Ventricular Catheters Market
Ventricular Catheters Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Ventricular Catheters Market Size, Market Forecast and Outlook By FMI
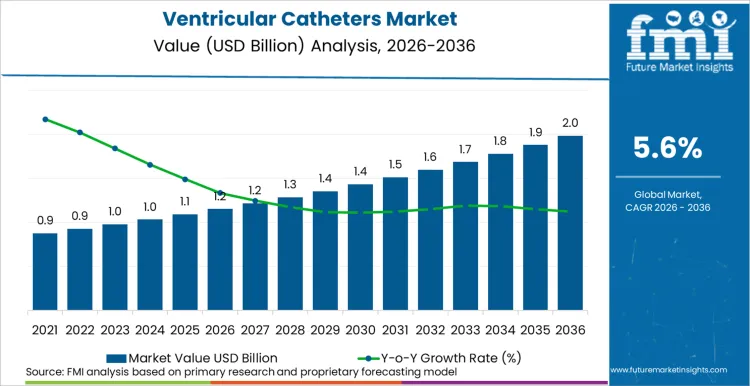
As per Future Market Insights, the ventricular catheters market is expected to grow from USD 1,150.0 million in 2026 to USD 1,980.0 million by 2036. This reflects a growth rate of 5.6% during the forecast period. This growth is supported by the consistent demand for emergency and neurocritical care procedures for hydrocephalus, traumatic brain injuries, intracerebral hemorrhage/intraventricular hemorrhage, and subarachnoid hemorrhage. FMI analysis implies that in the acute phase, external ventricular drain systems serve as the basis for all emergency procedures. Ventriculoperitoneal shunts serve as the basis for long-term management.
Quick Stats for Ventricular Catheters Market
- Market Snapshot
- The ventricular catheters market is valued at USD 1,150.0 million in 2026.
- The ventricular catheters market is projected to reach USD 1,980.0 million by 2036.
- The market is expected to grow at a CAGR of 5.6% during 2026 to 2036.
- Demand and Growth Drivers
- Demand is driven by sustained neurocritical care procedures for hydrocephalus, traumatic brain injury, and intracranial hemorrhage.
- External ventricular drainage systems are the primary devices used in emergency intracranial pressure management.
- Infection prevention requirements are accelerating adoption of antibiotic-impregnated and antimicrobial-coated catheters.
- Product and Segment View
- External ventricular drain (EVD) catheters hold approximately 42.0% share due to their use in acute care settings.
- Hydrocephalus accounts for about 46.0% of total ventricular catheter usage by indication.
- Hospitals remain the dominant end users due to concentration of neuro-ICU and neurosurgical procedures.
- Geography and Competitive Outlook
- The United States leads the market supported by advanced neurocritical care infrastructure and high procedure volumes.
- India is projected to be the fastest-growing market with a CAGR of 6.4% during 2026 to 2036.
- Competition is driven by coating technologies, infection control performance, and ICU-focused catheter usability among key players such as Medtronic and Integra Life Sciences.
FMI cites infection mitigation strategies, including catheter performance engineering, as the significant structural change during the period leading to 2036. Antibiotic or antimicrobial coated catheters continue to take priority in the neuro-ICU settings where infection danger from ventriculostomy catheters is most prominent. At the same time, use case dependency remains, with emphasis on compatibility with ICP requirements, ease of bedside management, and catheter drainage characteristics. “This regulatory milestone highlights the broader impact of our ReFlow innovation beyond hydrocephalus management, we continue to hear from neurosurgeons, nurses, patients, and families that ReFlow’s simple, seconds-long manual flush helps prevent blockages improving outcomes and providing peace of mind. Expanding our ReFlow portfolio reinforces our commitment to advancing neurocritical care and patient well-being.” said Elsa Chi Abruzzo, CEO and President of Anuncia Medical.
FMI also underlines the fact that competitive advantage is increasingly determined by how effectively manufacturers can convert catheter innovation into defensible clinical performance in particular by strategies related to coating, system compatibility, and ICU-centered usability. Though hospitals are the major end user, ambulatory and specialty neuro centers continue to have an impact on procurement patterns as far as elective and follow-up pathways go. FMI forecasts capability-driven strategies to further intensify differentiation and facilitate continued adoption through 2036 as neurocritical care teams prioritize outcome stability, infection reduction, and repeatable drainage performance.
Summary of Ventricular Catheters Market
What Is Growth Outlook for Ventricular Catheters Market as per Future Market Insights Projection?
The ventricular catheters market to expand at a CAGR of 5.6% from 2026 to 2036, increasing from USD 1,150.0 million in 2026 to USD 1,980.0 million by 2036. Growth is being supported by sustained neurosurgical demand for cerebrospinal fluid diversion and drainage, where hydrocephalus management, neurocritical care volumes, and hospital-based procedural concentration continue to drive recurring catheter utilization.
FMI Research Approach: Historical market size, neurosurgical procedure volumes, product-type mix across EVD and shunt catheters, infection-risk management practices, and country-level adoption trends were used to model demand scenarios through 2036.
How Do FMI Analysts Perceive Ventricular Catheters Market to Evolve?
Therefore, infection prevention and neurocritical settings device-performance consistency are increasingly shaping demand. Adoption is being steered toward catheter options that support predictable drainage performance and reduced complication risk, with greater procurement emphasis on coated variants and standardized ICU protocols that emphasize safety and continuity of care.
FMI Research Approach: Evaluation focused on neurocritical care workflow standards, coated-catheter adoption curves, infection-control practices, and procurement decisions across hospitals and high-acuity centers.
Which Country Holds Largest Share in Global Ventricular Catheters Market?
The market share of the global ventricular catheters market is held by the USA, which is due to the high level of neurocritical care facilities, established neurosurgical infrastructure, and the continued utilization of external ventricular drainage systems as part of the ICU management pathway.
FMI Research Approach: Country-level inputs included neurosurgery and ICU bed capacity, hydrocephalus and TBI care intensity, device adoption patterns, and hospital procurement concentration.
How Large Will Ventricular Catheters Market Be by 2036?
The global ventricular catheters market is projected to reach USD 1,980.0 million by 2036.
FMI Research Approach: Market size was calculated by modeling demand across the leading catheter type and dominant indication using weighted utilization factors by end user.
How are Infection-Control Expectations Fueling Ventricular Catheter Adoption?
Infection-control expectations and growing attention to ventriculitis risk are accelerating demand for ventricular catheters that support safer neurocritical drainage workflows. Adoption is being strengthened where coated options and standardized handling protocols are being used to reduce device-associated infection risk and support consistent outcomes in high-acuity care.
FMI Research Approach: Incorporated infection-prevention practices, coated-device penetration trends, ICU protocol standardization, and neurosurgical caseload dynamics into forecasting models to refine demand estimates.
Ventricular Catheters Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 1,150.0 million |
| Industry Value (2036) | USD 1,980.0 million |
| CAGR (2026 to 2036) | 5.6% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Why is the Demand for Ventricular Catheters Growing?
Demand for ventricular catheters is set to increase as hospital-based neurocritical care services are likely to endure at high levels. In addition, demand for CSF drainage and diversion is likely to endure at high levels. All this demand is being anchored by EVD ventricular catheters and VP shunt catheters. The demand for these devices is a result of preference for standard coating products. In other cases, antibiotic-impregnated products are used where there is prioritization of risk mitigation. In terms of indications used to generate this demand, hydrocephalus indications appear to dominate. Other indications used are those related to traumatic brain injury and intracranial hemorrhage. In terms of demand from end users, hospitals are likely to dominate demand.
How is the Ventricular Catheters Market Segmented?
The ventricular catheters market is segmented by product type, coating, indication, and end user. By product type, the market includes EVD ventricular catheters, VP shunt ventricular catheters, with ICP monitoring, and VA / other shunt catheters. Based on coating, products are segmented into standard, antibiotic-impregnated, antimicrobial (silver), and heparin / anti-thrombogenic variants. By indication, demand is categorized into hydrocephalus, traumatic brain injury, ICH / IVH, SAH, and CNS infection / other. By end user, the market is segmented into hospitals, ASCs, and specialty neuro centers.
How Do EVD Ventricular Catheters Secure Market Leadership?
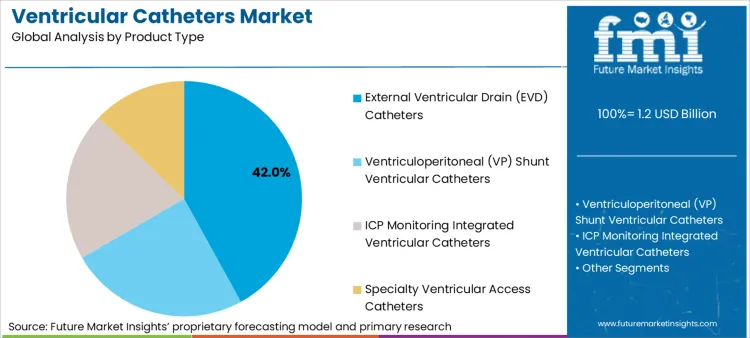
According to Future Market Insights, EVD ventricular catheters are leading in the global market for ventricular catheters due to the inevitable requirement for these devices in the management of acute intracranial pressure conditions. EVD catheters hold an approximate share in the market based on the total demand for the products, estimated to be around 42.0%. They are used in the management of situations where emergency care for cerebrospinal fluid diversion is required.
The EVD catheter is mainly used by high-acuity hospital settings that have neurosurgical and neuro-ICU capabilities integrated into their infrastructure and operations. Therefore, this product is a favorite choice for procedures owing to its use and preference by clinicians for standardized use, wherein acute neurological events are reliant on immediate CSF drainage and pressure normalization. The EVD ventricular catheter is a primary product anchor for this market space.
How Does Hydrocephalus Drive Ventricular Catheter Demand?
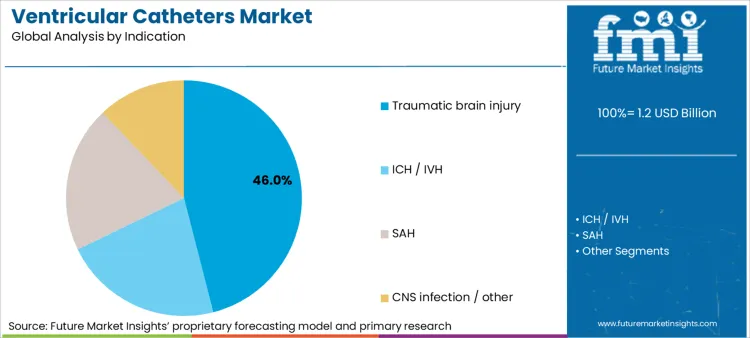
According to Future Market Insights, the future of healthcare will see hydrocephalus topping the pack as the leading indication, contributing 46.0% to total ventricular catheter usage. This is because vesicular fluid diversion might need to be done through shunting catheters to avert death from elevated intracranial pressure. This indicates that the healthcare system usage levels will include children and adults.
Treatment options for hydrocephalus sometimes also involve a variety of treatments that the patient needs to undergo over the course of a lifetime. In this respect, the selection criterion is affected by the need to ensure the catheters remain patent over a rather extended period and the risks of infection that need to be mitigated. The chronic nature of hydrocephalus, coupled with the need for catheter-based treatments, ensures that the condition remains the driving force for the demand for ventricular catheter products.
How Are Infection Control Protocols Influencing Catheter Selection?
Infection control is considered an important factor in the selection of the ventricular catheters, particularly for neurocritical care settings, because infections with catheters could affect patient outcomes. Most coating mechanisms reduce bacterial colonization, and hospital protocols usually emphasize these mechanisms, therefore clinically tested and antibiotic-impregnated/antimicrobial catheters are much important.
Purchase decisions are being aligned with institutional infection surveillance metrics and quality standards. Since infection prevention remains the top priority of neurosurgical care standards, catheter designs that help meet these ends are being prioritized.
Why Is Neurocritical Care Concentration Reshaping Demand Patterns?
This centralization of complex neurological care in specially equipped medical facilities has a significant impact on the demand for ventricular catheters. Cases involving traumatic brain injuries, hemorrhagic patients, and care of patients with hydrocephalus present acutely often receive care in specially equipped medical facilities that are used for neuro-ICU patients, which are facilities that use the majority of the ventricular catheters used.
Although these care pathways are being progressively more advanced, the overall demands are being biased towards hospital-based settings, where the hospital segment remains a significant end user category. This is impacting the overall behavior of suppliers.
How Are Coating and Material Refinements Driving Market Evolution?
Market evolution of ventricular catheters is largely being dictated by step-wise improvements to coating technology as well as materials used to make the catheters themselves. These advancements are mainly focused on securing reliable fluid flow, reducing thrombogenicity, as well as preventing infections while sticking to traditional catheter designs. Heparin/anti-thrombogenic as well as antimicrobial coatings are being used.
These developments focus more on reliability/portfolio risk reduction than disruptive innovation, which is a reflection of the critical nature of the performance characteristics for a ventricular catheter. With the ongoing evolution in neurocritical care devices, material and coating optimisation is key to differentiation and advancement in the future.
How Will Ventricular Catheters Market Expansion Unfold Across Key Global Regions?
Future Market Insights identifies the United States (CAGR ~5.2%), Germany (~4.6%), the United Kingdom (~4.3%), France (~4.1%), Japan (~3.7%), China (~6.0%), and India (~6.4%) as the core demand drivers shaping ventricular catheter utilization globally. The USA drives demand through high neuro procedure volumes, developed hydrocephalus treatment paradigms, and high hospital penetration of external ventricular drainage devices. Germany, the United Kingdom, and France underpin European demand through established neurocritical care pathways, consistent treatment volumes for traumatic brain injury and hemorrhagic stroke, and disciplined adoption of catheter upgrades prioritized for infection-risk mitigation.
Japan’s demand is also being supported by demographic dynamics, consistent neuro availability, and ongoing high control neurocritical care inpatient algorithms. China and India are driving new demand via neurocritical care developments and the ability of their tertiary hospitals to increase their management capabilities for hydrocephalus, traumatic brain injury, and intracranial hemorrhage. In summarizing the global demand drivers, FMI concludes that these countries are also prioritizing neurocritical care developments with reductions in infection risk through catheter upgrades, resulting in a structurally strong demand profile for ventricular catheters through 2036.
Ventricular Catheters Market Regional CAGR (2026 to 2036)
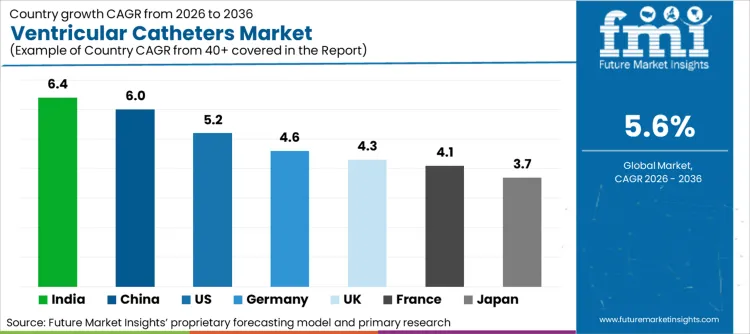
| Country | CAGR (2026 to 2036) |
|---|---|
| United States | 5.2% |
| Germany | 4.6% |
| United Kingdom | 4.3% |
| France | 4.1% |
| Japan | 3.7% |
| China | 6.0% |
| India | 6.4% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research.
Is Neurocritical Care Standardization Redefining Ventricular Catheter Demand in the United States?
The overall US ventricular catheters market is projected to expand at a 5.2% CAGR. Growth was attributed to existing mature neurocritical care pathways and increased use of external ventricular drainage for management of hydrocephalus and acute management of intracranial pressure. Moreover, increased demand in the US market was fueled by high volumes within tertiary neurocenters and trauma facilities where there is a critical need for acute cerebrospinal fluid diversion and critical management of intracranial pressure within neurologic emergencies. According to Future Market Insights, apart from neurocritical care pathways, overall market demand for catheters is currently fueled by purchasing decisions that prioritize infection prevention guidelines, where antibiotic-impregnated and antimicrobial catheters are increasingly being adopted along with standard catheters.
Does Germany’s Evidence-Led Neurocare Sustain High-Value Catheter Adoption?
German ventricular catheter market is set to witness a 4.6% CAGR, underpinned by an evidence-based neurocritical care model and an orderly process of technology adoption in the country’s hospitals. Demand for the devices is being driven by the consistent application of ventricular drainage and shunting for hydrocephalus and hemorrhage-associated management of elevated intracranial pressure. FMI research experts comment that currently, the selection of catheters is being driven by the requirements of quality assurance and infection risks, and this is resulting in the consistent adoption of high-quality catheters.
How Is the United Kingdom’s Neurocritical Workflow Discipline Influencing Market Growth?
UK ventricular catheters market will experience a growth rate of 4.3%. It is informed by the ongoing structure of neurocritical care service delivery, where ventricular catheters feature as a mainstay in the management of trauma-related brain injury and hemorrhage. It is noteworthy that the need for ventricular catheter use continues to be strengthened by the push towards standardized hospital protocols, which aim to reduce the risk of complications while driving consistency within high-intensity environments. As highlighted by analysts at Future Market Insights, procurement disciplines, alongside infection control objectives, continue to influence the incorporation of coated catheter use.
Is France’s Controlled Adoption Sustaining Steady Ventricular Catheter Demand?
France is expected to expand at a 4.1% CAGR for its market consisting of ventricular catheters, driven by carefully managed adoption dynamics along structured hospital-based neurocare procedural routes. The market is being driven by continued requirements for CSF diversion and ICP management for hydrocephalic and acute neurotraumatic patient groups. Future Market Insights analysts have commented that the market is being driven by a number of factors related to infection control and OR efficiency, thereby driving upgrades to coated catheter products at a steady rate.
Does Japan’s Aging Population Influence Long-Term Ventricular Catheter Utilization?
Japan’s ventricular catheter demand is expected to expand at a 3.7% CAGR, supported by an aging population and sustained hospital-led management of neurovascular events and chronic neurological conditions where CSF diversion or intracranial pressure monitoring may be required. Utilization is being reinforced by Japan’s high clinical rigor, structured inpatient neurocritical care pathways, and preference for controlled, protocol-driven decision-making. FMI analysts note that adoption is being shaped by reliability expectations and conservative technology validation, resulting in stable progression rather than rapid scaling. Growth is expected to remain supported by steady neurosurgical capacity and continued emphasis on complication prevention.
How Is China’s Expansion of Neurocritical Infrastructure Accelerating Demand?
The china ventricular catheters market is projected to register a high CAGR of 6.0%, owing to increase in capacity expansion within tertiary care facilities. Additionally, such increasing capacities of neurocritical care services have also acted as a major contributing factor towards the high demand being witnessed for such services. This increasing demand is attributed to a high level of management of services for hydrocephalic patients, and those affected by hemorrhages. This high-level demand is anticipated to be structurally upheld during 2036 and beyond, with more emphasis being placed on care standards and the delivery of such care services. More so, infection prevention measures are having a high impact on antibiotic-coated catheters.
Why Is India Emerging as a High-Growth Ventricular Catheters Market?
Future Market Insights has identified the Indian healthcare market as an area of strong growth, reaching an estimated 6.4% CAGR. This is due to the expansion of neurocritical care services, an increasing trauma burden, and an improving capability within hospitals to address hydrocephalus and intracranial pressures. This trend continues to be bolstered as specialized and tertiary care facilities expand their neurosurgical and neurocritical care capabilities and rely upon EVD and shunt therapy. Future Market Insights has cited the trend toward reducing the infection risk as an increasingly important contributor to purchasing, which is bolstering the move toward coated catheter models. This trend will continue to have operational support through the course of the forecast period as neurocare services extend beyond the higher-tier facilities.
What Strategic Moves Are Defining Competitive Landscape?
The competition in the ventricular catheters market is now characterised by infection risk mitigation reliability in long-term cerebrospinal fluid management and consistency with neurocritical care practices, rather than just availability of catheters. The manufacturers in this space are now focused on ensuring the integrity of materials, consistency in flow, and coating technology to help mitigate issues commonly associated with catheters in neurocritical care cases, especially in hydrocephalus and traumatic brain injury management.
The extension of the standard coating differentiation strategy to encompass antibiotic-impregnated, antimicrobial (silver), and heparin/anti-thrombogenic coatings to meet hospital-based infection control initiatives. Medtronics and Integra LifeSciences, among other players, have strengthened their portfolios as they relate to EVD ventricular catheters and VP shunt ventricular catheters for acute and chronic cerebrospinal diversion, and the business strategy of these firms is the extension of coating-based differentiation to include antibiotic-impregnated/ antimicrobial (silver coated) and heparin/anti-thrombogenic products specifically to address the hospital-acquired infection control opportunities driven by the hospital. Firms such as Medtronic and Integra LifeSciences are further differentiating their products around EVD ventricular catheters and VP Shunt Ventricular Catheters that accommodate both acute and chronic cerebrospinal fluid diversion opportunities.
Recent Developments:
- In August 2025, Integra Life Sciences, a global medical device manufacturing company announced FDA approval for The Codman Libertis EVD Catheter with Bactiseal and Endexo Technology for external cerebrospinal fluid (CSF) drainage.
- In August 2025, Rhovica Neuroimaging AG has raised CHF 2.3 million (EUR 2.5 million) in a funding round led by M2care, with participation from Kickfund and Valuemaker. The investment will accelerate development of SoNav™, a bedside navigation system designed to improve accuracy and safety of extraventricular drainage (EVD) catheter placement in emergency neurosurgery.
- In March 2025, Anuncia Medical, Inc., a company in CSF management and neurocritical care, has received Device Designation from the FDA for its ReFlow EVD, an innovative solution for external ventricular drains (EVDs) used to manage brain swelling and elevated intracranial pressure.
Key Players in Ventricular Catheters Market
- Medtronic
- Integra LifeSciences
- Johnson & Johnson (Codman Neuro)
- B. Braun
- Sophysa
- Natus Medical
- Spiegelberg
- Neuromedex GmbH
- RAUMEDIC AG
- Vygon
- Teleflex
- Mizuho Medical
Market Definition
The ventricular catheters market refers to the global industry involved in the development, manufacture, and clinical use of catheter systems designed for cerebrospinal fluid drainage, monitoring, and diversion within the ventricular system of the brain. These devices play a critical role in the management of elevated intracranial pressure and neurological conditions requiring controlled CSF flow. The market size is measured in USD million and analyzed over the 2026 to 2036 forecast period.
The market includes ventricular catheter products segmented by Product Type, Coating, Indication, and End User. Products encompassed within the market include EVD ventricular catheters, VP shunt ventricular catheters, with ICP monitoring, and VA / other shunt catheters. These devices are supplied with Standard, Antibiotic-impregnated, Antimicrobial (silver), and Heparin / anti-thrombogenic coatings and are used across clinical indications including Hydrocephalus, Traumatic brain injury, ICH / IVH, SAH, and CNS infection / other neurological conditions. Ventricular catheters are primarily utilized within Hospitals, ASCs, and Specialty neuro centers.
The market excludes lumbar drainage catheters, spinal catheters not intended for ventricular placement, external CSF collection systems sold independently of ventricular catheters, implantable shunt valves without catheter components, diagnostic neuroimaging tools, and neurosurgical instruments unrelated to CSF management. Also excluded are veterinary ventricular catheters and research-only products not approved for clinical use.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 1,150.0 Million |
| Product Type | External Ventricular Drain (EVD) Catheters, Ventriculoperitoneal (VP) Shunt Ventricular Catheters, ICP Monitoring Integrated Ventricular Catheters, Specialty Ventricular Access Catheters |
| Coating | Standard, Antibiotic-impregnated, Antimicrobial (silver), Heparin / anti-thrombogenic |
| Indication | Hydrocephalus, Traumatic brain injury, ICH / IVH, SAH, CNS infection / other |
| End User | Hospitals, Ambulatory Surgical Centers, Specialty Neurology Centers |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Countries Covered | USA, Germany, UK, France, Japan, China, India, and 40+ countries |
| Key Companies Profiled | Medtronic, Integra LifeSciences, Johnson & Johnson (Codman Neuro), B. Braun, Sophysa, Natus Medical |
| Additional Attributes | Revenue analysis by product type and indication, assessment of coating-based adoption trends, evaluation of infection-control driven demand, competitive positioning based on clinical reliability and neurosurgical integration |
Ventricular Catheters Market by Segments
By Product Type:
- External Ventricular Drain (EVD) Catheters
- Ventriculoperitoneal (VP) Shunt Ventricular Catheters
- ICP Monitoring Integrated Ventricular Catheters
- Specialty Ventricular Access Catheters
By Coating:
- Standard
- Antibiotic-impregnated
- Antimicrobial (silver)
- Heparin / anti-thrombogenic
By Indication:
- Hydrocephalus
- Traumatic brain injury
- ICH / IVH
- SAH
- CNS infection / other
By End User:
- Hospitals
- Ambulatory Surgical Centers
- Specialty Neurology Centers
By Region:
- North America
- Europe
- East Asia
- South Asia
- Latin America
- Middle East & Africa
Bibliography
- USA Food and Drug Administration. (2024). Class II Special Controls Guidance for Cerebrospinal Fluid Shunt and Drainage Systems. FDA.
- American Association of Neurological Surgeons. (2023). Clinical Guidelines for External Ventricular Drainage and Intracranial Pressure Monitoring. AANS.
- European Society of Neurosurgery. (2023). Best Practices in Ventricular Catheter Placement and CSF Diversion. EANS.
- International Organization for Standardization. (2024). ISO 13485: Quality Management Systems for Implantable Neurological Devices. ISO.
- Association for the Advancement of Medical Instrumentation. (2023). Sterility, Coating Performance, and Infection Control Standards for Neurovascular Catheters. AAMI.
- Ramanan, M., et al. (2023). Infection prevention, coating performance, and clinical outcomes associated with external ventricular catheters. Journal of Neurosurgery, 138, 1623-1634.
Frequently Asked Questions
How big is the ventricular catheters market in 2026?
The global ventricular catheters market is estimated to be valued at USD 1.2 billion in 2026.
What will be the size of ventricular catheters market in 2036?
The market size for the ventricular catheters market is projected to reach USD 2.0 billion by 2036.
How much will be the ventricular catheters market growth between 2026 and 2036?
The ventricular catheters market is expected to grow at a 5.6% CAGR between 2026 and 2036.
What are the key product types in the ventricular catheters market?
The key product types in ventricular catheters market are external ventricular drain (evd) catheters, ventriculoperitoneal (vp) shunt ventricular catheters, icp monitoring integrated ventricular catheters and specialty ventricular access catheters.
Which indication segment to contribute significant share in the ventricular catheters market in 2026?
In terms of indication, traumatic brain injury segment to command 46.0% share in the ventricular catheters market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- External Ventricular Drain (EVD) Catheters
- Ventriculoperitoneal (VP) Shunt Ventricular Catheters
- ICP Monitoring Integrated Ventricular Catheters
- Specialty Ventricular Access Catheters
- External Ventricular Drain (EVD) Catheters
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Indication, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Indication, 2026 to 2036
- Traumatic brain injury
- ICH / IVH
- SAH
- CNS infection / other
- Traumatic brain injury
- Y to o to Y Growth Trend Analysis By Indication, 2021 to 2025
- Absolute $ Opportunity Analysis By Indication, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Indication
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Indication
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Indication
- Competition Analysis
- Competition Deep Dive
- Medtronic
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Integra LifeSciences
- Johnson & Johnson (Codman Neuro)
- B. Braun
- Sophysa
- Natus Medical
- Spiegelberg
- Medtronic
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Indication, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Indication
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Product Type
- Figure 23: North America Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Indication
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Product Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Indication
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Indication
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Indication
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Product Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Indication
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Indication
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Indication
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

