Ursolic Acid Market
Ursolic Acid Market Analysis - Size, Share, and Forecast 2026 to 2036
Ursolic Acid Market Forecast and Outlook 2026 to 2036
The ursolic acid category is set to expand from USD 10.34 billion in 2026 to USD 19.39 billion by 2036, progressing at a 6.5% CAGR. Growth is shaped by increasing adoption of plant-derived actives across pharmaceuticals, cosmetics, and functional nutrition products where developers want a well-defined bioactive that can be standardized, tested, and delivered in repeatable formats.
Ursolic acid continues to move from niche botanical positioning toward structured ingredient procurement, where buyers compare it by extraction pathway, purity specification, and stability performance rather than by plant origin alone. A key commercial driver is the shift toward specification-led purchasing. Procurement teams and formulation owners are prioritizing consistent quality markers such as assay range, residual solvent compliance, and batch reproducibility.
These factors matter because ursolic acid is increasingly supplied into structured applications that require process discipline, including finished dosage forms, cosmetic actives systems, and fortified product lines that must meet label consistency across production runs. Once a supplier is qualified, purchasing tends to stabilize because changing purity grade or extraction method can require renewed validation across sensory, stability, and efficacy expectations.
Quick Stats for Ursolic Acid Market
- Ursolic acid value in 2026: USD 10.34 billion
- Ursolic acid value in 2036: USD 19.39 billion
- Forecast CAGR from 2026 to 2036: 6.5%
- Leading application share: Pharmaceuticals 30.3%
- Leading extraction share: Solvent Extraction 29.0%
- Leading purity share: 95% to 99% 44.0%
- Leading form share: Powder 37.5%
- Key growth countries: USA, Spain, India
- Key players: Sabinsa Corporation, Sami Labs Limited, Changsha Staherb Natural Ingredients Co., Ltd., Changsha Luyuan Bio-Tech Co., Ltd., Others
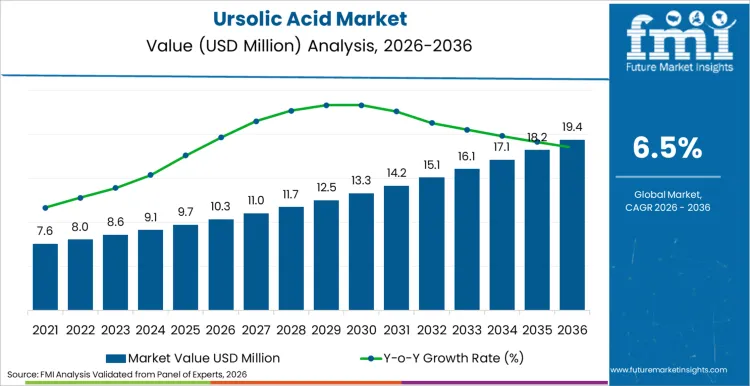
Ursolic Acid Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 10.34 billion |
| Market Forecast Value (2036) | USD 19.39 billion |
| Forecast CAGR (2026 to 2036) | 6.5% |
Why is Ursolic Acid Transitioning into a Specification-Led Botanical Active?
Ursolic acid demand is rising because buyers increasingly want plant-derived actives that behave like engineered inputs. That does not mean the ingredient loses its botanical identity. It means its commercial value is now tied to what can be measured and controlled. Purity range, batch-to-batch consistency, and extraction traceability influence which suppliers get repeat orders, particularly when the ingredient is supplied into finished dosage formats or high-visibility consumer products within the ursolic acid ingredient space.
A second driver is cross-category usability. Ursolic acid is being evaluated across pharmaceuticals, cosmetics, and functional foods, which expands addressable demand beyond any single downstream industry. This multi-application footprint encourages suppliers to strengthen standardized offerings, since the same purity grade can serve multiple customer types with different product goals. In practice, the ingredient fits portfolio building in a way that is consistent with broader activity in botanical ingredients and standardized extracts used across modern wellness categories.
Commercial buyers are also adjusting how they assess risk. Instead of focusing only on price per kilogram, many purchasers now evaluate supplier documentation, compliance readiness, and the ability to support audits. This is especially relevant when products are distributed through regulated channels or sold with structured benefit positioning. The preference for standardized actives aligns naturally with how brands manage pipelines in nutraceutical ingredient systems and related formulated products.
Segmental Analysis
Why does Pharmaceuticals hold the Leading Application Share?
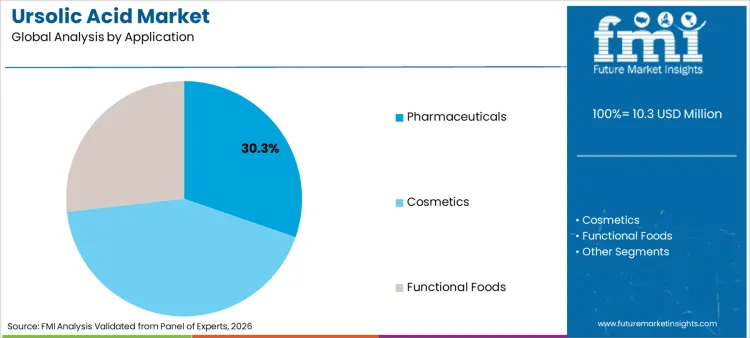
Pharmaceuticals account for a 30.3% share, positioning it as the strongest application segment. This reflects demand for ursolic acid where controlled concentration, documentation depth, and reproducible outcomes are required. In pharmaceutical-facing programs, buyers generally prioritize high assay consistency and stable impurity control because performance expectations are tied to defined dosing and repeatability. That creates stronger demand for standardized grades, especially those that sit within high-purity ranges that simplify formulation planning and compliance checks.
Pharmaceutical demand also tends to be less sensitive to seasonal retail swings. Once development programs validate material performance, purchasing becomes structured and contract-driven. This improves demand stability and reinforces long-term supplier relationships.
How does Solvent Extraction remain the Most Utilized Method at Scale?
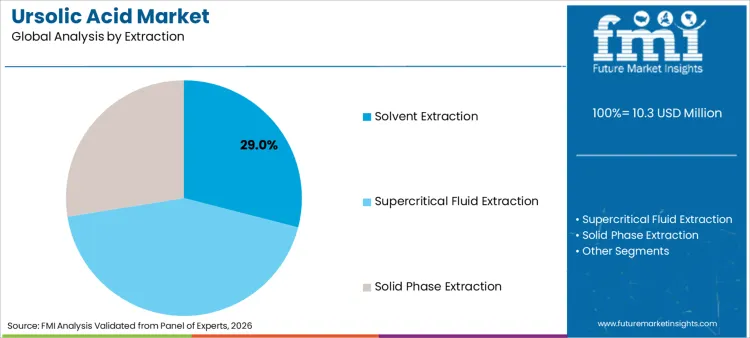
Solvent extraction leads with a 29.0% share, supported by its compatibility with industrial-scale output and cost-performance balance. Solvent extraction remains relevant because it can be integrated into scalable manufacturing workflows and can support high-volume supply programs when controlled properly. Buyers still evaluate solvent extraction outputs through residual solvent controls, consistent yield performance, and stable assay outcomes, especially when the ingredient is used in regulated or premium positioning.
Other extraction approaches such as supercritical fluid extraction and solid phase extraction remain important for specific supplier strategies and customer preferences. These methods can support differentiated positioning or improved selectivity in production, depending on the end-use specification requirements.
Why does 95% to 99% Purity Dominate Purchasing Patterns?
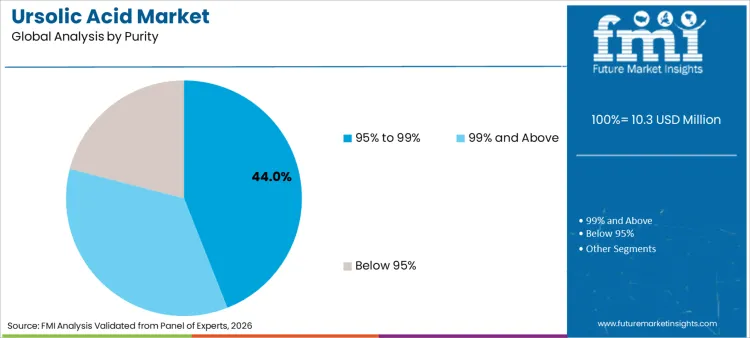
95% to 99% purity holds a 44.0% share, making it the leading purity band. This dominance reflects the balance buyers seek between high assay strength and scalable procurement. This range fits a wide set of applications, including pharmaceutical workstreams, cosmetics formulations, and structured nutrition products that require predictable active loading. It also aligns with formulation needs where high purity improves dosage control and reduces unwanted variability from co-extracted compounds.
Other purity segments remain relevant based on application. 99% and above may be preferred in highly controlled programs, while below 95% may be purchased for applications where cost targets and broader extract composition are acceptable.
Why does Powder lead the Form Mix Even with Growing Finished Dosage Formats?
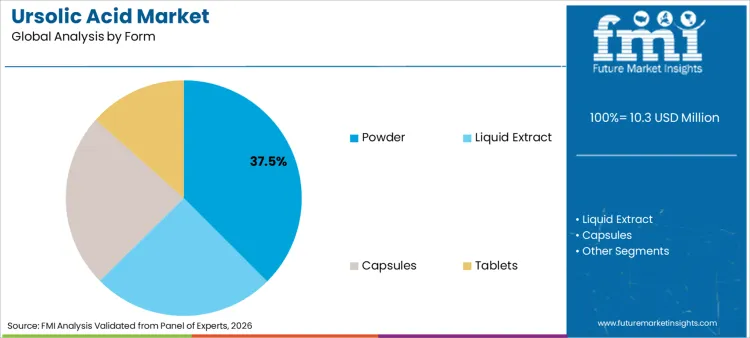
Ursolic acid powder holds a 37.5% share, reflecting practicality in storage, handling, and flexible downstream conversion. Powder formats support broad customer needs because they can be used directly in blending, encapsulation, tableting, and further formulation work. They also simplify shipping and warehousing efficiency, particularly for B2B supply chains that depend on stable inventory management across botanical extracts demand themes and botanical bioactive ingredient positioning.
Capsules and tablets represent finished dosage pathways that offer convenience and dosing clarity. Liquid extract formats remain relevant for certain cosmetics and functional product designs, particularly where rapid incorporation and processing speed matter.
What is Supporting Steady Volume Growth In High-Purity Grades?
High-purity demand is supported by buyer preference for ingredients that can be incorporated into multiple product lines with minimal variability. When purity sits in the leading band of 95% to 99%, manufacturers gain better control over loading levels and can maintain consistent outcomes across repeated production runs. This matters in finished dosage formats and in cosmetic actives systems where minor variation can affect product performance perception.
How is Cosmetics’ Demand Strengthening Commercial Relevance?
Cosmetics adoption supports volume growth because it is tied to ongoing portfolio refresh cycles. Skin and personal care formulations frequently rotate actives and product themes, creating repeat procurement opportunities for suppliers that can maintain consistency and provide formulation support. Cosmetics buyers tend to prioritize stability, compatibility with base systems, and consistent sensory outcomes, which supports continued demand for standardized grades.
What creates friction in procurement and supplier qualification?
Two factors commonly slow procurement cycles. First, buyers require documentation and quality assurance that matches their compliance systems. Second, supplier qualification often includes performance testing across multiple batches to confirm reproducibility. These steps increase sales cycle length but also strengthen long-term customer retention once suppliers are qualified.
Where are the Strongest Opportunities Emerging across Functional Foods?
Functional foods create incremental demand by incorporating standardized plant actives into wellness-led formats. These include fortified product designs where ingredient differentiation supports premium positioning.
For suppliers, the opportunity expands when they can provide the right purity grade, support stable blending performance, and maintain consistent specifications across higher-volume production runs aligned with nutraceutical product development pipelines.
How Do Growth Drivers Differ Across the USA, Spain, and India?
Global momentum differs by end-use specialization, production scale, and the maturity of botanical active supply networks. While demand spans pharmaceuticals, cosmetics, and functional products, each country reflects a distinct balance of adoption drivers.
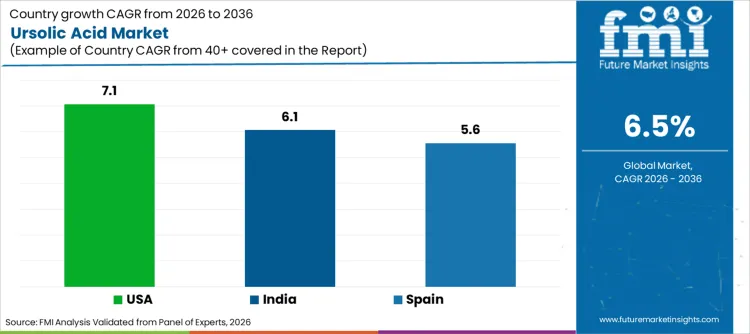
| Country | CAGR (%) |
|---|---|
| USA | 7.1% |
| Spain | 5.6% |
| India | 6.1% |
Why is the USA set to expand at a faster pace than the broader average?
The USA is forecast to grow at a 7.1% CAGR, supported by a strong base of nutraceutical development, structured supplementation habits, and active innovation pipelines across pharmaceuticals and wellness products. Demand strength also benefits from a mature ecosystem of botanical ingredient purchasing, where buyers are comfortable with standardized actives and formulation-led product positioning. This encourages steady procurement of high-purity grades that fit regulated and premium product lines.
Why does Spain show measured expansion driven by structured botanical adoption?
Spain is expected to progress at a 5.6% CAGR, reflecting balanced demand across pharmaceutical and consumer wellness channels. Growth in Spain is supported by regulated quality expectations and structured adoption of plant-derived inputs in formulations. Buyers typically emphasize documentation readiness and consistent specifications, which favors suppliers that can demonstrate reproducible purity and extraction control.
Why does India maintain strong growth through manufacturing expansion and export relevance?
India is projected to expand at a 6.1% CAGR, supported by expanding manufacturing capacity in botanical actives and growing demand across pharmaceuticals, cosmetics, and functional food product programs. Supplier competitiveness is influenced by the ability to deliver consistent grades at scale and support both domestic demand and international customer expectations. As product development activity grows, procurement becomes increasingly structured around standardized quality requirements.
Competitive Landscape
Competition is shaped by manufacturing control, purification capability, and quality documentation strength. Companies differentiate through their ability to supply consistent assay ranges, manage impurity profiles, and deliver stable batch performance that matches formulation expectations across pharmaceuticals, cosmetics, and functional foods.
Another competitive factor is the ability to support customers with application guidance. Buyers often need help aligning purity grades with target product formats such as powders for blending, capsules for finished dosage, or liquid extracts for cosmetics systems. Suppliers that provide dependable technical support and clear specification transparency tend to earn repeat procurement, especially in long-term programs where ingredient substitution increases formulation risk.
Supply reliability also matters. Customers prefer partners that can maintain consistent output over extended cycles, provide stable packaging and storage guidance, and support procurement schedules that match production planning. This encourages long-term supply relationships and reduces churn once qualification is completed.
Key Players
- Sabinsa Corporation
- Sami Labs Limited
- Changsha Staherb Natural Ingredients Co., Ltd.
- Changsha Luyuan Bio-Tech Co., Ltd.
- Shaanxi Hopeland Chem-Tech Co., Ltd.
- Xuancheng Baicao Pharmaceutical Co., Ltd.
- Shaanxi Huike Botanical Development Co., Ltd.
- Changsha E.K Herb Co., Ltd.
- Hunan NutraMax Inc.
- Shaanxi Jintai Biological Engineering Co., Ltd.
- Zhangjiajie Hengxing Biological Technology Co., Ltd.
- Xi’an TonKing Biotech Co., Ltd.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD Billion |
| Application | Pharmaceuticals; Cosmetics; Functional Foods |
| Extraction | Solvent Extraction; Supercritical Fluid Extraction; Solid Phase Extraction |
| Purity | 95% to 99%; 99% and Above; Below 95% |
| Form | Powder; Liquid Extract; Capsules; Tablets |
| Key Countries | USA; Spain; India |
| Key Companies Profiled | Sabinsa Corporation; Sami Labs Limited; Changsha Staherb Natural Ingredients Co., Ltd.; Changsha Luyuan Bio-Tech Co., Ltd.; Shaanxi Hopeland Chem-Tech Co., Ltd.; Xuancheng Baicao Pharmaceutical Co., Ltd.; Shaanxi Huike Botanical Development Co., Ltd.; Changsha E.K Herb Co., Ltd.; Hunan NutraMax Inc.; Shaanxi Jintai Biological Engineering Co., Ltd.; Zhangjiajie Hengxing Biological Technology Co., Ltd.; Xi’an TonKing Biotech Co., Ltd. |
Ursolic Acid Market Segmentation
By Application:
- Pharmaceuticals
- Cosmetics
- Functional Foods
By Extraction:
- Solvent Extraction
- Supercritical Fluid Extraction
- Solid Phase Extraction
By Purity:
- 95% to 99%
- 99% and Above
- Below 95%
By Form:
- Powder
- Liquid Extract
- Capsules
- Tablets
By Region:
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East and Africa
Bibliography
- European Medicines Agency. (2022). Guideline on quality of herbal medicinal products and traditional herbal medicinal products (Revision 3).
- National Center for Biotechnology Information. (2025). PubChem compound summary for ursolic acid.
- National Institutes of Health. (2023). Anti-inflammatory and antioxidant activity of ursolic acid systematic review and meta-analysis.
- National Institutes of Health. (2025). Recent developments on ursolic acid and its potential biological activities.
Frequently Asked Questions
What is the projected ursolic acid market size for 2026?
The ursolic acid market is expected to total USD 10.34 billion in 2026.
What value is expected for ursolic acid in 2036?
In 2036, demand for ursolic acid is forecast to reach USD 19.39 billion.
At what rate will the ursolic acid market progress from 2026 to 2036?
Ursolic acid demand is expected to grow at a 6.5% CAGR during 2026 to 2036.
Which application leads demand and what share does it hold?
Pharmaceuticals lead by application with a share of 30.3%.
Which extraction method leads demand and what share does it hold?
Solvent extraction dominates by extraction with a share of 29.0%.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application , 2026 to 2036
- Pharmaceuticals
- Cosmetics
- Functional Foods
- Pharmaceuticals
- Y to o to Y Growth Trend Analysis By Application , 2021 to 2025
- Absolute $ Opportunity Analysis By Application , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Extraction
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Extraction, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Extraction, 2026 to 2036
- Solvent Extraction
- Supercritical Fluid Extraction
- Solid Phase Extraction
- Solvent Extraction
- Y to o to Y Growth Trend Analysis By Extraction, 2021 to 2025
- Absolute $ Opportunity Analysis By Extraction, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Purity
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Purity, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Purity, 2026 to 2036
- 95% to 99%
- 99% and Above
- Below 95%
- 95% to 99%
- Y to o to Y Growth Trend Analysis By Purity, 2021 to 2025
- Absolute $ Opportunity Analysis By Purity, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Form
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Form, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Form, 2026 to 2036
- Powder
- Liquid Extract
- Capsules
- Tablets
- Powder
- Y to o to Y Growth Trend Analysis By Form, 2021 to 2025
- Absolute $ Opportunity Analysis By Form, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Application
- By Extraction
- By Purity
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Extraction
- By Purity
- By Form
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Application
- By Extraction
- By Purity
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Extraction
- By Purity
- By Form
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Application
- By Extraction
- By Purity
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Extraction
- By Purity
- By Form
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Application
- By Extraction
- By Purity
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Extraction
- By Purity
- By Form
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Application
- By Extraction
- By Purity
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Extraction
- By Purity
- By Form
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Application
- By Extraction
- By Purity
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Extraction
- By Purity
- By Form
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Application
- By Extraction
- By Purity
- By Form
- By Country
- Market Attractiveness Analysis
- By Country
- By Application
- By Extraction
- By Purity
- By Form
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Extraction
- By Purity
- By Form
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Extraction
- By Purity
- By Form
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Extraction
- By Purity
- By Form
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Extraction
- By Purity
- By Form
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Extraction
- By Purity
- By Form
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Extraction
- By Purity
- By Form
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Extraction
- By Purity
- By Form
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Extraction
- By Purity
- By Form
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Extraction
- By Purity
- By Form
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Extraction
- By Purity
- By Form
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Extraction
- By Purity
- By Form
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Extraction
- By Purity
- By Form
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Extraction
- By Purity
- By Form
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Extraction
- By Purity
- By Form
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Extraction
- By Purity
- By Form
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Extraction
- By Purity
- By Form
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Extraction
- By Purity
- By Form
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Extraction
- By Purity
- By Form
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Extraction
- By Purity
- By Form
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Extraction
- By Purity
- By Form
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Extraction
- By Purity
- By Form
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Application
- By Extraction
- By Purity
- By Form
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Application
- By Extraction
- By Purity
- By Form
- Competition Analysis
- Competition Deep Dive
- Sabinsa Corporation
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Sami Labs Limited
- Changsha Staherb Natural Ingredients Co., Ltd.
- Changsha Luyuan Bio-Tech Co., Ltd.
- Shaanxi Hopeland Chem-Tech Co., Ltd.
- Xuancheng Baicao Pharmaceutical Co., Ltd.
- Shaanxi Huike Botanical Development Co., Ltd.
- Changsha E.K Herb Co., Ltd.
- Hunan NutraMax Inc.
- Shaanxi Jintai Biological Engineering Co., Ltd.
- Zhangjiajie Hengxing Biological Technology Co., Ltd.
- Xi’an TonKing Biotech Co., Ltd.
- Sabinsa Corporation
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Extraction, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Purity, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Extraction, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Purity, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Extraction, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Purity, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by Extraction, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Purity, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by Extraction, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by Purity, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by Extraction, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by Purity, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by Extraction, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by Purity, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by Application , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by Extraction, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by Purity, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by Form, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Application , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Application
- Figure 6: Global Market Value Share and BPS Analysis by Extraction, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Extraction, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Extraction
- Figure 9: Global Market Value Share and BPS Analysis by Purity, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Purity, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Purity
- Figure 12: Global Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Form
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Application , 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Application
- Figure 29: North America Market Value Share and BPS Analysis by Extraction, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Extraction, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Extraction
- Figure 32: North America Market Value Share and BPS Analysis by Purity, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Purity, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Purity
- Figure 35: North America Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Form
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Application , 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Application
- Figure 42: Latin America Market Value Share and BPS Analysis by Extraction, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by Extraction, 2026-2036
- Figure 44: Latin America Market Attractiveness Analysis by Extraction
- Figure 45: Latin America Market Value Share and BPS Analysis by Purity, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Purity, 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Purity
- Figure 48: Latin America Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Form
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by Application , 2026-2036
- Figure 54: Western Europe Market Attractiveness Analysis by Application
- Figure 55: Western Europe Market Value Share and BPS Analysis by Extraction, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by Extraction, 2026-2036
- Figure 57: Western Europe Market Attractiveness Analysis by Extraction
- Figure 58: Western Europe Market Value Share and BPS Analysis by Purity, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by Purity, 2026-2036
- Figure 60: Western Europe Market Attractiveness Analysis by Purity
- Figure 61: Western Europe Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Form
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by Application , 2026-2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by Application
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by Extraction, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by Extraction, 2026-2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by Extraction
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by Purity, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by Purity, 2026-2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by Purity
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by Form
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by Application , 2026-2036
- Figure 80: East Asia Market Attractiveness Analysis by Application
- Figure 81: East Asia Market Value Share and BPS Analysis by Extraction, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by Extraction, 2026-2036
- Figure 83: East Asia Market Attractiveness Analysis by Extraction
- Figure 84: East Asia Market Value Share and BPS Analysis by Purity, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by Purity, 2026-2036
- Figure 86: East Asia Market Attractiveness Analysis by Purity
- Figure 87: East Asia Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 89: East Asia Market Attractiveness Analysis by Form
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by Application , 2026-2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by Extraction, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by Extraction, 2026-2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by Extraction
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by Purity, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by Purity, 2026-2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by Purity
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by Form
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by Application , 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by Application , 2026-2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by Extraction, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by Extraction, 2026-2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by Extraction
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by Purity, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by Purity, 2026-2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by Purity
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by Form
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

