The sterilization in place bioreactors market is projected to reach USD 2,339.2 million in 2026 and expand to USD 5,437.0 million by 2036, at a CAGR of 8.8%. Value behavior reflects structural dependence on automated sterilization for aseptic biomanufacturing, where contamination events carry disproportionate financial and regulatory consequences. Spending concentrates on stainless steel and hybrid bioreactor systems that support repeated sterilization cycles without disassembly, enabling high asset utilization across multiproduct facilities. Capital allocation is driven by long operating lifecycles, validation durability, and compatibility with evolving biologics pipelines rather than short-term capacity additions.
Adoption patterns are governed by regulatory scrutiny, production continuity requirements, and integration with automated cleaning and control architectures. Utilization intensity increases where monoclonal antibody, vaccine, and microbial fermentation operations require frequent turnaround with documented sterility assurance. High capital cost, utility infrastructure demands, and validation workload moderate uptake among smaller producers, reinforcing concentration within established manufacturers and CDMOs. Market structure remains shaped by compliance discipline, engineering complexity, and lifecycle service dependence, sustaining predictable growth linked to biomanufacturing scale-up and process standardization rather than episodic facility expansion.

| Metric | Value |
|---|---|
| Market Value (2026) | USD 2,339.2 million |
| Market Forecast Value (2036) | USD 5,437.0 million |
| Forecast CAGR 2026 to 2036 | 8.8% |
Demand for sterilization in place (SIP) bioreactors is rising as biopharmaceutical manufacturers and contract development and manufacturing organizations (CDMOs) require robust systems that support high product integrity while minimizing contamination risk. SIP bioreactors incorporate automated steam generation and circulation protocols that sterilize internal surfaces, valves, and connections without disassembly, which reduces manual handling and exposure to environmental contaminants. Process engineers specify SIP capability because it supports reproducible sterility assurance across successive batches of vaccines, monoclonal antibodies, and cell and gene therapies where even trace bioburden can compromise yield or safety. Procurement teams evaluate vessel design, validation documentation, control system integration, and compliance with current good manufacturing practice expectations to ensure that selected units align with production scale and quality benchmarks. These criteria are especially relevant where regulatory oversight demands documented sterility control and rapid turnover between campaigns.
Growth in complex biologic pipelines and intensified manufacturing schedules reinforces adoption of SIP bioreactors that improve facility throughput and lower risk of cross contamination between product lines. Facilities with flexible manufacturing strategies benefit from automated SIP cycles that reduce downtime and labor costs associated with manual sterilization and cleaning. Integration of SIP with automated cleaning in place (CIP) sequences enhances overall process efficiency and supports data capture for electronic batch records. Engineering teams also consider thermal distribution, cycle reproducibility, and energy usage when comparing SIP platforms because predictable performance under varied operating conditions reduces cycle variability. These application-specific operational and quality drivers are contributing to sustained demand growth in the sterilization in place bioreactors market.
Demand for sterilization in place bioreactors is driven by contamination control requirements, scale-up reliability, and regulatory expectations for aseptic processing. Bioprocessing operations prioritize validated steam sterilization, repeatable cycle performance, and integration with upstream automation. Adoption aligns with biologics pipeline expansion, flexible manufacturing strategies, and lifecycle cost control. Purchasing decisions emphasize material compatibility, documentation support, and upgrade pathways. Segmentation clarifies how system components, production use cases, and organizational maturity influence deployment across biopharmaceutical manufacturing environments.

Stainless steel SIP bioreactor systems account for 30.0%, reflecting their role as the core asset enabling robust, repeatable sterilization cycles. These systems support high-pressure steam exposure, long operational lifetimes, and regulatory familiarity. SIP and CIP skids with steam systems at 18.0% enable centralized sterilization utilities supporting multiple vessels. Control systems and automation software at 16.0% coordinate cycle execution and data capture. Sensors and PAT instrumentation at 14.0% ensure real-time verification. Installation and validation services at 12.0%, alongside retrofit kits at 10.0%, support compliance and lifecycle extension.
Key Points:

Monoclonal antibody manufacturing represents 26.0%, driven by large-volume upstream processing requiring strict sterility assurance. Vaccine production at 20.0% relies on SIP systems to manage batch-to-batch contamination risk. Microbial fermentation at 18.0% applies SIP for high-cell-density processes with frequent turnaround. Biosimilars manufacturing at 16.0% emphasizes process comparability and documentation rigor. Cell and gene therapy upstream at 12.0% uses SIP to protect sensitive cultures within smaller batches. Process development and pilot scale at 8.0% focuses on flexibility and method optimization. Application segmentation reflects alignment between contamination risk and sterilization intensity.
Key Points:
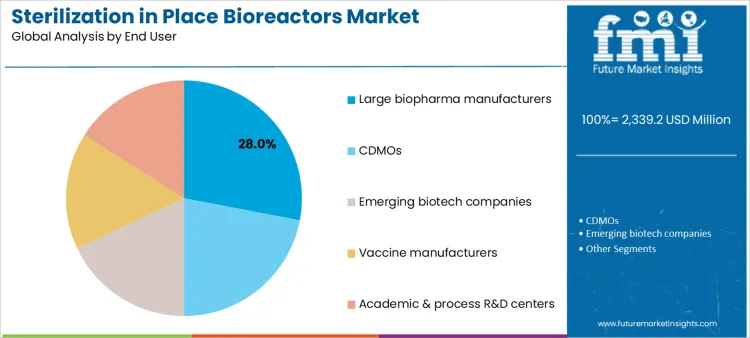
Large biopharma manufacturers account for 28.0%, reflecting established facilities with integrated sterilization infrastructure. CDMOs at 22.0% deploy SIP bioreactors to serve multiple client programs under strict quality frameworks. Emerging biotech companies at 18.0% adopt modular systems supporting phased capacity expansion. Vaccine manufacturers at 16.0% integrate SIP to maintain biosafety and throughput reliability. Academic and process R&D centers at 16.0% use SIP platforms for translational studies and scale-up validation. End-user segmentation highlights concentration where operational scale, regulatory accountability, and portfolio diversity shape equipment adoption.
Key Points:
Demand for sterilization in place bioreactors reflects bioprocessing requirements to maintain aseptic conditions without disassembly across repeated production cycles. Adoption concentrates in biopharmaceutical manufacturing, vaccine production, cell culture operations, and microbial fermentation facilities. Global scope aligns with regulatory expectations for contamination control in biologics manufacturing. Usage centers on stainless steel and advanced single-use hybrid bioreactors integrating automated SIP cycles to sterilize internal surfaces, piping, and valves.
Biologic manufacturing processes remain highly sensitive to microbial contamination that can invalidate entire batches. Demand increases where frequent cleaning and sterilization are required between runs without compromising uptime. SIP bioreactors enable validated sterilization of contact surfaces using controlled steam or chemical cycles while remaining fully assembled. Automated SIP sequences reduce operator intervention and variability, supporting reproducible aseptic conditions. Facilities producing monoclonal antibodies, vaccines, and enzymes rely on SIP to meet sterility assurance levels mandated by regulators. Integration with clean in place systems streamlines turnaround between batches. Adoption reflects operational priority to protect high-value products while maintaining consistent production schedules.
SIP bioreactors require robust pressure-rated vessels, steam infrastructure, and precise control systems, increasing initial capital investment. Demand sensitivity rises where smaller manufacturers face cost barriers relative to disposable alternatives. System qualification and repeated validation add engineering and documentation workload. Energy consumption during steam cycles affects operating cost. Retrofitting existing facilities presents space and utility constraints. Maintenance of valves, seals, and instrumentation impacts long-term reliability. Regulatory audits increase scrutiny on SIP performance data. Scalability remains constrained by high upfront cost, technical complexity, and extensive validation requirements across diverse biomanufacturing environments.
Demand for sterilization in place bioreactors is expanding globally as biopharmaceutical manufacturers prioritize contamination control, batch integrity, and operational efficiency. Single-use and stainless-steel bioprocessing facilities integrate SIP systems to ensure validated sterilization without dismantling equipment. Growth in biologics, vaccines, and cell-based therapies increases reliance on reproducible sterilization cycles. Regulatory expectations for aseptic assurance reinforce adoption across development and commercial scales. Growth rates in India at 11.3%, Brazil at 10.8%, China at 10.3%, the USA at 7.3%, and Germany at 7.2% indicate sustained expansion driven by biomanufacturing capacity build-out, compliance requirements, and process standardization rather than basic laboratory expansion.
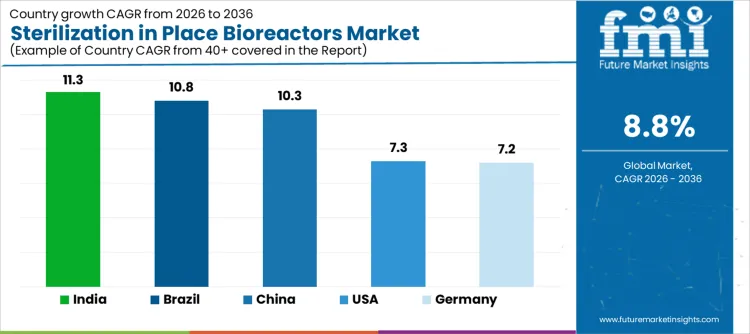
| Country | CAGR (%) |
|---|---|
| India | 11.3% |
| Brazil | 10.8% |
| China | 10.3% |
| USA | 7.3% |
| Germany | 7.2% |
Demand for sterilization in place bioreactors in India is growing at a CAGR of 11.3%, supported by rapid expansion of biologics and vaccine manufacturing capacity. Contract development and manufacturing organizations scale multiproduct facilities requiring validated SIP protocols. Increased focus on export-oriented biologics strengthens adherence to global regulatory standards. Automation investments improve cycle control and documentation. Local engineering firms supply customized bioreactor systems for diverse process scales. Growth reflects biopharmaceutical capacity expansion, regulatory alignment, and multiproduct manufacturing needs rather than early-stage research activity.
Sterilization in place bioreactor demand in Brazil is expanding at a CAGR of 10.8%, driven by public sector biologics production and biosimilar development. State-supported manufacturing facilities upgrade sterilization systems to ensure aseptic reliability. Increasing domestic vaccine and therapeutic protein production elevates need for integrated SIP solutions. Technology transfer partnerships introduce standardized bioprocessing platforms. Growth reflects institutional investment, domestic capacity strengthening, and quality assurance priorities rather than rapid diversification into novel modalities.
Sterilization in place bioreactor demand in China is growing at a CAGR of 10.3%, aligned with large-scale biologics manufacturing and process industrialization. High-volume production facilities require reliable, repeatable sterilization cycles. Regulatory oversight strengthens validation and documentation requirements. Domestic equipment manufacturers supply integrated SIP-capable bioreactors at scale. Continuous process optimization sustains replacement and upgrade demand. Growth reflects manufacturing scale, regulatory enforcement, and system standardization rather than pilot-stage experimentation.
Sterilization in place bioreactor demand in the United States is expanding at a CAGR of 7.3%, supported by mature biopharmaceutical manufacturing and regulatory rigor. Commercial biologics facilities rely on SIP for contamination control and batch consistency. Process intensification and closed-system designs increase sterilization complexity. Equipment upgrades focus on automation, monitoring, and data integrity. Replacement demand arises from lifecycle management of installed assets. Growth reflects process refinement and compliance adherence rather than expansion of manufacturing footprints.
Sterilization in place bioreactor demand in Germany is growing at a CAGR of 7.2%, shaped by precision biomanufacturing and strict regulatory frameworks. Biopharmaceutical producers emphasize validated sterilization performance and documentation accuracy. Engineering-led system design ensures integration with cleanroom and utility infrastructure. Procurement decisions prioritize reliability and compliance over rapid scale-up. Incremental upgrades support long-term operational stability. Growth remains steady, reflecting structured investment, quality governance, and process discipline rather than aggressive capacity expansion.
Demand for sterilization in place bioreactors is driven by biologics manufacturing scale-up, contamination risk control, and regulatory expectations for aseptic processing. Biopharmaceutical manufacturers evaluate sterilization reliability, cycle repeatability, cleanability, material compatibility, and automation integration. Buyer assessment includes validation documentation, temperature uniformity, downtime minimization, and alignment with GMP requirements. Procurement behavior reflects long capital planning cycles, preference for platform standardization, and reliance on suppliers offering strong commissioning and lifecycle support. Trend in the sterilization in place bioreactors market shows increasing adoption in monoclonal antibody and cell culture facilities emphasizing operational efficiency and compliance consistency.
Sartorius leads competitive positioning through integrated bioreactor platforms with proven sterilization performance and strong adoption in large-scale biologics production. Thermo Fisher Scientific competes by offering flexible bioreactor systems aligned with modular facility designs and rapid deployment needs. Cytiva supports demand with bioprocessing solutions emphasizing scalability, automation, and standardized sterilization workflows. Merck KGaA maintains relevance through bioreactors integrated within comprehensive upstream processing portfolios and strong regulatory support. GEA Group participates by supplying engineered bioreactor systems focused on thermal control, cleanability, and long-term operational robustness. Competitive differentiation centers on sterilization reliability, validation support depth, automation capability, and supplier experience in regulated biomanufacturing environments.
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Products | Stainless steel SIP bioreactor systems; SIP/CIP skids & steam systems; Control systems & automation software; Sensors & PAT instrumentation; Installation & validation services; Retrofit & upgrade kits |
| Applications | Monoclonal antibody manufacturing; Vaccine production; Microbial fermentation; Biosimilars manufacturing; Cell & gene therapy upstream; Process development & pilot scale |
| End User | Large biopharma manufacturers; CDMOs; Emerging biotech companies; Vaccine manufacturers; Academic & process R&D centers |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, Brazil, China, USA, Germany, and 40+ countries |
| Key Companies Profiled | Sartorius; Thermo Fisher Scientific; Cytiva; Merck KGaA; GEA Group; Others |
| Additional Attributes | Dollar sales by product category, application, and end user; sterilization reliability and contamination control performance in stainless steel SIP bioreactor operations; integration of CIP, steam, automation, and PAT systems for compliant bioprocessing; scalability across pilot, clinical, and commercial manufacturing; validation and qualification requirements aligned with GMP regulations; procurement dynamics influenced by biopharma capacity expansions, CDMO investments, and long-term service and upgrade contracts. |
How big is the sterilization in place bioreactors market in 2026?
The global sterilization in place bioreactors market is estimated to be valued at USD 2,339.2 million in 2026.
What will be the size of sterilization in place bioreactors market in 2036?
The market size for the sterilization in place bioreactors market is projected to reach USD 5,437.0 million by 2036.
How much will be the sterilization in place bioreactors market growth between 2026 and 2036?
The sterilization in place bioreactors market is expected to grow at a 8.8% CAGR between 2026 and 2036.
What are the key product types in the sterilization in place bioreactors market?
The key product types in sterilization in place bioreactors market are stainless steel sip bioreactor systems, sip/cip skids & steam systems, control systems & automation software, sensors & pat instrumentation, installation & validation services and retrofit & upgrade kits.
Which applications segment to contribute significant share in the sterilization in place bioreactors market in 2026?
In terms of applications, monoclonal antibody manufacturing segment to command 26.0% share in the sterilization in place bioreactors market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.