Touch Activated Phlebotomy Devices Market
Touch Activated Phlebotomy Devices Market Size and Share Forecast Outlook 2026 to 2036
Touch Activated Phlebotomy Devices Market Forecast and Outlook 2026 to 2036
The touch activated phlebotomy devices market is expected to grow from USD 288.7 million in 2026 to USD 819.7 million by 2036, reflecting a compound annual growth rate (CAGR) of 11%. These devices are designed to simplify the process of blood collection by automating the venipuncture process, reducing the risk of needlestick injuries, and improving the efficiency and safety of phlebotomy procedures. The market’s growth is driven by the increasing adoption of automated and safer blood collection technologies, as well as the rising awareness of healthcare worker safety and patient comfort during procedures.
The demand for touch-activated phlebotomy devices is also fueled by the ongoing focus on improving healthcare practices and reducing complications associated with manual blood collection. The growing prevalence of chronic diseases, which require regular blood tests, and the rising number of outpatient procedures are expected to further support market expansion. Technological advancements that improve the precision and ease of blood collection procedures are also contributing to the market's growth.
Quick Stats of the Touch Activated Phlebotomy Devices Market
- Touch Activated Phlebotomy Devices Market Value (2026): USD 288.7 million
- Touch Activated Phlebotomy Devices Market Forecast Value (2036): USD 819.7 million
- Touch Activated Phlebotomy Devices Market Global Forecast CAGR (2026-2036): 11%
- Touch Activated Phlebotomy Devices Market Leading Segment: Touch-Activated Safety Needles 46%
- Key Growth Regions in Touch Activated Phlebotomy Devices Market: North America, Asia Pacific, and Europe
- Top Players in Touch Activated Phlebotomy Devices Market: BD, Greiner Bio-One, Terumo, Nipro, B. Braun, Sarstedt, Cardinal Health, Medline, Smiths Medical, Vacuette (Greiner)
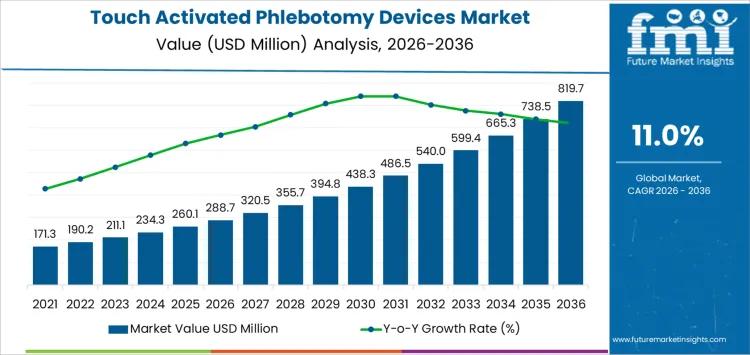
What is the Growth Forecast for the Touch Activated Phlebotomy Devices Market through 2036?
The 10-year growth comparison for the touch-activated phlebotomy devices market shows a consistent and steady increase over the forecast period. Starting at USD 288.7 million in 2026, the market grows to USD 320.9 million in 2027 and USD 356.3 million in 2028, reflecting gradual adoption and increasing recognition of the benefits of touch-activated phlebotomy devices in reducing manual errors and improving safety.
From 2028 to 2030, the market continues to expand steadily, reaching USD 395.8 million in 2029 and USD 439.6 million in 2030. As the technology becomes more widely used in clinical and outpatient settings, the adoption accelerates. From 2030 to 2035, the market sees faster growth, reaching USD 488.1 million in 2031, USD 541.5 million in 2032, and USD 600.5 million in 2033, driven by the rising demand for safer, more efficient blood collection solutions.
By 2035, the market is projected to reach USD 738 million, with the final market value of USD 819.7 million expected by 2036. The comparison highlights steady growth, with a notable acceleration towards the latter part of the forecast period as healthcare systems increasingly adopt these devices for blood collection, improving both the safety and efficiency of procedures across hospitals and clinics globally.
Touch Activated Phlebotomy Devices Market Key Takeaways
| Metric | Value |
|---|---|
| Industry Sales Value (2026) | USD 288.7 million |
| Industry Forecast Value (2036) | USD 819.7 million |
| Industry Forecast CAGR (2026-2036) | 11% |
What Is Driving the Demand for the Touch Activated Phlebotomy Devices Market Globally?
The global demand for touch activated phlebotomy devices is being driven by the need to improve safety, efficiency, and patient experience in blood collection procedures. Traditional venipuncture carries risks such as needlestick injuries for healthcare workers and discomfort for patients, especially those with difficult veins or in paediatric and geriatric populations. Touch activated phlebotomy devices incorporate mechanisms that stabilise the needle and collection assembly when contact with the skin is made, which enhances control during insertion and reduces unintended needle movement. This can lower the risk of needlestick injury, reduce sample haemolysis, and improve first-stick success rates, making these devices attractive to hospitals, diagnostic centres, and outpatient laboratories. Growing emphasis on healthcare worker safety, reinforced by occupational safety guidelines and campaigns, has increased adoption of engineered safety devices. In addition, expansion of routine diagnostic testing due to chronic disease monitoring, preventive health screening, and broader access to laboratory services supports increased utilization of advanced blood collection tools.
Future demand for touch activated phlebotomy devices is expected to grow as healthcare systems continue prioritising safety, quality, and operational efficiency. Advances in device ergonomics, user feedback features, and compatibility with existing blood collection systems will make these solutions more appealing to clinical teams. Integration with training programmes for nurses and phlebotomists can further improve adoption by reducing procedural variability and enhancing confidence in blood draws. As point-of-care testing and decentralized healthcare services expand, the ability to perform reliable blood draws outside the traditional lab setting will create additional demand for user-friendly, safety-enhancing devices. Regulatory environments that incentivise adoption of safer medical devices and reimbursement models that recognise the value of reducing workplace injuries and repeat procedures will also influence how widely these devices are deployed. Consumer expectations for less painful and safer blood collection, particularly in preventive health and chronic disease contexts, may further accelerate market growth. The emphasis on reducing occupational hazards, improving patient comfort, and streamlining clinical workflows is expected to sustain steady global expansion of the touch activated phlebotomy devices market.
What is the Segment Analysis for the Touch Activated Phlebotomy Devices Market?
The touch activated phlebotomy devices market is influenced by two primary segments: device types and use settings. Among the device types, touch-activated safety needles lead the market, accounting for 46% of the share, followed by touch-activated blood collection sets and touch-activated lancets. These devices are designed to improve the safety and efficiency of blood collection procedures. The market is also shaped by use settings such as hospitals, diagnostics/reference labs, clinics, and physician offices, with hospitals representing the largest share at 44%. The demand for touch activated phlebotomy devices is driven by the growing emphasis on patient and healthcare worker safety, as well as the need for more efficient, minimally invasive procedures. Regional factors, particularly in North America and Europe, play a significant role in the adoption of these devices. As healthcare facilities seek safer alternatives to traditional blood collection methods, the market for touch activated devices is expected to expand further.
What is Driving Growth in the Touch-Activated Safety Needles Segment of the Phlebotomy Devices Market?
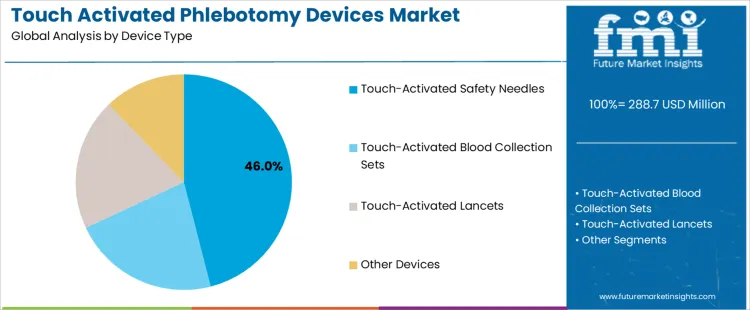
Touch-activated safety needles represent the leading device type in the touch activated phlebotomy devices market, comprising 46% of the market share. The growing demand for these needles is largely driven by their ability to reduce the risk of needlestick injuries, which are a significant concern for healthcare workers. Touch-activated safety needles feature a mechanism that automatically shields the needle after use, minimizing the risk of accidental pricks. As hospitals and clinics continue to prioritize healthcare worker safety, the adoption of safety needles is rising.
In addition, touch-activated safety needles are designed to be user-friendly, enabling faster and more efficient blood collection, which is crucial in high-volume healthcare settings. The shift toward minimizing occupational health risks and improving the safety of phlebotomy procedures is a key driver behind the growth of this segment. Furthermore, regulatory standards in many regions, such as OSHA in the United States, mandate the use of safety-engineered devices, further boosting the demand for touch-activated safety needles. With their ability to enhance both safety and efficiency, touch-activated safety needles are expected to maintain their dominant position in the market, particularly in hospital settings.
What is Contributing to the Growth of the Hospital Use Setting in the Phlebotomy Devices Market?
The hospital use setting holds the largest share in the touch activated phlebotomy devices market, accounting for 44% of the total market. This growth is driven by the high volume of blood collection procedures performed in hospitals, where the need for efficient, safe, and rapid phlebotomy tools is critical. Hospitals require devices that can handle a wide range of patient demographics, including those in emergency care, critical care, and general medical procedures.
Touch-activated devices, particularly safety needles, are becoming the preferred choice due to their ability to minimize risk and improve the speed of procedures, which is crucial in high-pressure hospital environments. The adoption of touch-activated phlebotomy devices in hospitals is also supported by stringent health and safety regulations aimed at reducing healthcare-associated infections and injuries. Furthermore, as hospitals seek to improve operational efficiency and patient safety, the demand for advanced, easy-to-use blood collection tools is expected to rise. With healthcare facilities prioritizing both safety and efficiency, the hospital use setting is poised to remain the dominant sector in the touch activated phlebotomy devices market, driving its continued growth.
What are the Key Dynamics of the Global Touch Activated Phlebotomy Devices Market?
The global touch activated phlebotomy devices market is expanding as healthcare providers adopt safer and more efficient blood collection tools. These devices reduce needlestick risk and improve consistency in venous access, supporting quality and safety goals in clinical settings. Demand grows with rising diagnostic testing, outpatient care volume and focus on infection prevention. Use of touch activated technology enhances clinician control during draws, lowering complications and improving patient comfort. Growth spans hospitals, blood collection centres, labs and outpatient clinics where safer phlebotomy practice is increasingly prioritised.
What are the Key Drivers for the Global Touch Activated Phlebotomy Devices Market?
Patient and clinician safety is a primary driver that encourages adoption of touch activated phlebotomy devices. Hospitals and clinics focus on lowering needlestick injuries and reducing exposure to bloodborne pathogens, which supports investment in engineered safety solutions. Rising volume of diagnostic tests and preventive screenings increases frequency of blood draws, prompting facilities to adopt tools that improve procedure reliability. Regulatory standards and workplace safety protocols in many regions reinforce use of devices with integrated safety mechanisms. Advances in ergonomic design and user control enhance appeal among phlebotomists and nursing staff seeking consistent performance.
What are the Restraints for the Global Touch Activated Phlebotomy Devices Market?
One restraint is the higher cost of touch activated devices compared with traditional phlebotomy needles and systems. Budget constraints in some healthcare facilities, particularly smaller clinics or rural providers, may slow adoption. Some purchasers remain reluctant to switch from familiar tools unless clear cost benefit in safety or efficiency is demonstrated. Variation in reimbursement policies and budget cycles across regions influences purchase timing and scale. Training requirements for clinicians to adopt new devices and integrate them into workflow add time and resource needs. Supply chain challenges for safety devices in certain markets can also affect consistent availability.
What is the Key Trends in the Global Touch Activated Phlebotomy Devices Market?
A key trend is development of devices with improved tactile feedback and ease of use that support consistent vein access across diverse patient populations. Manufacturers are focusing on innovations that combine safety features with intuitive design to reduce learning curves for clinical staff. Integration with safety protocols and clinician training programs is becoming more common to ensure optimal use and reduce complications. Growing interest in sustainable device options and recyclable components influences product development. Data from real world use and safety reporting is being used to refine design and support purchasing decisions by health systems aiming to improve outcomes and operational efficiency.
What are the Key Insights from the Touch Activated Phlebotomy Devices Market Analysis?
The touch activated phlebotomy devices market is expected to grow across multiple regions, with varying compound annual growth rates (CAGRs) reflecting the adoption and demand in each country. Increasing healthcare awareness, the need for safer blood collection methods, and technological advancements in medical devices are driving the growth of this market. The devices offer improved safety features, ease of use, and efficiency, making them increasingly popular in healthcare settings. With rising demand for minimally invasive procedures and safer blood draw options, touch activated phlebotomy devices are gaining traction globally. By understanding country-specific growth trends, businesses can optimize their strategies to cater to the growing need for these advanced phlebotomy solutions.
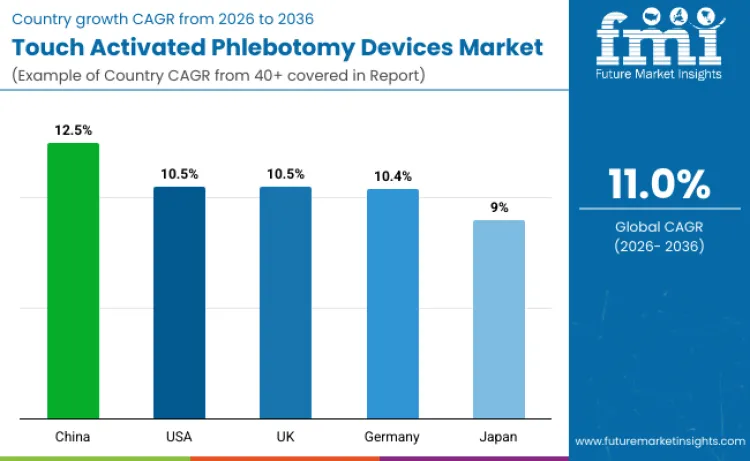
| Country | CAGR (2026-2036) |
|---|---|
| China | 12.5% |
| United States | 10.5% |
| United Kingdom | 10.5% |
| Germany | 10.4% |
| Japan | 9% |
What is the Growth Outlook for the Touch Activated Phlebotomy Devices Market in China?
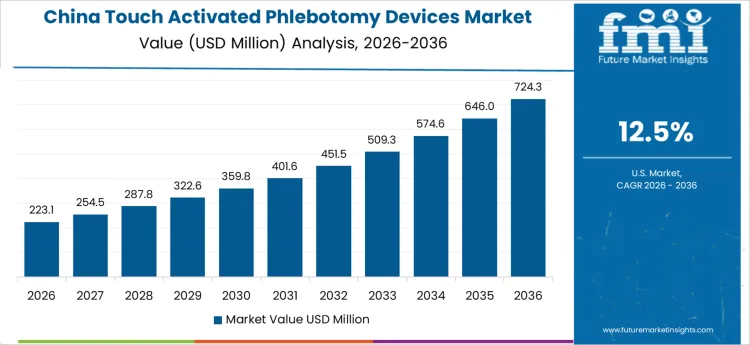
The touch activated phlebotomy devices market in China is projected to grow at the highest rate, with a CAGR of 12.5%. China’s growing healthcare needs, coupled with the increasing awareness of the importance of safe blood collection methods, are major drivers for this growth. The country’s expanding healthcare infrastructure and the rising demand for minimally invasive medical solutions contribute to the growing adoption of advanced phlebotomy devices. The Chinese government’s focus on improving healthcare services and technological advancements in medical devices further support the widespread adoption of touch activated phlebotomy devices. Additionally, China’s large population and the increasing incidence of medical procedures requiring blood collection create a significant market for these devices. As the healthcare system continues to modernize, the market for touch activated phlebotomy devices in China is expected to experience strong and sustained growth.
What is the Market Forecast for Touch Activated Phlebotomy Devices in the United States?
The touch activated phlebotomy devices market in the United States is expected to grow at a CAGR of 10.5%. The U.S. market benefits from a well-established healthcare infrastructure, high adoption of advanced medical technologies, and increasing focus on patient safety. The growing prevalence of chronic diseases, such as diabetes and cardiovascular conditions, which require frequent blood tests, is driving the demand for safer and more efficient blood collection methods. The widespread use of touch activated phlebotomy devices in hospitals, clinics, and laboratories supports the market’s growth. Additionally, the regulatory environment in the U.S. encourages the integration of advanced medical devices to enhance patient care and reduce the risks associated with traditional blood collection methods. As a result, the market for touch activated phlebotomy devices in the United States is expected to grow steadily, driven by these factors.
How is the Touch Activated Phlebotomy Devices Market Performing in the United Kingdom?
The touch activated phlebotomy devices market in the United Kingdom is projected to grow at a CAGR of 10.5%. The UK healthcare system, known for its high standards and focus on patient safety, is contributing to the adoption of advanced medical devices like touch activated phlebotomy solutions. As the need for safer blood collection methods increases, healthcare providers are increasingly adopting these devices to reduce the risk of needle-stick injuries and improve the efficiency of blood draws. The UK’s National Health Service (NHS) is a significant driver of medical device adoption, ensuring widespread access to these devices in public healthcare settings. Moreover, the growing demand for minimally invasive medical procedures supports the market growth. The UK is expected to continue experiencing steady growth in the adoption of touch activated phlebotomy devices in the coming years.
What is the Market Outlook for Touch Activated Phlebotomy Devices in Germany?
The touch activated phlebotomy devices market in Germany is expected to grow at a CAGR of 10.4%. Germany’s advanced healthcare system, along with its emphasis on medical innovation and patient safety, provides a strong foundation for the adoption of these devices. The increasing focus on reducing healthcare-associated risks, such as needle-stick injuries, is driving the demand for safer blood collection alternatives like touch activated phlebotomy devices. Germany’s large healthcare infrastructure, including hospitals and diagnostic centers, supports the widespread implementation of these devices. The regulatory environment in Germany also promotes the adoption of innovative medical technologies, ensuring that touch activated phlebotomy devices can be integrated into clinical practice effectively. As a result, Germany is expected to maintain steady growth in the adoption of touch activated phlebotomy devices.
What are the Growth Prospects for Touch Activated Phlebotomy Devices in Japan?
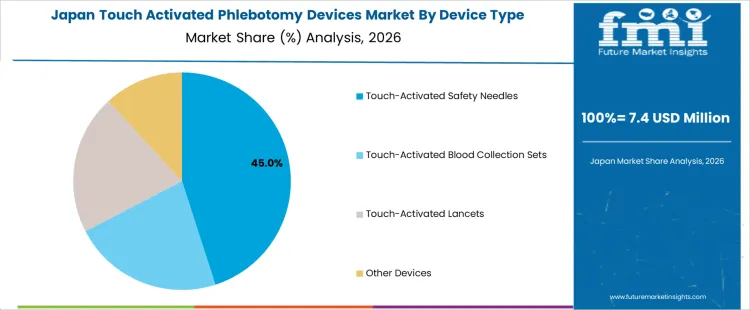
The touch activated phlebotomy devices market in Japan is forecasted to grow at a CAGR of 9%. Japan’s aging population and the increasing demand for safer medical practices are key factors contributing to the growth of the market. The country’s healthcare system is renowned for its high standards, and there is a growing focus on reducing the risks associated with blood collection procedures. The adoption of touch activated phlebotomy devices is expected to increase as healthcare providers seek to enhance safety and minimize the risk of infections. Japan’s strong regulatory environment for medical devices ensures that these devices will continue to be integrated into healthcare practices, providing a steady market for their use. While the growth rate may be slower than in some other countries, Japan remains a key player in the global touch activated phlebotomy devices market due to its technological sophistication and patient care priorities.
What is the Competitive Landscape for the Touch Activated Phlebotomy Devices Market?
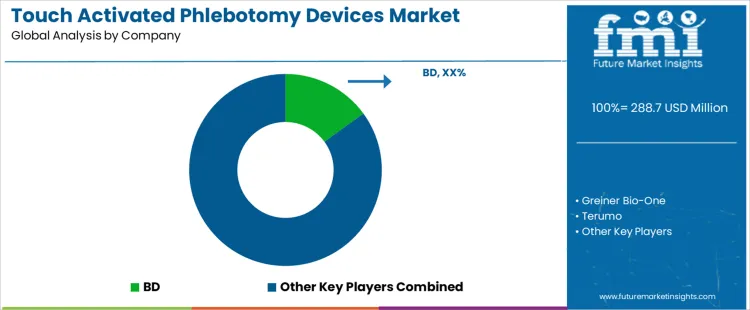
The touch activated phlebotomy devices market is growing as healthcare providers seek safer and more efficient methods for blood collection. BD leads the market with its innovative touch-activated phlebotomy devices, which are designed to improve safety, reduce the risk of needlestick injuries, and enhance the efficiency of blood collection procedures. BD’s advanced technologies have positioned the company at the forefront of this market, offering devices that are widely adopted across healthcare settings. Greiner Bio-One, Terumo, and Nipro are key competitors, each offering a range of touch-activated devices that prioritize ease of use and safety. Greiner Bio-One provides specialized solutions with its Vacuette line, while Terumo and Nipro focus on creating high-quality, cost-effective devices suitable for various clinical applications.
B. Braun, Sarstedt, Cardinal Health, Medline, and Smiths Medical add to the competitive landscape by offering products that enhance the blood collection process. B. Braun and Sarstedt focus on developing reliable, touch-activated phlebotomy devices that cater to both hospital and laboratory needs. Cardinal Health and Medline provide a broad range of medical products, including blood collection devices, while Smiths Medical brings specialized innovations in blood collection technologies. These companies compete by offering advanced features such as improved ergonomics, reduced needlestick risks, and faster collection times, ensuring better outcomes for both healthcare providers and patients.
Key Players of the Touch Activated Phlebotomy Devices Market
- BD
- Greiner Bio-One
- Terumo
- Nipro
- B. Braun
- Sarstedt
- Cardinal Health
- Medline
- Smiths Medical
- Vacuette (Greiner)
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD Million |
| Device Type | Touch-Activated Safety Needles, Touch-Activated Blood Collection Sets, Touch-Activated Lancets, Other Devices |
| Use Setting | Hospitals, Diagnostics/Reference Labs, Clinics & Physician Offices, Other Settings |
| Safety Mechanism | Passive/Auto-Engage, User-Activated (touch) mechanisms, Hybrid/Other |
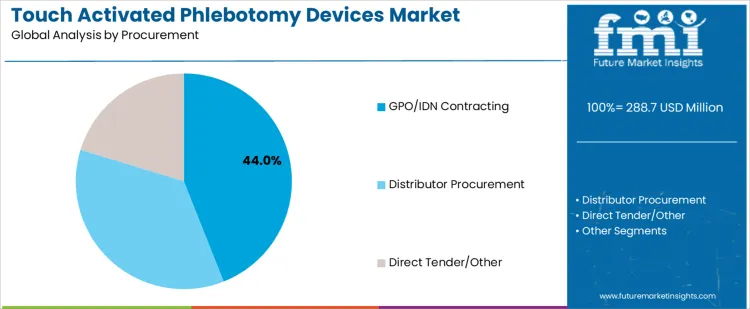
| Procurement | GPO/IDN Contracting, Distributor Procurement, Direct Tender/Other |
| Companies | BD, Greiner Bio-One, Terumo, Nipro, B. Braun, Sarstedt, Cardinal Health, Medline, Smiths Medical, Vacuette (Greiner) |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, South Asia and Pacific, East Asia, Middle East & Africa |
| Countries Covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, Netherlands, China, India, Japan, South Korea, ANZ, GCC Countries, South Africa |
| Additional Attributes | Dollar by sales by device type, use setting, safety mechanism, procurement model, and region. Includes market trends in touch-activated phlebotomy devices, focusing on safety needles, blood collection sets, and lancets. Highlights the role of passive/auto-engage, user-activated mechanisms, and hybrid safety features in improving phlebotomy safety. Focus on procurement models (GPO/IDN contracting, distributor procurement, direct tender) and the impact on market growth. |
Touch Activated Phlebotomy Devices Market Segmentation
Device Type:
- Touch-Activated Safety Needles
- Touch-Activated Blood Collection Sets
- Touch-Activated Lancets
- Other Devices
Use Setting:
- Hospitals
- Diagnostics/Reference Labs
- Clinics & Physician Offices
- Other Settings
Safety Mechanism:
- Passive/Auto-Engage
- User-Activated (touch) mechanisms
- Hybrid/Other
Procurement:
- GPO/IDN Contracting
- Distributor Procurement
- Direct Tender/Other
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Frequently Asked Questions
How big is the touch activated phlebotomy devices market in 2026?
The global touch activated phlebotomy devices market is estimated to be valued at USD 288.7 million in 2026.
What will be the size of touch activated phlebotomy devices market in 2036?
The market size for the touch activated phlebotomy devices market is projected to reach USD 819.7 million by 2036.
How much will be the touch activated phlebotomy devices market growth between 2026 and 2036?
The touch activated phlebotomy devices market is expected to grow at a 11.0% CAGR between 2026 and 2036.
What are the key product types in the touch activated phlebotomy devices market?
The key product types in touch activated phlebotomy devices market are touch-activated safety needles, touch-activated blood collection sets, touch-activated lancets and other devices.
Which use setting segment to contribute significant share in the touch activated phlebotomy devices market in 2026?
In terms of use setting, hospitals segment to command 44.0% share in the touch activated phlebotomy devices market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Device Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Device Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Device Type , 2026 to 2036
- Touch-Activated Safety Needles
- Touch-Activated Blood Collection Sets
- Touch-Activated Lancets
- Other Devices
- Y to o to Y Growth Trend Analysis By Device Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Device Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Use Setting
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Use Setting, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Use Setting, 2026 to 2036
- Hospitals
- Diagnostics/Reference Labs
- Clinics & Physician Offices
- Other Settings
- Y to o to Y Growth Trend Analysis By Use Setting, 2021 to 2025
- Absolute $ Opportunity Analysis By Use Setting, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Safety Mechanism
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Safety Mechanism, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Safety Mechanism, 2026 to 2036
- Passive/Auto-Engage
- User-Activated (touch) mechanisms
- Hybrid/Other
- Y to o to Y Growth Trend Analysis By Safety Mechanism, 2021 to 2025
- Absolute $ Opportunity Analysis By Safety Mechanism, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Procurement
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Procurement, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Procurement, 2026 to 2036
- GPO/IDN Contracting
- Distributor Procurement
- Direct Tender/Other
- Y to o to Y Growth Trend Analysis By Procurement, 2021 to 2025
- Absolute $ Opportunity Analysis By Procurement, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Device Type
- By Use Setting
- By Safety Mechanism
- By Procurement
- Competition Analysis
- Competition Deep Dive
- BD
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Greiner Bio-One
- Terumo
- Nipro
- B. Braun
- Sarstedt
- Cardinal Health
- Medline
- Smiths Medical
- Vacuette (Greiner)
- BD
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Use Setting, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Safety Mechanism, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Procurement, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Use Setting, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Safety Mechanism, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Procurement, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Use Setting, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Safety Mechanism, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Procurement, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by Use Setting, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Safety Mechanism, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Procurement, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by Use Setting, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by Safety Mechanism, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Procurement, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by Use Setting, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by Safety Mechanism, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by Procurement, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by Use Setting, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by Safety Mechanism, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by Procurement, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by Use Setting, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by Safety Mechanism, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by Procurement, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Device Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Device Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Device Type
- Figure 6: Global Market Value Share and BPS Analysis by Use Setting, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Use Setting, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Use Setting
- Figure 9: Global Market Value Share and BPS Analysis by Safety Mechanism, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Safety Mechanism, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Safety Mechanism
- Figure 12: Global Market Value Share and BPS Analysis by Procurement, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Procurement, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Procurement
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by Device Type , 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Device Type , 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Device Type
- Figure 29: North America Market Value Share and BPS Analysis by Use Setting, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Use Setting, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Use Setting
- Figure 32: North America Market Value Share and BPS Analysis by Safety Mechanism, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Safety Mechanism, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Safety Mechanism
- Figure 35: North America Market Value Share and BPS Analysis by Procurement, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Procurement, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Procurement
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by Device Type , 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Device Type , 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Device Type
- Figure 42: Latin America Market Value Share and BPS Analysis by Use Setting, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by Use Setting, 2026-2036
- Figure 44: Latin America Market Attractiveness Analysis by Use Setting
- Figure 45: Latin America Market Value Share and BPS Analysis by Safety Mechanism, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Safety Mechanism, 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Safety Mechanism
- Figure 48: Latin America Market Value Share and BPS Analysis by Procurement, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Procurement, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Procurement
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by Device Type , 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by Device Type , 2026-2036
- Figure 54: Western Europe Market Attractiveness Analysis by Device Type
- Figure 55: Western Europe Market Value Share and BPS Analysis by Use Setting, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by Use Setting, 2026-2036
- Figure 57: Western Europe Market Attractiveness Analysis by Use Setting
- Figure 58: Western Europe Market Value Share and BPS Analysis by Safety Mechanism, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by Safety Mechanism, 2026-2036
- Figure 60: Western Europe Market Attractiveness Analysis by Safety Mechanism
- Figure 61: Western Europe Market Value Share and BPS Analysis by Procurement, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Procurement, 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Procurement
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by Device Type , 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by Device Type , 2026-2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by Device Type
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by Use Setting, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by Use Setting, 2026-2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by Use Setting
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by Safety Mechanism, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by Safety Mechanism, 2026-2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by Safety Mechanism
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by Procurement, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by Procurement, 2026-2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by Procurement
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by Device Type , 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by Device Type , 2026-2036
- Figure 80: East Asia Market Attractiveness Analysis by Device Type
- Figure 81: East Asia Market Value Share and BPS Analysis by Use Setting, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by Use Setting, 2026-2036
- Figure 83: East Asia Market Attractiveness Analysis by Use Setting
- Figure 84: East Asia Market Value Share and BPS Analysis by Safety Mechanism, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by Safety Mechanism, 2026-2036
- Figure 86: East Asia Market Attractiveness Analysis by Safety Mechanism
- Figure 87: East Asia Market Value Share and BPS Analysis by Procurement, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by Procurement, 2026-2036
- Figure 89: East Asia Market Attractiveness Analysis by Procurement
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by Device Type , 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by Device Type , 2026-2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by Device Type
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by Use Setting, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by Use Setting, 2026-2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by Use Setting
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by Safety Mechanism, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by Safety Mechanism, 2026-2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by Safety Mechanism
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by Procurement, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by Procurement, 2026-2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by Procurement
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by Device Type , 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by Device Type , 2026-2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by Device Type
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by Use Setting, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by Use Setting, 2026-2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by Use Setting
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by Safety Mechanism, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by Safety Mechanism, 2026-2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by Safety Mechanism
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by Procurement, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by Procurement, 2026-2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by Procurement
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

