Absolute $ Opportunity Analysis By End Users, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Products
- By Applications
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End Users
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Products
- By Applications
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End Users
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Products
- By Applications
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End Users
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Products
- By Applications
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End Users
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Products
- By Applications
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End Users
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Products
- By Applications
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End Users
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Products
- By Applications
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End Users
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End Users
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End Users
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End Users
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End Users
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End Users
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End Users
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End Users
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End Users
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End Users
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End Users
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End Users
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End Users
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End Users
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End Users
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End Users
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End Users
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End Users
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End Users
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End Users
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End Users
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End Users
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End Users
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Products
- By Applications
- By End Users
- Competition Analysis
- Competition Deep Dive
- STERIS
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Getinge
- 3M
- Mesa Labs
- Fortive
- Others
- STERIS
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End Users, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Products
- Figure 6: Global Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Applications
- Figure 9: Global Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End Users, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End Users
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Products
- Figure 26: North America Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Applications
- Figure 29: North America Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End Users, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by End Users
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Products
- Figure 36: Latin America Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Applications
- Figure 39: Latin America Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End Users, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by End Users
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Products
- Figure 46: Western Europe Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Applications
- Figure 49: Western Europe Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End Users, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by End Users
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Products
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Applications
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End Users, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End Users
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Products
- Figure 66: East Asia Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Applications
- Figure 69: East Asia Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End Users, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by End Users
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Products
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Applications
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End Users, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End Users
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Products
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Applications
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End Users, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End Users
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

