The veterinary biologics market is valued at USD 14.8 billion in 2026 and is projected to reach USD 27.3 billion by 2036, reflecting a CAGR of 6.30%. Growth is driven by rising demand for disease prevention and health management in livestock, companion animals, and poultry, alongside expanding veterinary infrastructure. Products include vaccines, attenuated live, conjugate, inactivated, subunit, toxoid, DNA, recombinant, bacterial extracts, monoclonal antibodies, immunomodulators, cytokines, and allergenic extracts. Cost structures reflect R&D intensity, regulatory compliance, and distribution logistics, while margin concentration favors companies offering validated, multi-species biologics rather than bulk commodity solutions.
Between 2026 and 2036, distribution spans veterinary clinics, hospitals, research institutes, and retail pharmacies. Leading companies such as Elanco Animal Health, Ceva, Phibro Animal Health Corporation, Boehringer Ingelheim, Zoetis Inc., Merck and Co. Inc., Virbac, Hester Biosciences, Colorado Serum Company, Addison Biological Laboratory Inc., American Animal Health Inc., HIPRA, and Bimeda Inc. capture value through product portfolio breadth, regulatory alignment, and distribution scale. Fragmentation persists in regional or niche providers, while established players maintain pricing power, higher margins, and concentrated revenue streams through multi-species coverage and validated clinical efficacy.
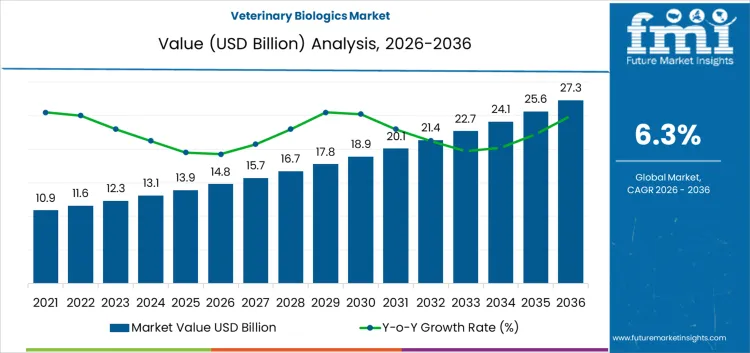
Between 2026 and 2031, the veterinary biologics market is projected to grow from USD 14.8 billion to USD 20 billion, generating an absolute increase of USD 5.2 billion and reflecting a CAGR of 6.3%. Early-stage growth is driven by increasing livestock populations, rising adoption of animal vaccines and immunotherapies, and expanding veterinary healthcare infrastructure. Volume growth contributes approximately 68% of expansion, while price growth accounts for 32%, supported by premium biologic formulations and enhanced efficacy. Value capture favors manufacturers providing clinically validated vaccines and biologics with strong distribution networks rather than focusing solely on unit sales.
From 2031 to 2036, the market is expected to expand from USD 20 billion to USD 27.3 billion, adding USD 7.3 billion. Volume remains the dominant driver, representing roughly 67% of growth due to broader adoption, increased animal healthcare awareness, and expansion in emerging markets. Price growth accounts for 33%, supported by innovative formulations and differentiated biologics. Competitive advantage shifts toward firms offering reliable, high-quality products, strong veterinary partnerships, and broad geographic coverage, while late entrants focus on niche applications and cost efficiency to capture incremental market share.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 14.8 billion |
| Forecast Value (2036) | USD 27.3 billion |
| Forecast CAGR 2026 to 2036 | 6.30% |
The veterinary biologics market is expanding due to rising demand for vaccines, immunotherapeutics, and diagnostic products to prevent and control diseases in livestock, companion animals, and aquaculture. Historically, disease management relied heavily on conventional pharmaceuticals and reactive treatment, which offered limited herd immunity and variable effectiveness. Modern biologics include recombinant vaccines, monoclonal antibodies, and viral vector platforms that provide targeted immune protection and improved safety profiles. Livestock producers, veterinary clinics, and aquaculture operators prioritize efficacy, regulatory compliance, and cold chain stability. Early adoption focused on high-value livestock and companion animal populations, while current demand spans large-scale production systems and global pet care markets, driven by animal health awareness and biosecurity requirements.
Future growth is shaped by regulatory frameworks, disease prevalence, and increasing adoption of preventive healthcare rather than reactive interventions alone. Compared with initial biologics, contemporary products emphasize recombinant technologies, adjuvant optimization, and multi-species applicability to enhance protection and reduce treatment frequency. Cost structures depend on R&D, manufacturing, and distribution logistics, concentrating margins among suppliers capable of delivering consistent, high-quality biologics. Veterinarians and producers adopt biologics to improve herd health, reduce mortality, and comply with safety standards. By 2036, veterinary biologics are expected to become standard in animal healthcare, supporting disease prevention, productivity, and sustainable livestock management globally.
The demand for veterinary biologics is segmented by animal type and distribution channel. Animal types include livestock, companion animals, canine, feline, avian, bovine, porcine, ovine and caprine, poultry, and equine. Distribution channels cover veterinary clinics, veterinary hospitals, veterinary research institutes, and retail pharmacies. Segment adoption is influenced by disease prevalence, vaccination schedules, and animal health priorities. Uptake is driven by regulatory approvals, safety standards, and efficacy of biologics rather than marketing considerations. Selection of animal type and distribution channel is guided by herd size, species susceptibility, and access to veterinary services, ensuring effective immunization coverage across commercial and companion animal populations.
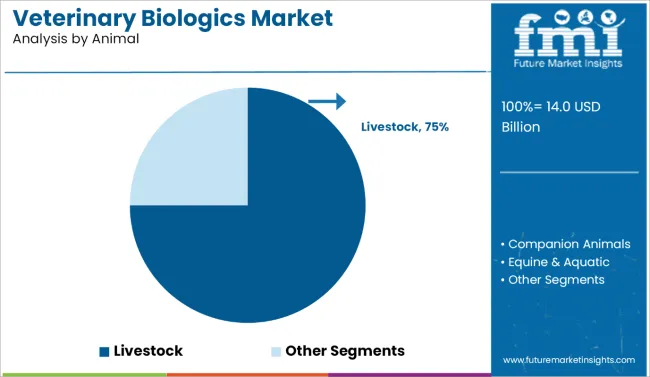
Livestock accounts for approximately 75% of total animal type demand, making it the dominant category. This reflects the high economic value and population density of cattle, poultry, swine, and small ruminants in commercial agriculture. Farmers and veterinarians prioritize immunization to prevent disease outbreaks, reduce mortality, and maintain productivity. Adoption is reinforced by large-scale vaccination programs, regulatory mandates, and the need for herd immunity. Biologics for livestock include vaccines, antitoxins, and immune modulators designed to protect against bacterial, viral, and parasitic infections, supporting operational efficiency and profitability in animal husbandry.
Demand for livestock biologics is further shaped by production scale and supply chain reliability. Clinics, mobile veterinary units, and farm-based distribution ensure effective coverage across extensive herds. Cold chain management, dosing accuracy, and vaccine stability are critical to maintain efficacy. The segment leads because livestock biologics combine high volume requirements, regulatory oversight, and measurable impact on animal health and farm economics, establishing them as the primary driver of global veterinary biologics demand.
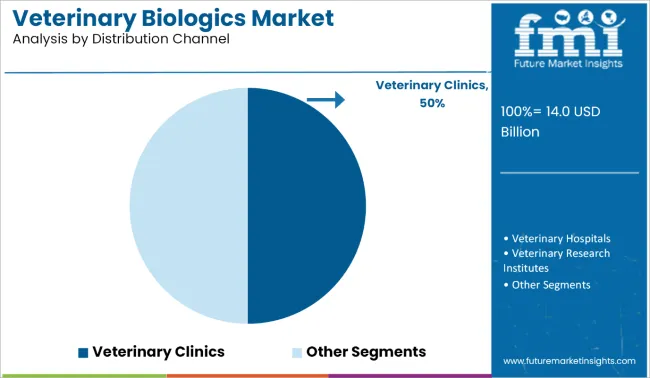
Veterinary clinics account for approximately 50% of total distribution channel demand, making them the largest segment. Clinics serve as the primary access point for routine vaccination, preventive care, and treatment of livestock and companion animals. They provide trained personnel, cold storage, and documentation systems to ensure safe and effective administration of biologics. Adoption is reinforced by high client trust, procedural oversight, and accessibility to both rural and urban animal populations.
Demand in veterinary clinics is driven by operational efficiency, regulatory compliance, and herd or pet health management. Clinics support both single-animal care and herd-based immunization, coordinating schedules and follow-up doses. They integrate biologic administration with diagnostic services, health monitoring, and owner education. The segment leads because veterinary clinics combine accessibility, professional expertise, and infrastructure to deliver consistent, regulated, and effective biologic coverage across multiple animal types, making them the primary distribution channel for veterinary biologics globally.
Veterinary biologics, including vaccines, antisera, and diagnostic immunogens, are increasingly adopted to prevent and control infectious diseases in livestock, poultry, and companion animals. Adoption is strongest in regions with high-density livestock farming, strict animal health regulations, and growing awareness of zoonotic disease risks. Manufacturers prioritize product efficacy, safety, and regulatory compliance rather than volume alone. Growth is driven by disease prevention, herd health management, and adherence to regional veterinary standards. Investment decisions focus on cold chain maintenance, quality assurance, and batch consistency. Biologics reduce mortality, improve productivity, and enhance animal welfare across commercial and smallholder operations.
Demand is influenced by local incidence of infectious animal diseases, livestock density, and government vaccination programs. Regions with frequent outbreaks of avian influenza, foot-and-mouth disease, or rabies show higher adoption. Regulatory approval for veterinary biologics, along with compliance to safety and efficacy standards, ensures consistent product quality. The driver is disease prevention and risk mitigation rather than cost. Suppliers capable of delivering validated, traceable biologics gain preference among veterinarians, farmers, and government programs. Regional livestock management practices and disease monitoring policies strongly shape production, distribution, and adoption strategies.
Barriers include high costs for advanced vaccines, storage and cold chain requirements, and limited access in rural or resource-constrained regions. Variability in animal immunity response, supply chain inefficiencies, and regulatory delays can affect product availability. Smaller farms or low-income regions may rely on traditional or locally produced vaccines. Limited awareness of disease prevention and lack of trained veterinary staff further constrain adoption. These factors concentrate early deployment in well-resourced commercial farms and organized veterinary networks, slowing broader uptake despite the critical need for biologics in disease management.
Trends focus on recombinant vaccines, combination biologics, and region-specific immunogens targeting prevalent diseases. Collaboration between biotech firms, veterinary research institutions, and government agencies supports product validation, distribution, and farmer education. Pilot programs validate efficacy and safety in local livestock populations before full-scale rollout. Digital tracking and batch certification improve traceability and regulatory compliance. Emphasis is on disease prevention, product reliability, and herd health outcomes rather than throughput or cost. Collaborative approaches enhance adoption in commercial and smallholder farms, improving livestock productivity, biosecurity, and regional animal health management.
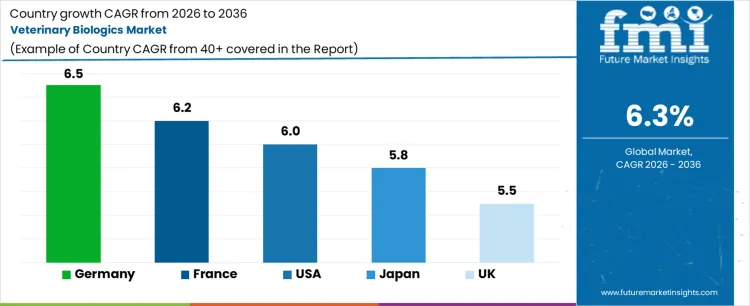
| Country | CAGR (%) |
|---|---|
| Germany | 6.5% |
| France | 6.2% |
| USA | 6.0% |
| Japan | 5.8% |
| UK | 5.5% |
Demand for veterinary biologics is rising as livestock and companion animal sectors increasingly adopt vaccines, immunotherapies, and biologically derived treatments to improve animal health and productivity. Germany leads with a 6.5% CAGR, supported by strong livestock farming, advanced veterinary infrastructure, and high adoption of preventive biologics. France follows at 6.2%, driven by regulatory support and widespread use in dairy and meat production. The USA grows at 6.0%, shaped by large-scale livestock operations, companion animal healthcare demand, and biologic innovation. Japan records 5.8% growth, supported by companion animal care and preventive livestock applications. The UK shows 5.5% CAGR, reflecting steady adoption in livestock and pet healthcare markets and gradual integration of advanced biologics.
Germany veterinary biologics market is expanding at a CAGR of 6.5%, supported by increasing adoption of vaccines, immunotherapies, and biologically derived treatments for livestock and companion animals. Veterinary clinics, livestock farms, and animal health distributors are implementing biologics to improve herd immunity and disease prevention. Demand is concentrated among commercial farms, veterinary hospitals, and animal health organizations. Investments focus on product quality, regulatory compliance, and cold chain logistics rather than broad-scale capacity expansion. Growth reflects rising awareness of animal health management and adoption of advanced biologic treatments.
France veterinary biologics market is growing at a CAGR of 6.2%, supported by increasing preventive care, adoption of biologics, and rising demand for disease control in livestock and pets. Veterinary clinics and animal health distributors are supplying vaccines, serum-based therapies, and biologically derived treatments. Demand is concentrated among commercial farms, veterinary hospitals, and companion animal care centers. Investments prioritize product efficacy, cold chain management, and regulatory adherence rather than production scale expansion. Growth reflects heightened animal health awareness and integration of biologic solutions in veterinary practice.
United States veterinary biologics market is expanding at a CAGR of 6%, supported by rising livestock and companion animal populations, regulatory support, and growing adoption of vaccines and immunotherapies. Animal health providers and distributors are supplying biologics to improve herd immunity, prevent disease outbreaks, and enhance animal wellness. Demand is concentrated among veterinary hospitals, livestock farms, and commercial distributors. Investments focus on product quality, regulatory compliance, and distribution efficiency rather than rapid capacity expansion. Growth reflects consistent adoption of preventive and therapeutic biologic treatments.
Japan veterinary biologics market is growing at a CAGR of 5.8%, supported by increasing preventive care for livestock and pets, rising animal health awareness, and adoption of biologic treatments. Veterinary clinics and livestock operations are implementing vaccines, serum therapies, and immunomodulatory products. Demand is concentrated in urban veterinary hospitals, commercial farms, and animal health distributors. Investments prioritize product reliability, regulatory adherence, and cold chain management rather than broad production expansion. Growth reflects gradual integration of biologic solutions to enhance animal disease prevention.
United Kingdom veterinary biologics market is expanding at a CAGR of 5.5%, supported by increasing adoption of vaccines and biologic therapies in companion animals and livestock. Veterinary hospitals, farms, and distributors are supplying biologics to prevent disease and enhance animal welfare. Demand is concentrated among urban veterinary clinics, commercial livestock farms, and specialized distributors. Investments focus on product quality, regulatory compliance, and supply chain reliability rather than large-scale expansion. Growth reflects consistent awareness of animal health and preventive care practices.
Competition in the veterinary biologics market is shaped by differences in product breadth, species coverage, disease targets, distribution networks, and regional regulatory approvals. Zoetis Inc. is widely recognised for its extensive portfolio of vaccines, immunotherapeutics, and diagnostics for companion animals and livestock, supported by broad global distribution and strong field support infrastructure. Merck and Co. Inc. competes with a comprehensive range of biologics, focusing on high impact livestock diseases and integrated preventive health programs. Boehringer Ingelheim differentiates through its targeted immunisation solutions and emphasis on respiratory and reproductive disease prevention in production animal systems.
Other firms bring varied depth and regional speciality. Elanco Animal Health and Ceva offer broad biologics for poultry, swine, and small animals, with Ceva’s portfolio tailored to certain regional markets and disease niches. Phibro Animal Health Corporation and Virbac provide vaccines and immunomodulators with strengths in specific segments like aquaculture, equine health, and companion animals. Smaller and regional players such as Hester Biosciences Limited, Colorado Serum Company, Addison Biological Laboratory Inc., American Animal Health Inc., HIPRA, and Bimeda Inc. (Texas Vet Lab Inc.) focus on niche disease targets, regional vaccine strains, or species specific immunogens. Competitive differences emerge in the depth of R&D pipelines, approval reach across geographies, and ability to tailor biologics to emerging disease pressures, with firms that combine robust disease coverage, regulatory agility, and strong practitioner support positioned to serve diverse veterinary needs.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD billion |
| Animal | Livestock, Companion Animals, Canine, Feline, Avian, Aquatic, Bovine, Porcine, Ovine and Caprine, Poultry, Equine |
| Product | Vaccines, Attenuated Live Vaccines, Conjugate Vaccines, Inactivated Vaccines, Subunit Vaccines, Toxoid Vaccines, DNA Vaccines, Recombinant Vaccines, Bacterial Extracts, Monoclonal Antibody, Immunomodulatory, Cytokines, Allergenic Extracts, Others |
| Distribution Channel | Veterinary Clinics, Veterinary Hospitals, Veterinary Research Institutes, Retail Pharmacies |
| Region | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Key Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Rest of Asia Pacific, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, Rest of Europe, United States, Canada, Mexico, Brazil, Chile, Rest of Latin America, Kingdom of Saudi Arabia, Other GCC Countries, Turkey, South Africa, Other African Union, Rest of Middle East & Africa |
| Key Companies Profiled | Elanco Animal Health, Ceva, Phibro Animal Health Corporation, Boehringer Ingelheim, Zoetis Inc., Merck and Co. Inc., Virbac, Hester Biosciences Limited, Colorado Serum Company, Addison Biological Laboratory Inc., American Animal Health Inc., HIPRA, Bimeda Inc. (Texas Vet Lab Inc.) |
| Additional Attributes | Dollar sales by animal type, product type, and distribution channel; regional CAGR and growth projections; volume vs. price contribution; margin concentration among suppliers offering multi-species validated biologics; competitive differentiation through R&D depth, regulatory approvals, cold chain reliability, and distribution reach; adoption driven by disease prevention, herd health management, and veterinary compliance |
How big is the veterinary biologics market in 2026?
The global veterinary biologics market is estimated to be valued at USD 14.8 billion in 2026.
What will be the size of veterinary biologics market in 2036?
The market size for the veterinary biologics market is projected to reach USD 27.3 billion by 2036.
How much will be the veterinary biologics market growth between 2026 and 2036?
The veterinary biologics market is expected to grow at a 6.3% CAGR between 2026 and 2036.
What are the key product types in the veterinary biologics market?
The key product types in veterinary biologics market are livestock, companion animals, canine, avian, feline, aquatic, bovine, porcine, ovine and caprine, poultry and equine.
Which distribution channel segment to contribute significant share in the veterinary biologics market in 2026?
In terms of distribution channel, veterinary clinics segment to command 50.0% share in the veterinary biologics market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.