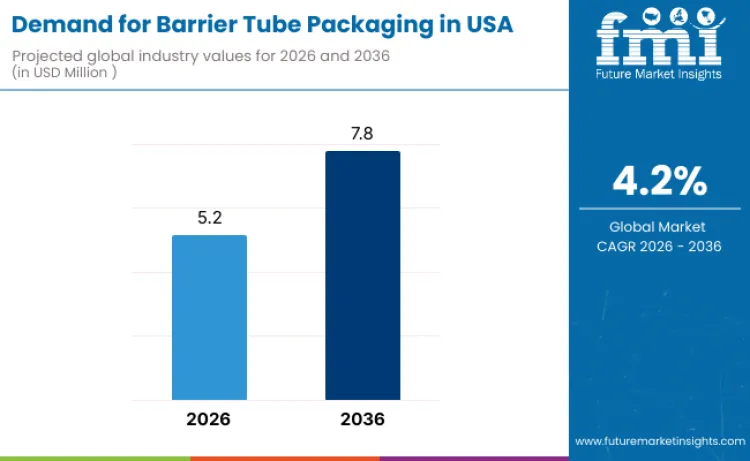
Demand for barrier tube packaging in USA is valued at USD 5.2 million in 2026 and is projected to reach USD 7.8 million by 2036, reflecting a CAGR of 4.2%. Demand grows due to steady consumption in personal care, cosmetics, pharmaceuticals, and oral care products where protection from oxygen, moisture, and light is essential. Brand owners prioritize packaging formats that extend shelf life while supporting controlled dispensing and portability.
Flexible packaging formats lead usage because multilayer barrier tubes combine lightweight construction with strong product protection and design flexibility. Aluminum-plastic and advanced polymer laminate structures prevent ingredient degradation and preserve formulation integrity. Improved sealing performance, print quality, and recyclability-focused material innovation support broader adoption across premium and mass-market product categories.
West USA, South USA, Northeast USA, and Midwest USA represent key growth regions supported by dense personal care manufacturing bases, pharmaceutical production hubs, and contract packaging operations. Berry Global, Albea Group, Essel Propack USA, Montebello Packaging, and CCL Container anchor competitive activity through multilayer barrier technologies, customization capabilities, and supply reliability aligned with evolving brand, regulatory, and ecofriendly requirements across USA packaging value chains.
| Metric | Value |
|---|---|
| USA Barrier Tube Packaging Sales Value (2026) | USD 5.2 million |
| USA Barrier Tube Packaging Forecast Value (2036) | USD 7.8 million |
| USA Barrier Tube Packaging Forecast CAGR (2026 to 2036) | 4.2% |
Demand for barrier tube packaging in the USA is shaped by product protection requirements, shelf-life stability needs, and compatibility with regulated consumer goods. Buyers evaluate barrier performance against oxygen, moisture, and light, along with material efficiency and process compatibility. Adoption patterns reflect usage across personal care, pharmaceuticals, food, and specialty products where controlled dispensing and extended product integrity are operational priorities within USA manufacturing and packaging systems.
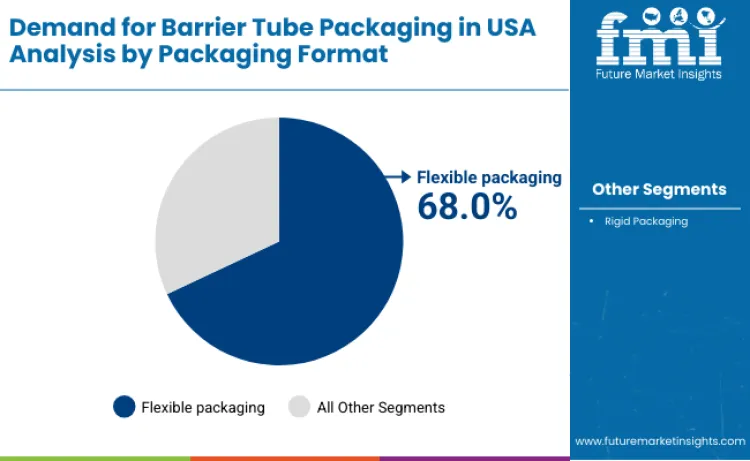
Flexible packaging holds 68.0%, making it the leading packaging-format segment in the USA. Flexible barrier tubes offer lightweight construction, reduced material usage, and efficient transportation characteristics. These formats support squeezability, controlled dispensing, and multilayer barrier integration suitable for personal care and pharmaceutical products. Rigid packaging holds 32.0%, serving applications requiring higher structural rigidity and premium positioning. Packaging-format distribution reflects preference for flexible tubes due to cost efficiency, consumer convenience, and compatibility with high-volume USA filling lines.
Key Points:
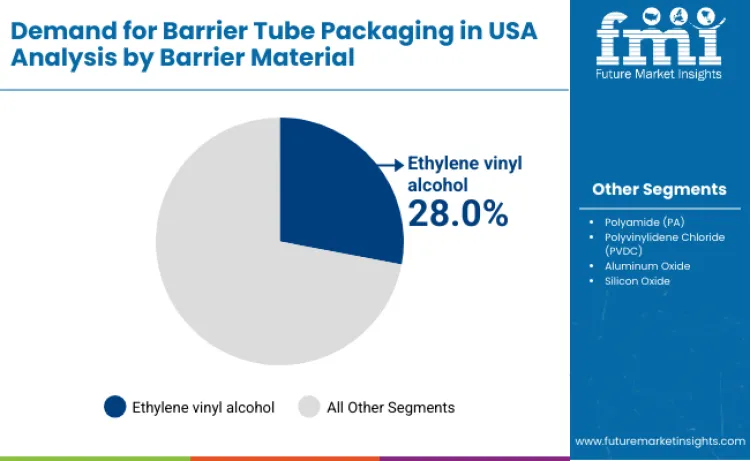
Ethylene vinyl alcohol holds 28.0%, making it the leading barrier-material segment in the USA. EVOH provides strong oxygen-barrier performance critical for shelf-life preservation in sensitive formulations. Aluminum oxide holds 25.0%, supporting high gas and aroma barrier requirements with thin coating layers. Polyvinylidene chloride holds 20.0%, offering moisture and oxygen resistance for specific product categories. Silicon oxide holds 15.0%, enabling transparent barrier coatings with recyclability advantages. Polyamide holds 12.0%, contributing mechanical strength and moderate barrier performance. Barrier-material distribution reflects balanced selection based on performance, processing compatibility, and regulatory considerations.
Key Points:
Plastic substrates hold 44.0%, making them the leading substrate segment in the USA. Plastic tubes support multilayer barrier integration, lightweight design, and compatibility with flexible packaging formats. Metal, paper and paperboard, and glass substrates serve limited applications requiring specific barrier, rigidity, or sustainability attributes. Substrate distribution reflects dominance of plastic due to processing versatility, cost efficiency, and alignment with high-speed tube manufacturing infrastructure across USA packaging operations.
Key Points:
Demand increases as personal care, pharmaceuticals, food, and industrial product manufacturers in USA adopt barrier tube packaging to protect formulations sensitive to oxygen, moisture, and light. Multilayer tubes support shelf-life stability, dosage control, and product integrity across regulated categories. Usage aligns with high-volume consumer goods production, prescription and OTC packaging needs, and premium product presentation standards within domestic manufacturing and distribution networks.
Personal care brands use barrier tubes for creams, gels, and ointments requiring protection against oxidation and moisture ingress. Pharmaceutical manufacturers rely on aluminium barrier laminate and EVOH-based tubes for topical medications to meet FDA stability and contamination control expectations. Food producers apply barrier tubes for condiments, pastes, and specialty products requiring extended shelf life after opening. Industrial adhesives and sealants use barrier tubes to prevent premature curing. Compatibility with automated filling lines supports adoption in large-scale US plants. Brand owners value controlled dispensing and reduced product waste. Growth in OTC healthcare and premium skincare sustains steady demand across domestic packaging operations.
Sustainability initiatives create pressure to reduce multilayer structures that complicate recycling within US waste systems. Material cost volatility for aluminium foils and specialty polymers affects procurement planning. Some brands evaluate mono-material tubes or alternative formats such as pumps and airless bottles. Recycling limitations for barrier laminates influence long-term packaging strategy. Regulatory scrutiny on packaging waste increases reporting and compliance requirements. Capital investment needed to modify filling equipment slows transition decisions. Demand remains stable where barrier performance is essential, while growth faces constraints from sustainability targets and material optimization efforts across USA packaging sectors.
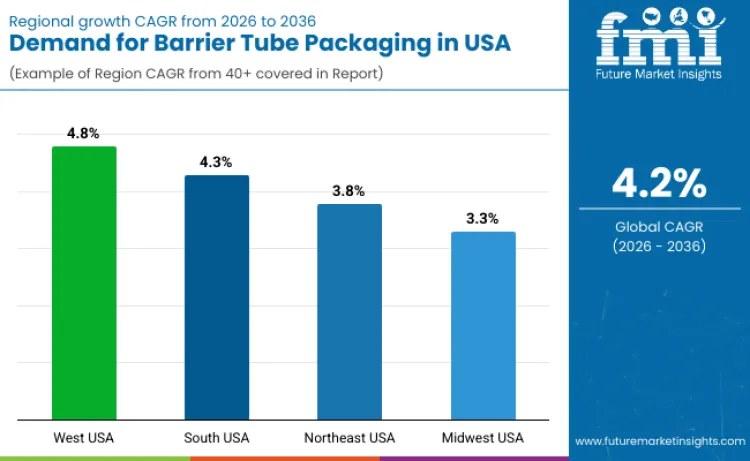
Demand for barrier tube packaging in the USA is expanding due to rising use in pharmaceuticals, personal care, cosmetics, and specialty food products requiring oxygen, moisture, and light protection. West USA leads with a 4.8% CAGR, supported by premium personal care brands, pharmaceutical innovation, and sustainability-led packaging upgrades. South USA follows at 4.3%, driven by large-scale oral care, cosmetics manufacturing, and contract packaging operations. Northeast USA records a 3.8% CAGR, shaped by pharmaceutical tube packaging, regulated OTC products, and controlled formulation environments. Midwest USA posts a 3.3% CAGR, reflecting stable demand from mass-market personal care, healthcare supplies, and industrial applications. Regional variation reflects brand positioning, regulatory exposure, and manufacturing scale across the USA.
| Region | CAGR (2026 to 2036) |
|---|---|
| West USA | 4.8% |
| South USA | 4.3% |
| Northeast USA | 3.8% |
| Midwest USA | 3.3% |
West USA demand is driven by high-value product positioning, formulation sensitivity, and premium brand requirements. The region’s CAGR of 4.8% reflects strong use of barrier tubes in dermatological products, cosmeceuticals, prescription topicals, and specialty nutraceuticals. Manufacturers prioritize multi-layer laminate and EVOH-based tubes to protect active ingredients from oxidation and moisture ingress. Sustainability initiatives influence adoption of recyclable barrier structures and downgauged materials. Contract manufacturers serving premium brands demand tight dimensional control and consistent barrier performance. Packaging formats favor small- to mid-diameter tubes with high-quality decoration for brand differentiation.
South USA demand is volume-driven and linked to large-scale consumer product manufacturing. The region’s CAGR of 4.3% reflects extensive use of barrier tube packaging in toothpaste, oral care gels, hair treatments, skin creams, and OTC pharmaceuticals. High-throughput production facilities require barrier tubes that maintain seal integrity under fast filling speeds. Contract packaging operations support national brands with standardized tube formats and consistent barrier performance. Cost efficiency, supply continuity, and machinability influence procurement decisions. Demand favors aluminum-plastic laminate and co-extruded barrier tubes supplied in bulk quantities.
Northeast USA demand reflects regulatory oversight, pharmaceutical concentration, and precision packaging requirements. The region’s CAGR of 3.8% is supported by use of barrier tubes for prescription ointments, OTC creams, medical gels, and specialty therapeutic products. Manufacturers emphasize barrier consistency, extractables compliance, and documentation traceability. Packaging suppliers provide validated tube structures meeting pharmaceutical standards. Smaller production batches increase demand for flexible order quantities and customized specifications. Growth remains steady, shaped by compliance-driven replacement and incremental product launches.
Midwest USA demand is stable and manufacturing-focused, reflecting established production of personal care, healthcare, and industrial-use products. The region’s CAGR of 3.3% reflects continued use of barrier tubes in hand creams, antiseptic gels, veterinary products, and institutional healthcare supplies. Manufacturers prioritize durability, shelf-life protection, and compatibility with existing filling equipment. Procurement decisions emphasize cost control and long-term supplier relationships. Innovation-led adoption remains limited, while replacement demand sustains baseline consumption. Packaging formats favor standard diameters and conventional barrier constructions.
Demand for barrier tube packaging in the USA is driven by usage across personal care, cosmetics, oral care, pharmaceuticals, and specialty food products requiring extended shelf stability. Applications depend on effective oxygen, moisture, and aroma barriers combined with squeeze performance and product compatibility. Buyers evaluate multilayer laminate structures, EVOH or aluminum barrier performance, tube integrity, and decoration quality aligned with brand requirements. Procurement teams prioritize suppliers with domestic manufacturing, reliable tooling support, and compliance with USA packaging and food-contact regulations. Trend in the USA industry reflects movement toward recyclable barrier structures, downgauged materials, and compatibility with mono-material initiatives while maintaining protection performance.
Berry Global operates extensive USA tube manufacturing facilities supplying laminated and barrier plastic tubes for personal care and healthcare brands. Albea Group maintains a strong USA footprint producing barrier tubes with advanced multilayer structures and high-quality decoration capabilities. Essel Propack USA supports demand through laminated tube solutions widely used in oral care and cosmetic applications requiring robust barrier properties. Montebello Packaging operates USA manufacturing serving pharmaceutical, cosmetic, and specialty product segments with aluminum and barrier tube formats. CCL Container supplies barrier tube packaging through integrated USA operations focused on personal care and healthcare end uses. Competitive positioning in the USA reflects barrier-material engineering, domestic production scale, decoration capability, and supply reliability supporting regulated and brand-sensitive applications.
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Packaging Format | Flexible Packaging; Rigid Packaging |
| Barrier Material | Ethylene Vinyl Alcohol (EVOH); Polyamide (PA); Polyvinylidene Chloride (PVDC); Aluminum Oxide; Silicon Oxide |
| Substrate | Plastic; Metal; Paper & Paperboard; Glass |
| End Use | Food; Beverages; Pharmaceutical; Cosmetics; Personal Care |
| Regions Covered | West USA; South USA; Northeast USA; Midwest USA |
| Key Companies Profiled | Berry Global; Albéa Group; Essel Propack USA; Montebello Packaging; CCL Container |
| Additional Attributes | Dollar sales by packaging format, barrier material, and end use; flexible plastic tubes dominate high-barrier adoption; pharmaceutical and personal care applications drive specification upgrades; demand influenced by shelf-life extension requirements, lightweight packaging preference, and compatibility with advanced multilayer barrier structures. |
How big is the demand for barrier tube packaging in USA in 2026?
The demand for barrier tube packaging in USA is estimated to be valued at USD 5.2 million in 2026.
What will be the size of barrier tube packaging demand in USA in 2036?
The demand size for barrier tube packaging in USA is projected to reach USD 7.8 million by 2036.
How much will the demand for barrier tube packaging in USA grow between 2026 and 2036?
The demand for barrier tube packaging in USA is expected to grow at a 4.2% CAGR between 2026 and 2036.
What are the key packaging formats in the barrier tube packaging demand in USA?
The key packaging formats in barrier tube packaging demand in USA include flexible packaging and other laminated tube structures.
Which barrier-material segment is expected to contribute a significant share in the barrier tube packaging demand in USA in 2026?
In terms of barrier material, ethylene vinyl alcohol (EVOH) is expected to command 28.0% share in the barrier tube packaging demand in USA in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.