Demand for Medical Bionic Implant and Artificial Organs in Japan
Demand for Medical Bionic Implant and Artificial Organs in Japan Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Demand for Medical Bionic Implant and Artificial Organs in Japan Forecast and Outlook 2025 to 2035
In 2025, demand for medical bionic implants and artificial organs in Japan is valued at USD 1.9 billion and is projected to reach USD 3.1 billion by 2035 at a CAGR of 5.1%. Early growth reflects Japan’s rapidly aging population and the steady rise in chronic cardiovascular, neurological, and mobility-related conditions. Cardiac assist devices, cochlear implants, and neurostimulation systems form the largest share of early demand. University hospitals and advanced specialty centers lead adoption due to surgical expertise and integrated rehabilitation infrastructure. Domestic patients show high acceptance of implant-supported mobility and sensory restoration, supported by structured insurance reimbursement and long post-operative care pathways. Device demand tracks closely with procedure volumes rather than discretionary spending.
After 2030, market expansion in Japan becomes more outcome-driven than procedure-driven. Demand rises from about USD 2.4 billion in 2030 toward USD 3.1 billion by 2035 as device performance, longevity, and revision reduction become central procurement factors. Artificial organ programs remain limited to controlled clinical use, with ventricular assist systems and hybrid renal support technologies receiving focused investment. Integration with home-based monitoring and rehabilitation platforms strengthens post-implant care continuity. Domestic medical device manufacturers and select global suppliers compete through miniaturization, reliability, and regulatory compliance under Japan’s strict clinical evaluation framework. Pricing dynamics increasingly reflect material science gains, long-term implant durability, and bundled care models rather than unit device cost alone.
Quick Stats of the Demand for Medical Bionic Implants and Artificial Organs in Japan
- Demand for Medical Bionic Implants and Artificial Organs in Japan Value (2025): USD 1.9 billion
- Demand for Medical Bionic Implants and Artificial Organs in Japan Forecast Value (2035): USD 3.1 billion
- Demand for Medical Bionic Implants and Artificial Organs in Japan Forecast CAGR (2025–2035): 5.1%
- Demand for Medical Bionic Implants and Artificial Organs in Japan Leading Product Type: Heart Bionics (22%)
- Demand for Medical Bionic Implants and Artificial Organs in Japan Key Growth Regions: Kyushu & Okinawa, Kanto, Kansai, Chubu, Tohoku, Rest of Japan
- Demand for Medical Bionic Implants and Artificial Organs in Japan Top Players: Touch Bionics Inc., Lifenet Health Inc., Cochlear Ltd., Sonova, Other Companies (combined)
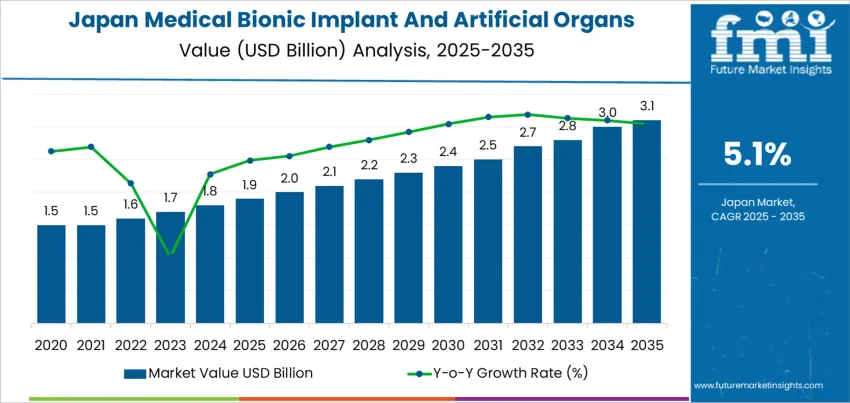
What is the Demand Forecast for Medical Bionic Implant and Artificial Organs in Japan through 2035?
The overall demand for medical bionic implant and artificial organs in Japan increases from USD 1.9 billion in 2025 to USD 2.3 billion by 2030, adding USD 0.4 billion in absolute value. This phase reflects steady growth anchored in an aging population, rising incidence of sensory impairment, cardiovascular disease, and orthopedic degeneration, along with stable reimbursement coverage for approved implant categories. Demand expansion remains clinically regulated, shaped by hospital procurement cycles, surgeon training capacity, and conservative device adoption pathways. Growth during this period is volume anchored, driven by incremental increases in cochlear implants, cardiac assist devices, and joint-related bionic systems rather than rapid technology substitution.
From 2030 to 2035, the market expands from USD 2.3 billion to USD 3.1 billion, adding a larger USD 0.8 billion in the second half of the decade. This back weighted acceleration reflects deeper integration of sensor-enabled implants, longer device life cycles, and improved biocompatible materials that reduce post-surgical complications. Demand also strengthens as artificial organ technologies move closer to routine bridge-to-transplant and long-term support applications. As Japan continues to face constrained organ donor availability and rising chronic disease burden, bionic implants and artificial organs shift from selective interventions to structurally embedded solutions within advanced clinical care pathways.
Medical Bionic Implant and Artificial Organs Industry in Japan Key Takeaways
| Metric | Value |
|---|---|
| Industry Value (2025) | USD 1.9 billion |
| Forecast Value (2035) | USD 3.1 billion |
| Forecast CAGR (2025–2035) | 5.1% |
Why Medical Bionic Implants and Artificial Organs Are Gaining Momentum in Japan
The increasing proportion of older citizens and a growing incidence of chronic illness in Japan support rising demand for bionic implants and artificial organs. As the share of people aged 65 and over climbs, cases of heart disease, kidney failure, hearing or vision loss, and other age-related disorders rise. Traditional organ donation and transplantation face persistent shortages in donor supply. These realities prompt patients and healthcare providers to seek mechanical, electronic or bioengineered solutions rather than wait for donor organs. The result is wider adoption of implants for heart support, joint replacement, sensory restoration, and organ substitution among patients unable to access transplants.
Looking ahead, demand is likely to expand as technological advances and demographic pressures converge. Improvements in biocompatible materials, miniaturized electronics, and modular implant design make artificial organs and bionic implants more reliable and less invasive. As aging continues and number of patients requiring organ replacement grows, implants may increasingly become the standard option for heart, kidney, joint or sensory failure. Expansion of regenerative medicine, implantable devices, and rehab-oriented prosthetics will further grow the market. Access constraints such as high cost, surgical complexity, and long-term monitoring burdens may temper growth, yet structural demographic change and organ supply challenges steer Japan toward greater reliance on implant- and bioengineered-based solutions.
How Are Product Type and Location Structuring the Demand for Medical Bionic Implants and Artificial Organs in Japan?
The demand for medical bionic implants and artificial organs in Japan is structured by product type and implantation location. Heart bionics, including ventricular assist devices, total artificial hearts, and artificial valves, account for 22% of total demand, followed by pacemakers, orthopedic bionics, bone anchored hearing aids, and cochlear implants. By location, implantable devices represent 69.0% of total consumption, while externally worn systems serve specific therapeutic needs. Demand behavior is shaped by aging population trends, chronic disease prevalence, surgical infrastructure capacity, and long term rehabilitation requirements. These segments reflect how clinical urgency, device durability, and patient mobility needs determine adoption across hospitals and specialty treatment centers in Japan.
Why Do Heart Bionics Lead the Medical Bionic Implant Product Demand in Japan?
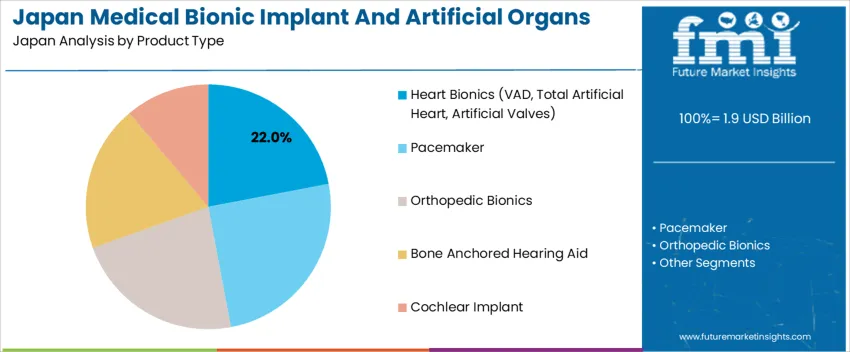
Heart bionics account for 22% of total medical bionic implant and artificial organ demand in Japan due to the high clinical priority of advanced cardiac care in an aging population. Ventricular assist devices and artificial valves are widely used in patients with end stage heart failure, valvular disease, and post myocardial infarction complications. These devices support both bridge to transplant and destination therapy approaches where donor organ availability remains limited. Cardiac centers across major urban regions rely on heart bionics to stabilize critically ill patients and improve survival outcomes under long term mechanical support.
Heart bionics also benefit from continuous clinical refinement and improved biocompatibility that support extended implantation periods. Reimbursement coverage through the national health insurance system further supports patient access. Surgical expertise in cardiovascular intervention remains strong across Japanese tertiary hospitals. These disease burden, reimbursement alignment, and procedural specialization factors sustain heart bionics as the leading product type within the medical bionic implant demand structure in Japan.
Why Do Implantable Devices Dominate the Location Demand for Medical Bionic Implants in Japan?
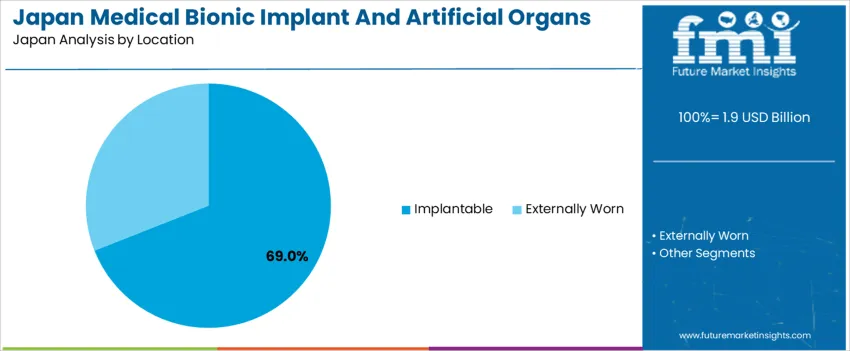
Implantable devices account for 69.0% of total medical bionic implant and artificial organ demand in Japan due to their ability to provide continuous physiological support without reliance on external attachments. Implantable systems reduce infection risk, improve patient mobility, and support long term therapeutic stability across cardiac, auditory, and orthopedic applications. Pacemakers, cochlear implants, orthopedic joint systems, and implanted heart valves all rely on internal placement for consistent functional performance. These advantages align with the clinical preference for permanent internal therapeutic solutions across hospital based treatment models.
Implantable devices also support better integration with body systems through improved biocompatible materials and long life power management. Post-surgery rehabilitation outcomes are more predictable with implanted systems compared with externally worn alternatives. National clinical guidelines favor implantable devices where long term disease management is required. These functional continuity, clinical reliability, and rehabilitation efficiency factors position implantable systems as the dominant location segment in the Japan medical bionic implant demand landscape.
Why Is Japan Approaching Bionic Implants and Artificial Organs as a Demographic Necessity Rather Than an Experimental Technology?
Demand for medical bionic implants and artificial organs in Japan is driven primarily by demographic pressure rather than elective innovation. Japan has one of the oldest populations globally, with a large share of citizens living with degenerative organ conditions, mobility loss, and chronic disease. Conventional transplant availability remains structurally limited, which elevates artificial organs from optional therapy to functional necessity. Cardiac assist devices, artificial joints, neural implants, and sensory prosthetics are increasingly applied to extend independent living rather than only to extend survival. This demographic reality anchors demand in public health continuity rather than frontier medicine.
How Do Hospital Robotics, Rehabilitation Culture, and Care Infrastructure Shape Adoption?
Japanese hospitals integrate bionic implants into long-term rehabilitation pathways rather than as isolated surgical interventions. Strong post-operative rehab systems allow continuous performance tuning of neural-controlled limbs, powered exoskeletons, and artificial joints. Robotics integration in surgery improves implantation precision, especially for spine, hip, and cochlear devices. Home-care compatibility also matters because many elderly patients recover outside hospital settings. Community-based physiotherapy and assistive mobility programs reinforce implant utilization. This infrastructure-driven adoption model differs from markets where bionics remain confined to elite surgical centers.
What Clinical Risk Tolerance and Cost Controls Restrain Wider Patient Access?
Despite technical capability, Japan applies strict patient selection and reimbursement control in advanced implant therapies. National insurance systems prioritize functional recovery probability and long-term cost balance before approving expensive artificial organs. High expectations for device reliability and low tolerance for foreign-body failure slow rapid adoption of experimental platforms. Long validation timelines and conservative clinical trials restrict early-stage device penetration. Import dependence for some high-end components also affects procurement timing. These financial and clinical safeguards slow large-scale deployment even as medical need continues to rise.
How Are Soft Robotics, Neural Interfaces, and Human-Machine Integration Redefining Future Demand?
Future demand in Japan is shifting toward soft robotics, bio-compatible materials, and machine interfaces that respond directly to neural signals. Myoelectric limbs, brain-computer interfaces, and sensory-feedback prosthetics are moving from lab testing toward pilot clinical use. Artificial organs are also evolving with tissue-integrated surfaces to reduce immune rejection and long-term clot risk. Domestic engineering firms collaborate closely with hospitals to tighten feedback between patient performance and device design. These trends indicate that demand is moving beyond replacement toward biological integration and adaptive human-machine synchronization.
What is the Demand for Medical Bionic Implant And Artificial Organs in Japan by Region?
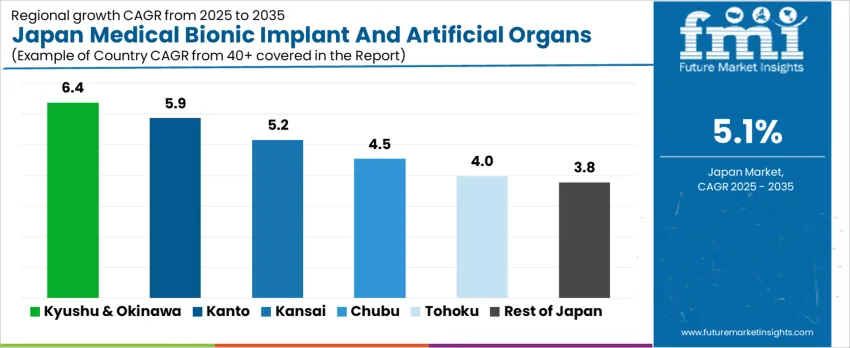
| Region | CAGR (%) |
|---|---|
| Kyushu & Okinawa | 6.4% |
| Kanto | 5.9% |
| Kansai | 5.2% |
| Chubu | 4.5% |
| Tohoku | 4.0% |
| Rest of Japan | 3.8% |
The demand for medical bionic implants and artificial organs in Japan is expanding steadily across all regions, led by Kyushu & Okinawa at a 6.4% CAGR. Growth in this region is supported by rising hospital investments, increasing surgical capacity, and growing adoption of advanced implant based therapies. Kanto follows at 5.9%, driven by its dense concentration of large hospitals, medical research centers, and specialist clinics that support high procedural volumes. Kansai records 5.2% growth, supported by established healthcare infrastructure and ageing population needs. Chubu at 4.5% shows moderate uptake linked to regional medical device manufacturing and hospital modernization. Tohoku and Rest of Japan, at 4.0% and 3.8%, reflect slower but stable growth shaped by lower population density and fewer tertiary care centers.
How is Advanced Surgical Infrastructure Shaping Demand in Kyushu And Okinawa?
Growth in Kyushu and Okinawa reflects a CAGR of 6.4% through 2035 for medical bionic implant and artificial organ demand, supported by expanding regional hospital capacity, medical tourism intake, and rising use of advanced reconstructive procedures. Cardiac assist devices, orthopedic bionics, and neurostimulation implants show steady uptake across tertiary care centers. Government backed healthcare modernization improves access to complex implant procedures. Aging population trends strengthen long term device utilization. Demand remains procedure driven, with stable growth tied to surgical volume expansion and specialist availability across regional clinical hubs.
- Tertiary hospitals anchor advanced implant procedures
- Medical tourism contributes incremental procedural demand
- Cardiac and orthopedic devices dominate utilization
- Aging population sustains long term replacement cycles
What is Driving Strong Adoption of Bionic Implants in Kanto?
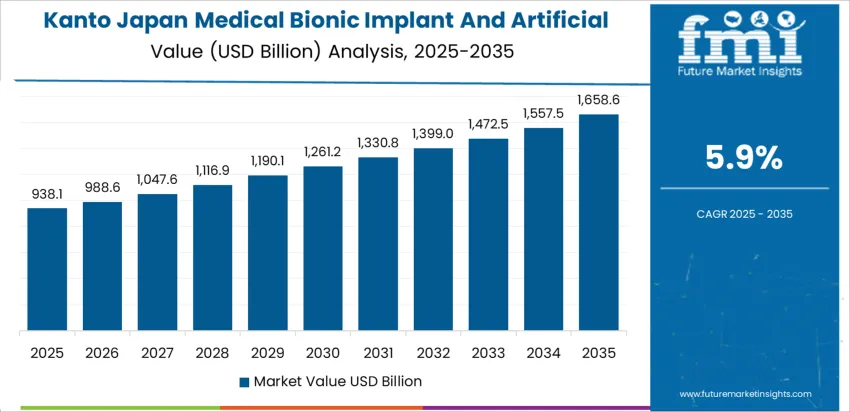
Expansion in Kanto registers a CAGR of 5.9% through 2035 for medical bionic implant and artificial organ demand, driven by dense concentration of university hospitals, research institutions, and high income patient populations. Robotic surgery integration supports precise implant placement and postoperative recovery. Neuroprosthetics, cochlear implants, and cardiac rhythm management systems remain core segments. Clinical trial activity also supports early adoption of next generation artificial organ systems. Demand remains innovation led and referral driven, shaped by complex case volumes and structured reimbursement frameworks across major metropolitan medical centers.
- University hospitals drive complex implant volumes
- Robotic surgery supports precision device placement
- Neuroprosthetics and cardiac systems anchor demand
- Clinical trials sustain early technology adoption
Why is Kansai Emerging as a Key Regional Market for Bionic Implants?
Kansai is expanding at a CAGR of 5.2% through 2035 for medical bionic implant and artificial organ demand, supported by strong presence of rehabilitation centers, trauma care hospitals, and orthopedic specialty clinics. Sports injury treatment and accident related reconstructive surgeries contribute steady implant usage. Limb prosthetics, spinal stimulators, and joint replacement bionics show consistent volume movement. Regional device distributors strengthen supply access for mid-sized hospitals. Demand remains recovery focused and referral based, with stable growth tied to rehabilitation intensity and regional accident treatment capacity.
- Trauma care centers support steady implant placement
- Orthopedic bionics dominate procedural volumes
- Rehabilitation networks sustain long term device usage
- Regional distributors improve hospital access
How is Industrial Healthcare Spending Supporting Demand in Chubu?
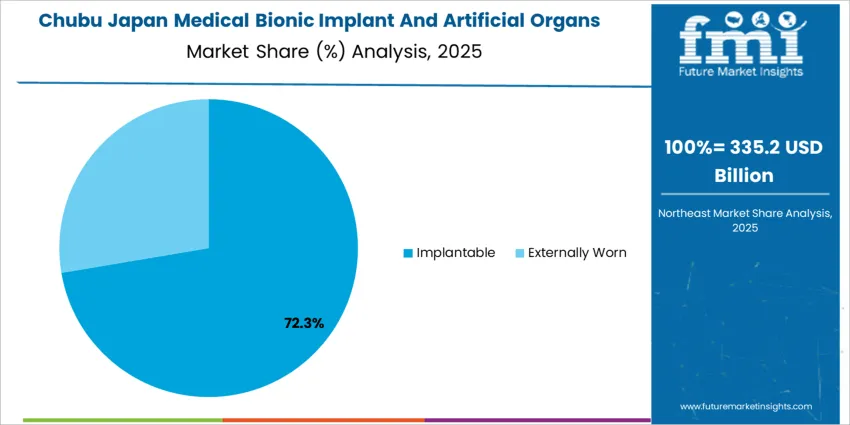
Progress in Chubu reflects a CAGR of 4.5% through 2035 for medical bionic implant and artificial organ demand, supported by industrial workforce healthcare coverage, occupational injury treatment, and expanding regional hospital investment. Manufacturing related trauma cases contribute to prosthetic and orthopedic implant demand. Cardiac assist devices show moderate adoption linked to chronic disease management. Employer sponsored health programs support inpatient procedure volumes. Demand remains treatment driven rather than innovation driven, with procurement aligned to hospital budget planning and predictable surgical throughput across industrial population clusters.
- Occupational injury treatment supports prosthetic demand
- Employer health coverage sustains surgical access
- Cardiac assist devices show moderate clinical uptake
- Hospital budgeting guides controlled procurement cycles
What is Sustaining Measured Growth of Bionic Implants in Tohoku?
Development in Tohoku records a CAGR of 4.0% through 2035 for medical bionic implant and artificial organ demand, shaped by regional hospital consolidation, aging rural populations, and gradual expansion of specialty surgical services. Orthopedic implants for mobility restoration remain the primary application area. Dialysis related artificial organ systems also contribute steady demand. Limited access to advanced research hospitals moderates high end device penetration. Demand remains necessity driven and clinically focused, guided by essential mobility restoration and chronic disease management needs across dispersed healthcare networks.
- Orthopedic mobility restoration anchors demand
- Dialysis systems support artificial organ utilization
- Aging rural populations sustain baseline procedure volumes
- Hospital consolidation guides regional access patterns
Why Does the Rest of Japan Show Gradual but Stable Adoption Trends?
Across the rest of Japan, growth reflects a CAGR of 3.8% through 2035 for medical bionic implant and artificial organ demand, supported by community hospital coverage, national insurance reimbursement stability, and steady chronic disease incidence. Implant procedures focus on essential cardiac devices, joint replacements, and basic prosthetic systems. Limited specialist density restrains high end device expansion. Patient referral flows toward major metros for advanced care. Demand remains stable and necessity oriented, with procurement driven by routine clinical needs rather than accelerated technology refresh across smaller regional healthcare facilities.
- Community hospitals anchor essential implant procedures
- National insurance stability supports predictable access
- Referral patterns concentrate complex cases in metros
- Basic prosthetics and cardiac devices dominate usage
What Is Driving the Demand for Medical Bionic Implants and Artificial Organs in Japan and Who Are the Key Players Shaping the Market?
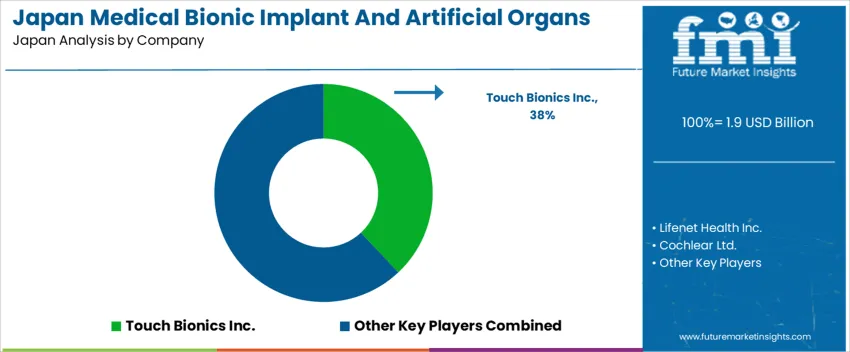
Demand for medical bionic implants and artificial organs in Japan is rising as the population ages and chronic diseases become more prevalent. Many elderly individuals require assistive or replacement devices for impaired hearing, mobility, heart function, or organ failure. Advances in bioengineering, robotics, neural interface technologies, and implantable electronics make devices safer, more reliable, and more effective than earlier generations. Tight supply of donor organs, long transplant waiting lists, and growing acceptance of prosthetic and artificial organ solutions also drive demand. Hospitals and specialty clinics expand capacity for implant surgeries, and growing patient awareness supports adoption across age groups.
Among firms active in or influencing the Japanese bionic implant and artificial organ market are Touch Bionics Inc., Lifenet Health Inc., Cochlear Ltd., Sonova and a range of other global and regional companies. Cochlear and Sonova lead in auditory prosthetics and hearing restoration implants. Touch Bionics and Lifenet Health contribute via prosthetics, implantable devices or tissue engineering solutions. Other medical device firms supply cardiac, neural, and orthopedic implants or artificial organ technologies. These companies influence the market by investing in R&D, gaining regulatory approvals, and establishing hospital and clinic distribution networks to deliver both bionic and artificial organ solutions across Japan.
Key Players in Medical Bionic Implants and Artificial Organs Industry in Japan
- Touch Bionics Inc.
- Lifenet Health Inc.
- Cochlear Ltd.
- Sonova
- Other Companies (combined)
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units (2025) | USD billion |
| Product Type | Heart Bionics (VAD, Total Artificial Heart, Artificial Valves), Pacemaker, Orthopedic Bionics, Bone Anchored Hearing Aid, Cochlear Implant |
| Location | Implantable, Externally Worn |
| Region | Kyushu & Okinawa, Kanto, Kinki, Chubu, Tohoku, Rest of Japan |
| Countries Covered | Japan |
| Key Companies Profiled | Touch Bionics Inc., Lifenet Health Inc., Cochlear Ltd., Sonova, Other Companies (combined) |
| Additional Attributes | Dollar sales by product type, Dollar by sales by implant location, Dollar by sales by region, Regional CAGR, Aging population and chronic disease prevalence, Surgical infrastructure and hospital adoption rates, Insurance reimbursement frameworks, Post-operative care and rehabilitation programs, Device miniaturization and biocompatibility, Clinical outcome-driven procurement, Robotics and neural interface integration, Artificial organ bridge-to-transplant programs, Domestic and global supplier competition |
Medical Bionic Implants and Artificial Organs Industry in Japan Segmentation
Product Type:
- Heart Bionics (VAD, Total Artificial Heart, Artificial Valves)
- Pacemaker
- Orthopedic Bionics
- Bone Anchored Hearing Aid
- Cochlear Implant
Location:
- Implantable
- Externally Worn
Region:
- Kyushu & Okinawa
- Kanto
- Kansai
- Chubu
- Tohoku
- Rest of Japan
Bibliography
- Ministry of Health, Labour and Welfare (Japan). (2024). Annual health, labour and welfare report 2024 (summary). Ministry of Health, Labour and Welfare.
- Ministry of Economy, Trade and Industry (Japan). (2024). Vision for the medical device industry 2024. Ministry of Economy, Trade and Industry.
- Organisation for Economic Co-operation and Development, & World Health Organization. (2024). Health at a glance: Asia/Pacific 2024. OECD Publishing.
Frequently Asked Questions
How big is the demand for medical bionic implant and artificial organs in Japan in 2025?
The demand for medical bionic implant and artificial organs in Japan is estimated to be valued at USD 1.9 billion in 2025.
What will be the size of medical bionic implant and artificial organs in Japan in 2035?
The market size for the medical bionic implant and artificial organs in Japan is projected to reach USD 3.1 billion by 2035.
How much will be the demand for medical bionic implant and artificial organs in Japan growth between 2025 and 2035?
The demand for medical bionic implant and artificial organs in Japan is expected to grow at a 5.1% CAGR between 2025 and 2035.
What are the key product types in the medical bionic implant and artificial organs in Japan?
The key product types in medical bionic implant and artificial organs in Japan are heart bionics (vad, total artificial heart, artificial valves), pacemaker, orthopedic bionics, bone anchored hearing aid and cochlear implant.
Which location segment is expected to contribute significant share in the medical bionic implant and artificial organs in Japan in 2025?
In terms of location, implantable segment is expected to command 69.0% share in the medical bionic implant and artificial organs in Japan in 2025.
Table of Content
- Executive Summary
- Japan Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Japan Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Japan Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- Japan Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2025 to 2035
- Heart Bionics (VAD, Total Artificial Heart, Artificial Valves)
- Pacemaker
- Orthopedic Bionics
- Bone Anchored Hearing Aid
- Cochlear Implant
- Y to o to Y Growth Trend Analysis By Product Type , 2020 to 2024
- Absolute $ Opportunity Analysis By Product Type , 2025 to 2035
- Japan Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Location
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Location, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Location, 2025 to 2035
- Implantable
- Externally Worn
- Y to o to Y Growth Trend Analysis By Location, 2020 to 2024
- Absolute $ Opportunity Analysis By Location, 2025 to 2035
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Location
- Competition Analysis
- Competition Deep Dive
- Touch Bionics Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Lifenet Health Inc.
- Cochlear Ltd.
- Sonova
- Other Companies (combined)
- Touch Bionics Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Japan Market Value (USD Million) Forecast by Region, 2020 to 2035
- Table 2: Japan Market Value (USD Million) Forecast by Product Type , 2020 to 2035
- Table 3: Japan Market Value (USD Million) Forecast by Location, 2020 to 2035
- Table 4: Japan Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 5: Japan Market Value (USD Million) Forecast by Product Type , 2020 to 2035
- Table 6: Japan Market Value (USD Million) Forecast by Location, 2020 to 2035
List of Figures
- Figure 1: Japan Market Pricing Analysis
- Figure 2: Japan Market Value (USD Million) Forecast 2020-2035
- Figure 3: Japan Market Value Share and BPS Analysis by Product Type , 2025 and 2035
- Figure 4: Japan Market Y-o-Y Growth Comparison by Product Type , 2025-2035
- Figure 5: Japan Market Attractiveness Analysis by Product Type
- Figure 6: Japan Market Value Share and BPS Analysis by Location, 2025 and 2035
- Figure 7: Japan Market Y-o-Y Growth Comparison by Location, 2025-2035
- Figure 8: Japan Market Attractiveness Analysis by Location
- Figure 9: Japan Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 10: Japan Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 11: Japan Market Attractiveness Analysis by Region
- Figure 12: Japan Market Incremental Dollar Opportunity, 2025-2035
- Figure 13: Japan Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 14: Japan Market Value Share and BPS Analysis by Product Type , 2025 and 2035
- Figure 15: Japan Market Y-o-Y Growth Comparison by Product Type , 2025-2035
- Figure 16: Japan Market Attractiveness Analysis by Product Type
- Figure 17: Japan Market Value Share and BPS Analysis by Location, 2025 and 2035
- Figure 18: Japan Market Y-o-Y Growth Comparison by Location, 2025-2035
- Figure 19: Japan Market Attractiveness Analysis by Location
- Figure 20: Japan Market - Tier Structure Analysis
- Figure 21: Japan Market - Company Share Analysis

