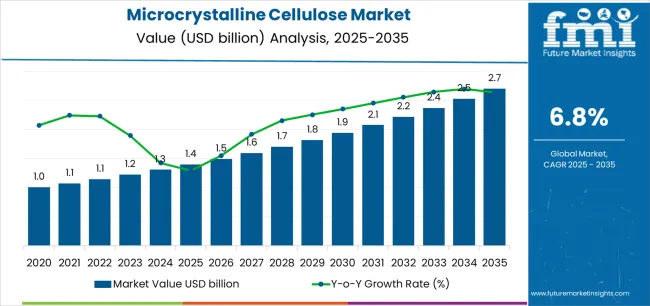
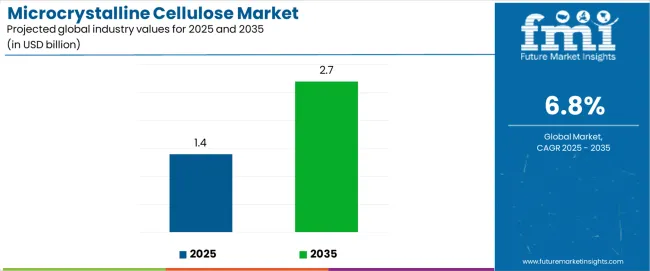
The microcrystalline cellulose (MCC) market is projected to grow from USD 1.4 billion in 2025 to USD 2.7 billion by 2035, registering a CAGR of 6.8%. Demand remains closely linked to expansion in pharmaceutical excipient usage, solid-dose formulation output, and rising adoption of clean-label food ingredients. Pharmaceuticals & nutraceuticals continue to represent the largest end-use segment at 46%, supported by the need for high-purity binders, disintegrants, and direct-compression excipients across generic drugs and nutraceutical tablets. Food & beverages follow with 33%, driven by the use of MCC as a fat replacer, texture modifier, and stabilizer in clean-label and reduced-calorie formulations. Wood-based MCC accounts for 65% of the market, supported by its consistent performance, well-established supply chains, and regulatory acceptance, while non-wood sources such as cotton linters, bamboo, and agricultural residues continue to gain traction as sustainable alternatives.
Product differentiation is increasing through advances in particle engineering, low-nitrite pharmaceutical grades, and functional performance optimization for high-speed tableting and orally disintegrating formulations. Regionally, Asia Pacific leads expansion, with India (8.3% CAGR) and China (7.4% CAGR) driven by generic drug production scale-up, excipient capacity development, and rising food processing activity. Brazil (6.5%) benefits from nutraceutical manufacturing and convenience foods growth, while mature markets such as Germany (6.2%), Japan (5.6%), and the United States (5.4%) show steady demand tied to quality-driven pharmaceutical production, premium personal care formulations, and functional foods adoption. Competitive positioning concentrates around pharmaceutical-grade quality consistency, sustainable sourcing, nitrosamine-risk-mitigating grades, and strong technical service capabilities, with leading players including IFF, Asahi Kasei, JRS PHARMA, DFE Pharma, Roquette Frères, and Mingtai Chemical.
A major dynamic shaping market expansion is the rapid growth of the global pharmaceutical sector. MCC is a preferred excipient in tablet and capsule formulations due to its excellent dry binding properties and ability to enhance mechanical strength without compromising disintegration time. With rising production of generics, OTC medicines, and prescription formulations, pharmaceutical manufacturers require consistent-quality MCC to support high-speed tableting, precision dosing, and controlled-release formulations. As drug manufacturing expands in India, China, the U.S., and Europe, the demand for MCC used in solid oral dosage forms continues to accelerate.
Technological advancements in production processes are reshaping competitive dynamics. Manufacturers are developing high-purity, modified, co-processed, and low-moisture MCC grades tailored for specialized pharmaceutical and food applications. Improved crystallization control, optimized particle morphology, and enhanced flow characteristics support high-speed manufacturing requirements. Co-processed MCC blends engineered for direct compression are gaining traction as they reduce processing steps and improve tablet uniformity.
| Metric | Value |
|---|---|
| Estimated Value in (2025E) | USD 1.4 billion |
| Forecast Value in (2035F) | USD 2.7 billion |
| Forecast CAGR (2025 to 2035) | 6.8% |
Market expansion is being supported by the increasing global demand for pharmaceutical excipients in solid-dose formulations and the corresponding need for multifunctional binders, disintegrants, and diluents that can support direct compression tableting, enhance drug bioavailability, and ensure robust manufacturing processes across various generic pharmaceuticals, branded drugs, and nutraceutical products. Modern pharmaceutical manufacturers and contract development organizations are increasingly focused on implementing excipient solutions that can simplify formulation development, reduce processing costs, and provide consistent quality across batch production while meeting stringent regulatory requirements. Microcrystalline cellulose's proven ability to deliver excellent compressibility, support rapid disintegration, and maintain chemical inertness makes it an essential excipient for contemporary solid-dose pharmaceutical manufacturing.
The growing emphasis on clean-label food formulations and natural ingredient preferences is driving demand for microcrystalline cellulose as a versatile texture modifier, fat replacer, and stabilizer that can support reduced-calorie products, improve mouthfeel in low-fat formulations, and provide natural origin claims appealing to health-conscious consumers. Food manufacturers' preference for multifunctional ingredients that combine technical performance with consumer-friendly labeling is creating opportunities for innovative microcrystalline cellulose applications across dairy alternatives, baked goods, and functional beverages. The rising influence of pharmaceutical quality improvements and nitrosamine risk mitigation initiatives is also contributing to increased adoption of low-nitrite microcrystalline cellulose grades that can minimize impurity-related risks while supporting comprehensive pharmaceutical quality assurance programs.
The market is segmented by material, product grade, end use, and region. By material, the market is divided into wood-based MCC and non-wood-based MCC (including cotton linters, bamboo, and agricultural residues). Based on product grade, the market is categorized into pharmaceutical grade, food grade, and industrial/technical grade. By end use, the market covers pharmaceuticals & nutraceuticals, food & beverages, personal care & cosmetics, and other industrial applications (including chemicals, electronics, and animal nutrition). Regionally, the market is divided into Asia Pacific, North America, Europe, Latin America, and the Middle East & Africa.
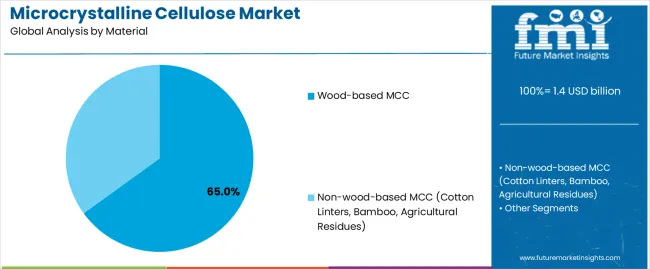
The wood-based MCC segment is projected to maintain its leading position in the microcrystalline cellulose market in 2025 with a commanding 65% market share, reaffirming its role as the preferred raw material source for commercial microcrystalline cellulose production due to established supply chains, consistent quality characteristics, and cost-competitive manufacturing economics. Pharmaceutical and food manufacturers increasingly utilize wood-based microcrystalline cellulose for its reliable functional properties, comprehensive regulatory acceptance across global markets, and well-documented safety profiles supported by decades of commercial use and extensive toxicological evaluation. Wood-based cellulose's proven effectiveness in delivering consistent particle morphology and predictable performance directly addresses industry requirements for reproducible excipient quality and manufacturing reliability in regulated applications.
This material segment forms the foundation of commercial microcrystalline cellulose production, as it represents the feedstock with the greatest supply availability and established processing infrastructure across major producing regions in North America, Europe, and Asia. Pharmaceutical investments in wood-based excipients continue to strengthen adoption among formulators and quality assurance professionals. With manufacturing processes requiring consistent raw material properties and predictable processing behavior, wood-based microcrystalline cellulose aligns with both quality objectives and supply security requirements, making it the central component of comprehensive excipient sourcing strategies supporting global pharmaceutical and food manufacturing operations.
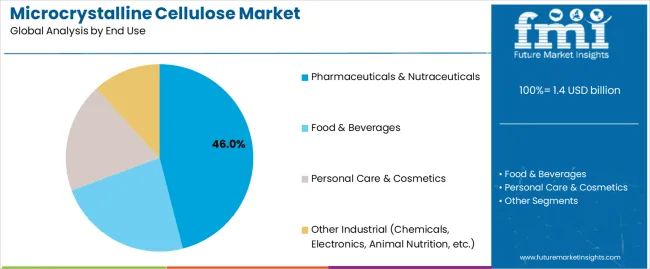
The pharmaceuticals & nutraceuticals end-use segment commands the largest market share at 46% in 2025, reflecting its established position as the most demanding and value-creating application category requiring stringent quality specifications, comprehensive analytical documentation, and consistent functional performance across direct compression tableting, wet granulation, capsule filling, and specialized delivery systems. This segment benefits from microcrystalline cellulose's unique combination of properties including excellent binding capacity, controlled disintegration behavior, chemical inertness with active pharmaceutical ingredients, and global regulatory acceptance enabling worldwide product distribution. The extensive use of microcrystalline cellulose as a primary excipient in immediate-release tablets, orally disintegrating formulations, and dietary supplements supports segment dominance.
Food & beverages follow with 33% share, utilizing microcrystalline cellulose for texture modification, fat replacement, stabilization, and clean-label ingredient positioning across processed foods, dairy alternatives, and functional beverages. Personal care & cosmetics account for 12%, leveraging microcrystalline cellulose for texture enhancement, suspension stabilization, and natural ingredient claims in skincare, color cosmetics, and oral care products. Other industrial applications hold 9% share, encompassing chemical processing aids, electronics applications, animal nutrition, and emerging uses in sustainable materials. The pharmaceuticals & nutraceuticals segment's leadership is reinforced by continuous growth in generic drug manufacturing, expanding nutraceutical markets globally, and increasing quality requirements driving adoption of premium excipient grades with superior purity and functionality supporting robust pharmaceutical manufacturing processes.
The microcrystalline cellulose market is advancing steadily due to increasing pharmaceutical manufacturing capacity expansion in emerging markets and growing adoption of clean-label food ingredients that provide natural functionality and consumer-friendly labeling across diverse pharmaceutical, nutraceutical, food, and personal care applications. However, the market faces challenges, including competition from alternative excipients and texture modifiers offering specialized functionality, price pressures from commodity ingredient markets affecting food-grade applications, and sustainability concerns regarding wood-sourcing practices driving demand for non-wood alternatives including agricultural residue-based cellulose. Innovation in low-nitrite grades and sustainable sourcing continues to influence product development and market expansion patterns.
The growing focus on nitrosamine impurity control in pharmaceutical products is enabling microcrystalline cellulose manufacturers to develop and commercialize ultra-low-nitrite grades that minimize nitrosamine formation risk in drug products containing secondary or tertiary amine functional groups, supporting pharmaceutical quality assurance and regulatory compliance with evolving ICH M7 guidelines. Advanced purification technologies and specialized manufacturing controls provide nitrite content below 0.1 ppm while maintaining functional properties required for tableting and capsule applications. Pharmaceutical manufacturers are increasingly recognizing the risk mitigation advantages of low-nitrite excipients for formulation safety, regulatory risk management, and proactive quality excellence across development and commercial manufacturing operations.
Modern microcrystalline cellulose producers are incorporating non-wood cellulose sources including cotton linters, bamboo fiber, and agricultural residues to enhance environmental sustainability credentials, reduce dependency on forest resources, and support circular economy principles through valorization of agricultural byproducts and textile industry waste streams. These alternative feedstocks improve environmental performance metrics while enabling differentiated product positioning addressing sustainability-focused customers and supporting corporate environmental responsibility objectives. Advanced processing technologies also allow manufacturers to achieve pharmaceutical and food-grade quality specifications from non-wood sources, creating competitive advantages in sustainability-conscious procurement decisions and supporting long-term feedstock diversification strategies mitigating supply chain concentration risks.
The emergence of advanced particle engineering techniques including spray drying, agglomeration, and surface modification is creating specialized microcrystalline cellulose grades with optimized flow properties for high-speed tableting, enhanced compressibility enabling reduced excipient loading, improved moisture management supporting stability in humid environments, and tailored dissolution profiles addressing bioavailability optimization requirements. Leading excipient suppliers are investing in proprietary manufacturing processes that create differentiated product portfolios addressing specific formulation challenges, supporting premium pricing realization, and strengthening technical partnerships with innovative pharmaceutical companies developing next-generation solid-dose products across therapeutic categories and patient populations.
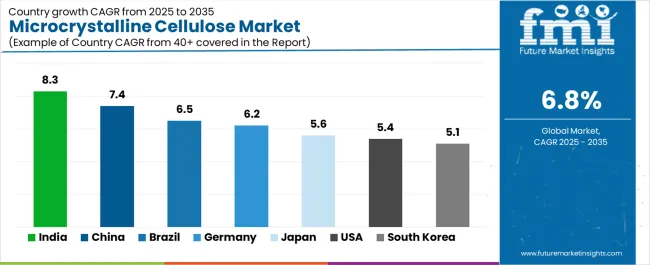
| Country | CAGR (2025-2035) |
|---|---|
| India | 8.3% |
| China | 7.4% |
| Brazil | 6.5% |
| Germany | 6.2% |
| Japan | 5.6% |
| USA | 5.4% |
| South Korea | 5.1% |
The microcrystalline cellulose market is experiencing robust growth globally, with India leading at an 8.3% CAGR through 2035, driven by expansion in generic pharmaceutical manufacturing and nutraceutical production, rising domestic excipient manufacturing capacity reducing import dependency, and fast growth in clean-label food categories addressing health-conscious consumer preferences. China follows at 7.4%, supported by large-scale pharmaceutical tableting operations serving domestic and export markets, processed foods growth driven by urbanization and convenience trends, and investments in bio-based cellulose production supporting domestic supply chain development. Brazil shows growth at 6.5%, emphasizing local pharmaceutical and nutraceutical capacity ramp-up, convenience foods penetration across expanding middle-class consumers, and import substitution strategies in pharmaceutical excipients. Germany records 6.2%, focusing on high-specification pharmaceutical demand and strong personal care formulation expertise with shift to certified low-nitrite microcrystalline cellulose grades. Japan demonstrates 5.6% growth, supported by aging-care formulations including orally disintegrating tablets, functional foods focus addressing health maintenance, and premium quality standards throughout pharmaceutical and food supply chains. The United States exhibits 5.4% growth, emphasizing strict FDA excipient control and protein/meal-replacement adoption. South Korea shows 5.1% growth, supported by K-beauty texture systems innovation and solid-dose innovation with functional foods and supplements expansion.
The report covers an in-depth analysis of 40+ countries, the top-performing countries are highlighted below.
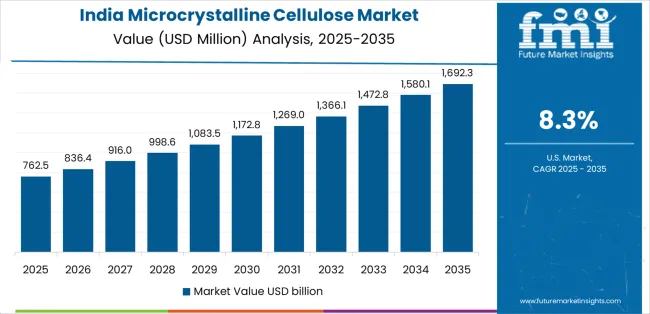
Revenue from microcrystalline cellulose in India is projected to exhibit exceptional growth with a CAGR of 8.3% through 2035, driven by rapid expansion in generic pharmaceutical manufacturing serving domestic healthcare needs and global export markets, rising domestic excipient production capacity reducing historical import dependency and supporting pharmaceutical industry cost competitiveness, and fast growth in clean-label food categories addressing increasingly health-conscious urban consumers demanding natural ingredients. The country's position as a global generics manufacturing hub and expanding food processing sector are creating substantial demand for pharmaceutical and food-grade microcrystalline cellulose across diverse applications and quality tiers. Major international excipient suppliers and domestic cellulose manufacturers are establishing comprehensive microcrystalline cellulose production and distribution capabilities to serve both domestic pharmaceutical markets and export opportunities.
Revenue from microcrystalline cellulose in China is expanding at a CAGR of 7.4%, supported by the country's enormous pharmaceutical manufacturing scale serving the world's largest population and significant generic drug export capabilities, processed foods growth driven by urbanization, rising incomes, and convenience-seeking consumers, and investments in bio-based cellulose production infrastructure supporting domestic raw material security and environmental sustainability objectives. The country's integrated pharmaceutical and food manufacturing ecosystems and government support for industry upgrading are driving demand for diverse microcrystalline cellulose grades. International excipient companies and Chinese cellulose producers are establishing extensive manufacturing and technical service capabilities to address growing microcrystalline cellulose demand.
Revenue from microcrystalline cellulose in Brazil is growing at a CAGR of 6.5%, driven by local pharmaceutical and nutraceutical manufacturing capacity ramp-up addressing domestic healthcare requirements and regional export opportunities, convenience foods penetration driven by urbanization and expanding middle-class purchasing power, and import substitution strategies in pharmaceutical excipients supporting domestic industry development and supply chain security. The country's large healthcare market and growing food processing sector are supporting demand for pharmaceutical and food-grade microcrystalline cellulose across applications. International excipient suppliers and domestic chemical companies are establishing comprehensive capabilities to serve Brazilian microcrystalline cellulose markets.
Revenue from microcrystalline cellulose in Germany is expanding at a CAGR of 6.2%, supported by the country's high-specification pharmaceutical demand requiring ultra-high purity and comprehensive analytical documentation for branded and generic drug manufacturing, strong personal care formulation expertise driving innovative texture system development, and shift to certified low-nitrite microcrystalline cellulose grades addressing proactive nitrosamine risk mitigation in pharmaceutical quality management. Germany's leadership in pharmaceutical quality standards and advanced cosmetic formulation are driving demand for premium microcrystalline cellulose grades. Leading excipient suppliers and specialty cellulose manufacturers are investing in specialized capabilities to serve German quality requirements and innovation-driven markets.
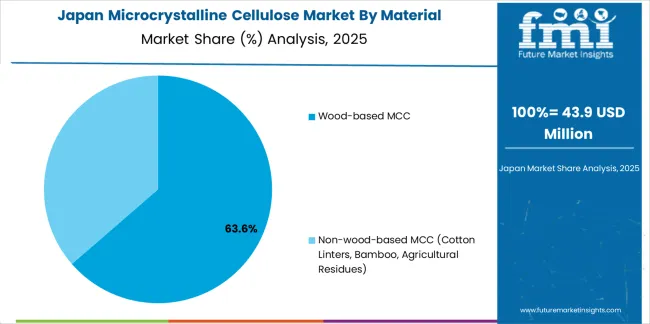
Revenue from microcrystalline cellulose in Japan is expanding at a CAGR of 5.6%, supported by the country's focus on aging-care formulations including orally disintegrating tablets addressing swallowing difficulties in elderly populations, functional foods emphasis supporting health maintenance and disease prevention through nutritional intervention, and premium quality standards requiring exceptional purity, consistent functionality, and comprehensive documentation throughout pharmaceutical and food supply chains. Japan's sophisticated healthcare system and quality-conscious consumers are driving demand for specialized microcrystalline cellulose grades. Leading Japanese pharmaceutical companies and excipient suppliers are investing in specialized capabilities to serve domestic quality requirements and demographic trends.
Revenue from microcrystalline cellulose in the United States is expanding at a CAGR of 5.4%, supported by the country's strict FDA excipient control requiring comprehensive quality documentation and regulatory compliance supporting pharmaceutical product quality and patient safety, protein and meal-replacement adoption driving functional beverage and nutritional bar development utilizing microcrystalline cellulose for texture and suspension properties, and steady contract development and manufacturing organization demand supporting pharmaceutical outsourcing trends and formulation development services. The nation's mature pharmaceutical market and innovative food technology sectors are driving demand for diverse microcrystalline cellulose applications. Major excipient suppliers and food ingredient companies are investing in comprehensive microcrystalline cellulose supply chains to serve both pharmaceutical and food markets.
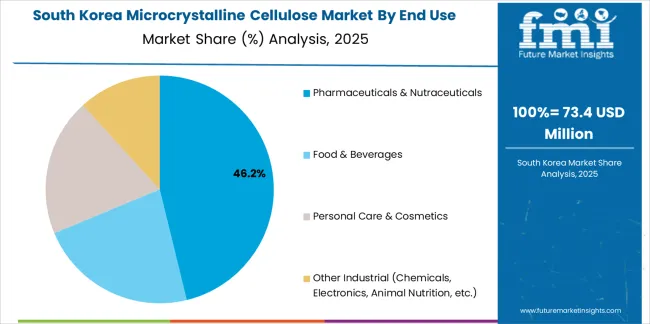
Revenue from microcrystalline cellulose in South Korea is growing at a CAGR of 5.1%, driven by the country's influential K-beauty industry's adoption of microcrystalline cellulose in texture systems for cushion foundations, pressed powders, and skincare formulations emphasizing smooth application and natural finish, solid-dose pharmaceutical innovation addressing domestic healthcare needs and export opportunities in generic and specialty pharmaceuticals, and functional foods and supplements expansion driven by health-conscious consumers and preventive healthcare culture. The country's dynamic beauty industry and advanced pharmaceutical sector are supporting investment in microcrystalline cellulose capabilities. Korean cosmetic companies and pharmaceutical manufacturers are establishing comprehensive microcrystalline cellulose formulation expertise to support product innovation and export growth.
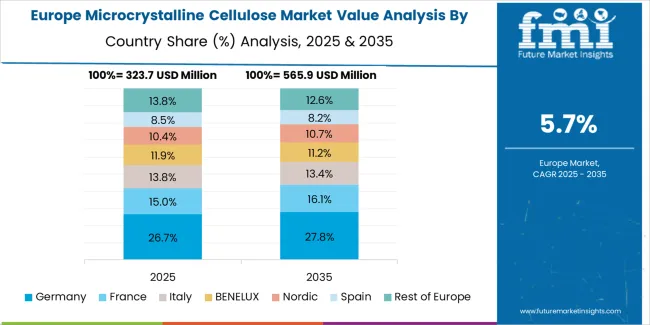
The microcrystalline cellulose market in Europe is projected to grow from USD 412 million in 2025 to USD 692 million by 2035, registering a CAGR of 5.3% over the forecast period. Germany is expected to maintain its leadership position with a 22.4% market share in 2025, holding at 22.4% by 2035, supported by its large prescription and over-the-counter pharmaceutical manufacturing base, strict excipient quality regimes, and strong tableting capacity in North Rhine-Westphalia and Bavaria industrial regions.
The United Kingdom follows with 16.3% in 2025, projected to reach 16.5% by 2035, driven by branded generics manufacturing, contract development and manufacturing organization activity, and high clean-label food adoption among health-conscious consumers. France holds 14.6% in 2025, rising to 14.8% by 2035, supported by broad pharmaceutical manufacturing and premium cosmetics usage across beauty and personal care applications. Italy commands 10.2% in 2025, projected to reach 10.4% by 2035, driven by solid-dose pharmaceutical hubs in Lombardy and Emilia-Romagna regions. Spain accounts for 7.5% in 2025, expected to reach 7.7% by 2035, supported by growing nutraceuticals sector and food applications. The Netherlands maintains 5.9% in 2025, growing to 6% by 2035, driven by food technology expertise and pharmaceutical distribution strengths. The Rest of Europe region, including Nordic countries, Central and Eastern Europe, Benelux excluding Netherlands, Ireland, Switzerland, Austria, and other markets, holds 23.1% in 2025, moderating to 22.2% by 2035, attributed to momentum in specialty pharmaceutical manufacturing, dairy applications, and sustainable packaging fiber development across emerging markets implementing advanced excipient standards and clean-label food formulations.
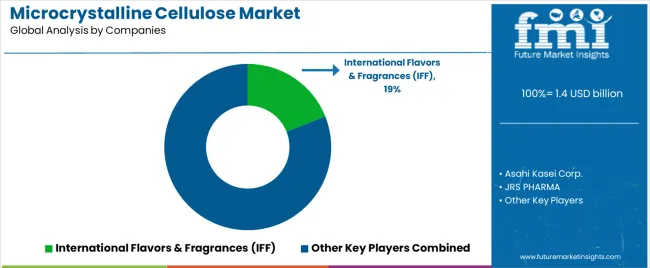
The Microcrystalline Cellulose (MCC) Market features approximately 30–40 players with moderate concentration, where the top three companies collectively account for around 44–50% of global share driven by advanced cellulose processing technologies, strong excipient portfolios, and entrenched relationships with leading oral solid dosage manufacturers. The leading company, International Flavors & Fragrances (IFF), holds an estimated 19% share supported by its globally recognized Avicel® product line, vertically integrated production capabilities, and proven performance across direct compression, wet granulation, and continuous manufacturing systems. Competition is shaped by excipient functionality, grade consistency, regulatory compliance, and technical support strength rather than price-based differentiation.
Market leaders including IFF, Asahi Kasei Corp., and JRS PHARMA maintain their competitive edge through GMP-compliant facilities, proprietary co-processed MCC technologies, and multi-compendial certifications (USP–NF, EP, JP) that enable broad global adoption. Their strengths include superior compressibility, controlled particle-size distribution, low bioburden levels, and extensive Drug Master File (DMF) support, which create high switching barriers for pharmaceutical manufacturers with validated tablet formulations. These companies continue expanding into next-generation MCC grades suited for high-speed tableting, orally disintegrating formats, and nutraceutical applications requiring enhanced flow characteristics.
Challenger companies such as DFE Pharma, Roquette Frères, Mingtai Chemical, and Sigachi Industries compete through diversified excipient portfolios, regionally optimized production footprints, and specialized MCC grades tailored for moisture-sensitive formulations, high-load direct compression blends, and dietary supplement manufacturing. Their expertise in tailoring particle morphology and flowability provides competitive traction across mid-sized pharmaceutical firms and contract manufacturers.
Additional pressure comes from regional and specialty excipient suppliers including Accent Microcell, Anhui Shanhe Pharma Accessories, Chemfield Cellulose, Blanver Farmoquímica, Avantor, Spectrum Chemical, and Juku Orchem. These firms build advantages through cost-effective production, agile regulatory support, localized distribution networks, and the ability to supply pharmacopeia-compliant MCC to fast-growing markets in Asia and Latin America. Overall, competitive dynamics favor manufacturers capable of combining high-purity cellulose processing, strong regulatory documentation, and application-specific technical support across the global solid-dose formulation ecosystem.
Microcrystalline cellulose represents a specialized functional ingredient segment within pharmaceutical excipients and food additives, projected to grow from USD 1.4 billion in 2025 to USD 2.7 billion by 2035 at a 6.8% CAGR. This purified, partially depolymerized cellulose primarily wood-based pharmaceutical grades for solid-dose applications is produced through acid hydrolysis and spray drying processes to serve as a multifunctional excipient providing binding, disintegration, and flow properties in pharmaceutical tablets and capsules, a texture modifier and fat replacer in food products, and a rheology modifier in personal care formulations. Market expansion is driven by increasing generic pharmaceutical manufacturing capacity, growing clean-label food ingredient demand, expanding nutraceutical production, and rising emphasis on pharmaceutical quality improvement including nitrosamine risk mitigation across diverse pharmaceutical, food, and personal care applications.
| Items | Values |
|---|---|
| Quantitative Units (2025) | USD 1.4 billion |
| Material | Wood-based MCC, Non-wood-based MCC (Cotton Linters, Bamboo, Agricultural Residues) |
| Product Grade | Pharmaceutical Grade, Food Grade, Industrial/Technical Grade |
| End Use | Pharmaceuticals & Nutraceuticals, Food & Beverages, Personal Care & Cosmetics, Other Industrial (Chemicals, Electronics, Animal Nutrition) |
| Regions Covered | Asia Pacific, North America, Europe, Latin America, Middle East & Africa |
| Countries Covered | India, China, Brazil, Germany, Japan, United States, South Korea, and 40+ countries |
| Key Companies Profiled | International Flavors & Fragrances (IFF), Asahi Kasei Corp., JRS PHARMA, DFE Pharma, Roquette Frères, Mingtai Chemical, Sigachi Industries, Accent Microcell, Anhui Shanhe Pharma Accessories, Chemfield Cellulose, Blanver Farmoquímica, Avantor, Spectrum Chemical, Juku Orchem |
| Additional Attributes | Dollar sales by material, product grade, and end-use categories, regional demand trends, competitive landscape, technological advancements in particle engineering, low-nitrite grade development, sustainable sourcing innovation, and pharmaceutical quality optimization |
The global microcrystalline cellulose market is estimated to be valued at USD 1.4 billion in 2025.
The market size for the microcrystalline cellulose market is projected to reach USD 2.7 billion by 2035.
The microcrystalline cellulose market is expected to grow at a 6.8% CAGR between 2025 and 2035.
The key product types in microcrystalline cellulose market are wood-based mcc and non-wood-based mcc (cotton linters, bamboo, agricultural residues).
In terms of end use, pharmaceuticals & nutraceuticals segment to command 46.0% share in the microcrystalline cellulose market in 2025.
Our Research Products

The "Full Research Suite" delivers actionable market intel, deep dives on markets or technologies, so clients act faster, cut risk, and unlock growth.

The Leaderboard benchmarks and ranks top vendors, classifying them as Established Leaders, Leading Challengers, or Disruptors & Challengers.

Locates where complements amplify value and substitutes erode it, forecasting net impact by horizon

We deliver granular, decision-grade intel: market sizing, 5-year forecasts, pricing, adoption, usage, revenue, and operational KPIs—plus competitor tracking, regulation, and value chains—across 60 countries broadly.

Spot the shifts before they hit your P&L. We track inflection points, adoption curves, pricing moves, and ecosystem plays to show where demand is heading, why it is changing, and what to do next across high-growth markets and disruptive tech

Real-time reads of user behavior. We track shifting priorities, perceptions of today’s and next-gen services, and provider experience, then pace how fast tech moves from trial to adoption, blending buyer, consumer, and channel inputs with social signals (#WhySwitch, #UX).
Partner with our analyst team to build a custom report designed around your business priorities. From analysing market trends to assessing competitors or crafting bespoke datasets, we tailor insights to your needs.
Supplier Intelligence
Discovery & Profiling
Capacity & Footprint
Performance & Risk
Compliance & Governance
Commercial Readiness
Who Supplies Whom
Scorecards & Shortlists
Playbooks & Docs
Category Intelligence
Definition & Scope
Demand & Use Cases
Cost Drivers
Market Structure
Supply Chain Map
Trade & Policy
Operating Norms
Deliverables
Buyer Intelligence
Account Basics
Spend & Scope
Procurement Model
Vendor Requirements
Terms & Policies
Entry Strategy
Pain Points & Triggers
Outputs
Pricing Analysis
Benchmarks
Trends
Should-Cost
Indexation
Landed Cost
Commercial Terms
Deliverables
Brand Analysis
Positioning & Value Prop
Share & Presence
Customer Evidence
Go-to-Market
Digital & Reputation
Compliance & Trust
KPIs & Gaps
Outputs
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE
Cellulose Diacetate Film Market Size and Share Forecast Outlook 2025 to 2035
Cellulose Fiber Market Forecast and Outlook 2025 to 2035
Cellulose Derivative Market Size and Share Forecast Outlook 2025 to 2035
Cellulose Film Packaging Market Size and Share Forecast Outlook 2025 to 2035
Cellulose Ether and Derivatives Market Size and Share Forecast Outlook 2025 to 2035
Cellulose Esters Market Size and Share Forecast Outlook 2025 to 2035
Cellulose Nanocrystals and Nanofibers Market Size and Share Forecast Outlook 2025 to 2035
Cellulose Gel Market Growth, Forecast, and Analysis 2025 to 2035
Market Share Insights for Cellulose Fiber Providers
Key Players & Market Share in Cellulose Film Packaging Market
Microcrystalline Wax Market
Nanocellulose Coating Market Analysis - Size and Share Forecast Outlook 2025 to 2035
Nanocellulose Barrier Coating Market Analysis - Size and Share Forecast Outlook 2025 to 2035
Nanocellulose Market Report - Demand, Growth & Industry Outlook 2025 to 2035
Ethyl Cellulose Market
Methyl Cellulose Market
Powdered Cellulose Market Analysis - Size, Share, and Forecast Outlook 2025 to 2035
Demand for Cellulose Derivative in Japan Size and Share Forecast Outlook 2025 to 2035
Demand for Cellulose Derivative in USA Size and Share Forecast Outlook 2025 to 2035
Demand for Cellulose Diacetate Film in Middle East & Africa Size and Share Forecast Outlook 2025 to 2035

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.
Chat With
MaRIA