Centesis Catheters Market
Centesis Catheters Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Centesis Catheters Market Forecast and Outlook 2026 to 2036
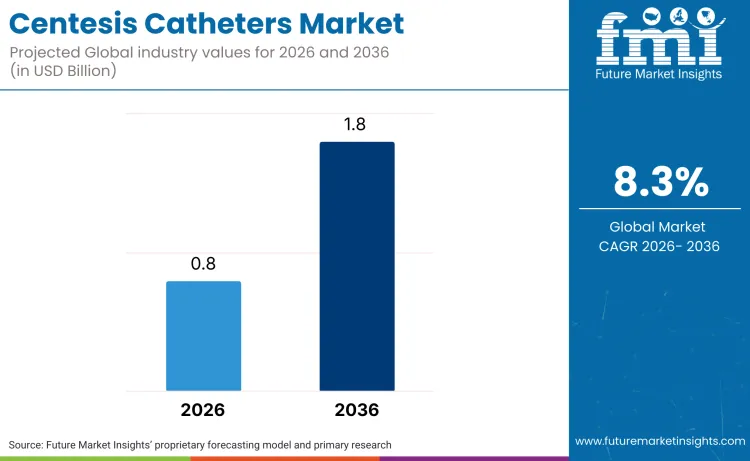
The global centesis catheters market is expected to cross USD 0.8 billion in 2026, advancing toward USD 1.8 billion by 2036, representing a robust 8.3% CAGR. Based on FMI’s analysis, growing clinical need for minimally invasive fluid‑drainage procedures continues to accelerate market expansion. Clinical demand is further bolstered by the rising need for effective pleural diseases therapeutics to manage symptoms in late-stage oncological patients.
Liam Kelly, Chairman, President and CEO of Teleflex Incorporated, stated: "The benefits of our diversified portfolio were evident as strong performances from Interventional and Surgical helped offset softness in Interventional Urology revenues. Palette Life Sciences revenues exceeded USD 75 million, which was above the guidance." This performance validates the industry’s momentum as manufacturers pivot toward interventional solutions.
The rising burden of chronic liver disease presents a critical growth vector, necessitating frequent fluid drainage. Companies that can bundle access sheaths with drainage catheters for hepatic procedures will dominate the paracentesis segment.
Summary of Centesis Catheters Market
What Is Growth Outlook for Centesis Catheters Market as per Future Market Insights Projection?
Future Market Insights projects the centesis catheters market to expand at a CAGR of 8.3% from 2026 to 2036, increasing from USD 0.8 billion in 2026 to USD 1.8 billion by 2036.
FMI Research Approach: Based on FMI’s bottom‑up revenue forecasting model, procedure volume modeling for paracentesis and thoracentesis, primary interviews with interventional radiology device manufacturers, and scenario analysis aligned with outpatient procedure migration.
How Do FMI Analysts Perceive Centesis Catheters Market to Evolve?
FMI analysts perceive the market evolving from generic drainage consumables toward procedure‑specific, safety‑engineered, and organ‑focused centesis systems optimized for outpatient and palliative care workflows.
FMI Research Approach: Evidenced by the shift toward bundled procedure kits, increased adoption of indwelling and safety‑engineered catheter designs, manufacturer focus on liver‑specific and oncology‑driven drainage workflows, and growing preference for ambulatory‑ready catheter platforms.
Which Country Holds the Largest Share in the Global Centesis Catheters Market?
The United States holds the largest share of the global centesis catheters market by value.
FMI Research Approach: Supported by country‑level revenue modeling across hospitals and ASCs, high prevalence of pleural effusion and chronic liver disease, stable reimbursement for paracentesis and thoracentesis, and strong penetration of branded and safety‑engineered catheter systems.
How Large Will the Centesis Catheters Market Be by 2036?
The global centesis catheters market is projected to reach USD 1.8 billion by 2036.
FMI Research Approach: Derived from long‑term forecasts by product type and procedure mix, scaling assumptions for paracentesis volumes and pigtail catheter adoption, exclusion of imaging capital equipment and professional fees, and cross‑validation with historical interventional consumables growth.
What Is the Definition of Centesis Catheters Market?
The centesis catheters market includes commercially sold catheters, needles, and drainage systems used for percutaneous removal of fluid or air from pleural and peritoneal cavities in diagnostic and therapeutic procedures.
FMI Research Approach: Built on FMI’s taxonomy and inclusion-exclusion framework, including pigtail, straight, and safety catheters, mapping across paracentesis, thoracentesis, and arthrocentesis, and exclusion of capital imaging devices and non‑procedure‑specific tools.
What Are Globally Unique Trends Shaping the Centesis Catheters Market?
Key global trends include outpatient migration of drainage procedures, specialization toward liver and oncology‑focused access systems, and consolidation around safety‑engineered interventional portfolios.
FMI Research Approach: Supported by rising ASC procedure volumes, development of organ‑specific access and drainage systems, strategic acquisitions/divestments by major medtech players, and procurement emphasis on needlestick prevention and device safety.
Centesis Catheters Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 0.8 billion |
| Industry Value (2036) | USD 1.8 billion |
| CAGR (2026-2036) | 8.3% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
What is Driving the Demand for Centesis Catheter?
Increase in malignancy rates is fundamentally reshaping the centesis market, manifesting as a surge in palliative drainage needs for conditions like malignant pleural effusion. NHS England data from July 2024 revealed a record 12,695 urgent cancer referrals per working day, directly increasing the incidence of fluid buildup cases requiring intervention. While demand rises, supply chain reliability remains critical; Rocket Medical's November 2024 recall of thoracentesis catheters due to adhesive failure highlights the market's sensitivity to manufacturing quality and the need for robust alternatives.
How is the Centesis Catheters Market Segmented?
The centesis catheters industry is segmented by product type, procedure, end user, and region to address specific clinical requirements. By product, the market includes pigtail catheters, straight catheters, and safety needles, reflecting the diverse anatomical needs of patients. By procedure, the market is divided into paracentesis, thoracentesis, and arthrocentesis, with adoption varying based on disease prevalence. By end user, utilization spans hospitals, ambulatory surgery centers (ASCs), and specialty clinics, highlighting the shift toward outpatient care models. Manufacturers are increasingly integrating closed loop centesis systems to enhance safety and reduce contamination risks during these critical drainage procedures.
Why does Paracentesis hold a Dominant Share?
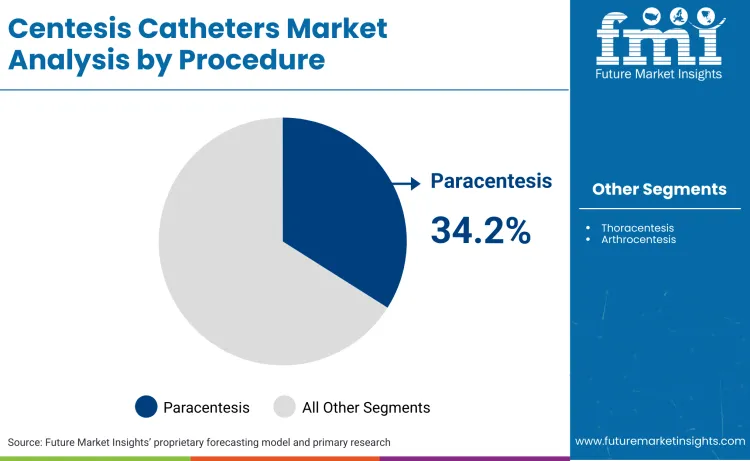
Paracentesis commands a 34.2% share, driven by the chronic management of ascites in patients with end-stage liver disease. The CDC reported 52,222 deaths from chronic liver disease in 2024, indicating a large patient pool requiring frequent, palliative drainage to maintain quality of life. This high procedure volume forces facilities to stock reliable malignant ascites management tools. George Leondis, President & CEO of Argon Medical Devices, noted: "Our company is dedicated to applying innovation to improve the devices interventionalists use to diagnose and treat liver diseases. This is demonstrated by our cadence of new product introductions in this space, including the BioPince Ultra Full-Core Biopsy Instrument, the Scorpion Portal Vein Access Sets, and the Traveler Portal Vein Access Sets."
Why are Pigtail Catheters gaining Traction?
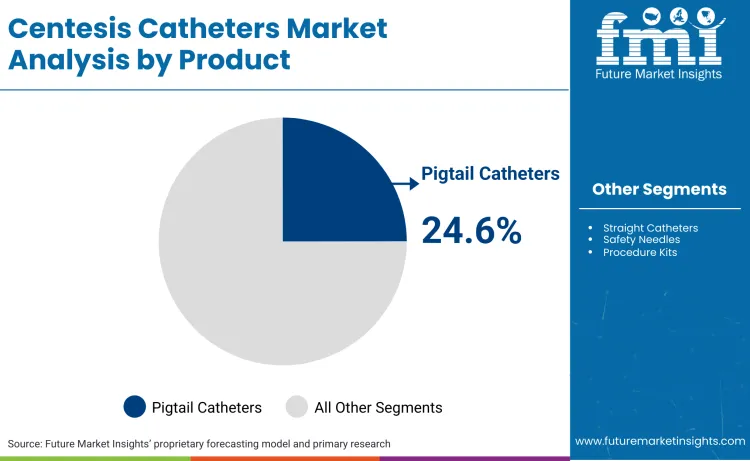
Pigtail catheters are capturing significant market interest, particularly the 24.6% share in drainage categories, due to their superior retention capabilities and kink resistance. Clinical preference is shifting toward these designs for long-term drainage as they minimize catheter occlusion, a common failure point in standard straight tubes. Merit Medical addressed this technical need by highlighting their "Micro ACE system" in April 2024, which offers enhanced kink resistance compared to legacy drainage catheter options. A 2025 clinical review further supported this shift, confirming that indwelling catheters provide effective dyspnea relief with shorter hospital stays, encouraging physicians to utilize pigtail designs for ambulatory management of recurrent effusions.
Why Is the Shift to Ambulatory Care Accelerating?
Procedures are aggressively moving from inpatient wards to outpatient clinics to reduce systemic healthcare costs. This trend drives demand for pre-packed chest drainage system kits that allow for rapid turnover in non-hospital settings. Teleflex confirmed this commercial reality in February 2025, reporting that their Interventional segment grew 18.2% year-over-year, partially fueled by the adoption of devices suitable for these efficient, lower-acuity care environments.
How Is Liver Access Specialization Changing Product Development?
Manufacturers are moving beyond generic drainage tools to develop organ-specific access systems, particularly for liver interventions. Tom Younker, Senior Vice President of Global Marketing at Argon Medical, emphasized this focused approach: "Liver diseases (cirrhosis, hepatitis, and cancer) account for 1 out of every 25 deaths worldwide, illustrating the need for faster, easier, and more accurate diagnosis and treatment options. We listened to the advice of physician experts in this field to guide development of the Intara Introducer Sheath." This specialization requires integrating aspiration and biopsy needles into drainage workflows to offer comprehensive diagnostic and therapeutic solutions.
How is Centesis Catheters Demand Outlook in Various Countries?
While mature regions prioritize 'Zero Harm' safety protocols and outpatient efficiency to drive value, emerging markets are propelled by fundamental access expansion and government-backed insurance schemes. India leads this high-velocity trajectory with a projected 11.0% CAGR, fueled by public health initiatives that are rapidly commercializing previously underserved patient populations. Germany's steady 3.0% growth reflects a saturated environment where market value is generated through the adoption of premium, safety-engineered devices rather than pure volume increases.
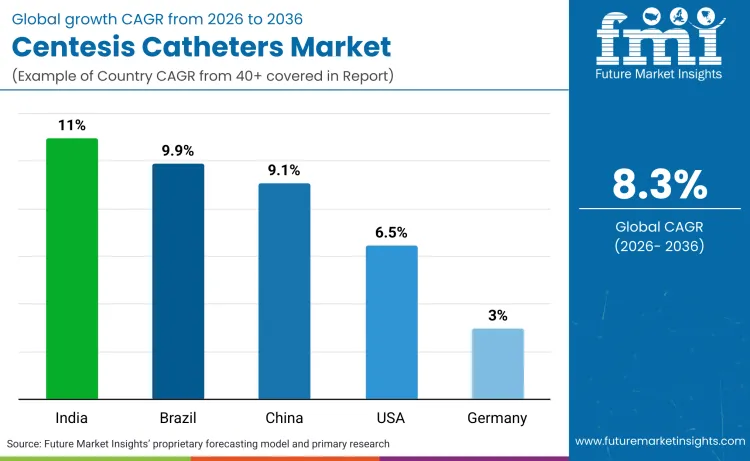
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 11.0% |
| Brazil | 9.9% |
| China | 9.1% |
| USA | 6.5% |
| Germany | 3.0% |
Source: FMI historical analysis and forecast data.
Why is India emerging as a high-growth market?
The centesis catheters industry in India is poised to register a commanding 11.0% CAGR. This surge is supported by the Ayushman Bharat scheme, which as of November 2024 subsidized surgical drainage devices and interventional procedures for millions of previously underserved patients. A press release from the Ministry of Health & Family Welfare confirmed the scale of this impact: "As of November 2024, 42 crore Ayushman cards issued and 8.39 crore hospital admissions authorized." Government focus on Non-Communicable Diseases (NCDs) in the 2024-25 budget further validates this growth trajectory, ensuring funding for cancer and liver disease management, allowing expanding hospital networks to procure advanced catheter systems that were previously cost-prohibitive.
What fuels the Demand for Centesis Catheters in Brazil?
Brazil is projected to expand at a robust 9.9% CAGR, driven principally by shifting epidemiological factors that demand frequent intervention. The centesis catheters market is structurally constrained by the financial realities of the Unified Health System (SUS); reported inefficiencies in resource allocation favor cost-effective, manual steerable needle and catheter kits over expensive automated alternatives.
How is China accelerating Regulatory Approvals?
Demand in China is set to rise at a rapid 9.1% CAGR, fundamentally reshaped by regulatory modernization. Recent milestones, such as Glaukos Corporation receiving approval for micro-invasive devices, set a critical precedent for the entry of minimally invasive drainage tools that were previously delayed by red tape. This newfound regulatory efficiency is forcing domestic companies to innovate rapidly to defend market share, creating a hyper-competitive environment that benefits from the adoption of high-tech peripherally inserted central catheters and specialized drainage solutions. As local barriers to entry lower for premium imports, the entire market is pivoting toward higher-quality, compliant interventional devices.
Why is the USA Pivoting to Safety and Outpatient Care?
The USA anticipates a steady 6.5% CAGR, driven by a decisive structural shift from inpatient wards to ambulatory settings. With approximately 1.5 million pleural effusion cases annually, the clinical focus has moved to efficient, reimbursement-friendly procedures performed in Ambulatory Surgery Centers (ASCs) where turnover time is critical. The 2024 CMS Physician Fee Schedule maintained stable reimbursement for paracentesis, providing the economic certainty needed to support this decentralized model.
Yongji Fu, Vice President & Head of R&D at B. Braun, highlighted this industry-wide prioritization of safety: "The awards for Introcan Safety 2 underscore B. Braun employees who have contributed to this success with their expertise. It represents a leap forward in ensuring the safety of both patients and healthcare professionals during vascular access procedures." This focus aligns perfectly with the nation's zero-tolerance policy for needlestick injuries, making safety-engineered devices a mandatory procurement standard in high-throughput clinics.
How is Germany integrating Digital Health with Intervention?
Germany is forecast to grow at a moderate 3.0%, characterized by market maturity and a distinct pivot toward digital integration. The German Digital Act (DigiG), effective March 2024, has expanded reimbursement pathways for Class IIb medical devices, effectively opening the door for "smart" drainage systems equipped with digital monitoring capabilities. This legislative support encourages the adoption of advanced central venous catheter technologies and drainage systems that can seamlessly integrate with hospital electronic records to track fluid output automatically. In a low-growth environment, the centesis catheters market prioritizes these premium devices that offer documented clinical benefits and safety features.
How is the Competition in the Centesis Catheters Market?
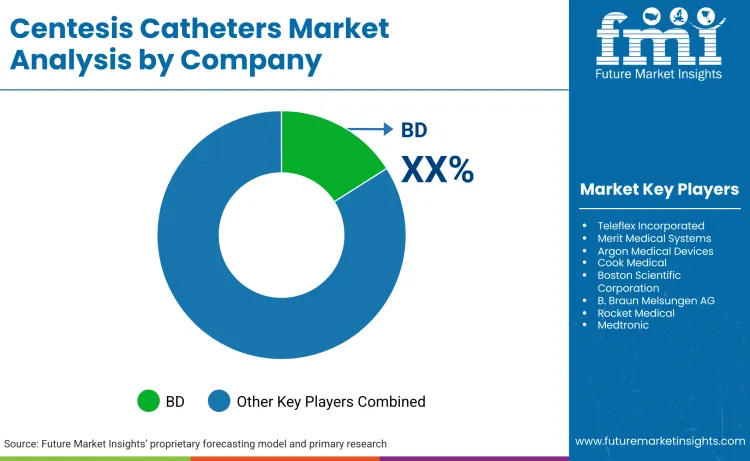
The centesis catheters market is moderately concentrated, with BD (16.2% share) and Teleflex leading the sector. BD's dominance is reinforced by its massive scale and recent strategic pivot to focus solely on MedTech, allowing for concentrated R&D in interventional spaces. Teleflex challenges this with an 18.2% growth in its Interventional segment (Q4 2024), leveraging a diversified portfolio to offset weakness in other areas.
Fred Lampropoulos, Chairman and CEO of Merit Medical Systems Inc., stated: "We are pleased with a solid start to the fiscal year and remain confident in our team's ability to deliver continued strong execution, stable constant currency growth, improving profitability, and solid free cash flow generation in 2024." Competitors are shifting toward specialized access and safety to gain market share. Argon Medical's launch of the Intara Sheath in September 2024 signals a move into niche liver management, moving away from generic catheter offerings. Meanwhile, Cook Medical's November 2024 investment in Zenflow suggests a continued appetite for minimally invasive technologies that can be cross-sold to interventional radiologists. The recall by Rocket Medical in November 2024 creates a short-term share capture opportunity for agile competitors in the UK and Europe who can quickly supply alternative thoracentesis kits to fill the void.
Recent Developments:
- In September 2024, BD completed the USD 4.2 billion acquisition of Edwards Lifesciences' Critical Care product group, including the Swan-Ganz pulmonary artery catheter for advanced patient monitoring. This bolsters BD's smart connected care solutions and expands its monitoring catheter offerings.
- In January 2025, Argon Medical Devices acquired the SeQure and DraKon microcatheters from Interventional Systems. These microcatheters are designed for precise delivery of therapeutic agents in interventional oncology procedures, expanding Argon's portfolio from diagnostic to therapeutic devices and strengthening its position in the interventional market.
- In January 2025, BD announced a USD 30 million investment to expand manufacturing capacity for IV lines and catheters at its Utah facility, building on a 2024 investment that increased IV catheter output by over 40 million units annually. This enhances BD's ability to meet demand for critical vascular access devices.
Key Players in Centesis Catheters Market
- Becton, Dickinson and Company (BD)
- Teleflex Incorporated
- Merit Medical Systems
- Argon Medical Devices
- Cook Medical
- Boston Scientific Corporation
- B. Braun Melsungen AG
- Rocket Medical
- Medtronic
- Cardinal Health
Market Definition
The centesis catheters market represents revenue generated from the sale of catheters, needles, and drainage systems used for the removal of fluid or air from body cavities, specifically the pleural space (thoracentesis) and peritoneal cavity (paracentesis). As operationally defined in the article, the market measures commercial sales of disposable and indwelling catheters, including safety-engineered variants, pigtail catheters, and straight catheters. Market sizing reflects the value of these devices sold to hospitals, ambulatory surgery centers, and specialty clinics, analyzed by procedure type and region, and expressed in USD million. The market includes procedure-specific kits that bundle catheters with drainage bags, tubing, and introductory needles.
It covers devices used for both diagnostic sampling and therapeutic drainage of effusions associated with cancer, liver disease, and infection. Revenue from interventional radiology related drainage tools is included where specific to centesis applications. The scope counts adoption across major global regions including North America, Europe, Asia Pacific, and Latin America. The market excludes general-purpose surgical suction devices not designed for percutaneous centesis procedures. Capital equipment such as ultrasound machines used for guidance are not included in the catheter market sizing. Services fees, professional physician charges, and hospital facility fees associated with the procedure are excluded. Drug-delivery catheters and urinary catheters are also outside the scope of this analysis.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 0.8 billion |
| Product Type | Pigtail Catheters, Straight Catheters, Safety Needles |
| Procedure Type | Paracentesis, Thoracentesis, Arthrocentesis |
| End User | Hospitals, Ambulatory Surgery Centers, Specialty Clinics |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Countries Covered | United States, Germany, United Kingdom, India, China, Brazil, and 40+ countries |
| Key Companies Profiled | BD, Teleflex, Merit Medical, Argon Medical, Cook Medical, Boston Scientific, B. Braun |
| Additional Attributes | Revenue analysis by product type and procedure, adoption trends across acute and outpatient care settings, regulatory and compliance landscape (FDA, CE marking), procedural volume impact analysis, safety-engineered device penetration, pricing and reimbursement considerations, and competitive positioning analysis |
Source: FMI historical analysis and forecast data.
Centesis Catheters Market by Segments
By Product:
- Pigtail Catheters
- Straight Catheters
- Safety Needles
- Procedure Kits
By Procedure:
- Paracentesis
- Thoracentesis
- Arthrocentesis
By End User:
- Hospitals
- Ambulatory Surgery Centers (ASCs)
- Specialty Clinics
By Region:
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa
Bibliography
- National Center for Biotechnology Information. (2025, January). Pleural effusion. StatPearls Publishing.
- NHS England. (2025). Hospital outpatient activity data 2024-2025. NHS Digital.
- Ministry of Health & Family Welfare, Government of India. (2024, December). Health initiatives and achievements 2024. Press Information Bureau.
- NHS Supply Chain. (2024, November). Field safety notice: Rocket Medical chest drain devices. NHS.
- National Medical Products Administration. (2024). Medical device approval announcements and regulatory updates. NMPA, China.
Frequently Asked Questions
What is the current global market size for Centesis Catheters?
The global industry is valued at USD 0.8 billion in 2026, with significant expansion driven by the rising prevalence of liver and pleural diseases.
What is the projected CAGR for the Centesis Catheters market?
Market revenues are projected to grow at an 8.3% CAGR from 2026 to 2036.
Which segment currently dominates the industry?
Paracentesis currently holds the dominant share of 34.2%, driven by the chronic management needs of ascites patients.
What are the primary risks or barriers to adoption?
Product recalls due to manufacturing defects, as seen with Rocket Medical in 2024, and strict reimbursement criteria in emerging markets create significant barriers.
Who are the leading players in the Centesis Catheters space?
BD and Teleflex lead the sector, leveraging massive distribution networks and recent strategic pivots toward interventional medtech.
Table of Content
- Executive Summary
- Centesis Catheters Market Introduction
- Market Definition
- Market Scope
- Market Taxonomy
- Research Methodology
- Study Framework
- Assumptions and Limitations
- Data Sources
- Market Estimation and Forecasting Approach
- Centesis Catheters Market Dynamics
- Drivers
- Restraints
- Opportunities
- Trends
- Global Centesis Catheters Market Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Centesis Catheters Market Analysis by Product Type
- Global Centesis Catheters Market Analysis by Procedure Type
- Global Centesis Catheters Market Analysis by End User
- Global Centesis Catheters Market Analysis by Region
- North America Centesis Catheters Market Analysis
- United States
- Canada
- Latin America Centesis Catheters Market Analysis
- Brazil
- Rest of Latin America
- Europe Centesis Catheters Market Analysis
- Germany
- United Kingdom
- France
- Italy
- Spain
- Rest of Europe
- Asia Pacific Centesis Catheters Market Analysis
- China
- India
- Japan
- Rest of Asia Pacific
- Middle East & Africa Centesis Catheters Market Analysis
- GCC
- South Africa
- Rest of Middle East & Africa
- Competitive Landscape Analysis
- Key Players Profiled
- Market Definition and Scope
- Market Segmentation Analysis
- Bibliography
- Frequently Asked Questions
List of Tables
- Global Centesis Catheters Market Size (USD Billion), 2021-2036
- Global Centesis Catheters Market Y-o-Y Growth (%), 2021-2036
- Global Centesis Catheters Market Absolute $ Opportunity (USD Billion), 2026-2036
- Global Centesis Catheters Market Size by Product Type, 2021-2036
- Global Centesis Catheters Market Size by Procedure Type, 2021-2036
- Global Centesis Catheters Market Size by End User, 2021-2036
- Global Centesis Catheters Market Size by Region, 2021-2036
- North America Centesis Catheters Market Size by Country, 2021-2036
- Latin America Centesis Catheters Market Size by Country, 2021-2036
- Europe Centesis Catheters Market Size by Country, 2021-2036
- Asia Pacific Centesis Catheters Market Size by Country, 2021-2036
- Middle East & Africa Centesis Catheters Market Size by Country, 2021-2036
- Global Centesis Catheters Market Share (%) by Product Type, 2025
- Global Centesis Catheters Market Share (%) by Procedure Type, 2025
- Global Centesis Catheters Market Share (%) by End User, 2025
- Global Centesis Catheters Market Share (%) by Region, 2025
List of Figures
- Global Centesis Catheters Market Growth Framework
- Centesis Catheters Market Value Chain Analysis
- Centesis Catheters Market Product and Technology Landscape
- Centesis Catheters Market Opportunity Map
- Centesis Catheters Market Scenario Forecast Model
- Global Centesis Catheters Market Share by Product Type, 2025
- Global Centesis Catheters Market Share by Procedure Type, 2025
- Global Centesis Catheters Market Share by End User, 2025
- Global Centesis Catheters Market Share by Region, 2025
- North America Centesis Catheters Market Structure
- Latin America Centesis Catheters Market Structure
- Europe Centesis Catheters Market Structure
- Asia Pacific Centesis Catheters Market Structure
- Middle East & Africa Centesis Catheters Market Structure
- Centesis Catheters Market Competitive Landscape Mapping
- Centesis Catheters Market Strategic Positioning Matrix
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

