Vascular Sealant Market
Vascular Sealant Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Vascular Sealant Market Forecast and Outlook 2026 to 2036
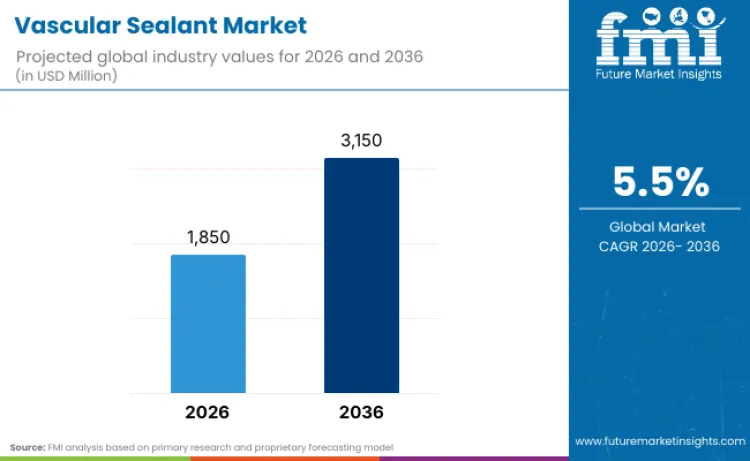
The global vascular sealant market is projected to reach USD 1,850.0 million in 2026 and expand to USD 3,150.0 million by 2036, registering a CAGR of 5.5% over the forecast period. According to the opinion of FMI, market growth is associated with the growing number of vascular, endovascular, and cardiac procedures, as well as the growing importance of intraoperative bleeding control and the reduction of post-procedural complications. The use of fibrin-based, collagen-based, PEG-based synthetic and cyanoacrylate-based sealants is being driven by their ability to complement sutures and staples and enhance procedural efficiency. Improvements in sealant composition, delivery systems, and applicators are making it possible for surgeons to reliably seal.
"Acquiring Vivasure expands Haemonetics' complete range of closure devices with new and clinically differentiated technology to bolster our presence in the large-bore closure market and our impact in fast-growing structural heart and endovascular procedures." said Ken Crowley, Vice President & General Manager, Interventional Technologies at Haemonetics.
Large firms are also consolidating their market share through innovation in sealant technology, formulation, and clinical application. Baxter International is a leading firm in the fibrin-based surgical sealant market for vascular and cardiovascular surgery. Johnson & Johnson (Ethicon) has been exploring adjunctive hemostatic and sealant therapies that complement its overall surgical business. Medtronic is also developing vascular sealing applications for open and minimally invasive procedures. However, B. Braun, CSL Behring, and Grifols are also developing biologic sealants, which show a trend in the industry toward an overall surgical hemostasis and sealing solution.
Market development is further aided by the growing use of vascular sealants in hospitals, ambulatory surgery centers, and specialty cardiac and vascular centers. Open vascular surgery and endovascular or hybrid procedures are both fueling demand, with sealants being employed for anastomotic sealing, graft reinforcement, needle-hole bleeding, soft tissue adjunct sealing, and access site management. Demand from end-users is still largely channeled through hospitals, aided by growing procedural volumes and an increasing adoption of advanced surgical adjuncts. In sum, the market dynamics are shifting towards more diversified product lines of sealants that integrate biological and synthetic chemistries with optimal delivery systems.
Summary of the Vascular Sealant Market
What Is Growth Outlook for Vascular Sealant Market as per Future Market Insights Projection?
Future Market Insights projects the vascular sealant market to expand at a CAGR of 5.5% from 2026 to 2036, increasing from USD 1,850.0 million in 2026 to USD 3,150.0 million by 2036.
FMI Research Approach: Bottom-up revenue estimation mapped across sealant chemistry, form factor, use case, clinical setting, and end-user categories, with procedure-volume anchoring through 2036.
How Do FMI Analysts Perceive Vascular Sealant Market to Evolve?
Future Market Insights analysts note that demand is increasingly being shaped by a shift from conventional hemostatic solutions toward performance-oriented sealing adjuncts embedded into standardized vascular workflows.
FMI Research Approach: Assessed product differentiation through application ease, biocompatibility, degradation predictability, and compatibility across open, endovascular, and hybrid procedures.
Which Country Holds Largest Share in Global Vascular Sealant Market?
Future Market Insights notes that high vascular intervention volumes, mature cardiovascular infrastructure, are supporting the United States and stronger adoption of adjunct technologies aligned with protocol-driven surgery.
FMI Research Approach: Gathered country-level inputs on procedure density, hospital capability, purchasing behavior, and utilization intensity across high-volume vascular centers.
How Large Will Vascular Sealant Market Be by 2036?
The global vascular sealant market is projected to reach USD 3,150.0 million by 2036.
FMI Research Approach: Forecasts were derived using segment-weighted utilization assumptions across key surgical use environments and application frequency across procedure mix.
How are Regulatory Tailwinds Fueling Vascular Sealant Adoption?
According to Future Market Insights analysts, rising scrutiny on safety, biocompatibility, and repeatable clinical performance is accelerating preference for sealing solutions that reduce bleeding and leakage variability.
FMI Research Approach: Incorporated quality expectations, risk-reduction priorities, and standard-of-care alignment into models to refine adoption curves across sealant chemistries and ready-to-use formats.
Vascular Sealant Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 1,850.0 million |
| Market Forecast Value (2036) | USD 3,150.0 million |
| Forecast CAGR (2026-2036) | 5.5% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research.
How Do Surgical Standardization and Outcome-Focused Care Frameworks Drive Demand for Vascular Sealants?
The increasing focus on surgical standardization and outcomes-driven accountable care is driving the adoption of vascular sealants. The reduced acceptance of variability in bleeding control, anastomotic security, and complication rates is accelerating the adoption of complementary sealing approaches that ensure predictable outcomes. This has led to the adoption of sealants as workflow-enabling technologies that facilitate intraoperative efficiency and outcomes, rather than being viewed as complements or add-ons. Commercially available fibrin-based and synthetic sealants are being increasingly positioned for controlled sealing in open and hybrid vascular surgery, which is in line with institutional approaches to optimize operative efficiency and accelerate recovery.
How is the Vascular Sealant Market Segmented?
The vascular sealant market has been segmented based on sealant chemistry, form factor, use case, setting, end user, and geography. By sealant chemistry, the market is divided into fibrin-based sealants, PEG-based synthetic sealants, cyanoacrylates, collagen or gelatin-based sealants, and polysaccharide or other formulations. By form factor, the market includes liquid, patch, spray or foam, and pre-filled applicator kits. Based on use case, the market is categorized into anastomotic sealing, graft sealing, needle-hole bleeding control, adjunct soft-tissue sealing, and access-site adjunct. By setting, the market spans open vascular surgery, endovascular or hybrid procedures, and cardiac surgery. By end user, the market is segmented into hospitals, ambulatory surgery centers (ASCs), and specialty cardiac or vascular centers. Regionally, the market covers the USA, Germany, the UK, France, Japan, China, and Saudi Arabia.
Why Do Fibrin-Based Sealants Remain Dominant?
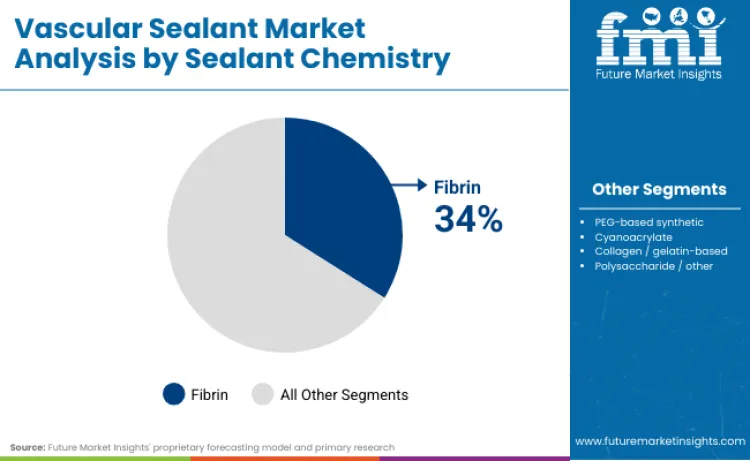
Fibrin-based sealants currently dominate the chemistry category with a 34.0% market share, thanks to their biomimetic approach and high level of clinical acceptance in vascular procedures. These products work by recreating the end-process of coagulation, allowing for the quick achievement of clotting and hemostasis in high-pressure arterial and venous environments. From a clinical perspective, they are often chosen for their predictable performance and versatility, especially in anastomotic reinforcement and graft closure in open vascular surgery. Their widespread use has also helped to establish them as the standard against which new synthetic and hybrid products are measured.
How Do Use-Case and Setting Requirements Shape Sealant Adoption?
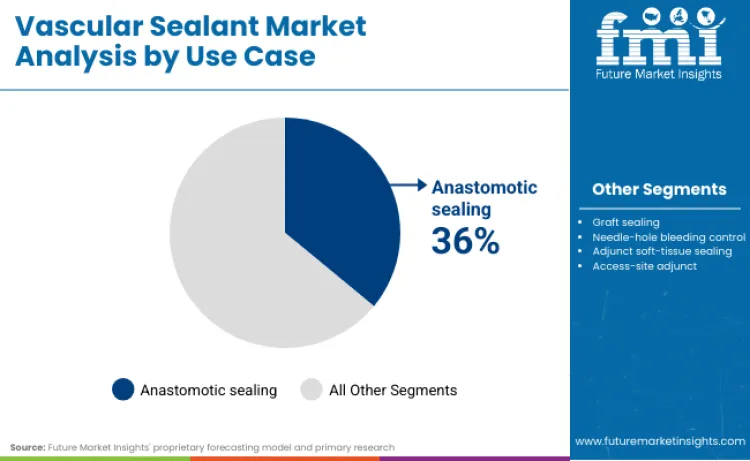
Anastomotic sealing is the largest use-case segment, accounting for 36.0% of the total demand, due to the large demand for leak and bleeding prevention at vascular junctions. Precision sealing at anastomotic sites is a highly required application for reducing postoperative complication rates and re-intervention rates, thus making sealants a crucial adjunct rather than a discretionary product in vascular surgery.
How Are Advanced Sealant Systems Replacing Conventional Hemostasis Methods?
The market for vascular sealants is gradually moving away from the traditional dependence on sutures and mechanical closure alone to more sophisticated sealant systems that offer immediate and localized hemostasis. The traditional approach is gradually being perceived as inadequate in complex vascular procedures, where the friability of tissue and high intraluminal pressures increase the risk of bleeding. Sealants are thus being incorporated directly into the traditional vascular closure strategies rather than being selectively used. This has also increased the expectations of sealant performance in terms of consistency, deliverability, and functionality in different anatomical settings. Sealants that can be easily incorporated into surgical practice without prolonging surgery times are being given preference, thus reemphasizing the importance of sealants as critical surgical tools.
Why Is Surgical Outcome Accountability Reshaping Sealant Selection?
The growing focus on postoperative care, complication prevention, and reducing length of stay is also changing the way sealants are chosen. Vascular procedures are now being evaluated based on parameters such as bleeding-related re-interventions and graft integrity, which is forcing the surgical community to adopt tools that can improve the reliability of these procedures. Sealants that have proven clinical performance and a reliable degradation profile are now being preferred because of these accountability-based models of care. The choice of sealants is no longer based solely on their hemostatic performance but also on their effects on recovery and resource utilization, thereby solidifying the position of sealants in value-based surgical care.
What Structural Shift Will Define Long-Term Market Leadership?
In the period from 2026 to 2036, the long-term leadership position in the vascular sealant market is expected to remain in favor of those suppliers who can support high-volume hospital-based procedures and also meet the demands of hybrid and endovascular procedures.
The vascular sealant market is experiencing a shift towards becoming a standardized part of the surgical infrastructure, rather than a discretionary part. Those suppliers who can integrate clinical efficacy, workflow, and applicability are expected to unlock long-term value as the volume of vascular interventions continues to grow.
How Is the Vascular Sealant Market Evolving Globally?
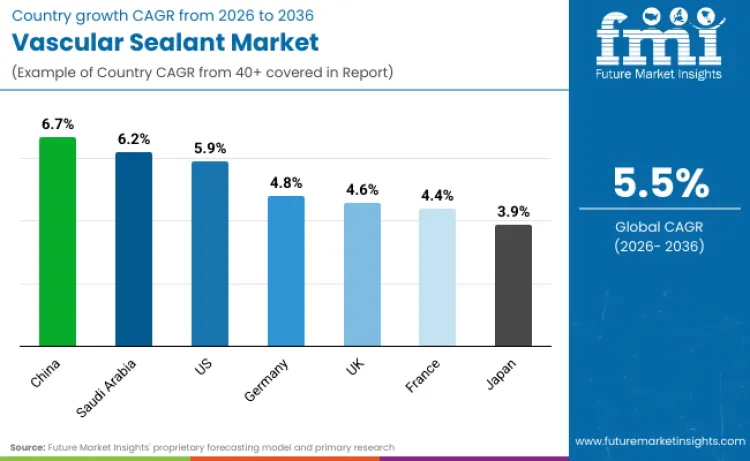
The global market for vascular sealant products is increasing because of the growing optimization of surgical and interventional procedures for faster hemostasis, less blood loss, and efficient procedural delivery. The market for products is being driven by the growing number of cardiovascular, endovascular, and minimally invasive procedures, and the growing interest in optimizing complications and hospital stays associated with procedures. The market for products is being driven by the growing interest in sealing, biocompatibility, ease of use, and compatibility with different surgical work flows and not the number of procedures. China is expected to have a CAGR of 6.7%, Saudi Arabia 6.2%, the USA 5.9%, Germany 4.8%, the UK 4.6%, France 4.4%, and Japan 3.9% over the 2026-2036 period. The development of the market is firmly grounded in clinical outcomes, work flow integration, and predictive outcomes and not rapid unit growth.
| Country | CAGR (2026-2036) |
|---|---|
| USA | 5.9% |
| Germany | 4.8% |
| UK | 4.6% |
| France | 4.4% |
| Japan | 3.9% |
| China | 6.7% |
| Saudi Arabia | 6.2% |
Source: FMI historical analysis and forecast data.
How is expanding surgical capacity in China driving sealant adoption?
In the Chinese market, the demand for vascular sealants is being fueled by the fast development of surgical infrastructure and the adoption of minimally invasive and catheter-based procedures in large tertiary hospitals. As a result of the increasing number of procedures and the need for efficiency, vascular sealants are being increasingly adopted in order to achieve faster vessel closure and minimize post-procedure complications. The increasing focus on the development of Chinese clinical capabilities is also fueling the adoption of vascular sealants in public healthcare settings. As a result of this focus, the Chinese market for vascular sealants is expected to register a 6.7% CAGR during the forecast period.
Looking ahead into the next five years, there will be an increased adoption of vascular sealants in secondary hospitals due to the increasing standardization of interventional procedures and familiarity with the technology. There will be a growing preference for vascular sealants that can achieve rapid polymerization, predictable performance, and are appropriate for high-volume procedural settings.
Is healthcare modernization in Saudi Arabia accelerating advanced hemostasis solutions?
In the case of Saudi Arabia, the market is being driven by the modernization of the healthcare system and increased investment in highly advanced surgical and intervention capabilities. Vascular sealants are also being increasingly used to support complex cardiovascular procedures, reduce intraoperative bleeding, and improve post-operative recovery outcomes. The expansion of specialty hospitals and centers of excellence is also driving demand for highly advanced hemostatic solutions. In line with this trend, the Saudi Arabia vascular sealant market is expected to register a 6.2% CAGR during the study period.
Going forward, there is expected to be increased usage in high acuity care environments, driven by the adoption of international surgical standards and ongoing investment in clinician education and advanced medical technology.
Is precision-oriented surgical practice in Germany sustaining market stability?
In Germany, the vascular sealant market is being sustained by a precision-oriented surgical culture where standardised technique, tight documentation, and outcomes benchmarking keep adoption steady rather than cyclical. Hospitals and surgical centres increasingly treat haemostasis as a controllable quality variable, using sealants to reduce intraoperative bleeding, support complex cardiac and vascular reconstructions, and minimise downstream complications that extend length of stay. Demand is reinforced by structured procurement and reimbursement environments that favour clinically validated products with repeatable performance across routine and high-complexity procedures. In line with this stability dynamic, the Germany vascular sealant market is expected to register a 4.8% CAGR during the study period.
Going forward, market continuity is expected to be maintained through protocol-led expansion in high-acuity operating rooms and interventional suites, supported by surgeon education, simulation-based skills training, and wider adoption of international surgical standards. As German providers intensify focus on complication avoidance and faster recovery, vascular sealants will remain reliability tools embedded in defined care pathways.
How is value-based care in the United Kingdom influencing sealant utilization?
In the United Kingdom, the adoption of vascular sealants is being driven by the growing need for value-based healthcare, procedural optimization, and the minimization of post-operative complications in the public and private healthcare settings. The use of vascular sealants is being considered for their potential to reduce procedure time and enable rapid patient recovery in cardiovascular and minimally invasive procedures. In this setting, the UK vascular sealant market is expected to register a 4.6% CAGR during the forecast period.
Future adoption is expected to be driven by strategic adoption in complex and high-risk procedures, with broader adoption tempered by procurement and budgetary factors.
Is controlled adoption in France supporting sustainable growth?
The development of the market in France is also being influenced by the controlled launch of vascular sealants. Hospitals are embracing technologies that have already been validated to offer clinical benefits, are cost-effective, and are compatible with current practices before mass adoption. With the continued validation of the clinical effectiveness of vascular sealants in the prevention of complications from bleeding, their adoption is gradually gaining traction. Thus, the vascular sealant market in France is expected to register a CAGR of 4.4% during the forecast period.
In the next few years, incremental growth is expected as pilot projects and acceptance gain momentum in cardiovascular and general surgery.
How is clinical caution in Japan shaping market progression?
In the Japanese market, the demand is being driven by a highly regulated healthcare setting where the aspects of clinical reliability, safety, and standardization are of utmost importance. The demand for vascular sealants is being driven in the most specialized surgical centers, with a cautious adoption that is in line with the conservative adoption of technology. Although the adoption is being done at a relatively slower pace compared to other geographies, the integration with the existing workflows is helping to sustain the demand. In this setting, the Japanese market for vascular sealants is expected to register a CAGR of 3.9% during the forecast period.
The future demand is expected to be driven by improvements in the formulation and usability of these products, which will help them align with the efficiency-oriented surgical practices in Japan.
How is the USA Market Advancing Through Clinical and Economic Maturity?
In the United States, market development is being fueled by the need to improve surgical efficiency, reduce the rate of complications, and manage procedural costs in high-volume healthcare environments. Vascular sealants are being increasingly employed to enable rapid hemostasis, decrease procedural times, and improve post-procedure outcomes in cardiovascular and interventional procedures. Healthcare environments in the United States are favoring products that have demonstrated stable performance and compatibility with existing surgical practices. In this mature market and environment, the USA vascular sealant market is expected to grow at a 5.9% CAGR during the forecast period.
Market development over the next five years is expected to be fueled by the adoption of expanded indications, incremental innovation, and growing usage in minimally invasive and catheter-based procedures.
Who are the Major Players Active in the Vascular Sealant Market?
The competitive environment in the vascular sealant market is fueled by the rising importance of ease of use, hemostasis efficacy, and compatibility with open and minimally invasive vascular procedures. The rising number of cardiovascular and peripheral vascular procedures, as well as the need to lower procedural time and complications is fueling the market dynamics. The different types of Sealant Chemistry such as fibrin, PEG-based synthetic, cyanoacrylate, and collagen/gelatin-based vascular sealants are fueling the market demand.
The leading companies are enhancing their market presence with diversified product offerings across various Form Factors, such as liquid, patch, spray/foam, and pre-filled applicator kits. Baxter International has a strong market presence with fibrin-based sealants that are mainly used in open vascular surgery environments. Johnson & Johnson (Ethicon) continues to enhance its market presence across both liquid and patch-type sealants, which target anastomotic sealing and graft sealing. Medtronic has utilized its cardiovascular device portfolio to support the adoption of sealants in open and endovascular or hybrid procedures.
Concurrently, companies such as B. Braun, CSL Behring, and Grifols are also strengthening their position in the market with biologic and plasma-derived sealants. In the USA, Germany, the UK, France, China, Japan, and Saudi Arabia, competitive differentiation is being fueled by adaptability in use cases, clinical versatility, and a strong penetration of end-users in the hospital setting.
Key Industry Developments:
- On January 9, 2026, Haemonetics Corporation, a global medical technology company focusedon delivering innovative solutions designed to improve patient outcomes, announced the acquisition of Vivasure Medical Limited, a Galway, Ireland-based company pioneering next-generation technology for percutaneous vessel closure.
- In June 2024, Advanced Medical Solutions Group plc, a world-leading specialist in tissue-healing technologies, announced its announcement of the proposed acquisition of Peters Surgical in March 2024, it has now received regulatory approvalto complete the acquisition of Peters Surgical.
Key Players in the Vascular Sealant Market
- Baxter International
- Johnson & Johnson (Ethicon)
- Medtronic
- B. Braun
- CSL Behring
- Grifols
- Integra LifeSciences
- Artivion (CryoLife)
- Smith+Nephew
- Teleflex
Market Definition
The market for vascular sealants is divided into segments as the global market demand for surgical sealant products used for hemostasis and tissue sealing in vascular surgery. The market value is expressed in USD million and forecasted from 2026 to 2036.The vascular sealants market comprises the demand for sealant products used in vascular surgery for anastomotic sealing, graft sealing, bleeding control, and access site adjunct use. The products are used in open vascular surgery, endovascular or hybrid procedures, and cardiac surgery.
The vascular sealants market is segmented by sealant chemistry, including fibrin, PEG-based synthetic, cyanoacrylate, collagen/gelatin-based, and polysaccharide/other sealants. The form factors, including liquid, patch, spray/foam, and pre-filled applicator kits. The use cases included in the analysis are anastomotic sealing, graft sealing, needle-hole bleeding, adjunct soft-tissue sealing, and access site adjunct. The end users included in the analysis are Hospitals, ASCs, and Specialty cardiac/vascular centers.
Conventional sutures, staples, clips, and mechanical closure devices that are not intended for hemostasis through the use of a sealant. Topical wound care adhesives, non-vascular surgical sealants, diagnostic hemostasis devices, and therapeutic biologics that are not intended for use in vascular sealing are excluded from the vascular sealant market.
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD 1,850.0 Million |
| Sealant Chemistry | Fibrin, PEG-based synthetic, Cyanoacrylate, Collagen / gelatin-based, Polysaccharide / other |
| Form Factor | Liquid, Patch, Spray / foam, Pre-filled applicator kits |
| Use Case | Anastomotic sealing, Graft sealing, Needle-hole bleeding control, Adjunct soft-tissue sealing, Access-site adjunct |
| Setting | Open vascular surgery, Endovascular / hybrid, Cardiac surgery |
| End User | Hospitals, ASCs, Specialty cardiac/vascular centers |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Countries Covered | USA, Germany, UK, France, China, Japan, Saudi Arabia, and 40+ countries |
| Key Companies Profiled | Baxter International; Johnson & Johnson (Ethicon); Medtronic; B. Braun; CSL Behring, Grifols, Integra LifeSciences, Artivion (CryoLife), Smith+Nephew and Teleflex |
| Additional Attributes | Dollar sales by sealant chemistry and use case; adoption trends across settings; impact of form-factor innovation and hospital procurement dynamics |
Source: FMI historical analysis and forecast data.
Vascular Sealant Market by Segment
Sealant Chemistry:
- Fibrin
- PEG-based synthetic
- Cyanoacrylate
- Collagen / gelatin-based
- Polysaccharide / other
Form Factor:
- Liquid
- Patch
- Spray / foam
- Pre-filled applicator kits
Use Case:
- Anastomotic sealing
- Graft sealing
- Needle-hole bleeding control
- Adjunct soft-tissue sealing
- Access-site adjunct
Setting:
- Open vascular surgery
- Endovascular / hybrid
- Cardiac surgery
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- Thailand
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- Netherlands
- Rest of Europe
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Colombia
- Argentina
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- Society for Vascular Surgery. (2024). Clinical Practice Guidelines for the Use of Vascular Sealants in Open and Endovascular Procedures. SVS.
- European Society for Vascular Surgery. (2023). Guidelines on Hemostasis and Sealant Use in Vascular and Cardiac Surgery. ESVS.
- USA Food and Drug Administration. (2023). Technical and Clinical Considerations for Fibrin and Synthetic Surgical Sealants in Vascular Applications. FDA.
- Patel, R. M., et al. (2023). Comparative performance of fibrin, PEG-based, and cyanoacrylate sealants in vascular anastomotic sealing. Journal of Vascular Surgery, 77, 1684-1696.
- Hoffmann, L. J., et al. (2024). Safety, efficacy, and workflow impact of topical vascular sealants across open and hybrid vascular procedures. Annals of Vascular Surgery, 94, 112-124.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Sealant Chemistry
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Sealant Chemistry , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Sealant Chemistry , 2026 to 2036
- Fibrin
- PEG-based synthetic
- Cyanoacrylate
- Collagen / gelatin-based
- Polysaccharide / other
- Fibrin
- Y to o to Y Growth Trend Analysis By Sealant Chemistry , 2021 to 2025
- Absolute $ Opportunity Analysis By Sealant Chemistry , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Form Factor
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Form Factor, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Form Factor, 2026 to 2036
- Liquid
- Patch
- Spray / foam
- Pre-filled applicator kits
- Liquid
- Y to o to Y Growth Trend Analysis By Form Factor, 2021 to 2025
- Absolute $ Opportunity Analysis By Form Factor, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Use Case
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Use Case, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Use Case, 2026 to 2036
- Anastomotic sealing
- Graft sealing
- Needle-hole bleeding control
- Adjunct soft-tissue sealing
- Access-site adjunct
- Anastomotic sealing
- Y to o to Y Growth Trend Analysis By Use Case, 2021 to 2025
- Absolute $ Opportunity Analysis By Use Case, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Setting
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Setting, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Setting, 2026 to 2036
- Open vascular surgery
- Endovascular / hybrid
- Cardiac surgery
- Open vascular surgery
- Y to o to Y Growth Trend Analysis By Setting, 2021 to 2025
- Absolute $ Opportunity Analysis By Setting, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- ASCs
- Specialty cardiac/vascular centers
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Sealant Chemistry
- By Form Factor
- By Use Case
- By Setting
- By End User
- Competition Analysis
- Competition Deep Dive
- Baxter International
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Johnson & Johnson (Ethicon)
- Medtronic
- B. Braun
- CSL Behring
- Grifols
- Integra LifeSciences
- Artivion (CryoLife)
- Smith+Nephew
- Teleflex
- Baxter International
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Sealant Chemistry , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Form Factor, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Use Case, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Setting, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Sealant Chemistry , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Form Factor, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Use Case, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Setting, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Sealant Chemistry , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Form Factor, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Use Case, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Setting, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Sealant Chemistry , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Form Factor, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Use Case, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Setting, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Sealant Chemistry , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Form Factor, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Use Case, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Setting, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Sealant Chemistry , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Form Factor, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Use Case, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Setting, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Sealant Chemistry , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Form Factor, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Use Case, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Setting, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Sealant Chemistry , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Form Factor, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Use Case, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Setting, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Sealant Chemistry , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Sealant Chemistry , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Sealant Chemistry
- Figure 6: Global Market Value Share and BPS Analysis by Form Factor, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Form Factor, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Form Factor
- Figure 9: Global Market Value Share and BPS Analysis by Use Case, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Use Case, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Use Case
- Figure 12: Global Market Value Share and BPS Analysis by Setting, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Setting, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Setting
- Figure 15: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by End User
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Sealant Chemistry , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Sealant Chemistry , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Sealant Chemistry
- Figure 32: North America Market Value Share and BPS Analysis by Form Factor, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Form Factor, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Form Factor
- Figure 35: North America Market Value Share and BPS Analysis by Use Case, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Use Case, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Use Case
- Figure 38: North America Market Value Share and BPS Analysis by Setting, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Setting, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Setting
- Figure 41: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by End User
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Sealant Chemistry , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Sealant Chemistry , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Sealant Chemistry
- Figure 48: Latin America Market Value Share and BPS Analysis by Form Factor, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Form Factor, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Form Factor
- Figure 51: Latin America Market Value Share and BPS Analysis by Use Case, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Use Case, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Use Case
- Figure 54: Latin America Market Value Share and BPS Analysis by Setting, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Setting, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Setting
- Figure 57: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by End User
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Sealant Chemistry , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Sealant Chemistry , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Sealant Chemistry
- Figure 64: Western Europe Market Value Share and BPS Analysis by Form Factor, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Form Factor, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Form Factor
- Figure 67: Western Europe Market Value Share and BPS Analysis by Use Case, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Use Case, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Use Case
- Figure 70: Western Europe Market Value Share and BPS Analysis by Setting, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Setting, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Setting
- Figure 73: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by End User
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Sealant Chemistry , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Sealant Chemistry , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Sealant Chemistry
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Form Factor, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Form Factor, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Form Factor
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Use Case, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Use Case, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Use Case
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Setting, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Setting, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Setting
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by End User
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Sealant Chemistry , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Sealant Chemistry , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Sealant Chemistry
- Figure 96: East Asia Market Value Share and BPS Analysis by Form Factor, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Form Factor, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Form Factor
- Figure 99: East Asia Market Value Share and BPS Analysis by Use Case, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Use Case, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Use Case
- Figure 102: East Asia Market Value Share and BPS Analysis by Setting, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Setting, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Setting
- Figure 105: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by End User
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Sealant Chemistry , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Sealant Chemistry , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Sealant Chemistry
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Form Factor, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Form Factor, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Form Factor
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Use Case, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Use Case, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Use Case
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Setting, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Setting, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Setting
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Sealant Chemistry , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Sealant Chemistry , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Sealant Chemistry
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Form Factor, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Form Factor, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Form Factor
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Use Case, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Use Case, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Use Case
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Setting, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Setting, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Setting
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

