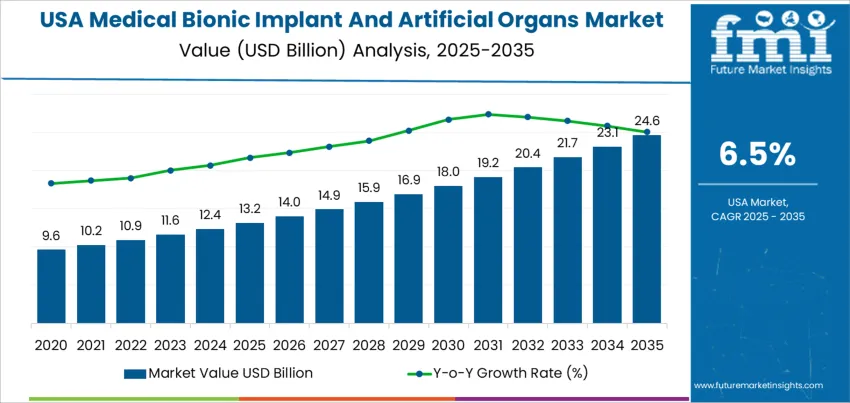
The demand for medical bionic implants and artificial organs in the USA is valued at USD 13.2 billion in 2025 and is forecasted to reach USD 24.6 billion by 2035, reflecting a CAGR of 6.5%. Demand is influenced by a rising population living with chronic cardiac, auditory, and musculoskeletal impairments that require long-term functional restoration. Aging demographics, greater clinical acceptance of implantable assistive technologies, and advancements in regenerative and biointegrated systems are driving adoption across hospitals, specialty care networks, and transplant programs. Reimbursement pathways supporting life-sustaining devices are also improving patient access. Heart bionics represent the leading category. Ventricular assist devices and related circulatory-support technologies are used for patients with advanced heart failure who are not eligible or are awaiting heart transplantation. Increasing clinical focus on improving survival rates and quality of life in high-risk cardiac patients supports continued adoption.
Demand is concentrated across West USA, South USA, and Northeast USA because of significant cardiac care infrastructure, major transplant centers, and stronger procedural volume capabilities. These regions also support ongoing clinical studies and advanced device-integration programs. Key suppliers include Össur, Cochlear Limited, Sonova, Abiomed (Johnson & Johnson), and LifeNet Health. Their development strategies emphasize improved biocompatibility, sensor-enabled performance monitoring, and durability to reduce device failure risks. Companies are also expanding collaboration with healthcare providers to enhance patient rehabilitation and long-term outcomes.
Growth contribution for demand for medical bionic implants and artificial organs in the United States is primarily driven by adoption in cardiovascular and orthopedic applications. Pacemakers, ventricular assist devices, and artificial joints account for a large share of total revenue contribution due to established reimbursement pathways and high procedure volumes. These categories provide consistent year-to-year gains that shape much of the overall growth profile. Cochlear implants and advanced prosthetics add supplementary contributions through technology upgrades and expanded eligibility criteria.
Emerging products deliver rising but smaller contributions in the early period. Developments in bioengineered tissues, artificial pancreas systems, and implantable vision aids generate incremental acceleration as clinical validation and regulatory approvals advance. Their influence on growth increases in the later stage as specialized centers expand patient access and long-term outcome data improve payer support. Demographic factors such as aging populations and chronic disease prevalence form the core contribution base across the forecast timeline. Technology-driven improvements in device performance and durability sustain upward movement without relying on cyclical demand changes. The index indicates that foundational implant categories remain the primary contributors while fast-evolving innovations add layered growth over time.
| Metric | Value |
|---|---|
| USA Medical Bionic Implant And Artificial Organs Sales Value (2025) | USD 13.2 billion |
| USA Medical Bionic Implant And Artificial Organs Forecast Value (2035) | USD 24.6 billion |
| USA Medical Bionic Implant And Artificial Organs Forecast CAGR (2025-2035) | 6.5% |
Demand for medical bionic implants and artificial organs in the USA is increasing because hospitals face a rising number of patients with organ failure, advanced cardiovascular disease and severe musculoskeletal impairment. Longer life expectancy and higher prevalence of diabetes and chronic kidney disease strengthen the need for long term solutions that restore function and reduce dependence on frequent medical interventions. Cardiac assist devices, cochlear implants and orthopedic bionics allow patients to maintain mobility, communication and daily independence, which aligns with rehabilitation goals after injury or surgery.
Transplant waiting lists remain lengthy, so artificial organs and implantable devices provide clinical options when donor organs are unavailable. Research centers and medical device companies in the USA develop advanced materials, improved power systems and refined neural interfaces to enhance device compatibility with the human body. Insurance coverage expands for selected implants, supporting wider patient access through specialist clinics and surgical teams experienced in complex procedures. Constraints include high device cost, rigorous regulatory pathways and the need for lifelong follow up care. Not all patients qualify due to underlying health conditions or adaptation challenges. Workforce training and infrastructure requirements can limit adoption to major medical centers.
Demand for medical bionic implants and artificial organs in the United States is driven by the high prevalence of chronic diseases, trauma-related disabilities, and rising replacement procedures for vital functions such as cardiac rhythm, limb mobility, and sensory restoration. Patient preference for durability, improved quality of life, and minimally invasive implant procedures strengthens utilization. Technology adoption also benefits from reimbursement access, skilled surgical infrastructure, and continuous enhancement of device longevity and biocompatibility.
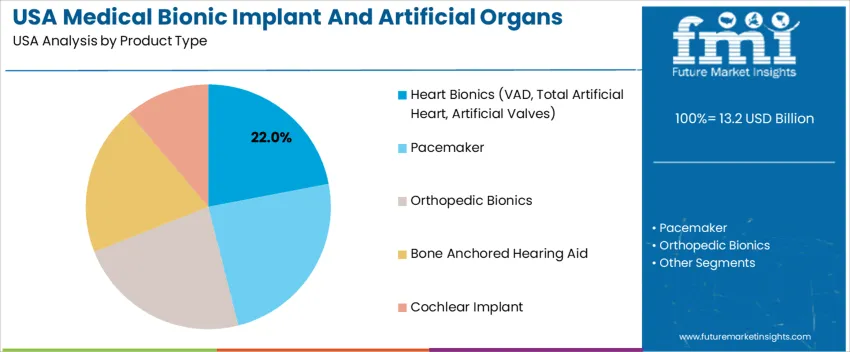
Heart bionics represent 22.0%, making them the most widely adopted product group among reported categories. This segment includes ventricular assist devices (VADs), total artificial hearts, and artificial heart valves, all essential in addressing heart failure and structural cardiac disorders. Demand is supported by a significant patient pool requiring temporary or long-term circulatory support and improved survival outcomes. Other product categories, including pacemakers, orthopedic bionics, bone-anchored hearing aids, and cochlear implants, contribute the remaining share. Each addresses functional impairments ranging from hearing restoration to musculoskeletal mobility but without specified percentage distribution. Adoption depends on implant reliability, patient age profile, and therapy urgency in U.S. clinical practice.
Key Points:
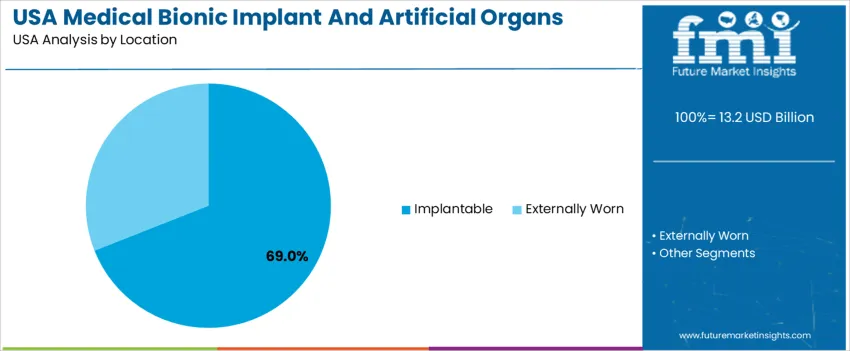
Implantable devices account for 69.0%, reinforced by permanent therapeutic use that improves survival, sensory function, and mobility. These implants are preferred for advanced cardiac care, cochlear surgery, and orthopedic reconstruction where continuous physiological integration is required. Adoption benefits from surgeon familiarity, proven clinical results, and regulatory support for high-risk devices improving long-term patient outcomes. Externally worn solutions represent the remaining share and serve patients requiring transitional, non-invasive, or monitoring-only support. These products extend access for individuals awaiting surgery or those unable to undergo invasive procedures. The USAge split reflects strong investment in implant-grade materials and automation technologies within U.S. healthcare.
Key Points:
Growth of chronic disease prevalence, increased demand for mobility restoration and expansion of transplant alternatives are driving demand.
In the United States, medical bionic implants and artificial organs gain adoption as cardiovascular disease, diabetes and kidney failure generate sustained clinical need for long-term restorative solutions. Orthopedic and spinal implants support aging adults and injured workers who require mobility and pain-relief interventions to remain active in home and workplace settings. Artificial hearts and ventricular assist devices are used as bridge-to-transplant or permanent therapy for patients on long waitlists. Cochlear and retinal implants help restore sensory function in individuals with profound impairment, supported by private insurance and Medicare reimbursement pathways. Hospitals with advanced surgical programs in major states such as California, Texas and New York adopt bionic technologies to improve survival rates and reduce disability burdens. These medical requirements maintain stable procurement of implantable and extracorporeal systems.
High procedural cost, limited surgeon specialization and risk management for long-term device function restrain demand.
Implant surgeries involve expensive devices and extended hospitalization that may create out-of-pocket costs for patients even with insurance coverage. Availability of surgeons trained in complex bionic implantation varies by region, leading to access disparities in rural areas. Artificial organs require strict monitoring to prevent infection, clotting or device malfunction, and some patients may hesitate to commit to lifelong follow-up care. Device recalls or safety concerns can slow adoption of newer models as hospitals prioritize reliability history. These economic and clinical considerations contribute to cautious expansion.
Shift toward personalized implant design, increased integration with digital monitoring platforms and rising focus on regenerative-bionic hybrid solutions define key trends.
Custom implants designed using 3D imaging improve anatomical fit and reduce surgical revision rates. Remote monitoring systems enable clinicians to track artificial organ performance and detect complications early, strengthening home-care support. Research collaborations explore regenerative scaffolds that work with electronic components to enhance biological compatibility for long-term organ replacement. Rehabilitation programs emphasize coordinated recovery to maximize the functional benefits of neural and musculoskeletal bionics. These developments indicate sustained, innovation-driven demand for medical bionic implants and artificial organs across the United States healthcare system.
Demand for medical bionic implants and artificial organs in the United States reflects adoption of advanced prosthetics, cardiovascular support systems, neural implants, and organ-replacement technologies linked to chronic disease prevalence and surgical innovation. Reimbursement access, clinical-trial activity, and proximity to specialized hospitals strongly influence deployment patterns. Medical device manufacturers evaluate biocompatibility, miniaturization, and durability as primary procurement criteria. The West USA leads with 7.4% CAGR, followed by the South USA at 6.7%, Northeast USA at 5.9%, and Midwest USA at 5.2%, indicating differences in patient-care capacity, regulatory engagement, and commercialization infrastructure.
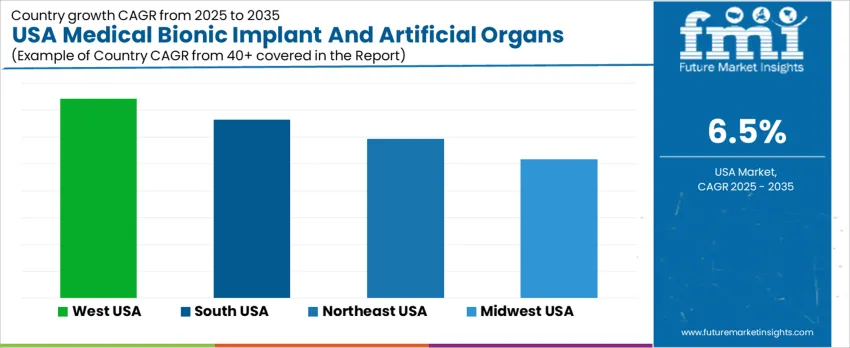
| Region | CAGR (2025-2035) |
|---|---|
| West USA | 7.4% |
| South USA | 6.7% |
| Northeast USA | 5.9% |
| Midwest USA | 5.2% |
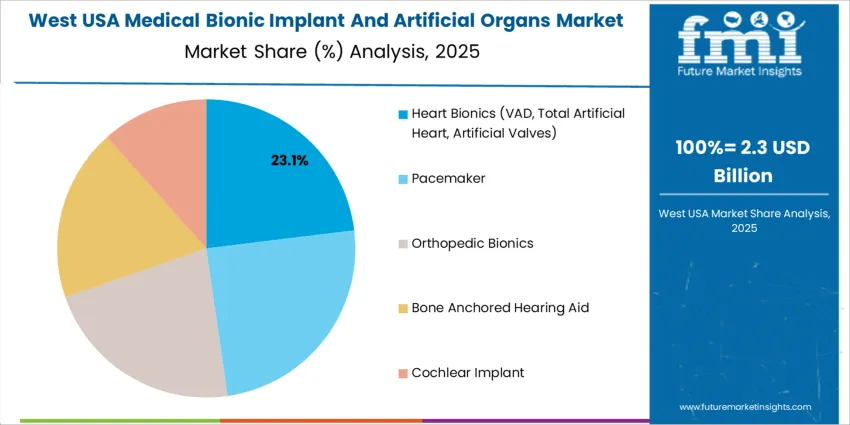
West USA demonstrates the strongest national expansion at 7.4% CAGR, supported by innovation ecosystems in California and Washington where translational medical-device engineering and surgical robotics are advancing implant capabilities. Hospitals specializing in heart-failure therapies expand USAge of ventricular-assist devices and artificial heart modules to address an aging population with complex conditions. Neural-interface implants are evaluated within rehabilitation centers serving traumatic-injury cases linked to outdoor lifestyles and sports activity. Procurement focuses on high-precision manufacturing quality, tissue-integration outcomes, and wireless-monitoring compatibility. Venture-backed startups collaborate with university medical hubs to accelerate FDA submissions and pilot programs.
Key adoption drivers
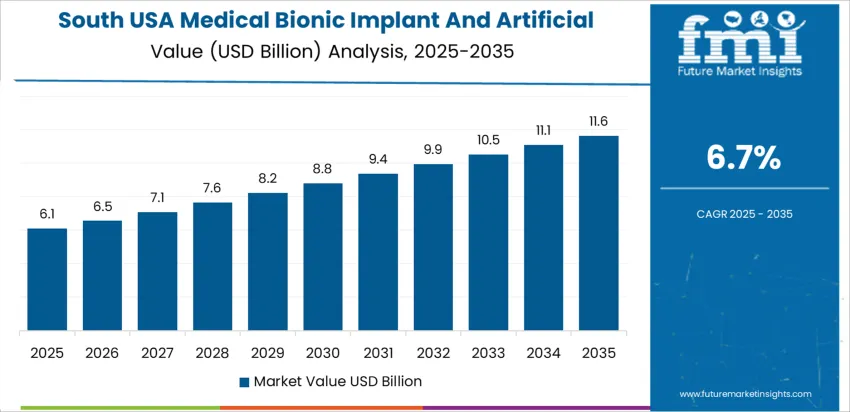
South USA expands at 6.7% CAGR, influenced by rising orthopedic implant surgeries in Texas, Florida, and Georgia, driven by higher obesity-related mobility decline and increased joint-replacement volume. Specialized trauma hospitals adopt advanced bionic limb solutions to treat accident-related amputations common on regional freight and industrial sites. Heart-transplant support programs deploy artificial circulatory systems to reduce patient wait-list risks. Reimbursement decision-makers evaluate life-extension benefit and readmission-reduction indicators to justify capital procurement. Medical-device logistics networks across Gulf ports improve supply stability for implant components sourced internationally.
Key adoption drivers
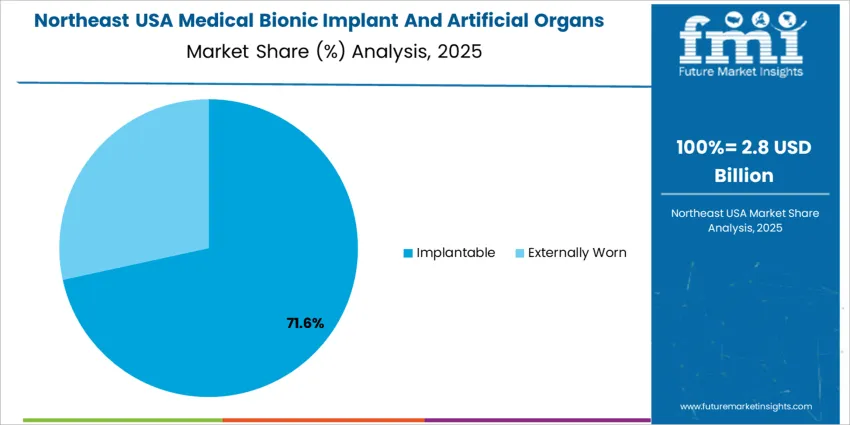
Northeast USA posts 5.9% CAGR, anchored by leading academic hospitals in New York, Massachusetts, and Pennsylvania conducting research in artificial-organ biocompatibility and immune-response minimization. Neurological-rehabilitation facilities deploy bionic assistive implants for stroke-recovery pathways driven by an aging population in dense urban settings. Heart-failure management programs use implantable support systems designed to reduce surgery complexity. Procurement criteria emphasize FDA-approved reliability data and long-term functional survival rates. Regional insurance providers evaluate cost-offset potential linked to fewer hospital days and improved independence of high-acuity patients.
Key adoption drivers
Midwest USA expands steadily at 5.2% CAGR, supported by strong manufacturing-engineering capabilities and centralized transplant-care networks in Illinois, Ohio, and Michigan. Chronic-disease treatment programs adopt artificial-organ solutions for patients with diabetes-linked kidney and circulation complications. Orthopedic prosthetics demand continues in regions with greater occupation-related musculoskeletal injuries. Procurement leaders assess device reliability under high physical-stress conditions required for active and industrial workers. Regional medical suppliers balance affordability with durability, ensuring patient suitability across broad demographic segments.
Key adoption drivers
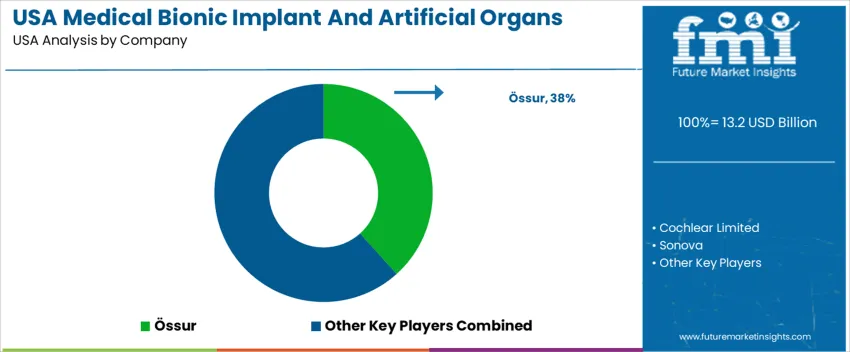
Medical bionic implants and artificial organs in the United States serve patients requiring functional restoration after limb loss, organ failure, or progressive sensory impairment. Hospitals and specialized surgical centers remain the primary decision-makers, guided by clinical evidence, surgeon familiarity, and reimbursement eligibility. Applications cover upper-limb prosthetics, cochlear implantation, ventricular assist devices, and bioengineered tissues that support long-term independence. Adoption increases as device reliability and patient usability improve across daily-living activities. Össur holds an estimated 38.3% share in advanced prosthetic systems. Its U.S. presence includes upper-limb robotics integrated with microprocessor control and adaptive movement support.
Clinical service networks reinforce continuity of fitting and post-operative rehabilitation. Cochlear Limited contributes significantly through implantable hearing systems used in severe or profound hearing loss, supported by established audiology programs across the country. Sonova participates with implant and bone-conduction technologies that integrate speech-processing platforms used in pediatric and adult care. Abiomed (Johnson & Johnson) strengthens demand for circulatory support devices used in cardiology and transplant pathways where mechanical assist solutions extend patient survival. LifeNet Health supports regenerative applications including bio-tissue used in reconstructive procedures, injury recovery, and graft-based structural repairs. Competitive positioning in the United States depends on FDA-cleared performance evidence, rehabilitation access, surgical training support, and durable insurance coverage enabling long-term device use for improving functional outcomes.
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Product Type | Heart Bionics (VAD, Total Artificial Heart, Artificial Valves), Pacemaker, Orthopedic Bionics, Bone Anchored Hearing Aid, Cochlear Implant |
| Location | Implantable, Externally Worn |
| Regions Covered | West USA, South USA, Northeast USA, Midwest USA |
| Key Companies Profiled | Össur, Cochlear Limited, Sonova, Abiomed (Johnson & Johnson), LifeNet Health |
| Additional Attributes | Dollar sales by product type and implant location; regional demand drivers across hospital-based implantation and outpatient bionic solutions; adoption trends in cardiovascular support, auditory restoration, and musculoskeletal mobility enhancement; technology improvements in biocompatibility, neural integration, and powered assistive systems; reimbursement environment shaping access to advanced cardiac and orthopedic implants; major U.S. manufacturers expanding innovation pipelines through bio-integration, remote monitoring, and customized prosthetic design. |
How big is the demand for medical bionic implant and artificial organs in USA in 2025?
The demand for medical bionic implant and artificial organs in USA is estimated to be valued at USD 13.2 billion in 2025.
What will be the size of medical bionic implant and artificial organs in USA in 2035?
The market size for the medical bionic implant and artificial organs in USA is projected to reach USD 24.6 billion by 2035.
How much will be the demand for medical bionic implant and artificial organs in USA growth between 2025 and 2035?
The demand for medical bionic implant and artificial organs in USA is expected to grow at a 6.5% CAGR between 2025 and 2035.
What are the key product types in the medical bionic implant and artificial organs in USA?
The key product types in medical bionic implant and artificial organs in USA are heart bionics (vad, total artificial heart, artificial valves), pacemaker, orthopedic bionics, bone anchored hearing aid and cochlear implant.
Which location segment is expected to contribute significant share in the medical bionic implant and artificial organs in USA in 2025?
In terms of location, implantable segment is expected to command 69.0% share in the medical bionic implant and artificial organs in USA in 2025.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.